Ophiuroids display diverse feeding strategies, often combined, and ranging from predation and scavenging to deposit- and suspension feeding (Warner, 1982; Harris et al., 2009). Based on the size of the ingested particles, Warner (1982) proposed the division between macrophagous and microphagous species, with the former acquiring large particles through predation and scavenging while the latter include suspension feeding and the acquisition of small particles from the substratum. Some species exhibit high degrees of flexibility in their feeding mechanisms and their prey spectra (Fontaine, 1965; Warner, 1982), and it has been proposed that for both, shallow water and deep-sea species, differences in diets between species also reflect differences in lifestyles. Hereby they may act as non-specialized opportunists with respect to diet (Pearson & Gage, 1984).
Information available on the diet of brittle stars has been established through field observations estimating the feeding preferences by the observed feeding behavior (Warner, 1982), or by stomach content analyses (Harris et al., 2009). Depending on their behavior it is possible to identify active predators or suspensionfeeding species (Davoult & Gounin, 1995), while is difficult to directly observe those species that feed on deposited material from the sediment surface or beneath it (Gielazyn et al., 1999). Prey items that are found in stomach content studies allow for the identification of an ophiuroid being microphagous, but also for the general source of the respective food particles: planktonic prey items suggest a filter feeding mode on water-borne items, whereas considerable amounts of sediment may indicate a surface- or subsurface deposit feeding mode (Ferrari & Dearborn, 1989; Dahm, 1999). A third approximation of brittle star feeding preferences is relatively poorly developed and is based on the analysis of the stereom microstructure of the teeth (i.e. how calcite is arranged or disposed in the teeth). Medeiros-Bergen (1996) analyzed the stereom microstructure of teeth from several ophiuroid species using light and scanning electron microscopy. She defined two tooth types: a uniform teeth, in which the tooth stereom is completely fenestrated, and compound teeth, where the base of the tooth is fenestrated, while the distal edge is composed of imperforated calcite. With a few exceptions, macrophagous feeders possess uniform teeth, while microphagous species reveal compound teeth (Medeiros-Bergen, 1996).
The brittle star Ophioplocus januarii (Lütken, 1856) (Ophiolepididae) is distributed along the Western Atlantic coast from Golfo Nuevo, Argentina, to northern Brazil, with a doubtful record from Barbados (Thomas, 1975; Bernasconi & D’Agostino, 1977). It is found from the tidal zones down to 74 meters depth, both on hard substrates and on soft sediments rich in detritus (Bartsch, 1982). To date very little is known about the feeding biology of O. januarii (Brogger et al., 2013). Bartsch (1982) analyzed stomach contents of some individuals finding only sediment grains. Medeiros-Bergen (1996) observed the microstructure of the teeth of O. januarii under a light microscope and described it as completely fenestrated, similar to those teeth she observed in Ophioplocus esmarki Lyman, 1874 with the scanning electron microscope.
Here we report on the diet of O. januarii and discuss its feeding biology based on stomach content analysis, feeding mechanisms observed in the field, and tooth stereom microstructure analysis using scanning electron microscopy. For comparison, we also describe and discuss the microstructure of the teeth of other brittle stars species and their relations to the respective feeding habits.
Materials and methods
Ophioplocus januarii were collected monthly from Playa Villarino (42º 24’ S - 64º 17’ W), Golfo San José (northern Patagonia), from April 2005 to April 2007 comprising a total of 25 consecutively taken samples. During each sampling event, the specimens were captured from the same location by SCUBA diving at depths between 2 and 7 m (depending on the tides) and collected in plastic bags. At the laboratory, they were fixed in Bouin’s solution for 24 h and then preserved in 70 % ethanol. When individual remaining particles were found in the collection bags they were also preserved in 70% ethanol for later observation. While sampling, brittle stars were observed and photographed in situ in order to identify and determine feeding activities. All sampling and observations were conducted during daylight.
Stomach contents
The discs were cut off along with the stomach lining, and the contents picked out. Stomach contents of 10 brittle stars from each month (total N = 250) were examined under the light microscope, and all content items were separated. When at least one item from a stomach was identified, the respective brittle star was considered ‘with stomach content’, including unidentifiable and/ or digested remains. Only those ophiuroids without any stomach content were considered ‘empty’. Some portions of the unidentifiable or digested remains were examined with the light and scanning electronic microscope (SEM).
Tooth microstructure analysis
The jaws of O. januarii were dissected and placed for a few minutes in a diluted solution of commercial house bleach in order to remove the epidermal layer. Longer maceration allowed for the dissociation of individual teeth from the dental plates. In order to observe the internal calcite microstructure, one tooth was fractured. For comparative purposes, the jaws and teeth of five other brittle star species were examined as well. These included Ophiomyxa vivipara, Amphipholis squamata, Ophiactis asperula, Ophiacantha vivipara and Ophiocten amitinum. All jaws and teeth were prepared for SEM observations. At least 7 adult individuals of each species were dissected during the tooth microstructure analysis.
Results
The remaining particles from the individual collection bags resulted to be small shell fragments mixed with algal fragments originating from the sediment. These particles were similar to objects that are sometimes retained or hooked by the ophiuroid’s arm spines and other body parts. In no case there were signs of these particles being egested stomach contents from prior to the animals’ fixation.
Typical passive suspension-feeding activities were observed in the field. While feeding, the individuals raised two or three arms into the passing currents. Small particles were trapped by the tube feet and collected into a bolus that was passed down along the arm to the mouth.
Stomach contents
Of the 250 individuals examined, 31.2 % had stomachs with contents. Of these, 64.1 % presented only one food item, and far lower percentages referred to stomachs containing two to five different items (Table 1). The most frequent item found corresponded to macroalgal fragments, mainly from filamentous algae. Food particle sizes were up to 0.5 mm with the exception of two larger structures: a macroalgal fragment of 6.0 mm, and a cuticular structure of 7.5 mm. The average number of food items was 1.64 per individual.
Table 1 Total number of stomachs with content and percentage of stomachs with distinguishable amounts of food item
| Number | Percentage | ||
| Stomachs analyzed | 250 | - | |
| With contents | 78 | 31.2 | |
| With one food item | 50 | 64.1 | |
| With two food items | 13 | 16.6 | |
| With three food items | 9 | 11.5 | |
| With four food items | 5 | 6.4 | |
| With five food items | 1 | 1.3 | |
Macroalgal fragments were present in 60 % of the stomachs with contents, and in 64 % of the studied months. Other frequent items found were unidentifiable material (31%) and small terrestrial plant debris (28 %). Less frequent items were cuticular animal structures (13%), unidentifiable laminar structures (8 %), spicules (4 %), three foraminiferans, three ostracods, one amphipod, other crustaceans, one juvenile bivalve and one hydrozoarian colony. No sediment particles were found inside the stomachs.
Tooth microstructure analysis
All teeth in O. januarii revealed a fenestrated stereom microstructure (Fig. 1). However, it was possible to clearly identify two distinct regions: the basal parts of the teeth which are in contact with the dental plate, are more loosely perforated than the distal parts. There the calcite is much more compacted, presenting a clear different arrangement (Fig. 1 A, B, C, D). This superficial distinction is visible also internally in the calcite microstructure as can be seen in a fractured tooth (Fig. 1 F, G). The basal surface presents little serrated edges, but at the distal portion these edges are less prominent because of the tightly packed calcite (Fig. 1 E).
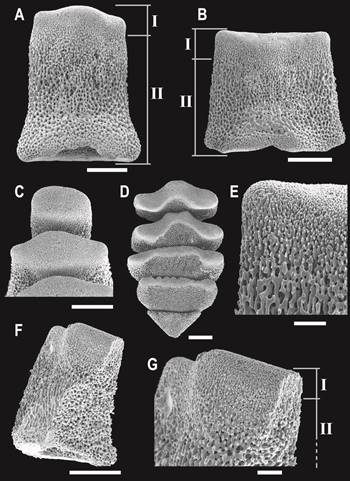
Fig. 1 Scanning electron microscopy images of teeth in Ophioplocus januarii. A) Dorsalmost tooth. B) Mid-positioned tooth. C) Proximal view of dorsal teeth. D) Proximal view of ventral teeth. E) Detail of B, showing the different calcite compaction. F) Fractured tooth. G) Detail of F, showing the internal differentiation in microstructure. I and II indicate regions of more compaction (I) and more porosity (II) of the calcite, respectively. Scale bars: A, B, C, D = 200 μm; E, G = 50 μm.
Under light microscope, the teeth of Ophiomyxa vivipara presented translucent crystalline edges with small spines protruding from the distal edges; they are semicircle in shape and are perforated apart from the distally protruding spines (Fig. 2 A). The teeth of Ophiacantha vivipara and Ophiocten amitinum presented spine like shapes with uniform fenestrated surfaces and sharply serrated edges (Fig. 2 D, E). Amphipholis squamata carried teeth with a fenestrated base, but distally the calcite was imperforate (Fig. 2 B). The tooth stereom microstructure of Ophiactis asperula is similar to that described here for O. januarii, and consisted of two regions with differentiated calcite compactions (Fig. 2 C). The tooth types of the six species analyzed presently are summarized in the Table 2.

Fig. 2 Scanning electron microscopy images of tooth stereom microstructure in different ophiuroid species (mid-positioned teeth). A) Ophiomyxa vivipara. B) Amphipholis squamata. C) Ophiactis asperula. D) Ophiacantha vivipara. E) Ophiocten amitinum. Scale bars: A, C, D = 200 μm; B, E = 100 μm.
Table 2 Tooth stereom microstructure for six analyzed ophiuroid species: uniform (completely fenestrated), compound (fenestrated base and imperforate calcite tip) and intermediate (fenestrated with two distinct regions) tooth types
| Species | Family | Source | Tooth type |
|---|---|---|---|
| Ophioplocus januarii (Lütken, 1856) | Ophiolepididae | 42º 24’ S; 64º 17’ W (2 - 7 m) | intermediate |
| Amphipholis squamata (Delle Chiaje, 1828) | Amphiuridae | 44º 53 ’S; 65 º40’ W (0 m) | compound |
| Ophiacantha vivipara Ljungman, 1870 | Ophiacanthidae | 38º 51’ S; 55º 39’ W (115 m) | uniform |
| Ophiactis asperula (Philippi, 1858) | Ophiactidae | 38º 51’ S; 55º 35’ W (145 m) | intermediate |
| Ophiocten amitinum Lyman, 1878 | Ophiuridae | 38º 51’ S; 55º 35’ W (145 m) | uniform |
| Ophiomyxa vivipara Studer, 1876 | Ophiomyxidae | 43º 36’ S; 60º 05’ W (96 m) | uniform |
Discussion
In the present study, Ophioplocus januarii from Playa Villarino fed ingesting small-suspended particles through suspension feeding from the water-sediment interface. Contrarily to previously made assumptions (Warner, 1982; Medeiros-Bergen, 1996), O. januarii is a microphagous species. It fed opportunistically, mainly ingesting fragments of macroalgae, but also small plant and animal structures, and other suspended material. When analyzing stomach contents from a 60 meters depth O. januarii population collected in a nearby location on the continental shelf (42° S - 62° W), Bartsch (1982) observed stomachs lacking food but filled partly with sediment grains. In the present study, no sediments were found, suggesting differences in feeding preferences to be dependent of habitat diversity. This was also observed for different populations of Ophiura ophiura (Blegvad, 1914 in Warner, 1982) and of Ophionotus victoriae (Dearborn, 1977). The main distinction between feeding types in brittle stars used to be drawn between carnivory and microphagy (Warner, 1982). Those species that capture large particles -typically of animal origin- and, thus, feed as microphagous feeders, are generally described as carnivorous, while microphagous species feed on a mixture of minute animal and vegetal material. The diet presently observed for O. januarii indicates that this species is an unselective omnivorous species. Others also feeding mainly on vegetal origin material are mostly associated with sediments. Ophionereis reticulata, for example, has been indicated as exclusively herbivorous (May, 1925 in Warner, 1982) and more recently as an omnivore with algal feeding preference (Yokoyama & Amaral, 2008). Algal and calcareous fragments are the most frequent items in the diets of Ophiocoma wenditii, O. echinata and O. pumila (Sides & Woodley, 1985).
In stomach contents analyses conducted with different species, the percentages of empty stomachs found were highly variable between the species. For example, Harris et al. (2009) observed 66 % of empty stomachs in Ophiura sarsii, Yokoyama and Amaral (2008) 23 % in Ophionereis reticulata, Hendler (1982) found variations from 100 to 5% with a dependence of the months under study, and Hendler and Miller (1984) observed differences in percentage for Asteroporpa annulata according to whether the individuals were captured during day or nighttime. In the present study, the percentage of empty stomachs in O. januarii was close to 70 % and included only samples that were collected during the daylight. The fact that brittle stars may egest their stomach contents in response to collecting procedure or handling (Pearson & Gage, 1984; Hendler, pers. comm.), could explain the high proportions of empty stomach found in some deep-sea investigations (Warner, 1982). This, however, does not seem to be the case in the presently investigated O. januarii because no egested material was found when analyzing the collection bags.
The tooth stereom microstructure in O. januarii is distinctly different from that in teeth of macrophagous as well as microphagous species (Fig. 1; Table 2). The presently found intermediate fenestrated arrangement of the stereom was also here observed for Ophiactis asperula, and confirmed as such when analyzing the teeth with the scanning electron microscope (SEM). The fact that SEM reveals structures which might remain disguised under the light microscope could explain that Medeiros-Bergen (1996) did not recognize differences in the tooth microstructure between Ophioplocus esmarki and O. januarii. Therefore, it is possible that other species previously described as carrying uniform teeth (macrophagous) could, in fact, possess intermediate tooth types. In the present study we identified the intermediate tooth type, and the previously described uniform and compound types. However, it would be interesting to analyze additional species in order to recognize other possible variations in the tooth stereom microstructure previously overlooked.
The present results observed for the diet of O. januarii represent, to our knowledge, the first trustworthy report of microphagy in the family Ophiolepididae. Dietary studies on Ophiolepis elegans suggested this species to be a macrophagous species (Warner, 1982), while Medeiros-Bergen (1996), based on tooth microstructure, estimated three Ophioplocus species and Ophiolepis impressa to be microphagous as well. Two other species from the same family (Ophioplocus incipiens and Ophiomusium lymani) are reported to conduct suspension-feeding activities (Warner, 1982), and little information about their diets is available. Pearson and Gage (1984) suggested O. lymani to have an omnivorous diet. Because there are no records of passive suspension feeding in macrophagous species, it is likely that both O. incipiens and O. lymani being representatives of the Ophiolepididae, are microphagous as well.
Much progress on the knowledge of the diet of ophiuroids has been made since Warner’s summary in 1982. Yet, much is still obscure. For example, the role that brittle stars play in trophic transfers of nutrients within benthic communities and between benthic and pelagic communities still remains highly unknown (Gielazyn et al., 1999). The utility of analyzing teeth microstructure may surpass not only knowledge on feeding habits and diet in ophiuroids, but may also provide a helpful tool for phylogenetic issues, most likely in combination with tooth macrostructure analyzes as proposed by Stöhr (2005), and with studies on jaw morphology already used by Murakami (1963), Smith et al. (1995) and Stöhr and Muths (2010).












 uBio
uBio 

