Introduction
The family Loranthaceae comprises 73 genera and around 900 species of hemiparasitic plants distributed mainly in the tropics (Nickrent, Malécot, Vidal-Russell, & Der, 2010; Kuijt, 2015). In the aforementioned most of the New World genera comprise a clade corresponding to the tribe Psittacantheae, which also includes Tupeia antarctica (New Zealand), but in Nickrent, Anderson, and Kuijt (2019) this species is excluded from the tribe. Gaiadendron, a genus of widespread distribution in the New World, is outside of this clade, being basal in the family.
The Psittacantheae tribe is made up of four subtribes: Psittacanthinae, Ligarinae, Notantherinae (South of Chile), and Tupeinae (New Zealand), that form a polytomy. Almost all Psittacanthinae species are of tropical distribution, spanning a wide altitudinal range. Ligarinae includes two genera, Ligaria with species distributed in the temperate and sub-temperate zone of South America and Tristerix, which includes a Southern clade of species from the temperate zone of Argentina and Chile and a northern clade with tropical high Andean species (Amico, Vidal-Russell, &Nickrent, 2007).
The Kuijt monograph of Tristerix (1988) describes the flowers as bisexual and penta- or tetramerous. The isomorphic stamens are yellow-red, even black or dark violet; their anthers are medifixed, versatile and long (Kuijt, 2015). Kuijt (1988) subdivided the genus Tristerix into two subgenera: Tristerix, with only two species, each with bracteoles and tetramerous flowers, and Metastachys, which includes the remaining species and is characterized by pentamerous flowers that lack bracteoles. In contrast, Amico et al. (2007) generated a molecular phylogeny in which they differentiated two main clades within Tristerix, the first consisting of six species, distributed in northern South America (Colombia, Peru and Ecuador), and the other clade composed of species that inhabit the southernmost areas of the continent (Bolivia, Chile and Argentina). Their study indicates that speciation in this genus is related to the emergence of the Andes and the origin of cloud forests.
Tristerix longebracteatus is the highest altitude parasitic plantin the world, reaching 4 618 m above sea level in the cordillera of Vilcabamba in Peru, parasitizing trees of Polylepis pepei and Gynoxys cuzcoensis (Sylvester, Sylvester, & Kessler, 2014). According to Graves (1982), this species is pollinated by hummingbirds and presents highly synchronous flowering in Northern Peru. Acording to Kuijt (1988), Tristerix longebracteatus is closely related to T. peytonii and T. pubescens. This is consistent with the results obtained by Amico et al. (2007) in whose phylogeny the three species make up a clade. Studies in the genus Tristerix have concentrated mainly on aspects such as morphology, phylogeny, biogeography and pollination, as in the recent work of González and Pabón (2017) where some floral and inflorescence characters were studied in relation to their ornithophyllous pollination.
Within the tribe Psittacantheae, the structure of the gynoecium presents variation, including: (1) trilocular ovaries with ategmic ovules in the genus Tripodanthus (Cocucci, 1983; Venturelli, 1983), (2) unilocular ovaries with a central conical structure called a mamelon or collective ovules in the genera Peristethium (Robles, Raz, & Marquínez, 2015), Pasovia (= Phthirusa; Kuijt, & Weberling, 1972) and Strutanthus (Venturelli, 1984a, 1984b) and (3) gynoecium formed by five congenitally fused carpels with a central solid ovary around a simple mamelon in Tristerix (González & Pabón, 2017).
The tribe Psittacantheae also presents variation in the structure of the floral nectaries: (1) annular nectaries between the petals and style in species of the Psittacanthinae subtribe (for example in Peristethium, Robles et al., 2015), (2) stylar nectaries in Ligaria (Galetto, Bernardello, & Juliani, 1990; Kuijt, 1990) and (3) supraovary nectary disks in Tristerix spp. (González & Pabón, 2017).
The objectives of this research are to describe the floral anatomy of Tristerix longebracteatus,compare the results with studies of other genera in the Psitticantheae, including vascularization pattern, nectary, androecium and gynoecium structure, megasporogenesis and microsporogenesis.
Materials and methods
Floral buds and mature individual flowers of Tristerix longebracteatus were collected near laguna La Cubierta, department of Risaralda, Colombia at 3 400 m in August 2015, voucher specimens were deposited in HUQ (F. Castaño 560 Fig. 1A). The determination of the material was carried out using taxonomic keys, descriptions and comparison with herbarium specimens.
Flower buds and open flowers from 1.7 to 7.0 cm in length, were fixed in FAA (formaldehyde: acetic acid: 70 % ethanol, 10:5:85), stored in 70 % ethanol, and subsequently treated following the protocol of Robles et al. (2015). This involved standard methods of dehydratation using a clearing agent (Histoclear), paraffin infiltration, sectioning with a rotary microtome (820 Spencer, American Optical Company, NY) and attachment to microscope slides. The slides were stained with astra-blue and basic fuchsin and deposited in the Department of Biology collection at the Universidad Nacional de Colombia, Bogotá. They were analyzed and photographed using an Olympus BX-50 microscope with a Moticam Pro 282B camera. Fixed flowers were studied with a Leica M205A stereoscope using multifocus mode and photographed with a Leica MC 170 HC camera. Digital images were processed and edited with the Adobe programs Photoshop CC and InDesign.
Results
Morphology of the inflorescence and the flower
Tristerix longebracteatus is a profusely branched hemiparasite with terminal simple racemose inflorescences whose flowers develop in an acropetal order (Fig. 1A, Fig. 1B). The perianth is biseriate, actinomorphic, although with the apex slightly curved in the floral bud; the maximum length of the flower is 7 cm, including a 6.1 cm corolla tube and a 0.9 cm inferior ovary (Fig. 1B, Fig. 1G).
Each flower is subtended by a single lanceolate foliaceous bract that is approximately 4 x 0.7 cm at anthesis (Fig. 1C), with reddish-purple coloration at the apex. The bracts are fused at the base with the floral pedicels. The pedicels reach about 6 mm in length, surmounted by the ovoid inferior ovary of ca. 3-5 mm, which is, in turn, surrounded by the hypanthium. The calyculus is reduced to a ring with toothed margins (Fig. 1C), which surrounds the base of the corolla tube and is very prominent in young fruit (Fig. 1F). The corolla is scarlet red at the apex and base, and orange in the central part (Fig. 1C, Fig. 1G). The number of petals varies between 4 and 5, even in the same inflorescence, although the pentamerous condition is the most frequent. Shortly before anthesis, the petals are fused for a third of their length by interdigitation of the epidermal cells, while the distal two thirds are slightly separated forming a fenestrated floral tube. The apical lobes of the corolla are strongly reflexed at anthesis displaying yellow and pink tonalities on the adaxial surface (Fig. 1D).
The flower is bisexual, with an inferior ovary (Fig. 1G). The androecium is composed of 4-5 epipetalous stamens with reddish, cylindrical, papillate filaments and with yellow, versatile, bithecal, and tetrasporangiate anthers. They are medifixed with longitudinal dehiscence (Fig. 1E). The dentate calyculus is very prominent in the fruit (Fig. 1F).

Fig.1 Adult growth habit, macroscopic view of the inflorescence, morphology and anatomy of the flower in Tristerix longebracteatus. A. Adult specimen in its natural habitat, the paramo ecosystem (Colombia); B. Inflorescence at anthesis; showing acropetal development; C. Flower at late bud stage showing the inferior ovaries and subtending foliaceous bracts (lateral view); D. Flower at anthesis showing the reflected petals and the bases of the filaments (lateral view); E. Detail of the stamens revealing anther dehiscence (lateral view); F. Fruit at an intermediate developmental stage showing the persistent bract and the toothed calyculus (upper view); G. Median longitudinal section of the flower shortly before anthesis (the lines correspond to the approximate level of figures 2A-D, 3D-E). Scale bars: 5 cm in B, 1 cm in C, 5 mm in D, E, F, 2 mm in G. Abbreviations: An: Anther, Br: Bract, Ca: Calyculus, Fi: Filament, Fr: Fruit, Hy: Hypostase, Io: Inferior ovary, Pd: Pedicel, Pe: Petal, Sg: Stigma.
Anatomy of the bract and pedicel: Each pedicel measures approximately 6 mm in length (Fig. 1C, Fig. 1G), and is covered by a uniserial epidermis of quadrangular cells and thin cuticle, the cortex and medulla are parenchymatic with 6-9 vascular bundles arranged concentrically (Fig. 2A). At the base of the bract, a large vascular bundle that is divided in the laminar portion and form a large central vascular bundle and one to two small lateral bundles on each side. The epidermis is unistratified, formed by quadrangular coating cells with a thin cuticle and the parenchymal mesophyll consists of 6-8 homogenous layers of rounded cells (Fig. 2A).
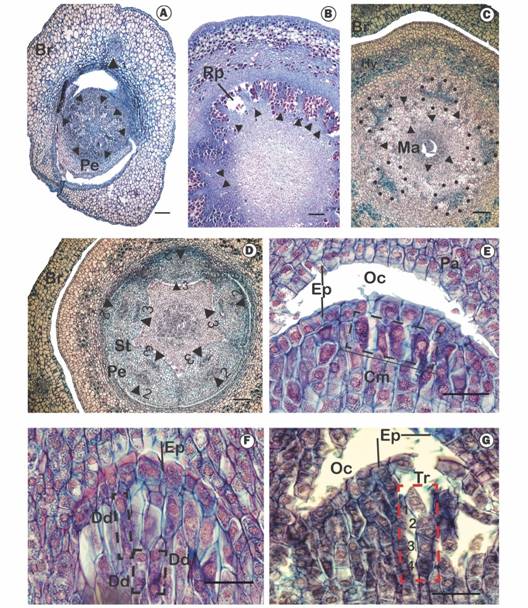
Fig. 2 Serial transverse sections of the flower and longitudinal sections of the mamelon in Tristerix longebracteatus. A. Transverse section of the basal region of the flower around mid-height of the pedicel (arrowheads = vascular bundles); B. Transverse section of the flower corresponding to the basal region of the ovary (arrowheads = vascular bundles of the gynoecium); C. Transverse section of the flower at the level of the mamelon (dotted circle = vascular bundles of the hypanthium, arrowheads = vascular bundles of the gynoecium); D. Transverse section of the flower towards the basal region of the petals and the style (arrowheads 2 = external vascular bundles of the petals, arrowheads 3 = internal vascular bundles of the petals); E. Longitudinal section of the flower showing the mamelon with arquesporial cells (dotted circle); F. Longitudinal section of the mamelon showing the dyads (dotted circles); G. Longitudinal section of the flower showing the mamelon with tetrads (numbers corresponding to cells 1, 2, 3, 4. Scale bars: 1 mm in A, B, C, D. 50 µm in E, F, G, H. Abbreviations: Br: Bract, Cm: Megagametophyte cells, Dd: Dyad, Ep: Epidermis, Es: Embryo sac, Hy: Hypanthium, Ma: Mamelon, Nu: Embryo sac nucleus, Oc:: Ovarian cavity, Pa: Amyliferous parenchyma, Pe: Petal, Rp: Parenchymal rays, St: Stamen, Tr: Tetrad.
Anatomy of the ovary, floral vascularization: The mature ovary of T. longebracteatus is about 4-5 x and 2-3 mm and, being inferior, consists of a projection of the receptacle (hypanthium), which supports the outer whorls of the flower and surrounds the ovary itself (Fig. 2A). The base of the ovary, below the pelvis or hypostase, is a uniserial epidermis of quadrangular cells with a thin cuticle, followed by one to two layers of collenchyma and then 13 to 16 layers of tanniniferous parenchyma consisting of large, ovoid cells. Interior to this are 4-6 layers of parenchyma formed by smaller cells that surround about 18-20 concentric vascular bundles separated from each other by tanniniferous parenchymal rays. The medullary parenchyma is composed of about 26 layers of small cells (Fig. 2B).
At the level of the pelvis, towards the sclerenchyma structure on which the ovary develops, the vascular bundles divide again and reorganize. Five groups of three vascular bundles surrounded by tanniniferous parenchyma externally traverse the hypanthium and innervate the five petals, along with their adnate stamens. Another ten gynoecium vascular bundles are located around the ovary, five of which continue into the style (Fig. 2C, Fig. 2D). In the area above the pelvis is the ovary itself, corresponding to a reduced structure called the mamelon or collective ovary, which means that megagametophytes (embryo sacs) develop directly from an oval, amyliferous central structure without the formation of ovules (Fig. 1G). The mamelon is located below the canal of the style (Fig. 1G).
The mamelon is initially composed of an epidermis and a layer of 4-6 archesporial subepidermal cells (Fig. 2E), which lengthen and divide meiotically giving rise to dyads (Fig. 2F) and then linear tetrads (Fig. 2G). Before anthesis, the embryo sacs invade the amyliferous tissue that surrounds the mamelon and the transmitting tissue of the style (Fig. 3A). Unfortunately, no flowers were collected with mature embryo sacs.
The style is long and straight, being almost the same length as the petals (ca. 6 cm, Fig. 1G). At its widened base is a nectary formed by an epidermis with abundant sunken stomata, below which there is parenchymal tissue with dense cytoplasm and abundant intercellular spaces surrounding the vascular bundles (Fig. 3A, Fig. 3B). The distal portion of the style is formed by a unistratified epidermis composed of rounded cells, 6-8 layers of parenchyma, among which there are five collateral vascular bundles and a central, solid, thick-walled collenchymal tissue with abundant amyloplasts (Fig. 3D). The stigma is capitate, reddish, approximately 1 mm thick; the epidermis consists of elongated cells with a thick reddish cuticle (Fig. 1G, Fig. 3C).
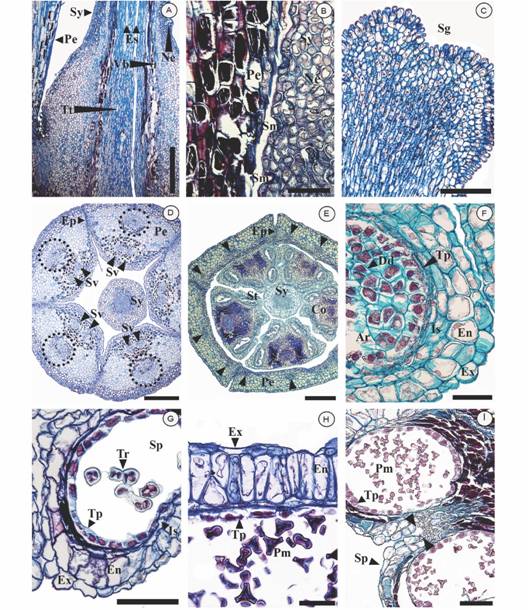
Fig. 3 Anatomy of the flower in Tristerix longebracteatus. A. Longitudinal section of the flower towards the center of the gynoecium revealing the stylar nectary; B. Detail of the nectary stomata (longitudinal section); C. Detail of the capitate stigma (transverse section); D. Transverse section of the flower around corolla mid-height (dotted circles = central vascular bundles of the petal); E. Transverse section of the flower at the level of the anthers (arrows indicate the vascular bundles of the petals); F. Detail of a pollen sac showing arquesporial tissue (transverse section); G. Detail of a pollen sac showing tetrads (transverse section); H. Detail of a pollen sac with mature pollen (transverse section); I. Transverse section of the anther revealing longitudinal dehiscence of the anther (arrows indicate rupture point of the septum). Scale bars: 1 mm in A, D, E. 200 µm in B, C. 50 µm in F, H. 100 µm in G, I. Abbreviations: Ar: Archesporium, Co: Connective, Dd: Dyad, En: Endotecium, Ep: epidermal cells joined by interdigitation, Es: Embryo sac, Ex: exothecium, Is: Intermediate strata, Ne: Nectary, Pe: Petal, Pm: Mature pollen, Sg: Stigma, Sm: Stoma, Sp: Pollen Sac, St: Stamen, Sv: Stamen vascular bundles, Sy: Style, Tp: Tapetum, Tr: Tetrad, Tt: Transmitting tissue, Vb: Vascular bundle.
Perianth anatomy: The calyculus or calyx has a single layer of epidermal cells, which are small and elongated on the adaxial side and large and rounded on the abaxial side. The mesophyll consists of 11-12 undifferentiated layers of parenchyma, without any vascularization. On the abaxial side the cells of the parenchyma are rounded and on the adaxial face they are cubic and smaller.
The corolla is formed by four or five petals joined by interdigitation of the epidermal cells in the floral bud. Towards the basal half the petals are thicker, since they include the adnate filaments of the corresponding stamens (Fig. 3D). The distal half of the petal is laminar. Anatomically, the petals are formed by an adaxial epidermis formed by quadrangular cells and few poorly differentiated stomata and the adaxial epidermis is formed by round cells (Fig. 3E).
Toward the basal half, the mesophyll includes about 22 layers of tanniniferous parenchyma, two vascular bundles of the stamen and the vascular bundle of the petal (Fig. 3D, Fig. 3E). The distal portion of the petals has 9-11 layers of parenchyma cells of similar size and shape. The vascular bundle of the epipetalous portion divides into three toward the distal laminar portion. Some sclereids are also isolated or in small groups in the second or third layer below the adaxial epidermis (Fig. 3D).
Androecium: This whorl consists of 4-5 epipetalous, isomorphic stamens, with irregularly shaped filaments vascularized by two bundles. The filaments are formed by a uniseriate epidermis of more or less quadrangular, thick-walled cells, followed by four to seven layers of parenchymal tissue with tannins in the innermost layers that surround the two collateral vascular bundles. The filaments are not completely cylindrical, presenting lobes orientated toward the base (Fig. 1E, Fig. 1G).
The anthers are about 5-8 x 1.2 mm, being medifixed, bithecal and tetrasporangiate (Fig. 1E, Fig. 3E, Fig. 3I). The exothecium consists of a single layer of quadrangular cells, elongated and flat, followed by a fibrous endothecium with O-shaped thickenings and 2-3 intermediate layers of elongated cells. No amyloplasts were observed in any of the layers of the wall at any stage of anther development; however, the connective presents tanniniferous parenchyma (Fig. 3E).
The secretory-type tapetum is unistratified, consisting of uni or binuclear rectangular cells with dense cytoplasm, that remain in the original position and maintain their identity during microsporogenesis until their eventual degeneration in situ at the end of pollen development (Fig. 3F, Fig. 3I). The cells of the archesporium are polygonal and thin-walled, with prominent nuclei and dense cytoplasm (Fig. 3F). Simultaneous microsporogenesis occurs, generating dyads (Fig. 3F) and then tetrahedral tetrads (Fig. 3G). Subsequently, the four trilobate pollen grains are formed inside the callose wall of the tetrad. (Fig. 3H). The dehiscence of the anthers is longitudinal and occurs by rupture between the central septum and the walls of the pollen sacs of each theca.
Discussion
Taxonomic identification and morphological variability: The material studied corresponds to the species Tristerix longebracteatus, according to the criteria established by Kuijt (1988), Dueñas-Gómez and Franco-Roselli (2001), Amico et al. (2007). The axes of the inflorescences and pedicels are terete with foliaceous bracts, mainly pentamerous flowers, petals up to 6 cm long and fruits ripening black with a prominent calyculus and conical apex. The maximum size of the petals of our material (6.1 cm) is close to the maximum length indicated by Kuijt (1988) in his identification key.
Nevertheless, the presence in the same inflorescence of pentamerous and tetramerous flowers makes this character less useful for distinguishing among Tristerix species, and even less so for separating the subgenera Tristerix (tetramerous) and Metastachys (pentamerous) proposed by Kuijt (1988) and Dueñas-Gómez and Franco-Roselli (2001).
On the other hand, it should be said that the characteristic floral coloration in alternating bands of red-yellow-red, reported by Amico et al. (2007) for T. longebracteatus and closely related species, did not appear clearly in our material, in which the general reddish coloration acquires orange, but not yellow tones in the floral tube. The proposed coloration of the stamens, yellow in the filament and reddish in the anther (Kuijt, 1988), which are clearly observed in Contero and Dehesa (2007) for T. longebractatus specimens from Cayambe (Ecuador), contrasts with the reddish coloration of the filaments and yellow of the anthers in our material. Also, the pendant position of the inflorescences in González and Pabón (2017) contrasts with the erect position observed in this study. Kuijt (1988) described variation in leaf size and floral length across this species’ broad geographic distribution, but it should be noted that the characters reported above are also variable.
Vascularization of the flower: González and Pabón (2017) indicate that in the Tristerix species that they studied, “five vascular bundles enter the base of the pedicel [….] above which they radially split into an outer ring of five traces that irrigate the common petal-stamen bases and an inner ring that serves the gynoecium.” In our specimens we observed a more complex pattern of vascularization, with 6-7 pedicel bundles that subdivide into 18-20 vascular bundles separated from each other by tanniniferous parenchyma (Fig. 2B) that González and Pabón (2017) identify as a collenchyma cap. The traces that irrigate the petal-stamens bases in our case are not solitary, but are formed by three minor vascular bundles (Fig. 2C); and there are ten gynoecial bundles of which five continue in style (Fig. 3D). This pattern of floral vascularization is much more complex than that observed in other species of the subtribe Psittacantheae, such as Passovia (= Phthrirusa in Kuijt and Weberling, 1972), Struthanthus (Venturelli 1981, Venturelli 1984a, Venturelli 1984b), Peristhethium (Robles et al, 2015), Passovia, Aetanthus and Oryctanthus (Suaza, Pabón, & González, 2016), and can be explained by the large size of the flowers and the adaptation to pollination by hummingbirds that requires increased production of nectar.
Nectary: The most diverse subtribe is the neotropical Psittacanthinae, characterized by flowers with annular nectaries between the petals and the base of the floral style in the genera Peristhethium (Robles et al., 2015), Passovia,Aetanthus,Oryctanthus (Suaza et al., 2016), Strutanthus (Venturelli, 1981, Venturelli 1984a), and Cladocolea (Kuijt, 1975); although Cid-Villamil (2006) reports nectaries at the base of the petals in Cladocolea loniceroides.
On the other hand, nectaries are also reported as being due to modification and thickening of the base of the style proper in Nuytsia, the basal genus of the family (Narayana, 1958) and genera of the tribe Elytrantheae such as Lepeostegeres (Schaeppi & Steindl, 1942; Raj, 1970). In the present study we found a nectary formed by thickening of the base of the style in T. longebracteatus (named supraovary nectary disk by González and Pabón (2017) in Tristerix spp.). In the genus Ligaria, the other genus that together with Tristerix makes up the subtribe Ligarinae, this kind of nectary is also present (Galetto et al., 1990; Kuijt, 1990).
Although the type of nectaries in the subtribes Psittacanthinae (annular nectary) and Ligarinae (stylar nectary) appear to be of taxonomic importance, unfortunately, the absence of information in the subtribe Nothantherinae does not allow a clear interpretation of the evolution of this character in the tribe.
Gynoecium: González and Pabón (2017) affirm “The ovary [of Tristerix spp.] is solid and served by a poorly differentiated vascular ring around a simple mamelon” and that “the gynoecium is formed by five congenitally fused carpels, which are evident by the five vascular bundles and the edges alternating the petals and stamens”. According to our observations, the ovary of T. longebracteatus has a more complex vascularization compared to the rest of the tribe Psittacantheae or even the rest of the family. We also observed a mamelon clearly separated from the surrounding parenchymatic tissues in the ovary chamber, both in cross section (Fig. 2C), and in longitudinal section (Fig. 2E, Fig. 2G), across all developmental stages, which implies a single ovarian cavity, not a solid ovary.
The number of vascular bundles or their position alone is insufficient to determine the number of carpels that make up the gynoecium of this or other Loranthaceae. The evolution of the gynoecium in Loranthaceae and the identity of the ovary-ovule complex and mamelon (sensuJohri, Ambegaokar, & Srivastava, 1992), or the “collective ovary” sensuCocucci (1982) is currently unclear and new approaches in developmental biology, such as those used in the pioneering work of Brown, Nickrent, and Gasser (2010), will be required to shed light on the matter.
Ethical statement: authors declare that they all agree with this publication and made significant contributions; that there is no conflict of interest of any kind; and that we followed all pertinent ethical and legal procedures and requirements. All financial sources are fully and clearly stated in the acknowledgements section. A signed document has been filed in the journal archives.












 uBio
uBio 

