Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.62 suppl.3 San José Sep. 2014
Possible recovery of Acropora palmata (Scleractinia:Acroporidae) within the Veracruz Reef System, Gulf of Mexico: a survey of 24 reefs to assess the benthic communities
Posible recuperación de Acropora palmata (Scleractinia:Acroporidae) en el sistema arrecifal de Vera Cruz, Golfo de México: evaluación de comunidades bentónicas en 24 arrecifes
Posible recuperación de Acropora palmata (Scleractinia:Acroporidae) en el sistema arrecifal de Vera Cruz, Golfo de México: evaluación de comunidades bentónicas en 24 arrecifes
Abstract
Recent evidence shows that Acropora palmata within the Veracruz Reef System, located in the southwestern Gulf of Mexico, may be recovering after the die off from the flooding of the Jamapa River and a dramatic cold water event in the 1970s. Since this decline, few surveys have documented the status of A. palmata. The 28 named reefs in the system are divided into 13 northern and 15 southern groups by the River. Between 2007 and 2013, we surveyed 24 reefs to assess the benthic communities. Seven of the 11 reefs surveyed in the northern group and all in the southern group had A. palmata. Colonies were typically found on the windward side of the reefs in shallow waters along the reef edges or crest. We also recorded colony diameter and condition along belt transects at two reefs in the north (Anegada de Adentro and Verde) and two in the south (Periferico and Sargazo), between 2011 and 2013. In addition, eight permanent transects were surveyed at Rizo (south). A total of 1 804 colonies were assessed; densities ranged from 0.02 to 0.28 colonies/m² (mean (±SD), colony diameter of 58 ± 73cm, and 89 ± 18% live tissue per colony). Total prevalence of predation by damselfish was 5%, by snails 2%, and <1% by fireworms, disease prevalence was <3%. Size frequency distributions indicated that all of the sites had a moderate to high spawning potential, 15-68% of the colonies at each site were mature, measuring over 1 600cm². The presence of these healthy and potentially reproductive colonies is important for species recovery, particularly because much of the greater Caribbean still shows little to no signs of recovery. Conservation and management efforts of these reefs are vital.
Key words: Acropora palmata, population recovery, Veracruz, Mexico, colony size class frequency.
Resumen
Evidencia reciente indica que las poblaciones de Acropora palmata del sistema arrecifal de Vera Cruz, ubicado al suroeste del Golfo de México, podría estarse recuperando después de las inundaciones del río Jamapa e intrusiones de agua fría que afectaron la región en los años setenta. Desde la disminución, pocos estudios han documentado el estado A. palmata. Consiste de 28 arrecifes divididos 13 al norte y 15 al sur del río. Entre el 2007 y 2013 se muestrearon 24 arrecifes para caracterizar las comunidades bentónicas. Siete de los 11 arrecifes del norte y en todos los arrecifes del sur tenían A. palmata. Las colonias se encontraron por lo general en el barlovento de los corales en agua sómeras a lo largo del borde del arrecife. También registramos los diámetros de las colonias y condición a lo largo de transectos tipo conturón en dos arrecifes al norte (Anegada de Adentro y Verde) y dos en el sur (Periférico y Sargazo), entre el 2011 y 2013. Además, ocho transectos se evaluaron en Rizo (al sur). Evaluamos un total de 1 804 colonias; las densidades variaron entre 0.02 a 0.28 colonias/m² (promedio (±DE) del diámetro de colonia 58± 3cm y 89±18% de tejido vivo por colonia). La depredación fue 5% por peces damisela, 2% por caracoles y <1% por gusanos, prevalencia de enfermedades <3%. La distribución de tamaños indicó que las poblaciones tienen un potencial moderado a alto de desove, 15-68 de las colonias eran maduras, y medían más de 1 600cm².La presencia de estas colonias saludables y potencialmente reproductivas son importantes para la recuperación de especies, particularmente porque el Caribe muestra muy pocas o ninguna señale de recuperación. Esfuerzos en conservación y manejo en estos arrecifes es vital.
Palabras clave: Acropora palmata, recuperación poblacional, Veracruz, Mexico, distribución de frecuencia de tamaño.
Acropora palmata and A. cervicornis (Lamarck, 1816) historically have been major reef framework builders in the greater Caribbean, Gulf of Mexico and Florida (Adey, 1978; Neigell & Avise, 1983). Their complex three-dimensional structure provides habitat to numerous invertebrate and vertebrate species and coastal protection during storms and hurricanes. Between the 1970’s and 1980’s populations across the species range were plagued by disease and in some locations researchers documented up to 100% loss by white band disease (Dustan & Halas, 1987; Knowlton, Lang & Keller, 1990; Aronson & Precht, 2001; Bruckner, 2002). There has been very little documentation of species recovery since these declines (Grober-Dunsmore, Bonito & Frazer, 2006; Mayor, Rogers & Hillis-Starr, 2006; Jones et al., 2008) thus, in 2006 they were listed as threatened by the US Endangered Species Act (NOAA, 2006) and critically endangered by the IUCN Red list (Aronson et al., 2008a; 2008b).
Since the listing, many populations have not returned to self-sustainable levels despite increased restoration activities and protection (Knowlton et al., 1990; Aronson & Precht, 2001; Bruckner, 2002; Miller, Bourque & Bohnsack, 2002; Acropora Biological Review Team, 2005); however, there are remnant populations throughout the Acropora spp. range. Large Acropora spp. populations have been reported in Florida (Vargas-Angel, Thomas & Hoke, 2003; Williams, Miller & Kramer, 2008; Walker, Larson, Moulding & Gilliam, 2012); Punta Rusia, Dominican Republic (Lirman et al., 2010); Roatan, Honduras (Keck, Houston, Purkis & Riegl, 2005); Venezuela (Zubillaga, Márquez, Cráquer & Bastidas, 2008); and St. John, US Virgin Islands (Grober-Dunsmore et al., 2006). Here we add to this list by reporting on the abundant A. palmata in Veracruz, Mexico.
The Veracruz Reef System (VRS) is located within the Parque Nacional Sistema Arrecifal Veracruzano (PNSAV) which encompasses 52 000ha of marine ecosystems off the coast of the major port city of Veracruz, Mexico and a small fishing village to the south, Antόn Lizardo. The VRS includes 28 shallow-water coral reefs, which are separated into two groups by the Jamapa River outflow. The northern group consists of 13 reefs near the city of Veracruz and the southern group has 15 reefs near Antόn Lizardo. Most of the reefs in this system are heavily impacted by urbanization, run-off, sedimentation, close proximity to coastline and fishing activities (Emery, 1963; Horta-Puga, 2007). In 1992, the VRS was declared a natural protected area and was converted into a National Park by the Mexican Government (Diario Oficial, 1992).
Since the late 1800’s, A. palmata and A. cervicornis have dominated the shallow-water coral reefs of the VRS (Heilprin, 1890). However, similar to other Caribbean populations, researchers observed local declines through the 1970’s and 1980’s. Mortality in this region was attributed to disease and the flooding of the nearby Jamapa and Papaloapan rivers (Rannefeld, 1972; Tunnell, 1988; 1992). In 1971, Rannefeld (1972) documented up to a 100% loss at Enmedio for both species and by 1973, abundance had decreased dramatically on many reef slopes (Tunnell, 1988; 1992). However, Jordan-Dahlgren (1992) documented re-colonization of A. palmata to old standing dead colonies in the late 1980’s. Another study by Lara, Padilla, García and Espejel (1992) observed Acropora spp. at 82% of the reefs surveyed (n=17) and recorded them as dominant (>20% live cover) on the fore reef edge and inner fore reef zones. Despite this, in 2002 population levels of A. palmata were reported as low (0–0.3% cover) at six reefs in the VRS (Jones et al., 2008), some of which had previously been documented as having colonies (Lara et al., 1992).
This study was part of a larger, long-term project in which we partnered with PNSAV staff to characterize the benthic communities in the VRS (Rangel Avalos et al., 2007). To date, 24 of the 28 reefs have been visited and it was during these visits that it was noted that a majority of the reefs had the presence of one or both species of Acropora. Using the knowledge gained of these populations during the benthic surveys, additional A. palmata assessments were completed to document the current status, health and possible recovery of A. palmata within the VRS. Population recovery herein is defined by an increase in colony abundance, size and distribution and the potential to sexually reproduce with low prevalence of recent mortality in comparison to previous studies in the VRS.
Materials and Methods
Benthic characterization assessment: Reefs within the VRS were visited between 2007 and 2013 as part of a larger benthic characterization project (Rangel Avalos et al., 2007). Fifty-five sites, targeting the windward side and northern and southern ends of the reefs, were surveyed across 24 of the 28 reefs. Of these sites, 28 were in the northern group and 27 were in the southern group. At each site, four or five 30m point intercept transects were completed for benthic characterization. In addition, notes and images were taken of the area surrounding the transects. Special attention was taken to note the presence of A. palmata. These data were utilized to document the distribution of A. palmata within the VRS and target sites for the A. palmata assessment.
Acropora palmata assessment: Five sites where A. palmata was found in greatest abundance were used to further investigate the status of A. palmata within the VRS. These sites were on two reefs in the northern group (Verde and Anegada de Adentro) and three reefs in the southern group (Rizo, Periférico, and Sargazo). Colony maximum length, height, percent mortality and presence or absence of disease and predation were collected along five or six parallel 10m belt transects at all reefs but Rizo. Divers spread across the reef approximately 10m apart, swimming parallel to each other, collecting data on colonies within 5m to each side of them. A hand-held GPS was used to mark the start and end of each transect to obtain transect length. Belt transects length varied between 15-170m and depended on the population size and time available. At Rizo, eight previously installed permanent transects, 7m x 30m, were utilized for the assessment. For all transects, a colony was only included if it had live tissue and measurements were made of the entire colony skeleton (live tissue + dead skeleton).
A live area index (LAI) was used to compare the amount of live tissue between sites and across years to better understand the population structure within the VRS. LAI indices typically use colony length, width and percent live tissue measurements. However, in order to maximize the number of colonies measured within the limited time available during each sampling, colony width was only measured for a subset of colonies (348) at four of the five sites; width measurements were not collected at Periférico. The mean ratio of length divided by the width of 348 colonies was 0.75, and this value was used as a conversion to estimate width for the LAI. Therefore, for this study LAI = (Length × (0.75) Length × % live tissue)/100, where length was the maximum dimension of the colony and percent live tissue was the percentage of the whole colony covered by live tissue.
The resulting size frequency distribution was used to describe the population structure. Soong and Lang (1992) reported that A. palmata was not fertile until colonies were >60cm2, and as size of the colony increased as did the frequency of fertility, 31% of the colonies between 250 and 1 000cm2, 43% of the colonies between 1 000 and 4 000cm2 and 88% of the colonies >4 000cm2 were fertile. In addition to determining the proportion of fertile colonies within each size class, they concluded that colonies greater than 1 600cm2 were reproductively mature. Therefore, our chosen size classes, similar to those of Schärer et al. (2008), are as follows: small colonies with no reproductive potential (<60cm2), medium colonies with low frequency of fertility (60-1 600cm2) and large mature colonies with moderate to high potential to spawn (>1 600cm2).
Results
Benthic characterization assessment: Based on the point intercept surveys and site descriptions, A. palmata was present at 20 of the 24 reefs surveyed and 34 of the 50 sites. In the northern group, A. palmata was present at seven of the 11 reefs and 13 of the 28 sites (Fig. 1). In the southern group, A. palmata was present at all reefs (n=13) and 21 of the 27 sites (Fig. 2). Where A. palmata was present, colony abundance varied by site ranging from one colony to multiple large overlapping colonies.
Five reefs were of particular interest because A. palmata was present in greater abundance. In the northern group, large overlapping A. palmata colonies were found at Verde and Anegada de Adentro reefs. Colonies were observed on the reef flat at Anegada de Adentro and on the leeward slope of Verde. Three reefs in the southern group, Rizo, Sargazo and Periférico also had greater abundance. Colonies at Rizo were observed on the reef flat and on the upper fore reef of Sargazo and Periférico.
Acropora palmata assessment: Belt transects targeting A. palmata were completed between 2011 and 2013 at Verde, Anegada de Adentro, Rizo, Periférico and Sargazo reefs, surveying a total area of 21 420m² (Table 1). A total of 1 804 colonies were measured across all sites and all years. The number of colonies ranged from 40 in 2011 at Rizo to 764 at Verde in 2012 (Table 1).
Colony density (±SD) ranged from 0.02±0.03colonies/m² to 0.28±0.08colonies/m² (Table 1). The greatest abundance of A. palmata was found at Anegada de Adentro where the density was significantly greater than that of the other four sites (Kruskal-Wallis, p<0.05). When reefs were grouped by location, the northern sites’ mean density (0.15±0.12colonies/m²) was significantly greater than the southern (0.06±0.06colonies/m²—Kruskal- Wallis, p<0.05). Verde and Rizo were visited during consecutive years and both had an increase in colony density from 2011 to 2012 (Table 1). The increased number of colonies at Rizo was likely due to the VRS being directly impacted by two storms between the 2011 and 2012 surveys. Rizo was surveyed once more in 2013 and density remained similar to that observed in 2012.
Mean colony length (±SD) for all reefs was 58±73cm. Anegada de Adentro had significantly larger colonies than the rest of the reefs (122±163cm —Kruskal-Wallis, p<0.05) and the largest colony recorded (length= 1 320cm). At Verde colony length increased from 2011 to 2012. At Rizo colony length decreased from 2011 to 2012, but increased slightly in 2013 (Table 1).
Mean percent live tissue (±SD) per colony for all reefs across all years was 89±18%. Mean percent live tissue per colony varied by reef and was greatest at Rizo in 2011, 96% (Table 1). However, from 2011 to 2013 colonies at Rizo decreased in mean percent live tissue per colony from 96% to 73%. Colonies at Verde also decreased in live tissue from 92% to 87% between the 2011 and 2012 survey.
Predation by the threespot damselfish, Stegastes planifrons (Cuvier 1830), snail, Coralliophilia abbreviata (Lamarck 1816) and bearded fireworm, Hermodice carunculata (Pallas 1766) were present in both groups. Damselfish predation, through the creation of algal gardens was the most common type, observed on 95 colonies. Its impact was difficult to quantify and was only recorded as present or absent on a colony since it was not considered a cause of recent mortality. The most common cause of recent mortality was predation by snails, affecting 35 colonies throughout the three years of data collection. Half of the colonies with snail predation were observed at Sargazo. Fireworm predation was recorded on ten colonies; eight of which were observed at Verde. The mean percent recent mortality of all colonies was 1.1%, and ranged from 3.3% at Sargazo to 0.1% at Rizo.
Disease prevalence was low. Although it was recorded at four of the five reefs, it was only observed on 2.7% of the colonies across the entire study. Disease was not observed at Rizo. Rapid tissue loss (RTL) was the most commonly recorded condition (Williams & Miller, 2005), observed affecting 1.4% of the colonies. White band disease (WB) and white pox (WP) were also observed in low levels 0.9% and 0.3% of the colonies respectively. WB was mainly observed at Verde affecting only one other colony at Periférico. RTL was observed at Verde, Anegada de Adentro and Sargazo and WP was only observed at Verde and Sargazo.
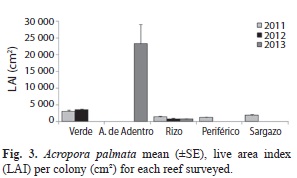
Mean LAI ranged from 828cm² to 23 448cm² per reef (Fig. 3). The reefs in the northern group had significantly larger mean LAI’s, Anegada de Adentro (23 448cm²) and Verde (3 097cm² in 2011 and 3 580cm² in 2012) than the southern group, where all reefs had a LAI under 2 000cm² (Kruskal-Wallis, p<0.05- Fig. 3).
In 2011, for all reefs combined, 6% of the measured colonies were small (<60cm²), 66% were medium (60-1 600cm²) and 28% were large (>1 600cm²). From 2011 to 2012, there was a small shift from small colonies to medium and large at Verde. At Rizo, there was a decrease in the number of large colonies and an increase in the number of small and medium colonies and by 2013, there was a small increase in medium colonies and decrease in small colonies. Anegada de Adentro was the only site dominated by large colonies with 69% having a mean LAI greater than 1 600cm² (Fig. 3, 4).
Discussion
This study is the most comprehensive study of A. palmata within the Veracruz Reef System to date. Its distribution through the VRS indicates that A. palmata may be recovering from the 1970’s and 1980’s die off. Acropora palmata was present at a majority of the sites surveyed in both reef groups. For those sites where it was not found it appears that either the habitat was not suitable (too deep) or the sites were within close proximity to shore and/or the major shipping port (northern group). The overall mean density of this study (0.08colonies/ m²) was greater than the previously described for the area (0.03), Puerto Rico (0.03), St. Croix (0.02) and St. John (0.06) and similar to Venezuela (0.08) (Jordan-Dahlgren, 1992; Mayor et al., 2006; Schärer et al., 2008; Zubillaga et al., 2008). Colony density and mean colony length were greatest in the northern group (Verde and Anegada de Adentro). This was contrary to what was found by Jordan-Dahlgren (1992), however the three reefs that they surveyed in the southern group were not included in our A. palmata assessment.
In order for a population to be recovering it needs to be dominated by ‘healthy’ colonies, which are characterized by a broad distribution of colony size frequencies (Knowlton, 2001) low prevalence of disease, predation and recent mortality and have reproductive potential (Richmond, 1997; Zubillaga et al., 2008). The mean colony length during this study was 58cm, whereas Jordan-Dahlgren (1992) reported a mean colony size of 22cm. The largest colony measured (1 320cm) was over ten times that of Jordan-Dahlgren (1992). Across the three years of data collection, the mean percent live tissue of all colonies was 89% and mean recent mortality was only 1.1%. Prevalence of disease (2.7%) and predation (2.5%) were observed at very low levels. Other studies have recorded similar or higher levels of disease and predation in other regions (Grober-Dunsmore, et al., 2006; Schärer et al., 2008; Zubillaga et al., 2008; Williams & Miller, 2011).
The A. palmata populations in the VRS have a high spawning potential, 38% of the colonies are considered mature (>1 600cm²), of those colonies 53% were >4 000cm2. According to Soong and Lang (1992) there is a high potential (88%) that colonies >4 000cm2 are fertile. All sites were dominated by medium sized colonies; only 4% of the colonies were classified as small across all years. This size frequency distribution indicates that these populations have a high potential for colonies to spawn, whereas two decades prior the majority of the colonies were in the small to medium size classes (Jordan-Dahlgren 1992). Large colonies were found across the VRS, increasing the potential for sexual reproduction, albeit the genetic diversity for this species is currently unknown for this region.
Although the A. palmata populations were ‘healthy’ as defined herein, there was a decrease in the percent live tissue cover (96% to73%) at Rizo from 2011 to 2013. We found no signs of disease and very little recent mortality, however, the VRS was directly hit by two storms between the 2011 and 2012 surveys, Tropical Storm Harvey (August) and Hurricane Nate (September). These storms caused an increase in wave height and flooding of the Jamapa river (Blake, 2011; Kimberlain, 2011). At Rizo in 2011, 40 A. palmata colonies were measured within the permanent monitoring transects and in 2012, the count increased to 89. Initially, there were no small colonies within this population, but in 2012, 7% of the colonies were less than 60cm². There was also a 20% drop in the number of large colonies, which may indicate that the energy from the storms caused some large colonies to fragment into smaller colonies. Williams (2011) found similar increases in fragmentation, decreases in colony size, and increases in colony tissue mortality following a hurricane. This shift was also apparent in the mean colony length, decreasing from 40cm to 33cm between 2011 and 2012. However, this trend seemed to be reverting in 2013, as we observed a decrease in small colonies and an increase in medium colonies. This could indicate that the small fragments formed in 2012 had survived and grown in the intervening period, showing some resilience, characteristic of a healthy population. We were not able to detect similar patterns at Verde because, unlike Rizo, the transects started in the same location both years, but the 2012 transect continued an additional 1 200m past the end of the 2011 transects, therefore including more colonies in the assessment. In addition, the colonies at Verde may be more protected as they are found on the leeward slope of the reef, whereas colonies at Rizo are more exposed on the windward edge of the reef.
These data suggest that A. palmata in the VRS is healthy, abundant, broadly distributed, and has a high spawning potential, whereas most of the Caribbean populations are not. Hence, the next question may be, why has this species been able to proliferate in this area where there are continuous terrestrial and environmental impacts? Especially if it is thought to have endured the same population decrease as the rest of the Greater Caribbean. During a time where this species is under review to be uplisted to Endangered under the US Endangered Species Act, proper management and conservation efforts should be made to protect these recovering populations.
Acknowledgments
We thank the staff of the Parque Nacional Sistema Arrecifal Veracruzano, M. Rangel Avalos and J. Santander Monsalvo for critical logistical support. Nova Southeastern University Oceanographic Center graduate students, V. Brinkhuis, S. Bush, A. Costaregni, N. D’Antonio, P. Espitia, D. Fahy, A. Halperin, M. Sathe, J. Walczak, and C. Walton, provided essential help with data collection. Partial funding for this research was provided by multiple Nova Southeastern University President Faculty Research and Development Grants, the Guy Harvey Research Institute, and a grant from the National Oceanic and Atmospheric Administration to the National Coral Reef Institute. We thank Margaret Miller and an anonymous reviewer for constructive comments on this work. This is NCRI Contribution 157.
References
Acropora Biological Review Team. (2005). Atlantic Acropora status review document. Report to National Marine Fisheries Service, Southeast Regional Office. [ Links ]
Adey, W. H. (1978). Coral reef morphogenesis: a multidimensional model. Science, 202, 831-837. [ Links ]
Aronson, R. B., Bruckner, A. W., Moore, J.,. Precht W. F., & Weil, E. (2008a). IUCN Red List of Threatened Species: Acropora cervicornis. Retrieved from www.iucnredlist.org. [ Links ]
Aronson, R. B., Bruckner, A. W., Moore, J., Precht W. F., & Weil, E. (2008b). IUCN Red List of Threatened Species: Acropora palmata. Version 2012.2. Retrieved from www.iucnredlist.org. [ Links ]
Aronson, R. B., & Precht, W. F. (2001). White-band disease and the changing face of Caribbean coral reefs. Hydrobiologia, 460, 25-38. [ Links ]
Blake, E. S. (2011). Tropical Cyclone Report. Tropical Cyclone Harvey. National Oceanic and Atmospheric Administration, USA. [ Links ]
Bruckner, A. W. (2002). Proceedings of the Caribbean Acropora Workshop: Potential Application of the U.S. Endangered Species Act as a Conservation Strategy. NOAA Technical Memorandum NMFS-OPR-24. [ Links ]
Diario Oficial, D. (1992). Decreto por el que se declara área natural protegida con el carácter de Parque Marino Nacional, la zona conocida como Sistema Arrecifal Veracruzano, ubicada frente a las Costas de los municipios de Veracruz, Boca del Río y Alvarado del estado de Veracruz Llave, con superficie de 52,238-91-50 hectáreas. Diario Oficial de la Federacion, Mexico. [ Links ]
Dustan, P. & Halas, J. C. (1987). Changes in the reef-coral community of Carysfort reef, Key Largo, Florida: 1974 to 1982. Coral Reefs, 6, 91-106. [ Links ]
Emery, K. O. (1963). Coral reefs off Veracruz, Mexico. Geofisica internacional, 3, 11-17. [ Links ]
Grober-Dunsmore, R., Bonito, V., & Frazer, T. K. (2006). Potential inhibitors to recovery of Acropora palmata populations in St. John, US Virgin Islands. Marine Ecology Progress Series, 321, 123-132. [ Links ]
Heilprin, A. (1890). The Corals and Coral Reefs of the Western Waters of the Gulf of Mexico. Proceedings of the Academy of Natural Sciences of Philadelphia, 42, 303-316. [ Links ]
Horta-Puga, G. (2007). Environmental Impacts, p. 126-141. In Tunnell J. W. Jr., Chavez E. A., & Withers K. (eds.). Coral Reefs of the Southern Gulf of Mexico. Texas: A&M University Press, College Station. [ Links ]
Jones, J., Withers, K., & Tunnell, J. W. Jr. (2008). Comparison of Benthic Communities on Six Coral Reefs inthe Veracruz Reef System (Mexico). Proceedings of the 11th International Coral Reef Symposium, Fort Lauderdale, FL USA, 757-760. [ Links ]
Jordan-Dahlgren, E. (1992). Recolonization Patterns of Acropora palmata in a Marginal Environment. Bulletin of Marine Science, 51, 104-117. [ Links ]
Keck, J., Houston, R. S., Purkis, S., & Riegl, B. M. (2005). Unexpectedly high cover of Acropora cervicornis on offshore reef in Roatan (Honduras). Coral Reefs, 24, 509. [ Links ]
Kimberlain, T. B. (2011). Tropical Cyclone Report. Hurricane Nate. National Oceanic and Atmospheric Administration, USA. [ Links ]
Knowlton, N. (2001). The future of coral reefs. Proceedings of the National Academy of Science of the United States of America, 5419-5425. [ Links ]
Knowlton, N., Lang, J. C. & Keller, B. D. (1990). Case study of natural population collapse: post-hurricane predation on Jamaican Staghorn coral. Smithsonian Contributions to the Marine Sciences, 36. [ Links ]
Lara, M., Padilla, C., Garcia, C., & Espejel, J. J. (1992). Coral Reef of Veracruz Mexico I. Zonation and community. Proceedings of the 7th International Coral Reef Symposium, Guam., 1, 535-544. [ Links ]
Lirman, D., Bowden-Kerby, A., Schopmeyer, S., Huntington, B., Thyberg, T., Gough, M., Gough, T., Gough, R., & Gough, Y. (2010). A window to the past: documenting the status of one of the last remaining ‘megapopulations’ of the threatened staghorn coral Acropora cervicornis in the Dominican Republic. Aquatic Conservation: Marine and Freshwater Ecosystems, 20, 773-781. [ Links ]
Mayor, P.A., Rogers, C.S. & Hillis-Starr, Z.M. (2006). Distribution and abundance of elkhorn coral, Acropora palmata, and prevalence of white-band disease at Buck island reef national monument, St. Croix, US Virgin Islands. Coral Reefs, 25, 239-242. [ Links ]
Miller, M. W., Bourque, J., & Bohnsack, J. (2002). An analysis of the loss of Acroporid corals at Looe Key, Florida, USA 1983-2000. Coral Reefs, 21, 179-182. [ Links ]
Neigell, J. E., & Avise, J. C. (1983). Clonal diversity and population structure in a reef-building coral, Acropora cervicornis: self recognition analysis and demographic interpretation. Evolution, 37, 437-453. [ Links ]
NOAA. (2006). Endangered and threatened species: final listing determinations for Elkhorn coral and Staghorn coral Federal Register 71: 26852-26861. [ Links ]
Rangel Avalos, M. A., Jordan, L. K. B., Walker, B. K., Gilliam, D. S., Carvajal Hinojosa, E., & Spieler, R. E. (2007). Fish and Coral Reef Communities of the Parque Nacional Sistema Arrecifal Veracruzano (Veracruz Coral Reef System National Park) Veracruz, Mexico: Preliminary Results. Proceedings of the 60th Annual Gulf and Caribbean Fisheries Institute Meeting, Punta Cana, Dominican Republic, 60, 427-435. [ Links ]
Rannefeld, J. W. (1972). The stony corals of Enmedio Reef off Veracruz, Mexico. M.S Thesis. Texas A & M University, College Station. [ Links ]
Richmond, R. H. (1997). Reproduction and recruitment in corals: critical links in the persistence of reefs, p. 175-197. In Brikeland, C. (eds.). Life and Death of Coral Reefs. New York: Chapman & Hall. [ Links ]
Schärer, M., Nemeth, M., Valdivia, A., Miller, M., Williams, D., & Diez, C. (2008). Elkhorn coral distribution and condition throughout the Puerto Rican Archipelago. Proc. of the 11th Int. Coral Reef Symp., Fort Lauderdale, FL USA., 815-819. [ Links ]
Soong, K., & Lang, J. C. (1992). Reproductive integration in reef corals. Biological Bulletin, 183, 418-431. [ Links ]
Tunnell, J. W. J. (1988). Regional comparison of southwestern Gulf of Mexico to Caribbean sea coral reefs. Proceedings of the 6th International Coral Reef Symposium, Townsville, Australia, 3, 303-308. [ Links ]
Tunnell, J. W. J. (1992). Natural Versus Human Impacts to Southern Gulf of Mexico Coral Reef Resources. Proceedings of the 7th International Coral Reef Symposium, Guam., 1, 300-306. [ Links ]
Vargas-Angel, B., Thomas, J. D., & Hoke, S. M. (2003). High-latitude Acropora cervicornis thickets off Fort Lauderdale, Florida, USA. Coral Reefs, 22, 465-473. [ Links ]
Walker, B. K., Larson, E. A., Moulding, A. L., & Gilliam, D. S. (2012). Small-scale mapping of indeterminate arborescent acroporid coral (Acropora cervicornis) patches. Coral Reefs, 31, 885-894. [ Links ]
Williams, D. E., & Miller, M. W. (2005). Coral disease outbreak: pattern. prevalence and transmission in Acropora cervicornis. Marine Ecology Progress Series, 301, 119-128. [ Links ]
Williams, D. E., Miller, M. W., & Kramer, K. L. (2008). Recruitment failure in Florida Keys Acropora palmata, a threatened Caribbean coral. Coral Reefs, 27, 697-705. [ Links ]
Williams, D. E., & Miller, M. W. (2011). Attributing mortality among drivers of population decline in Acropora palmata in the Flordia Keys (USA). Coral Reefs, 31, 369-382. [ Links ]
Zubillaga, A. L., Márquez, L. M., Cráquer, A., & Bastidas, C. (2008). Ecological and genetic data indicate recovery of the endangered coral Acropora palmata in Los Roques, Southern Caribbean. Coral Reefs, 27, 63-72. [ Links ]
Adey, W. H. (1978). Coral reef morphogenesis: a multidimensional model. Science, 202, 831-837. [ Links ]
Aronson, R. B., Bruckner, A. W., Moore, J.,. Precht W. F., & Weil, E. (2008a). IUCN Red List of Threatened Species: Acropora cervicornis. Retrieved from www.iucnredlist.org. [ Links ]
Aronson, R. B., Bruckner, A. W., Moore, J., Precht W. F., & Weil, E. (2008b). IUCN Red List of Threatened Species: Acropora palmata. Version 2012.2. Retrieved from www.iucnredlist.org. [ Links ]
Aronson, R. B., & Precht, W. F. (2001). White-band disease and the changing face of Caribbean coral reefs. Hydrobiologia, 460, 25-38. [ Links ]
Blake, E. S. (2011). Tropical Cyclone Report. Tropical Cyclone Harvey. National Oceanic and Atmospheric Administration, USA. [ Links ]
Bruckner, A. W. (2002). Proceedings of the Caribbean Acropora Workshop: Potential Application of the U.S. Endangered Species Act as a Conservation Strategy. NOAA Technical Memorandum NMFS-OPR-24. [ Links ]
Diario Oficial, D. (1992). Decreto por el que se declara área natural protegida con el carácter de Parque Marino Nacional, la zona conocida como Sistema Arrecifal Veracruzano, ubicada frente a las Costas de los municipios de Veracruz, Boca del Río y Alvarado del estado de Veracruz Llave, con superficie de 52,238-91-50 hectáreas. Diario Oficial de la Federacion, Mexico. [ Links ]
Dustan, P. & Halas, J. C. (1987). Changes in the reef-coral community of Carysfort reef, Key Largo, Florida: 1974 to 1982. Coral Reefs, 6, 91-106. [ Links ]
Emery, K. O. (1963). Coral reefs off Veracruz, Mexico. Geofisica internacional, 3, 11-17. [ Links ]
Grober-Dunsmore, R., Bonito, V., & Frazer, T. K. (2006). Potential inhibitors to recovery of Acropora palmata populations in St. John, US Virgin Islands. Marine Ecology Progress Series, 321, 123-132. [ Links ]
Heilprin, A. (1890). The Corals and Coral Reefs of the Western Waters of the Gulf of Mexico. Proceedings of the Academy of Natural Sciences of Philadelphia, 42, 303-316. [ Links ]
Horta-Puga, G. (2007). Environmental Impacts, p. 126-141. In Tunnell J. W. Jr., Chavez E. A., & Withers K. (eds.). Coral Reefs of the Southern Gulf of Mexico. Texas: A&M University Press, College Station. [ Links ]
Jones, J., Withers, K., & Tunnell, J. W. Jr. (2008). Comparison of Benthic Communities on Six Coral Reefs inthe Veracruz Reef System (Mexico). Proceedings of the 11th International Coral Reef Symposium, Fort Lauderdale, FL USA, 757-760. [ Links ]
Jordan-Dahlgren, E. (1992). Recolonization Patterns of Acropora palmata in a Marginal Environment. Bulletin of Marine Science, 51, 104-117. [ Links ]
Keck, J., Houston, R. S., Purkis, S., & Riegl, B. M. (2005). Unexpectedly high cover of Acropora cervicornis on offshore reef in Roatan (Honduras). Coral Reefs, 24, 509. [ Links ]
Kimberlain, T. B. (2011). Tropical Cyclone Report. Hurricane Nate. National Oceanic and Atmospheric Administration, USA. [ Links ]
Knowlton, N. (2001). The future of coral reefs. Proceedings of the National Academy of Science of the United States of America, 5419-5425. [ Links ]
Knowlton, N., Lang, J. C. & Keller, B. D. (1990). Case study of natural population collapse: post-hurricane predation on Jamaican Staghorn coral. Smithsonian Contributions to the Marine Sciences, 36. [ Links ]
Lara, M., Padilla, C., Garcia, C., & Espejel, J. J. (1992). Coral Reef of Veracruz Mexico I. Zonation and community. Proceedings of the 7th International Coral Reef Symposium, Guam., 1, 535-544. [ Links ]
Lirman, D., Bowden-Kerby, A., Schopmeyer, S., Huntington, B., Thyberg, T., Gough, M., Gough, T., Gough, R., & Gough, Y. (2010). A window to the past: documenting the status of one of the last remaining ‘megapopulations’ of the threatened staghorn coral Acropora cervicornis in the Dominican Republic. Aquatic Conservation: Marine and Freshwater Ecosystems, 20, 773-781. [ Links ]
Mayor, P.A., Rogers, C.S. & Hillis-Starr, Z.M. (2006). Distribution and abundance of elkhorn coral, Acropora palmata, and prevalence of white-band disease at Buck island reef national monument, St. Croix, US Virgin Islands. Coral Reefs, 25, 239-242. [ Links ]
Miller, M. W., Bourque, J., & Bohnsack, J. (2002). An analysis of the loss of Acroporid corals at Looe Key, Florida, USA 1983-2000. Coral Reefs, 21, 179-182. [ Links ]
Neigell, J. E., & Avise, J. C. (1983). Clonal diversity and population structure in a reef-building coral, Acropora cervicornis: self recognition analysis and demographic interpretation. Evolution, 37, 437-453. [ Links ]
NOAA. (2006). Endangered and threatened species: final listing determinations for Elkhorn coral and Staghorn coral Federal Register 71: 26852-26861. [ Links ]
Rangel Avalos, M. A., Jordan, L. K. B., Walker, B. K., Gilliam, D. S., Carvajal Hinojosa, E., & Spieler, R. E. (2007). Fish and Coral Reef Communities of the Parque Nacional Sistema Arrecifal Veracruzano (Veracruz Coral Reef System National Park) Veracruz, Mexico: Preliminary Results. Proceedings of the 60th Annual Gulf and Caribbean Fisheries Institute Meeting, Punta Cana, Dominican Republic, 60, 427-435. [ Links ]
Rannefeld, J. W. (1972). The stony corals of Enmedio Reef off Veracruz, Mexico. M.S Thesis. Texas A & M University, College Station. [ Links ]
Richmond, R. H. (1997). Reproduction and recruitment in corals: critical links in the persistence of reefs, p. 175-197. In Brikeland, C. (eds.). Life and Death of Coral Reefs. New York: Chapman & Hall. [ Links ]
Schärer, M., Nemeth, M., Valdivia, A., Miller, M., Williams, D., & Diez, C. (2008). Elkhorn coral distribution and condition throughout the Puerto Rican Archipelago. Proc. of the 11th Int. Coral Reef Symp., Fort Lauderdale, FL USA., 815-819. [ Links ]
Soong, K., & Lang, J. C. (1992). Reproductive integration in reef corals. Biological Bulletin, 183, 418-431. [ Links ]
Tunnell, J. W. J. (1988). Regional comparison of southwestern Gulf of Mexico to Caribbean sea coral reefs. Proceedings of the 6th International Coral Reef Symposium, Townsville, Australia, 3, 303-308. [ Links ]
Tunnell, J. W. J. (1992). Natural Versus Human Impacts to Southern Gulf of Mexico Coral Reef Resources. Proceedings of the 7th International Coral Reef Symposium, Guam., 1, 300-306. [ Links ]
Vargas-Angel, B., Thomas, J. D., & Hoke, S. M. (2003). High-latitude Acropora cervicornis thickets off Fort Lauderdale, Florida, USA. Coral Reefs, 22, 465-473. [ Links ]
Walker, B. K., Larson, E. A., Moulding, A. L., & Gilliam, D. S. (2012). Small-scale mapping of indeterminate arborescent acroporid coral (Acropora cervicornis) patches. Coral Reefs, 31, 885-894. [ Links ]
Williams, D. E., & Miller, M. W. (2005). Coral disease outbreak: pattern. prevalence and transmission in Acropora cervicornis. Marine Ecology Progress Series, 301, 119-128. [ Links ]
Williams, D. E., Miller, M. W., & Kramer, K. L. (2008). Recruitment failure in Florida Keys Acropora palmata, a threatened Caribbean coral. Coral Reefs, 27, 697-705. [ Links ]
Williams, D. E., & Miller, M. W. (2011). Attributing mortality among drivers of population decline in Acropora palmata in the Flordia Keys (USA). Coral Reefs, 31, 369-382. [ Links ]
Zubillaga, A. L., Márquez, L. M., Cráquer, A., & Bastidas, C. (2008). Ecological and genetic data indicate recovery of the endangered coral Acropora palmata in Los Roques, Southern Caribbean. Coral Reefs, 27, 63-72. [ Links ]
1. Nova Southeastern University Oceanographic Center, 8000 N. Ocean Dr. Dania Beach, FL, 33004 USA; goergen@nova.edu
Received 03-XI-2013 Corrected 12-III-2014 Accepted 24-III-2014