Rio Grande do Sul (RS) State is located in the Southernmost territory of Brazil. It coverseveral regions and environments diversified climatic and geomorphologically. The Pampa Biome is located in the Southern of the state and is made up of a set of distinct ecosystems containing vast biodiversity. Although it remains mostly unexplored, this area consists of an extremely rich gene pool (Boldrini et al., 2010).
These ecosystems have been affected by several human activities, such as inadequate use of the soil, inadequate disposal of industrial and domestic effluents, as well as the increasing use of agrochemicals (Esteves, 2011). This has contributed to biodiversity loss, jeopardizing the balance of aquatic and terrestrial biomes and, consequently, impacting human welfare (Cardinale et al., 2012). According to Merritt and Cummins (1996) invertebrates are one of the most common groups in freshwater environments, and, among them, insects stand out because of their association with other freshwater communities.
The Odonata order comprise insects commonly known as dragonflies, damselflies, lava-bunda, zig-zag, and, specifically in Rio Grande do Sul, Cicadas. This order is composed of paleopterous amphibiotic insects, under hemimetabolous development, and is divided into three suborders: Zygoptera, Anisoptera and Anisozygoptera (Rehn, 2003). Globally, this order contains 6 500 species (Trueman & Rowe, 2009), and about 800 of them from 15 different families were reported in Brazil alone (Neiss & Hamada, 2014).
These insects are relevant bioindicators for assessing and monitoring environmental conditions of specific locations. Thus, the greater the biodiversity, the better the health of that ecosystem (Ferreira-Peruquetti & De Marco, 2002; Foote & Rice, 2005; Silva, Marco, & Resende, 2010; Carvalho, Silva-Pinto, Oliveira-Júnior, & Juen, 2013; Monteiro Júnior, Couceiro, Hamada, & Juen, 2013). In this context, some researchers have recognized the need to list the diversity of these animals and develop models and criteria that can be applied in environmental change assessments (Reaka-Kudla, Wilson, & Wilson, 1997).
Species inventories are indispensable in management and conservation processes, in which the entomofauna diversity is an important tool for environmental sanity. Furthermore, the knowledge of insect diversity may provide valuable information on the integrity of the studied environments (Lutinski & Garcia, 2005).
According to the Red List of Threatened Species of the International Union for Conservation of Nature (IUCN), one in ten Odonata species is in critical danger of being extinct (Mace et al., 2008), and 35 % of them species lack incidence data (Clausnitzer et al., 2009). In Rio Grande do Sul (RS), there are very few studies that address the Odonata inventory and are limited almost exclusively to the reports made by Costa (1971) and Teixeira (1971) in Santa Maria and Porto Alegre cities, respectively. The last data concerning Odonato fauna in RS are present in the work of Kittel and Engels (2014), who studied the diversity of Zygotera in São Francisco de Paula. They included four new species in the list of registered species of the State. Furthermore, Renner, Périco, Sahlén, Dos Santos, and Consatt (2015) conducted an inventory study in 12 cities of the Rio Taquari valley region, where 50 Odonata species were found.
This study aims to widen the knowledge on the Odonata species and measure the species incidence in the aforementioned region. In addition, fauna indexes will be associated with the sampling areas.
Materials and methods
Field of study: Known as the “Southern fields”, the Pampa Biome covers approximately 62 % of Rio Grande do Sul (177 767 km2) along with part of Argentina and the entire territory of Uruguay, which is rich in water resources. Most of the water bodies are ecologically classified as shallow lakes, which are easily influenced by natural and human activity because of their shallow depth (Trindade, Furlanetto, & Silva, 2009; Zambrano, Contreras, Hiriart, & Arista, 2009). The sampling areas are located in the Lagoa Mirim and Lagoa dos Patos, both lakes with well defined seasons with cold and rainy winters and hot and dry summers. According to the Köppen-Geiger climate classification, local climate is considered Cfa (humid subtropical). In this region, average annual temperature varies between 16 ºC and 18 ºC and average annual rainfall is approximately 1 500 mm.
In the cities where the study was conducted, two collection sites were selected with three sampling points each. In Capão do Leão, sampling was conducted at Campus of the Federal University of Pelotas - UFPel (31º80’16” S - 52º41’94” W) and at Irmão Teodoro Luis Botanical Garden (31º81’27” S - 52º43’58” W). In Pelotas, the samples were collected in Vila Princesa (31º62’79” S - 52º32’66” W) and Balneário dos Prazeres (31º72’07” S - 52º19’14” W). In Rio Grande city, the samples collected in Arroio Bolaxa (32º16’02” S - 52º18’80” W) and Taim Ecological Station (32º 44’33” S - 52º 34’28” W).
Collected data: Odonata samples were obtained between November 2014 to October 2015; no samples were taken during the winter season because of the lack of activity among adult insects due to low temperatures. Each location was visited nine times, totalizing 54 samplings. In order to capture the insects, the active collection method was conducted with entomological nets. The samplings were performed on sunny days, between 9 am to 4 pm, during a period of three hours at each collection site.
Species identification: The collected specimens were kept alive in entomological envelopes for at least four hours and were later treated with Acetone, as methodology proposed by Lencioni (2005). Samples were identified using the taxonomic keys proposed by Lencioni (2005; 2006) and Heckman (2006; 2008). Damaged especimens or those that had no similarity with the descriptions offered in literature were classified at generic level. The specimens were deposited at the Ceslau Biezanko Entomological Museum, UFPEL.
Data analysis: The data analyses were conducted considering specific abundance (n), generating the species-accumulation curve. Relative frequency (Rf) was measured by dividing the total number of collected species by the total of collected insects, multiplied by 100 (Silveira Neto, Nakana, Barbin, & Vila Nova, 1976; Bianconi, Mikich, & Pedro, 2004). For this measurement, a 5 % confidence interval was used, and species were classified as very frequent (C ≥ 50 %), frequent (25 % ≤ C < 50 %) and infrequent (C < 25 %).
The abundance assessments and comparisons were conducted using the Chao-1 estimator. Based on abundance, this evaluation relates the number of species represented by only one individual (singletons), and the number of species with only two sample individuals (doubletons) (Colwell, 2012). Furthermore, it is calculated by the equation: Chao 1 = S + (a²/2b); in which S is the number of encountered species in the samples, a is the number of species represented by one specimen, and b is the number of species represented by two specimens (Chao, 1984; 1987; Ferraz, Gadelha, & Aguiar-Coelho, 2009).
The diversity measurement was performed using the Shannon-Wiener index: H’= -Σ (fi) log (fi); in which fi is the proportion of individuals belonging to the nth specie and ln is the neperian logarithm (Pielou, 1975).
For the species-diversity estimates and their confidence intervals (95 %), the analytical estimator first-order Jackknife index was used (Jack 1 = S + L (n-1/n), in which S is the sum of the species found in the samples, L is the number of species present in a single sample, and n is the total number of samples (Palmer, 1991). For calculations and graph and table elaboration, Excel and EstimateS 8.0 programs were used.
Results
Considering all our sample collections, 2 680 specimens were found, comprised by 45 species distributed into 22 genera and 6 families (Table 1). The species Progomphus complicatus, Lestes minutus, Homeoura ambigua and Tauriphila xiphea were recorded for the first time, in ascending order of frequency, in RS (Table 1).
Table 1: List of the species collected in the Brazilian Pampa Biome (Southern region of Rio Grande do Sul) divided by suborder and families
| Family / Species | Collection Sites1 | Total | % | Fr2 | |||||||
| Suborder Zygoptera | PBP | PVP | CHB | CCL | RAB | RET | |||||
| Calopterygidae | |||||||||||
| Hetaerina rosea Selys, 1853 | 5 | 9 | 0 | 3 | 2 | 0 | 19 | 0.671 | IF | ||
| Coenagrionidae | |||||||||||
| Acanthagrion gracile Rambur, 1842 | 2 | 1 | 0 | 1 | 2 | 0 | 6 | 0.223 | IF | ||
| Acanthagrion lancea Selys, 1876 | 0 | 0 | 0 | 3 | 1 | 0 | 4 | 0.149 | IF | ||
| Homeoura chelifera Selys, 1876 | 0 | 26 | 48 | 23 | 10 | 26 | 133 | 4.964 | VF | ||
| Homeoura ambígua Ris, 1904* | 7 | 2 | 2 | 4 | 0 | 0 | 15 | 0.559 | IF | ||
| Ischnura capreolus Hagen, 1861 | 36 | 26 | 41 | 47 | 29 | 40 | 219 | 8.137 | VF | ||
| Ischnura fluviatilis Selys, 1876 | 41 | 29 | 29 | 56 | 30 | 39 | 224 | 8.361 | VF | ||
| Oxyagrion terminale Selys, 1876 | 10 | 0 | 0 | 9 | 0 | 5 | 24 | 0.895 | IF | ||
| Oxyagrion simile Costa, 1978 | 0 | 0 | 0 | 5 | 0 | 0 | 5 | 0.186 | IF | ||
| Telebasis willinki Fraser, 1948 | 44 | 19 | 24 | 36 | 15 | 45 | 183 | 6.830 | VF | ||
| Lestidae | |||||||||||
| Lestes undulatus Say, 1839 | 8 | 10 | 0 | 9 | 8 | 0 | 35 | 1.306 | IF | ||
| Lestes tricolor Erichson, 1848 | 7 | 11 | 0 | 0 | 9 | 0 | 27 | 1.007 | IF | ||
| Lestes minutus Selys, 1862* | 0 | 0 | 0 | 0 | 0 | 8 | 8 | 0.298 | IF | ||
| Suborder Anisoptera | |||||||||||
| Aeshnidae | |||||||||||
| Coryphaeschna amazonica DeMarmels, 1989 | 4 | 0 | 5 | 1 | 0 | 1 | 11 | 0.410 | IF | ||
| Remartinia luteipennis Burmeister, 1839 | 0 | 6 | 3 | 4 | 5 | 0 | 18 | 0.671 | IF | ||
| Rhionaeschna bonariensis Rambur, 1842 | 11 | 18 | 10 | 22 | 9 | 19 | 89 | 3.322 | F | ||
| Rhionaeschna cornigera Brauer, 1865 | 0 | 0 | 6 | 11 | 0 | 0 | 17 | 0.634 | IF | ||
| Gomphidae | |||||||||||
| Progomphus complicatus Selys, 1854* | 0 | 6 | 0 | 0 | 0 | 0 | 6 | 0.223 | IF | ||
| Phyllocycla sp. Calvert, 1948 | 0 | 0 | 0 | 7 | 0 | 0 | 7 | 0.261 | IF | ||
| Libellulidae | |||||||||||
| Diastatops Intensa Montgomery, 1940 | 0 | 3 | 0 | 4 | 2 | 0 | 9 | 0.335 | IF | ||
| Erythemis attala Selys in Sagra, 1857 | 0 | 23 | 18 | 22 | 15 | 41 | 119 | 4.441 | VF | ||
| Erythemis plebeja Burmeister, 1839 | 0 | 7 | 4 | 16 | 8 | 10 | 45 | 1.679 | F | ||
| Erythemis peruviana Rambur 1842 | 0 | 35 | 20 | 30 | 13 | 7 | 105 | 3.919 | F | ||
| Erythemis vesiculosa (Fabricius, 1775) | 0 | 0 | 1 | 5 | 0 | 4 | 10 | 0.373 | IF | ||
| Erythrodiplax fusca Rambur, 1842 | 21 | 16 | 9 | 31 | 10 | 30 | 117 | 4.367 | VF | ||
| Erythrodiplax nigricans Rambur, 1842 | 0 | 18 | 0 | 24 | 0 | 2 | 44 | 1.642 | F | ||
| Erythrodiplax paraguayensisFörster, 1904 | 5 | 0 | 3 | 7 | 4 | 2 | 21 | 0.783 | IF | ||
| Erythrodiplax atroterminata Ris, 1911 | 30 | 20 | 27 | 28 | 12 | 33 | 150 | 5.559 | VF | ||
| Erythrodiplax chromoptera Borror, 1942 | 0 | 0 | 0 | 7 | 4 | 0 | 11 | 0.410 | IF | ||
| Erythrodiplax media Borror, 1942 | 14 | 38 | 32 | 20 | 29 | 23 | 156 | 5.823 | VF | ||
| Erythrodiplax hyalina Förster, 1907 | 10 | 6 | 8 | 19 | 1 | 5 | 49 | 1.829 | F | ||
| Erythrodiplax sp. Brauer, 1868 | 1 | 2 | 4 | 3 | 1 | 1 | 12 | 0.485 | IF | ||
| Miathyria marcella Selys in Sagra, 1857 | 28 | 58 | 40 | 31 | 27 | 75 | 259 | 9.667 | VF | ||
| Micrathyria tibialis Kirby, 1897 | 0 | 0 | 2 | 1 | 0 | 1 | 4 | 0.149 | IF | ||
| Micrathyria hypodidyma Calvert, 1906 | 33 | 51 | 39 | 30 | 30 | 22 | 205 | 7.652 | VF | ||
| Micrathyria pseudeximia Westfall, 1992 | 8 | 23 | 10 | 9 | 17 | 14 | 81 | 3.023 | F | ||
| Micrathyria stawiarskii Santos, 1953 | 0 | 0 | 4 | 1 | 1 | 2 | 8 | 0.289 | IF | ||
| Micrathyria catenata Calvert, 1909 | 0 | 2 | 0 | 5 | 1 | 0 | 8 | 0.289 | IF | ||
| Orthemis nodiplaga Karsch, 1891 | 1 | 8 | 0 | 16 | 9 | 12 | 46 | 1.717 | F | ||
| Orthemis ambinigra Calvert, 1909 | 3 | 5 | 0 | 2 | 3 | 6 | 19 | 0.709 | IF | ||
| Pantala flavescens Fabricius, 1798 | 0 | 13 | 0 | 8 | 17 | 9 | 47 | 1.754 | F | ||
| Perithemis mooma Kirby, 1889 | 2 | 9 | 1 | 5 | 7 | 3 | 27 | 1.007 | IF | ||
| Tauriphila risi Martin, 1896 | 12 | 4 | 0 | 8 | 11 | 6 | 41 | 1.530 | F | ||
| Tauriphila xiphea Ris, 1913* | 0 | 6 | 0 | 3 | 2 | 12 | 23 | 0.858 | IF | ||
| Tramea cophysa Hagen, 1867 | 2 | 5 | 0 | 3 | 1 | 3 | 14 | 0.522 | IF | ||
| Total | 345 | 515 | 390 | 579 | 345 | 506 | 2 680 | 100 | |||
| Total by County | 860 | 969 | 851 | ||||||||
1Pelotas, Balneário dos Prazeres (PBP) and Vila Princesa (PVP). Capão do Leão, Irmão Teodoro Luis Botanical Garden (CHB) and Capão do Leão Campus of the Federal University of Pelotas- UFPel (CCL). Rio Grande Arroio Bolaxa (RAB) and TAIM Ecological Station (RET).
2Frequency was indicated as follows: infrequent (IF), frequent (F) and very frequent (VF).
*Newly-reported species in RS.
The most speciose genera were Erythrodiplax and Micrathyria, with eight and five species, respectively. These genera, represented by two species (Rhionaeschna bonariensis and Telebasis willinki), along with Ischnura, were found at least once in all the visited sites. Gomphidae specimens were less frequent and exhibited low density in this study, since P. complicatus specimens were found only in Pelotas, specifically on sandbanks of Arroio Pelotas, located in Vila Princesa. Female Phyllocycla were collected close to the pools of UFPel Campus in Capão do Leão (Table 1).
Ten species, from the Libellulidae (6) and Coenagrionidae (4), were considered very frequent (VF), reaching a total of 65 % of the captured individuals. The Gomphidae and Calopterygidae were represented by 32 total specimens, corresponding to 1.19 % of the collected insects and considered infrequent (IF) (Table 1).
The Libellulidae family was the most abundant since it comprising 57.78 % of the samples (Table 2), in which Miathyria marcella was the most common specie, amounting to 9.66 % of total dragonfly samples (Table 1). The next most common families were Coenagrionidae and Aeshnidae, with a total of 20 % and 8.89 % of the specimens, respectively. Lastly, the least abundant families were Lestidade with 6.67 %, Gomphidae with 4.44 %, and Calopterygidae with 2.22 % of the sampled species. Concerning the sample density, the Libellulidae family comprised 60.82 % of total collected specimens, followed by Coenagrionidae of the Zigoptera suborder at 30.35 % of total collected Odonata (Table 2).
Table 2: Number of Odonata species collected in the the Brazilian Pampa Biome (Southern region of Rio Grande do Sul) from each family, their percentage (%) in relation to the total of collected species and to the total of collected specimens
| Suborder | Family | Number of species | Percentage (%) | Density (%) |
| Zigoptera | Coenagrionidae | 9 | 20.00 | 30.35 |
| Lestidae | 3 | 6.67 | 2.61 | |
| Calopterygidae | 1 | 2.22 | 0.70 | |
| Anisoptera | Libellulidae | 26 | 57. 78 | 60.82 |
| Aeshnidae | 4 | 8.89 | 5.03 | |
| Gomphidae | 2 | 4.44 | 0.49 | |
| Total | 45 | 100 | 100 | |
Based on the different faunistc indexes (faunistic) (Table 3), the highest number of specimens were collected in Capão do Leão, followed by Pelotas, and Rio Grande, with 969, 860, and 851 specimens respectively. In addition, the highest specific wealth was also found at Capão do Leão, where 42 species were reported, while in Rio Grande and Pelotas 40 and 36 species were collected, respectively.
Table 3: Fauna data analyses of the species collected in all three cities in the Brazilian Pampa Biome
| Faunistic indexes | Capão do Leão | Pelotas | Rio Grande |
| Individuals (n) | 969 | 860 | 851 |
| Species diversity | 42 | 36 | 40 |
| Shannon-Wiener (H’) | 1.62 | 1.49 | 1.54 |
| Chao -1 | 46.3 | 48.6 | 48.0 |
In the current study, the highest Shannon-Wiener diversity indexes were found in Capão do Leão (1.62) and Rio Grande (1.54), while Pelotas (1.49) exhibited the lowest result. On the other hand, regarding the species-diversity estimator, Chao 1 method, Pelotas reached the highest value, followed by Rio Grande (48.0) and Capão do Leão (46.3) (Table 3).
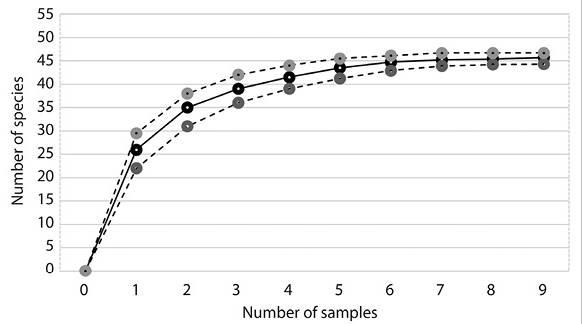
The species accumulation curve (Figure 1) was unable to reach the asymptote, indicating that the diversity in the region is relatively larger than the 45 species found in the current study. The first-order Jackknife index is 57.3 species, with standard deviation of ± 8.2.
Discussion
The heterogeneity of the sampled environments enabled us to collect 45 Odonata species. Regarding their frequency, 26 of 45 species occurred infrequently (IF), totaling 57.78 %, ten species were found very frequently (VF), comprising 1 765 collected specimens, and nine species were considered frequent (F) (Table 1).
Regarding genus diversity, De Figueiredo et al. (2013) found 34 Odonata genera in the Rio Ibicuí river basin in Central RS. Their results were higher than the results obtained in this study, which was conducted in the Southern region of the state. However, inferior diversity wealth in this region may be due to insufficient studies. In addition, the huge fields of the Pampa Biome, permeated with small forest areas and several types of water bodies, are ideal environments for Odonata (Maluf, 2000).
The UFPel Campus, located in Capão do Leão city, exhibited 42 species, the greatest diversity among all collection sites. Furthermore, Capão do Leão was also where the highest number of insects were captured (579) (Table 1). These data indicate that, despite flow of people in the university campus, the sampling area, that is far away, provide adequate conditions for the Odonata diversity found in the present study. In Rio Grande, the greatest diversity was observed in Arroio Bolaxa, with 34 collected species, while at the Taim Ecologic Station 31 species were observed. The difference in total species found in Arroio Bolaxa and in Taim, may have occurred because of the larger green area exhibited in the first (Juen & De Marco, 2012). Contraringly to diversity, 161 more specimens were collected at Taim Ecologic Station than in Arroio Bolaxa.
According to the Shannon-Wiener index (H’), the lowest diversity was found in Pelotas (Table 3). This may be attributed the fact that the collection sites in this city constantly suffer human disturbances, which, according to Clausnitzer & Jödicke (2004), occur when industrial and/or domestic effluents are inadequately disposed, i.e. in locations close to urban areas. This describes Vila Princesa, an area located near the BR116 road. Human disturbances are also found in the Balneário dos Prazeres collection site, which exhibits large flow of people, such as residents and bathers. The impacts caused on Odonata diversity by human actions in these areas are still unclear, especially because these types of studies are rare in Brazil (Ferreira-Peruquetti & De Marco, 2002; Clausnitzer et al., 2009). In Pelotas, although lower species diversity was found, its Chao 1 index was higher than Capão do Leão and Rio Grande, estimating 48.6 species. This difference may be because the latter index considers rare species, i.e. it analyzes the number of species represented by only one specimen and the number of species represented by only two sample specimens.
Dragonflies are outstanding insects that can be affected both by environmental (Juen & De Marco, 2011) and spacial factors (Juen, Cabette, & De Marco, 2007; Clausnitzer et al., 2009). This indicates that these insects are potential bioindicators for monitoring the quality of aquatic environments (Ferreira-Peruquetti & De Marco, 2002). Furthermore, dragonflies may be considered valuable tools of freshwater environments, since the diversity of immature Odonata in the environment elucidates the diversity of the entire macroinvertebrate community (Foote & Rice, 2005).
Twenty-six of the 45 collected species were from the Libellulidae family, which can be explained by the fact that most of these specimens are generalist and can be found in many different environments. Like the rest of the Anisoptera suborder, this family exhibits the largest insects of all Odonata, making them more capable of flying and distributing themselves geographically (Kalkman et al., 2008; Juen & De Marco, 2012).
M. marcella was the most common of this study with 259 captured specimens and is considered VF at 9.66 % frequency. This may be explained by the fact that it is an abundant and generalist specie. In fact, M. marcella is strongly associated with floating vegetation, such as Eichhornia and Pistia, commonly known as water hyacinth and water lettuce, respectively. Dragonflies deposit their eggs on these floating plants (Paulson, 2017) for reproduction, which is in agreement with the sampling environments studied here.
Concerning the species reported for the first time in Rio Grande do Sul state, P. complicatus was collected only six times, all of which occurred in the city of Pelotas. L. minutus specimens were collected only in Rio Grande and Taim, and all 15 specimens of the Homeoura ambígua were collected in Rio Grande. T. xiphea specimens were present in all the sampling locations, having been most frequent in Rio Grande, totaling 23 individuals. The aforementioned species expanded its geographic distribution since it was previously only found in Paraguay, Argentina and Rio de Janeiro (Heckman, 2006). Regarding frequency, all the specimens reported for the first time were classified as IF.
According to Juen et al., (2007), the distributions of some species are limited by physical and ecological factors. Having that in mind, Southern landscape of RS favors the species that are more agile and more capable of dispersion, since the vegetation is composed mainly by open fields. This may explain the low diversity of Zygoptera found during this study, considering that the small forest areas present in this region are mostly spread out and distant from one another. Due to their type of thermoregulation, these small specimens would have trouble taking long flights in open fields (De Marco & Resende, 2002). In addition, according to Carvalho, Oliveira-Junior, Faria, and Juen (2013), the Zygoptera would have trouble surviving in open spaces exposed to light and heat.
The sampling efficiency reached in this study was around 78 % (Jack 1) of the expected diversity and was far from reaching the asymptote. This shows that the number of existing species in the studied region is larger than the 45 species found in the present study and that more sampling efforts are required in all regions of the state to find the expected diversity.
Although this study presents the Odonata diversity found in a small sample portion of the Pampa Biome in the south of Rio Grande do Sul, the data are still limited. The species herein reported for the first time in the state show that more studies must be conducted so more precise data about the species of the region can be observed. Furthermore, aquatic sampling should be include the collection of immature stages.
In conclusion, Progomphus complicatus, Lestes minutus, Homeoura ambigua, and Tauriphila xiphea were reported for the first time in the State of Rio Grande do Sul. In regard to the Odonata diversity in the Brazilian pampa biome, 45 species were encompassed in 22 genera and the most commonly found genera were Erythrodiplax and Micrathyria. Concerning the occurrence frequency, 26 species were considered infrequent, 9 were frequent, and 10 were very frequent. The city of Capão do Leão exhibited the largest species diversity (wealth), the largest number of collected specimens, and greatest diversity, in comparison to Pelotas and Rio Grande. The Miathyria marcella species represented 9.6 % of all collected libellulidae and was the most abundant species.
Ethical statement: authors declare that they all agree with this publication and made significant contributions; that there is no conflict of interest of any kind; and that we followed all pertinent ethical and legal procedures and requirements. A signed document has been filed in the journal archives












 uBio
uBio