Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.63 n.2 San José Apr./Jun. 2015
Traditional utilization and biochemical composition of six mollusc shells in Nigeria
Utilización tradicional y composición bioquímica de seis conchas de moluscos en Nigeria
Utilización tradicional y composición bioquímica de seis conchas de moluscos en Nigeria
Abstract
The shells of molluscs protect them from physical damage, predators and dehydration. We studied various local uses of shells and their biochemical properties in Abeokuta, Nigeria. A standard structured questionnaire about use was applied to 100 snail and herb sellers and shells from 120 adult individuals of Archachatina marginata, Achatina achatina, Achatina fulica, Littorina littorea, Meretrix lusoria and Merceneria mercenaria were evaluated for their mineral components (Ca2+, Fe2+, Mg2+, Na+, Zn+, P+, K+) and proximate composition (crude protein, ash, fibre, crude fat and carbohydrate) using standard methods. Properties against fungi and bacteria isolates were also tested. These shells are used for bleaching, brushing, abrasion and others. The weight of the shells varied from 0.5g (L. littorea) to 25.00g (A. marginata) and thickness from 0.46mm in M .lusoria to 5.35mm in M. mercenaria. We found no inhibitory effect against fungi and bacterial isolates. The molluscs are high in carbohydrates (83.54-92.76g/100g) and low in protein (0.16-0.38g/100g). The fat content ranged between 0.42g/100g and 0.82g/100g, and ash between 2.14g/100g and 9.45g/100g. Ca2+ was the most abundant (10.25-96.35mg/g) while K+ was the least abundant (0.3-0.7mg/g) (p<0.05). Active ingredients of these shells can be used in the feed and construction industries.
Key words: shell, mollusc, proximate analysis, antimicrobial, antifungal, uses.
Resumen
Las conchas protegen a los moluscos del daño físico, los depredadores y la deshidratación. En este trabajo se estudiaron los diversos usos locales de las conchas y sus propiedades bioquímicas en Abeokuta, Nigeria. Se utilizó un cuestionario estructurado estándar sobre los usos de las conchas, aplicado a 100 vendedores de caracoles y hierbas, conchas de 120 individuos adultos de Archachatina marginata, Achatina achatina, Achatina fulica, Littorina littorea, Meretrix lusoria y Merceneria mercenaria fueron evaluados para la extracción de los compuestos minerales (Ca2+, Fe2+, Mg2+, Na+, Zn+, P+, K+) y la composición proximal (proteína cruda, materia seca, fibra, contenido graso y carbohidratos) usando métodos estándar. También se analizaron las propiedades de aislamientos contra hongos y bacterias. Las conchas se utilizan para el blanqueamiento, cepillado, abrasión y otros. El peso de las conchas varió de 0.5g (L. littorea) a 25.00g (A. marginata) y el espesor de 0.46mm en M. lusoria a 5.35mm en M. mercenaria. No se encontraron efectos inhibidores de los aislamientos contra hongos y bacterias. Los moluscos resultaron ricos en carbohidratos (83.54-92.76g/100g) y bajos en proteínas (0.16-0.38g/100g). El contenido graso osciló entre 0.42g/100g y 0.82g/100g y la materia seca entre 2.14g/100g y 9.45g/100g. Ca2+ fue el más abundante (10.25-96.35mg/g), mientras que K+ el menos abundante (0.3-0.7mg/g) (p<0.05). Los ingredientes activos de estas conchas se pueden utilizar en la industria alimentaria y de la construcción.
Palabras clave: concha, molusco, análisis proximal, antimicrobiano, antifúngico, usos.
The shell is made up of calcium carbonate and it encloses supports and protects the soft part of animals in the Phylum Mollusca. Its members include: snails, periwinkles and clam. Man consumes animal products especially milk, egg, meat and fish. These products are treasured in the diet because of their flavor, texture and nutrients. However, shells are disposed as wastes where they constitute environmental nuisance due to unpleasant odor and unsightly appearance (Adewuyi, & Ola, 2005).
Research studies confirm that land snail parts (flesh and haemolymph) are useful in the treatment of diseases like anemia, hypertension, labor pain and constipation (Akinnusi, 2004; Adikwu, 2012). Similarly, periwinkle shells serve as a coarse aggregate of concrete in areas where there are neither stones nor granite for paving of water logged areas (Falade, 1995). Clam shells were similarly used as a source of calcium supplements for lactating cows (Finkelstein, Wohlt, & Emmanuele, 1993). Agbelusi and Ejidike (1992) reported that snail shells were used not only as calcium source to farm animals but as an important ingredient in traditional medicine. Some rural dwellers store traditional medicines or concoctions in the shells of snails while some use them for washing utensils and teeth brushing.
Most of the literature focuses on the flesh and haemolymph of molluscs, while shell and especially the traditional use of mollusc shells have received very little attention. The aims of this study were to document and evaluate the various uses of mollusc shells and to analyze their chemical compositions.
Materials and Methods
Experimental site: The study was conducted at the Department of Biological Sciences, Federal University of Agriculture, Nigeria (7°10’00” N - 3°02’00” E). The ambient temperature was 27.0±0.05°C and relative humidity was 65.70% (12D:12L).
Experimental samples: Six members of Phylum Mollusca commonly found in Southwest Nigeria were used for this study. They were three snail species (Archachatina marginata, Achatina achatina, and Achatina fulica), periwinkle (Littorina littorea) which were purchased from Itoku market, Abeokuta, Nigeria and the two species of clam (Meretrix lusoria and Mercenaria mercenaria) were obtained from Lagos lagoon, Lagos state (6°40’94” N - 4°09’15” E). Twenty individuals of each species were de-shelled and the shell weighed by sensitive electronic balance (Mettler-PM 11K), and the shell thickness was measured using a micrometer screw gauge.
Questionnaire administration: A standard structured questionnaire (containing 25 questions) was administered to 100 persons (herb and snail sellers) at three major markets in Abeokuta, Nigeria. The questionnaire was administered so as to evaluate the various uses of mollusc shells in Abeokuta, Nigeria.
Chemical analysis: The proximate analysis of the mollusc shells was carried out by A.O.A.C (1990) methods. The content of ash, fibre, protein, fat and carbohydrate of the shells were all determined. Also, the mineral analysis of the shells (Fe2+, Zn2+, Ca2+, Mg2+, Na+, K+ and P) was also carried out using spectrophotometry method. All experiments were repeated three times.
Microbial analysis: The shells of the molluscs were sun dried and ground into fine particles using mortar and pestle. Anti-microbial analysis of the shell was determined by methods described by Sodipe, Osinowo, Onagbesan and Bankole (2012) using disc diffusion method. The anti-microbial activities were tested against bacterial (Proteus mirabitus, Pseudodomonas aeruginosa and Staphylococcus aureus) and fungal (Aspergillus flavus, Aspergillus fumigatus and Aspergillus niger) isolates. The plates were incubated and the zones of inhibition were measured (Sodipe et al., 2012).
Data collected were analyzed by one-way analysis of variance (ANOVA) and separation of significant values (p<0.05) was done by Duncan Multiple Range Test (DMRT).
Results
Shell uses: The various uses of mollusc shells as mentioned by respondents are shown in Table 1. Respondents stated that shells of various molluscs were used for brushing, washing of utensils and form part of ingredients for curing measles and other infections.
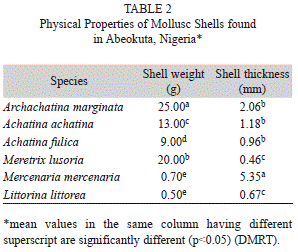
Physical and chemical composition of the shells: Table 2 presents the physical properties of the shells. The shell of A. marginata had the highest weight while the shell of L. littorea had the lowest value.
Table 3 shows the proximate composition of the mollusc shells. The shells contain no moisture content except M. mercenaria and L. littorea (2.22% and 3.37% respectively). Similarly, the shells had low fat and protein content. However, the shells had high carbohydrate content which ranged between 83.54g/100g (L. littorea) and 92.76/100g (A. achatina).
The mineral composition of the mollusc shells is shown in Table 4. Results showed that M. lussoria and M. mercenaria (clam species) had the highest Ca2+ and Fe2+ concentrations while A. achatina had the least.
Microbial analysis: The antimicrobial effects of the mollusc shells against bacteria and fungi isolates are shown in Table 5. The shells of the molluscs did not inhibit the isolates except the control.
Discussion
Responses from herb and snail sellers in Abeokuta shows that mollusc shells were used for washing utensils, as abrasive material and used in treating infections like cough, measles and tuberculosis by mixing with other plants materials. This parallels the reports of Agbelusi and Ejidike (1992) that in Ondo State, Nigeria snail shells form part of the material used in traditional medicine for treating gonorrhea, measles and cough. The shells were burnt into ashes, ground into fine powder and mixed with other ingredients to form herbal concoction. Similarly, other respondents mentioned that mollusc shells are used for aesthetic purpose. Amusan and Omidiji (1998) and Amubode and Fafunwa (2014) reported that snail shells are used to decorate offices, cars and beautify homes. This observed usage of the shells was due to the indelible unique markings or stripes on the mollusc shells especially A. achatina and clam.
The physical characteristics of mollusc shells revealed that a clam shell (M. mercenaria) has the highest thickness followed by A. marginata. This probably explains their being used for cutting objects and storing substances as earlier reported by Agbelusi and Ejidike (1992).
Falade (1995) reported that shells of molluscs (periwinkle) are used in coarse aggregate of buildings. The low moisture content of mollusc shells observed in this study might probably explain this function of mollusc shells. Recently too, Adewuyi (2014) discovered that mollusc shells are good replacement for Ordinary Portland Cement in concrete production. This is made possible because of their strong, hard and bristle properties.
The mollusc shells have high carbohydrate content. This is not surprising as the organic constituents of mollusc shell according to Hodasi (1982) are polysaccharides and glycoproteins. Jatto, Asia and Medjor (2010) similarly reported that shells of four snail species have high carbohydrate content. Thus, shell of molluscs can be added to animal feeds as energy source.
The low protein and fat content of the mollusc shell observed in this study agrees with the findings of Yusuff and Oseni (2004) on pond snail Lymnae stagnalis. The main function of mollusc shell is protection and carbohydrate unlike protein and fat has strong binding properties which do not decompose easily in the presence of bad elements of weather.
The mineral analysis of the mollusc shells showed that there were significant differences in the concentration of ions across the mollusc species. It was observed that clams (M. lusoria and M. mercenaria) had significantly higher Fe2+ and Ca2+ than other mollusc species. This might likely be due to their location, that is, marine environment, whereas other molluscs are terrestrial. Bertine and Goldberg (1972) observed that shell composition changes as a result of changing water chemistry. Subtidal anoxic marine sediments contain high amount of soluble sulphate (H2S) and acid labile sulphate (FeS) (Howarth, & Giblin, 1983). Furthermore, Ca2+was significantly more than other ions present in the shells. This might explain the use of mollusc shells as abrasive, brushing agents and for stopping bleeding as earlier reported by Agbelusi and Ejidike (1992). Blood clotting involves conversion of fibrinogen to fibrin which forms a mesh network and covers the wound (Muszbek, Bagoly, Bereczky, & Katona, 2008). This process depends on calcium availability for its proper mechanism. Similarly, Finkelstein et al. (1993) added clam shells to lactating cow feed as a calcium supplement because snail shell is known to have high concentration of calcium. Egonmwan (2008) reported that a positive relationship existed between snail (Limicolaria flammea) shell and calcium concentration. The presence of these vital minerals needed for strong teeth in snail shells warranted their inclusion in formula for washing teeth in rural areas of Ondo state. Here, the shell is oven-dried and ground into fine powder and kept in safe container.
The mollusc shells had low Na+ concentration. This is not unexpected as other parts of the molluscs-haemolymph and flesh are reported to have low concentration of Na+ (Ademolu, Idowu, Mafiana, & Osinowo, 2007; Idowu, Somide, & Ademolu, 2008). Thus, mollusc shells can be included in livestock and man’s diet without fear of complications arising from high Na+ intake.
Results showed that mollusc shells do not have antimicrobial property as they did not inhibit the growth of pathogenic isolates. Sodipe et al. (2012) and Kayode and Ademolu (2014) likewise observed that snails’ haemolymph did not resist the growth of fungal and bacterial isolates. This is in tandem with earlier observation of Akinloye & Olorode (2000) that the physiological state of the snails is reflected in the haemolymph as it has open circulatory system where the fluid bathes the entire organs and tissues. However, in a recent study by Abiona, Akinduti, Osinowo and Onagbesan (2013), the epiphragm of albino and normal skinned A. marginata inhibited four bacterial isolates better than the conventional antibiotics (streptomycin). In conclusion, this study has revealed the reason behind the use of mollusc shells in traditional medicine and industries. The high concentration of carbohydrate, Fe2+ Ca2+ and Mg2+ in the shells is the major reason for their usage in washing teeth, stopping bleeding and inclusion in animal feeds formulation.
References
Abiona, J. A., Akinduti, A., Osinowo, O. A., & Onagbesan, O. M. (2013). Comparative evaluation of inhibitory activity of epiphragm from albino and normal skinned giant African land snail (Archachatina marginata) against selected bacteria isolates. Ethiopia Journal of Environmental Studies and Management, 6(2), 177-181. [ Links ]
Ademolu, K. O., Idowu, A. B., Mafiana, C. F., & Osinowo, O. A. (2007). Performance, proximate and minerals analysis of African giant land snail (Archachatina marginata) fed different nitrogen sources. Tropical Veterinarian, 25(4), 124-131. [ Links ]
Adewuyi, A. P., & Ola, B. F. (2005). Application of waterworks sludge as partial replacement for cement in concrete production. Science Focus Journal, 10(1), 123-130. [ Links ]
Adikwu, M. U. (2012). Snail production for sustainable development and good health. In S. I. Ola, G. A. Dedeke, & A. O. Fafiolu (Eds.), Procceedings of the 1st International Conference on Giant African Land snails (NeTGALS) (pp. 3-7). Federal University of Agriculture, Abeokuta, Nigeria. [ Links ]
Adewuyi, A. P. (2014). Relevance of residues of snail farming to civil engineering construction works. In J. A. Abiona, & I. O. Osunsina (Eds.), Proceedings of the 3rd International Conference on Giant African Land Snails (NeTGALS) (pp. 18-25). Federal College of Education, Abeokuta, Nigeria. [ Links ]
Agbelusi, E. A., & Ejidike, B. N. (1992). Utilization of the Giant African Land Snail, Archachatina marginata in the humid area of Nigeria. Journal of Tropical Agriculture, 69, 88-92. [ Links ]
Akinloye, O. A., & Olorode, O. (2000). Effect of different feeding condition on performance, haemolymph biochemical and mineral value of Giant African Snail (Archachatina marginata). Journal of Agriculture and Environment, 1, 143-147. [ Links ]
Akinnusi, F. A. O. (2004). Introduction to Snails and Snail farming. Abeokuta, Nigeria: Gbemi Sodipo Press Ltd. [ Links ]
Amubode, A. A., & Fafunwa, F. (2014). Snail farming and hospitality industries. In In J. A. Abiona, & I. O. Osunsina (Eds.), Proceedings of the 3rd International Conference on Giant African Land Snails (NeTGALS)(pp. 31-36). Federal College of Education, Abeokuta, Nigeria. [ Links ]
Amusan, J. A., & Omidiji, M. O. (1998). Edible Land Snail: A Technical Guide to Snail Farming in the Tropics. lbadan, Nigeria: Verity Printer Limited. [ Links ]
Association of Official Analytical Chemist (A.O.A.C.). (1990). 13th edition. Washington, D.C.: Lexington Books Inc. [ Links ]
Bertine, K. K., & Goldgerg, E. D. (1972). Trace elements in clams, mussels and shrimps. Limnology and Oceanography, 17(6), 877-884. [ Links ]
Egonmwan, R. I. (2008). Effects of dietary calcium on growth and oviposition of the African land snail Limicolaria flammea (Pulmonata:Achatinidae). Revista de Biología Tropical, 56(1), 333-343. [ Links ]
Falade, F. (1995). An Investigation of Periwinkle Shell as Coarse Aggregate in Concrete. Building and Environment, 30(4), 573-577. [ Links ]
Finkelstein, A. D., Wohlt, J. E., & Emmanuele, S. M. (1993). Composition and nutritive value of ground sea clam shells as calcium supplements for lactating Holsten cows. Journal of Dairy Science, 76(2), 582-589. [ Links ]
Hodasi, J. K. N. (1982). The effect of different light regimes on the behavior and biology of Achatina achatina. Journal of Molluscan Studies, 48, 1-7. [ Links ]
Howarth, R. W., & Giblin, A. (1983). Sulphate reduction in salts marshes at Sapelo Island, Georgia. Limnology and Oceanography, 28(1), 70-82. [ Links ]
Idowu, A. B., Somide, O. M., & Ademolu, K. O. (2008). Comparative Analysis of the chemical composition of the haemolymph, flesh and the microflora content of the guts of some African land snails in Abeokuta, Nigeria. Tropical Veterinarian, 26(1&2), 20-29. [ Links ]
Jatto, O. E., Asia, I. O., & Medjor, W. E. (2010). Proximate and mineral composition of different species of snail shell. The Pacific Journal of Science and Technology, 11(1), 416-419. [ Links ]
Kayode, O. V., & Ademolu, K. O. (2014). Antimicrobial and Biochemical properties of Haemolymph from two snail ecotypes. In J. A. Abiona, & I. O. Osunsina (Eds.), Proceedings of the 3rd International conference on Giant African Land Snails (NeTGALS) (pp 45-48). Federal College of Education, Abeokuta, Nigeria. [ Links ]
Muszbek, L., Bagoly, Z., Bereczky, Z., & Katona, E. (2008). The involvement of blood coagulation factor XIII in fibrinolysis and thrombosis. Cardiovascular and Hematological Agents in Medical Chemistry, 6(3), 190-205. [ Links ]
Sodipe, O. G., Osinowo, O. A., Onagbesan, O. M., & Bankole, O. M. (2012). Evaluation of the haemolymph of the Giant African land Snails Achatina achatina and Archachatina marginata for bacteria sterility and inhibitory properties. Proceedings of the 1st International Conference on Giant African Land snails (NeTGALS), Federal University of Agriculture, Abeokuta. [ Links ]
Yusuff, A. A., & Oseni, O. A. (2004). Nutritional value and functional properties of pond snail (Lymnae stagnalis). I. C. Eromosele, & T. O. S. Popoola (Eds.), Proceedings of the 1st International Conference on Science and National Development (pp. 90-93). College of Natural Sciences, Federal University of Agriculture, Abeokuta, Nigeria. [ Links ]
Ademolu, K. O., Idowu, A. B., Mafiana, C. F., & Osinowo, O. A. (2007). Performance, proximate and minerals analysis of African giant land snail (Archachatina marginata) fed different nitrogen sources. Tropical Veterinarian, 25(4), 124-131. [ Links ]
Adewuyi, A. P., & Ola, B. F. (2005). Application of waterworks sludge as partial replacement for cement in concrete production. Science Focus Journal, 10(1), 123-130. [ Links ]
Adikwu, M. U. (2012). Snail production for sustainable development and good health. In S. I. Ola, G. A. Dedeke, & A. O. Fafiolu (Eds.), Procceedings of the 1st International Conference on Giant African Land snails (NeTGALS) (pp. 3-7). Federal University of Agriculture, Abeokuta, Nigeria. [ Links ]
Adewuyi, A. P. (2014). Relevance of residues of snail farming to civil engineering construction works. In J. A. Abiona, & I. O. Osunsina (Eds.), Proceedings of the 3rd International Conference on Giant African Land Snails (NeTGALS) (pp. 18-25). Federal College of Education, Abeokuta, Nigeria. [ Links ]
Agbelusi, E. A., & Ejidike, B. N. (1992). Utilization of the Giant African Land Snail, Archachatina marginata in the humid area of Nigeria. Journal of Tropical Agriculture, 69, 88-92. [ Links ]
Akinloye, O. A., & Olorode, O. (2000). Effect of different feeding condition on performance, haemolymph biochemical and mineral value of Giant African Snail (Archachatina marginata). Journal of Agriculture and Environment, 1, 143-147. [ Links ]
Akinnusi, F. A. O. (2004). Introduction to Snails and Snail farming. Abeokuta, Nigeria: Gbemi Sodipo Press Ltd. [ Links ]
Amubode, A. A., & Fafunwa, F. (2014). Snail farming and hospitality industries. In In J. A. Abiona, & I. O. Osunsina (Eds.), Proceedings of the 3rd International Conference on Giant African Land Snails (NeTGALS)(pp. 31-36). Federal College of Education, Abeokuta, Nigeria. [ Links ]
Amusan, J. A., & Omidiji, M. O. (1998). Edible Land Snail: A Technical Guide to Snail Farming in the Tropics. lbadan, Nigeria: Verity Printer Limited. [ Links ]
Association of Official Analytical Chemist (A.O.A.C.). (1990). 13th edition. Washington, D.C.: Lexington Books Inc. [ Links ]
Bertine, K. K., & Goldgerg, E. D. (1972). Trace elements in clams, mussels and shrimps. Limnology and Oceanography, 17(6), 877-884. [ Links ]
Egonmwan, R. I. (2008). Effects of dietary calcium on growth and oviposition of the African land snail Limicolaria flammea (Pulmonata:Achatinidae). Revista de Biología Tropical, 56(1), 333-343. [ Links ]
Falade, F. (1995). An Investigation of Periwinkle Shell as Coarse Aggregate in Concrete. Building and Environment, 30(4), 573-577. [ Links ]
Finkelstein, A. D., Wohlt, J. E., & Emmanuele, S. M. (1993). Composition and nutritive value of ground sea clam shells as calcium supplements for lactating Holsten cows. Journal of Dairy Science, 76(2), 582-589. [ Links ]
Hodasi, J. K. N. (1982). The effect of different light regimes on the behavior and biology of Achatina achatina. Journal of Molluscan Studies, 48, 1-7. [ Links ]
Howarth, R. W., & Giblin, A. (1983). Sulphate reduction in salts marshes at Sapelo Island, Georgia. Limnology and Oceanography, 28(1), 70-82. [ Links ]
Idowu, A. B., Somide, O. M., & Ademolu, K. O. (2008). Comparative Analysis of the chemical composition of the haemolymph, flesh and the microflora content of the guts of some African land snails in Abeokuta, Nigeria. Tropical Veterinarian, 26(1&2), 20-29. [ Links ]
Jatto, O. E., Asia, I. O., & Medjor, W. E. (2010). Proximate and mineral composition of different species of snail shell. The Pacific Journal of Science and Technology, 11(1), 416-419. [ Links ]
Kayode, O. V., & Ademolu, K. O. (2014). Antimicrobial and Biochemical properties of Haemolymph from two snail ecotypes. In J. A. Abiona, & I. O. Osunsina (Eds.), Proceedings of the 3rd International conference on Giant African Land Snails (NeTGALS) (pp 45-48). Federal College of Education, Abeokuta, Nigeria. [ Links ]
Muszbek, L., Bagoly, Z., Bereczky, Z., & Katona, E. (2008). The involvement of blood coagulation factor XIII in fibrinolysis and thrombosis. Cardiovascular and Hematological Agents in Medical Chemistry, 6(3), 190-205. [ Links ]
Sodipe, O. G., Osinowo, O. A., Onagbesan, O. M., & Bankole, O. M. (2012). Evaluation of the haemolymph of the Giant African land Snails Achatina achatina and Archachatina marginata for bacteria sterility and inhibitory properties. Proceedings of the 1st International Conference on Giant African Land snails (NeTGALS), Federal University of Agriculture, Abeokuta. [ Links ]
Yusuff, A. A., & Oseni, O. A. (2004). Nutritional value and functional properties of pond snail (Lymnae stagnalis). I. C. Eromosele, & T. O. S. Popoola (Eds.), Proceedings of the 1st International Conference on Science and National Development (pp. 90-93). College of Natural Sciences, Federal University of Agriculture, Abeokuta, Nigeria. [ Links ]
1. Department of Biological Sciences, Federal University of Agriculture, P.M.B.2240, Abeokuta, Nigeria; kennyademolu@yahoo.com, akintolamy@yahoo.com, funmiolayinka@yahoo.co.uk, bola.blessing@yahoo.com
Received 03-II-2014. Corrected 12-XII-2014. Accepted 23-I-2015.