Introduction
Elasmobranchs are important due to their ecological role as meso and top predators, as fisheries resources in several nations, and more recently because of the current threatened conservation status of many species (Dulvy et al., 2014; IUCN, 2020). They have a complex reproductive system, which includes specialized structures, internal fertilization and long gestation periods (Hamlett, 2005).
Much is known about elasmobranch reproduction and development, see Pratt and Carrier (2001) and Hamlett (2005) for reviews. However, we are just beginning to understand the elaborate pre-copulatory and copulatory behaviors exhibited by elasmobranchs from caught specimens, laboratory examinations of reproductive structures and physiological studies, as well as from animals kept in captivity. Copulation has been directly observed in captivity, but it is still limited in the wild, with some examples involving Hypanus americanus (Chapman et al., 2003), Ginglymostoma cirratum (Carrier et al., 1994; Pratt & Carrier, 2001), Taeniura meyeni (Ritter &Vargas, 2015), Triaenodon obesus; (Pratt & Carrier, 2005), Urolophus jamaicensis (Young, 1993) and Mobula alfredi and M. birostris (Stevens et al., 2018).
Although the reproductive behaviors of elasmobranchs are likely to be relatively common in nature, they are still rarely observed and poorly documented. Elasmobranchs in mating behavior are reclusive and rarely seen in human presence. Even if mating is witnessed, the entire event is rarely observed and recorded. This is especially true for the larger sharks, which are often reclusive, challenging to find and sometimes dangerous to approach when reproductively active. Thus, such records are mostly dependent on casual or one-of-a-kind observations, except for the only well-characterized mating ground used by nurse sharks, G. cirratum, in the Dry Tortugas, Florida (Pratt & Carrier, 2001). Consequently, there is a poor understanding of habitat requirements for mating of most shark species, hampering part of the management decisions and conservation efforts directed to this threatened group of fishes.
Knowledge on the reproductive behavior of the lemon shark, Negaprion brevirostris (Poey, 1868), is also scarce. Clark (1963) reported a copulation event in captivity, in which individuals of lemon shark performed coordinated pair swimming while copulating (‘parallel swimming in copula’ sensu) (Pratt & Carrier, 2001) for a period of at least 30 minutes. No other record of courtship and mating behavior in lemon sharks has been published ever since.
The lemon shark is a large (at least 340 cm total length (TL)) inshore Carcharhinidae that inhabits the continental and insular shelves in disjoint populations in the Western Atlantic (from New Jersey to southern Brazil), Eastern Atlantic and Eastern Pacific Oceans (Ebert et al., 2013). Globally, the species had its status recently changed from Near Threatened to Vulnerable (Carlson et al., 2021) and it is also listed as Vulnerable along the Brazilian coast, due to population reduction and decrease in its geographical distribution (Rosa et al., 2018).
Citizen science, the participation of non-experts in data collection, reporting and monitoring under the supervision of scientists, is emerging as one of the most important issues since the environmental movement of the 1960-70’s (Peter et al., 2021). The collaboration between scientists and those who are just curious or concerned and motivated with nature has become an established practice with an increasing number of projects in different disciplines, delivering information to support science and management (Cunha et al., 2017 and references therein), including shark research (Araujo et al., 2016; Bargnesi et al., 2020; Giovos et al., 2019; Vianna et al., 2014). In this sense, reports on the reproductive behavior of elasmobranchs provided by opportunistic observations and video and photo records of non-experts such as recreational divers and dive guides can be especially valuable to the documentation of interactions that are rarely observed by researchers.
Within this perspective and based on reports provided by citizen science, we describe an area in the Equatorial Atlantic used as a mating ground by N. brevirostris and describe part of the species pre-copulatory behaviors. We discuss the importance of fortuitous records made by non-scientists as a complementary tool for ecological studies of sharks in nature, especially in the context of Marine Protected Areas (MPAs) and the identification of critical habitats for shark conservation.
Materials and methods
Study site: Data come from the Fernando de Noronha Archipelago (FEN), an isolated group of volcanic islands located in the equatorial region of the Western South Atlantic, 345 km off the North-eastern coast of Brazil (03°52’W & 32°25’S) (Fig. 1). FEN is under the influence of the South Equatorial Current, with mean water temperature of 26 °C and a tropical oceanic climate with two distinct seasons, a wet season from February to July and a dry season during the rest of the year (Maida & Ferreira, 1997). FEN has been designated as an UNESCO World Heritage since 2001 and 70 % of its total area encompass a no-take Marine Protected Area (MPA) established in 1988. The archipelago is known for the occurrence of several species of elasmobranchs, which use the area as breeding and feeding grounds (Aguiar et al., 2009; Garla et al., 2006; Garla et al., 2017). This site is also a major lemon shark nursery in the South Atlantic Ocean (Garla et al. 2009).
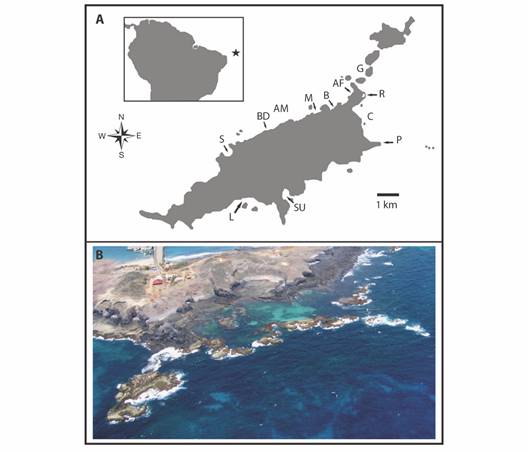
Fig. 1 A. Geographical localization of Fernando de Noronha Archipelago (depicted by the star in the inset) in the Atlantic Ocean and location of the sites where lemon sharks, Negaprion brevirostris, where observed between 2004 and 2019: AF (Air France), AM (Americanos), B (Biboca), C (Caieiras), G (Sela Gineta), L (Leão), P (Pontinha), M (Praia do Meio), S (Sancho), SU (Sueste), R (Buraco da Raquel lagoon). B. Indicates the lagoon (R), in which most of the adult sharks were sighted between 2004 and 2014 (Photograph by J. C. Marenga).
Data from citizen scientists: The information used in the present study was provided by two groups of citizen scientists and obtained through two means of observation between 2004 and 2019: i) underwater observations of divers (tourists and residents) and MPA rangers, and ii) observations made from one observatory on the top of a cliff and from the shoreline (i.e., above the water), respectively.
Underwater observations: Snorkeling surveys were conducted between 2004 and 2014 at different sites on the main island of FEN. However, most of the observations were concentrated in a lagoon with approximately 44 000 m2 located at the windward side of FEN, in which adult lemon sharks are known to aggregate (Fig. 1A, site R; Fig. 1B). Named as “Buraco da Raquel”, this lagoon is subject of rough seas and thus surveys were occasionally performed, depending on calmer sea conditions, which usually occur during the austral summer (from December to March). Surveys conducted in the lagoon and elsewhere usually consisted of one or two divers following random routes in depths ranging from 0.5 to 5 m, searching for lemon sharks. Upon sighting a shark, digital photographs and videos were taken. Each dive lasted approximately 50 min and was performed in slow and constant speed, with no changes in direction or itinerary. All dives were performed during daylight hours (06:00 to 18:00 h), in absence of large waves (> 1 m), and with water visibility of at least 4 m. The approximate total length (TL) of all sharks sighted was estimated at up to 2 m of total length (TL) or ≥ 2 m TL, based on comparison with objects such as rocks and patch reefs.
Observations above the water: Additional records were made from one observatory in January 2008 by a tourist, and in February 2007 and February 2019 by MPA rangers on the beach (Fig. 1A, sites R, L and AM, respectively). In those three occasions, observers provided videos, digital photographs and brief descriptions. Shark size estimates of the events in February 2007 and 2019 were made in the same manner as described above.
Data analysis: Digital photographs and videos were analyzed using Windows Photo Viewer and Windows Media Player to validate the identity of lemon sharks. For all records, the size (total length-TL) and the sex of the sharks were determined, using an object of known size and by assessing the presence or the absence of claspers, respectively. Behaviors were interpreted according to Pratt and Carrier (2001) and body marks that could indicate mating bites were based on Ritter and Amin (2019).
Results
Underwater observations: A total of 328 records of N. brevirostris were reported from 177 dives made by tourists and residents (approximately 148 h of underwater observations) (Table 1). Dives were concentrated during the austral summer (December to March), when sea conditions were better. The number of lemon sharks ≥ 2 m TL sighted per dive ranged from 1 to 12 individuals (mean= 1 ± 1). Solitary individuals were spotted in 42 dives. Most of the records came from the “Buraco da Raquel” lagoon (site R; 232 records in 122 dives) (Table 2).
Table 1 Underwater records of Negaprion brevirostris made by citizen scientists in different sites of Fernando de Noronha Archipelago (see Fig. 1A for locations).
| Site | 2004 | 2005 | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | 2014 | 2019* | Total |
| AirFrance (AF) | 1J (1) | - | - | - | 5J (3) | - | - | - | - | - | - | - | 6 |
| Americanos (AM) | - | - | - | - | - | - | - | - | - | - | - | 2A, 1J (1) | 3 |
| Biboca (B) | 5J (2) | 2J (2) | - | 17J (10) | 1J (1) | - | - | - | - | - | - | - | 25 |
| Caieiras (C) | 1J (1) | 1J (1) | 2J (2) | - | 5J (5) | 2J (2) | 12J (7) | 5J (3) | - | - | - | - | 28 |
| Gineta (G) | - | 1J (1) | - | - | - | - | - | - | - | - | - | - | 1 |
| Leão (L) | - | - | - | 2A (1) | - | - | - | - | - | - | - | - | 2 |
| Pontinha (P) | - | - | 1J (1) | - | - | - | - | - | - | - | - | - | 1 |
| Meio (M) | - | - | 9J (4) | - | - | - | - | - | - | - | - | - | 9 |
| Sancho (S) | - | - | - | - | - | - | - | - | 1A, 2J (2) | 8A, 8J (4) | - | - | 19 |
| Sueste (SU) | - | - | 1J (1) | 1J (1) | - | - | - | - | - | - | - | - | 2 |
| Raquel (R) | 2A, 2J (3) | - | 27A, 12J (20) | 13A, 27J (23) | 23A, 18J (23) | 15A, 19J (17) | 14A, 30J (21) | 13A, 5J (9) | 3A, 5J (4) | - | 2A, 2J (2) | - | 232 |
| Total | 11 | 4 | 52 | 60 | 52 | 36 | 56 | 23 | 11 | 16 | 4 | 328 |
A: number of sharks ≥ 2 m TL. J: number of sharks up to 2 m TL. Numbers in parentheses represent the number of dives at each site. *No sampling was performed between 2015 and 2018.
Table 2 Underwater records of Negaprion brevirostris made by citizen scientists in Buraco da Raquel lagoon, Fernando de Noronha Archipelago (see Fig. 1B for location).
| Month | 2004 | 2005* | 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013* | 2014 | Total |
| J | 1A, 2J (2) | - | 9A, 5J (6) | 4A, 1J (4) | 17A, 3J (11) | 7A, 5J (5) | 5A, 1J (3) | 6A, 1J (3) | 1A, 5J (3) | - | 2A (1) | 51A, 18J (36) |
| F | - | - | 15A, 5J (10) | 6A, 8J (8) | 1A (1) | - | 2A, 1J (2) | 6A, 4J (6) | 2A (1) | - | 2J (1) | 32A, 19J (30) |
| M | - | - | 1J (1) | 3J, (3) | 1A, 1J (2) | 8J (5) | 0 (1) | - | - | - | - | 1A, 13J (12) |
| A | 1A (1) | - | - | 2A, 11J (4) | 4J (2) | 3J (2) | 5J (2) | - | - | - | - | 3A, 21J (11) |
| M | - | - | - | - | 4J (2) | - | 3J (3) | - | - | - | - | 7J (5) |
| J | - | - | - | - | - | - | 1A (1) | - | - | - | - | 1A (1) |
| J | - | - | - | - | - | - | - | - | - | - | * | |
| A | - | - | - | 1A, 4J (4) | - | - | 1A (1) | - | - | - | - | 2A, 4J (5) |
| S | - | - | - | - | - | - | - | - | - | - | * | |
| O | - | - | - | - | - | - | 14J (3) | - | - | - | - | 14J (3) |
| N | - | - | 1A, (1) | - | 1A, 1J (2) | 8A, 3J (5) | 2J (1) | - | - | - | - | 10A, 6J (4) |
| D | - | - | 2A, 1J (2) | - | 3A, 5J (3) | - | 5A, 2J (4) | 1A (1) | - | - | - | 11A, 8J (10) |
| Total | 2A, 2J (3) | * | 27A, 12J (20) | 13A, 27J (23) | 23A, 18J (23) | 15A, 19J (17) | 14A, 30J (21) | 13A, 5J (9) | 3A, 5J (4) | * | 2A, 2J (2) | 112A, 120J (122) |
A: number of sharks ≥ 2 mTL. J: number of sharks up tp 2 m TL. Numbers in parentheses represent the number of dives reported per year. Asterisks indicate months or year with no observations/dives.
Females presenting fresh scars were recorded in 22 of the 181 dives (18 % of the total). These scars were considered as mating-related wounds since they were predominantly associated to gills or observed between the pectoral and pelvic girdles (Fig. 2A, Fig. 2B, Fig. 2C). Pregnant females were recorded in 24 dives (19 %) (Fig. 2D, Fig. 2E, Fig. 2F). Adult males were sighted in 24 dives (18 %), including an individual presenting fresh scars (Fig. 2G, Fig. 2H, Fig. 2I). A summary of the numbers of females with mating scars, pregnant females, and adult males observed per month between 2004 and 2014 is presented in Fig. 3.

Fig. 2 A-B. Lemon sharks, Negaprion brevirostris, females with mating scars; C. Female ≤ 2 m TL presenting mating scars; D-F. pregnant females; G-H. Adult males observed in Buraco da Raquel lagoon between 2004 and 2014 (site R); I. Adult male with bite scars (Photographs by L. B. Veras). J. Male and female sharks a few minutes before the mating activities recorded in site L in February 2007 (Photograph by F. S. Buloto). K-L. Three lemon sharks observed swimming in close contact nearby the shoreline of site AM in February 2019 (Photographs by D. O. Cavalheiro) (see Fig.1B for location of sites).
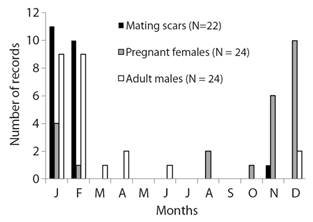
Fig. 3 Number of sightings of females with mating scars, pregnant females, and adult males of lemon sharks, Negaprion brevirostris, per month between 2004 and 2014 in Fernando de Noronha Archipelago.
Descriptions of mating events: A mating event was recorded in site R (Fig. 1A) on 26 January 2008 around 1:00 PM during the ebb tide. Two adult sharks close to the shoreline (< 0.5 m depth) were sighted by tourists from the top of an observatory in front of site R. Sharks swam slowly over the reef bottom, with an individual biting and holding the caudal and pelvic regions of the other. The shark that was held tried to escape. The tourists’ report suggested that this event was a mating attempt, although the female did not appear to be receptive. This interaction lasted for about one minute (Garla, 2021a). As soon as one of the authors (LBV) was informed about the event, he entered the water to record details of this interaction. It was possible to confirm the tourists’ perception, as an adult male and an adult female were observed close to each other. The mating event itself and the male’s attempts to hold the female were not observed during the dive. However, the female had fresh bite marks and swam in a slowly and apparent extenuated condition.
MPA rangers described similar events in sites L and AM (Fig. 1A): a) On February 2nd, 2007, at 4:00 PM during the flood tide, two lemon sharks were observed swimming in close contact nearby the shoreline of a sandy beach in site L (Fig. 2J). A shark measuring about 2.5 m TL was observed with several bite marks in the dorsum. The event was initially observed from above water, but after sighting the female with bite marks, two MPA rangers entered the water. The rangers identified the shark as a female, which was swimming slowly, dragging the fins at the bottom, in an apparent exhausted condition (Garla, 2021b). Then, an adult male with approximately 2 m TL quickly approached the female and bit it in the left pectoral fin and in the dorsum. The rangers referred to the sound produced by the bites as the sound of “wood sticks being broken”. The male managed to bite the female’s dorsum firmly and swam roughly trying to push it to the surface, while the female tried to return to the bottom. After 2-3 min the male released the female and both sharks remained motionless on the sand bottom for five min, apparently exhausted, and then swan to deeper waters out of sight. The whole event lasted approximately 20 min. Few minutes after the rangers had left the water, they saw both sharks again in the shallows; b) On March 5th, 2019, at 1:00 PM during the flood tide in site AM, a MPA ranger recorded three lemon sharks (two individuals > 2 m TL and a third shark measuring approximately 2 m TL) swimming frantically close to the shoreline. Sharks were constantly in physical contact and the larger individual was actively pursued and herded against the shoreline by the two other sharks (Fig. 2K, Fig. 2L). The larger pursuer shark swam with greater vigor and had a notable prevalence over the smaller one. The pursuers were more efficient in herding the pursued shark over rocky bottoms in comparison to sandy substrates. The ranger also mentioned that this episode was different from other “patrolling shoreline” events (Garla et al., 2017) previously witnessed by him elsewhere in FEN, due to the physical contact and the fact that two sharks insistently tried to herd the third shark into the shallows. The whole event lasted for about 10 min, after which the sharks apparently swam to deeper waters and were not sighted again.
Discussion
The events involving mating behavior of N. brevirostris were observed during distinct times of the day and in different sites of FEN, both in sandy beaches and reef environments. Although the mating event itself was not witnessed, the repeated sightings of females with fresh mating scars and pregnant females recorded during the austral summer in most of the sampled years attests that FEN, and particularly the Buraco da Raquel lagoon, serves as both as mating area and nursery ground for the species. Observations also suggest that lemon sharks were attempting to courtship and mate in very shallow waters (≤ 1 m depth).
The observation and captures of neonate individuals of N. brevirostris mainly occur between November and March in FEN (Garla et al., 2009), coinciding with the period with the highest number of records of females with fresh mating scars and pregnant females presented herein. Although sampling in Buraco da Raquel lagoon was concentrated within this period, it is noteworthy that females ≥ 2 m TL captured for tagging during other periods of the year in FEN did not show scars (Garla, 2020). Furthermore, adult females caught in late February have only well-healed scars. Thus, results suggest that mating and parturition probably occur simultaneously in FEN and are mainly concentrated during the peak of the austral summer, between December and mid-February.
Records of females with mating scars were relatively rare elsewhere in FEN. The Buraco da Raquel lagoon, located within the undisturbed MPA, a National Park, is currently known as the main adult aggregation site of N. brevirostris in shallow waters of FEN. Although similar efforts have been made in other locations, the number of observations of aggregations of adult sharks in other sites are comparatively lower. This lagoon is one of the few sites in FEN with sheltered and calm waters on the windward side of the archipelago. Therefore, all features mentioned above suggest that Buraco da Raquel lagoon is an essential habitat for lemon sharks. To date, aggregations of adult individuals of lemon sharks in FEN are only known for shallow habitats, although they might also congregate in deeper habitats around the archipelago for reproductive or other purposes, such as reported in the deeper waters off Florida (Kessel et al., 2014). Thus, identifying critical habitats to the life cycle of vulnerable species such as N. brevirostris is crucial for designing efficient protection strategies.
The pre-mating parallel swimming described by Clark (1963) was not observed in FEN, but the event recorded in site AM could be interpreted as the close-following behavior described by Johnson and Nelson (1978). The close contact and herding behavior recorded in that event suggest that pre-mating activities may also involve tactile stimuli as observed by Ritter and Amin (2019).
Records of females ≤ 2 m TL without the typical robust constitution of mature females, but presenting mating scars, were also obtained in Buraco da Raquel lagoon throughout the study period. It is reasonable that fishes can distinguish between virgins and mated females by differences in the composition of their pheromone releases (Guevara-Fiore et al., 2010). It is also known that some immature shark female (e.g., Prionace glauca) show mating scars and can store sperm until they reach adulthood (Calich & Campana, 2015; Stevens, 1976). Thus, the observation of apparently immature females with mating scars opens the question whether mature males are able to distinguish between receptive and non-receptive females and differentiate virgin and mated females, as in some situations they appear to simply try to copulate based on opportunity (Ritter & Amin, 2019). The records of apparent immature females with mating scars and the presence of a pursuing shark ≤ 2 m TL in site AM on February 2019 further suggest that immature individuals may be attracted by the reproductive frenzy of adult sharks, and in the case of females, even participate of the mating activities, voluntarily or not.
N. brevirostris is becoming increasingly rare along the Brazilian coast (Rosa et al., 2018). For this reason, FEN and the nearby oceanic MPA (Atol das Rocas, located 148 km westward) are currently regarded as the main sites to maintain the species in Northeastern Brazil as well as in the Equatorial Atlantic. Besides being important nurseries for N. brevirostris (Freitas et al., 2006; Garla et al., 2009), results presented herein and reports from Atol das Rocas researchers (Oliveira, 2003) attest that both locations are also used as mating areas by lemon sharks.
The reason why lemon sharks are attracted specifically to the lagoon in FEN is still speculative. For G. cirratum in FEN and in Dry Tortugas, the use of sheltered, shallow, and structurally complex bottoms may be related to mate selection of mating avoidance (Afonso et al., 2016; Carrier & Pratt, 1998). On the other hand, observations herein suggest that it might be easier to males to herd and grasp females against the shoreline and structured bottoms, although there is no information if they are able to resume copulation in this habitat. Whatever the case, mating activities in shallow sites are vulnerable to the disruptive effects of human presence and activities such as boat traffic, swimming and diving, and has the potential of interfering with successful reproduction of the species (Carrier & Pratt, 1998), which is already vulnerable. Both in FEN and in Atol das Rocas, the access to the mating areas is already restricted to researchers and guides accompanying them. For other mating sites that might be identified in the future, the suggestion is that specific proposals to protect mating grounds follow at least part of the recommendations of Carrier and Pratt (1998) for G. cirratum, including (1) restriction of the public during times identified as essential for mating, and (2) implementation a program of public information and education.
As the observations of mating behaviors are uncommon in nature, the citizen science could be crucial for a better understanding of this kind of interaction to improve the knowledge of sharks in the wild. In the past 15 years, the lead author (RCG) has unsuccessfully tried to start citizen science initiatives in FEN, probably because the volunteers needed to fill out and physically deliver paper forms at specific collection points. However, the current dissemination of smartphones, social media and citizen science projects greatly increases the successful execution of this kind of initiative nowadays.
In summary, the results demonstrate that (1) FEN is used as a mating area by lemon sharks in the Equatorial Atlantic, (2) the main adult aggregation and mating site of lemon sharks in FEN is the Buraco da Raquel lagoon during the austral summer, (3) shallow waters in FEN should be considered essential habitats for lemon shark in current and future management planning to avoid shark disturbance, with a particular emphasis in the Buraco da Raquel lagoon, and (4) data collection by citizen scientists should be encouraged to complement research data collection and monitoring in FEN.
Ethical statement: the authors declare that they all agree with this publication and made significant contributions; that there is no conflict of interest of any kind; and that we followed all pertinent ethical and legal procedures and requirements. All financial sources are fully and clearly stated in the acknowledgements section. A signed document has been filed in the journal archives.












 uBio
uBio 


