Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.61 n.3 San José Sep. 2013
Structural adaptations of two sympatric epiphytic orchids (Orchidaceae) to a cloudy forest environment in rocky outcrops of Southeast Brazil
Adaptaciones estructurales de dos orquídeas epífitas simpátricas (Orchidaceae) en un bosque nuboso de un campo rupestre del sureste de Brasil
Adaptaciones estructurales de dos orquídeas epífitas simpátricas (Orchidaceae) en un bosque nuboso de un campo rupestre del sureste de Brasil
*Dirección para correspondencia:
Abstract
The survival of plants in epiphytic environments depends on vegetative adaptations capable to defraud different stresses. Based on the structural diversity of the Orchidaceae, the current study has the objective of relating the anatomical structure of Dichaea cogniauxiana and Epidendrum secundum with the distinct environments where they live. It was expected that, despite structural similarities as strategies for resource acquisition, some peculiar variations related to the distinct light microenvironments (inside or in the edge of the nebular forest, near to “campo rupestre” area) might be found. Leaves and roots of both species were collected in a nebular forest located at a “campo rupestre” area at Serra da Piedade, Brazil), in January and February 2005. D. cogniauxiana is adhered to trunks, in sites with high atmospheric humidity and shaded, while E. secundum is located at the edge of the nebular forest, in more luminous sites. The leaves of E. secundum had thicker cuticle and higher number of stomata per area than those of D. cogniauxiana, characteristics coherent with their distinct pattern of exhibition to sun light. The suprastomatic chambers formed by the thicker cuticle may function as a barrier of resistance to water evaporation. The succulence of the leaves of E. secundum propitiates organic acids storage at night, and the storage of starch may be involved in PEP-carboxylase metabolism, both propitiating CAM mechanism. Roots with larger number of cell layers of the velamen, and specialized thick walled cortical cells (both in E. secundum) help water absorption and indicate better adaptation to an environment with intense solar radiation and a probable higher water deficit. The remarkable cell wall thickening of E. secundum exodermis can confer more efficient protection against the excess of transpiration at the border of the nebular forest. On the other hand, besides D. cogniauxiana be epiphyte, it is in a low position - in a shaded environment and with high relative humidity. Its thin thickened velamen permits the entrance of the low available light, and photosynthesis, producing oxygen and helping to avoid hypoxia condition. As features registered for D. cogniauxiana and E. secundum roots, we can depict the velamen, distinct exodermis and endodermis, and specialized thick walled cortical cells as characteristic of epiphytic plants.
Key words: Orchidaceae, anatomy, epiphytic environments, ecological anatomy, histochemistry.
Resumen
La sobrevivencia de las plantas epífitas a su ambiente depende de las adaptaciones vegetativas que les permiten afrontar diferentes tipos de estrés. Basado en la diversidad estructural de las Orchidaceae, el presente estudio tiene por objetivo relatar la estructura anatómica de Dichaea cogniauxiana y Epidendrum secundum con relación a los distintos ambientes donde viven. Hojas y raíces fueron recolectados en un bosque nuboso de un campo rupestre en la Serra da Piedade, Brasil. Dichaea cogniauxiana está adherida a los troncos, en sitios con alta humedad atmosférica y sombra, mientras que E. secundum está localizada en el margen del bosque nebular, en sitios más iluminados. Las hojas de E. secundum tienen una cutícula más gruesa y un mayor número de estomas por área que las de D. cogniauxiana, características coherentes con los diferentes patrones de exhibición a la luz del sol. Las cámaras supraestomáticas formadas por la cutícula pueden funcionar como una barrera de resistencia a la evaporación del agua. La suculencia de las hojas de E. secundum propicia el almacenamiento de ácidos orgánicos por la noche, y el almacenamiento de almidón puede estar implícito en el metabolismo de la PEP-carboxilasa. Las raíces con mayor velamen y especializadas paredes gruesas de las células corticales (ambas en E. secundum) ayudan a la absorción e indican una mejor adaptación al ambiente con intensa radiación solar y probable alto déficit de agua. El engrosamiento de la pared celular en la exodermis de E. secundum puede conferir una protección más eficaz contra el exceso de transpiración en el margen del bosque nebular. Por otra parte, D. cogniauxiana además de ser epífita, está en una posición baja, en un ambiente sombreado y con una alta humedad relativa. Su velamen delgado permite la entrada de la baja luz disponible y la fotosíntesis, produciendo oxígeno que ayuda a evitar la condición hipóxica.
Palabras clave: Orchidaceae, anatomía, ambientes epífitos, anatomía ecológica, histoquímica.
The most species-rich family in the epiphytic flora is Orchidaceae, represented by more individuals than all other taxa together (Benzing 1987, Zotz 2004). Two out of three epiphyte species are orchids, and about 70% of the family is canopy-dependent (Benzing 1987). These plants exhibit structural and physiological mechanisms for carbon fixation, and water and nutrients acquisition, resources scarce in epiphytic environment. Structural characteristics from roots, stems and leaves include tissues specialized in water absorption in roots, and leaf and stem succulence (Benzing et al. 1983, Moreira & Isaias 2008). Physiologically, the crassulacean acid metabolism (CAM) provides important water economy by stomata closure during the day (Lüttge 2004), and photosynthesis in roots helps themselves in the maintenance of oxygen supply (Dycus & Knudson 1957, Erickson 1957, Moreira et al. 2009).
Aerial roots often exhibit velamen, and a stele involved by parenchymatic cortex limited internally by an endodermis, and externally by an exodermis (Holtzmeier et al. 1998, Oliveira & Sajo 1999a, Stern & Whitten 1999, Moreira & Isaias 2008). The velamen is formed by dead compactly arranged cells with suberized walls. When the atmosphere is dry, its cells are completely air filled, but when it rains, and the air moisture is high, they fill with water, providing an efficient way of water absorption for epiphytic plants (Dycus & Knudson 1957, Benzing et al. 1982, Cockburn et al. 1985). In mature stage, the exodermis and endodermis may develop suberin lamellae and thick, secondary walls, preventing ion movement through the symplast. Some short cells without deposition of suberin lamellae are called passage cells, and may be responsible for symplast control, and transference of water and nutrients (Peterson & Enstone 1996, Ma & Peterson 2003).
For survivorship in epiphytic environment, not only water absorption is important, but also its storage, which may confer leaf succulence. This is a common feature in epiphytic Orchidaceae, and is provided by epidermis with thick cuticle, large mesophyll cells with specialized wall thickenings, and large vacuoles. Succulence is intimately linked to CAM, since vacuolated cells are necessary for water storage, and for organic acid accumulation at night (Griffiths et al. 1986, Jiménez & Morales 1987, Cushman 2001, Moreira et al. 2009).
Sympatric species belonging to the same plant family, such as Dichea cogniauxiana Schltr. and Epidendrum secundum Jacq., may constitute model species for studies on environmental dependent and independent anatomical features, and their variation in response to light and humidity, especially in an epiphytic environment. Both plant species occur at Serra da Piedade, Minas Gerais, Brazil. D. cogniauxiana is generally adhered to trunks covered by mosses, in sites with high atmospheric humidity, frequent fog, and almost never occurs 70cm above the soil (Alves 1990). E. secundum is located on trunks, 2m above the soil at the edge of the nebular forest, in more luminous sites. This species is representative of the flora of “campos rupestres”, being reported sometimes as an epilithic species (Barros 2004).
Some anatomical aspects of both species have been studied, in relation to their photosynthetic yield (Moreira et al. 2009) or to their anatomical features related to epiphytic habit (Oliveira & Sajo 1999a, b). In fact, Oliveira & Sajo (1999a, b) studies on E. secundum dealt with plants cultivated in greenhouse conditions, and described roots with five-six layered velamen, exodermal cells with wall thickenings, 10-12 layered cortex, O-thickened endodermis, and 17 protoxylem poles. The leaves measured 9.5 x 1.5cm, had simple and hypostomatic epidermis, hypoderm and groups of sclerified cells within mesophyll, and pericyclic fibers around vascular bundles.
In the current research, we assume that the epiphytic environment conditions, with the same general type of substratum and water availability, implies on similar mechanisms to acquisition of resources, and that D. cogniauxiana and E. secundum may present some structural adaptive convergences. Considering this environment, even discrete abiotic variations may be reflected in plant anatomy, and it is expected that either D. cogniauxiana or E. secundum will present anatomical and histochemical features related to epiphytic habit. However, some peculiar variations related to the distinct light microenvironments (inside or in the edge of the nebular forest, near a “campo rupestre” area) may be found.
Material and Methods
Leaves and roots of D. cogniauxiana (Fig. 1a), and E. secundum (Fig. 1b, c) were collected in a nebular forest located at a “campo rupestre” area at Serra da Piedade (Fig. 1d), Caeté/MG, Brazil (ca. 19°49'11" S - 43°40'12" W, at an elevation of approximately 1 300m), in January and February 2005 (vouchers BHCB 56452 and BHCB 56465, respectively). The species were collected in trunks 10-30cm in diameter of distinct tree species, with canopy reaching between two and four meters, approximately. Fragments of the samples were fixed in FAA50 (formaldehyde 37%, acetic acid and ethanol 50°GL) for at least 48 hours, and stored in ethanol 70°GL (Johansen 1940).
Free hand sections were clarified in 50% sodium hypochlorite, and stained with 1% safranin (C.I. 50 240) in 50% ethanol (Berlyn & Miksche 1976), and astra blue (C.I. not indicated) (1:9v/v), both with concentration modified for 0.5%. Semi-permanent slides were mounted in Kaiser’s jelly glycerin (Johansen 1940). Permanent slides of longitudinal (LS) and transverse sections (TS) of roots and leaves were prepared by ethanolic dehydration, and inclusion in Leica® historesin. Sections were stained with 0.05% toluidine blue in NaHPO4 0.1M (pH 6.8) (O’Brien & McCully 1981), and mounted in 50% glycerin.
Leaf fragments (1cm2) of totally expanded leaves were immersed in 50% sodium hypochlorite until the completely detachment of epidermis. These fragments were stained with 1% safranin in 50% ethanol (Berlyn & Miksche 1976), and mounted in Kaiser’s jelly glycerin for frontal view (FV) observations. Stomata and common epidermal cells density were quantified (n=40 fields) in six fragments from four leaves per species, with the aid of a clear camera coupled to a light microscope Olympus BH2-BHT.
Histochemical analyses were performed in fresh plant material. Lignins were detected using acid phloroglucin (Johansen 1940), neutral fats and wax acids were evidenced with Sudan III (Gahan 1984), starch grains with Lugol (Jensen 1962), pectic compounds with ruthenium red (Johansen 1940), and alkaloids with Dragendorff reagent (Johansen 1940). For phenolic derivatives, the sections were submitted to ferric chloride reaction (Johansen 1940). Flavonoidic derivatives were tested in sections previously fixed in caffeine-sodium benzoate, and submitted to DMACA (p-dimethylaminocinnamaldehyde) for two hours (Feucht et al. 1986). The slides were mounted in Kaiser’s jelly glycerin, except those for the test of DMACA in which 50% glycerin was used (Johansen 1940). For all histochemical analyses, the blank sections and control treatments were performed.
Root perimeter (P) was determined in four fragments (1cm in length) in ten distinct roots per species. The diameters from each fragment were measured in three distinct positions with the aid of a digital caliper. The values were applied in the formula P=2
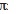 r, where (r) corresponds to the media of the three diameters of each fragment divided per two.
r, where (r) corresponds to the media of the three diameters of each fragment divided per two. Results
D. cogniauxiana plants were sampled on trunks covered by mosses, about 50cm above the soil. E. secundum was located on trunks 2m above the soil at the edge of the nebular forest, in more luminous sites.
Dichaea cogniauxiana: The roots are cylindrical in shape, with approximately 79.1±12.3mm in perimeter (Fig. 2a). The velamen is two-layered, with dead and thin-walled cells (Fig. 2b). Root portions in contact to the substratum surface have absorbent hairs (Fig. 2c). The exodermis has dead cells, outer thinthickened periclinal walls, impregnated with lipidic compounds (Table 1). The passage cells are small, quadrangular in TS, with dense cytoplasm and interpolated by the other exodermal cells (Fig. 2b). The parenchymatic cortex is 14-16 layered, with round cells, reduced in size when adjacent to exodermis and endodermis. Mycorrhizal fungi are scattered all over the cortex, mainly in cell surfaces in contact to the substratum (Fig. 2d). Intact or partially degraded pelotons are interlinked through thin hyphae (Fig. 2e). Idioblasts with raphids immersed in pectic matrix (Fig. 2f, Table 1) were detected in cortical region. The parenchyma cells may have starch grains (Fig. 2g, Table 1) or lipidic wall impregnations (Fig. 2g, Table 1). The endodermis is U-thickened, impregnated in external periclinal walls and in medium lamellae by lipidic substances (Fig. 2h, Table 1). Periclinal internal and anticlinal walls are lignified (Fig. 2i, Table 1). The passage cells may be isolated or two grouped interpolated to the other endodermal cells. Lignified pericyclic cells are interspersed by two-three layers of thin-walled cells, located towards each of the nine protoxylem poles.
The vascular parenchyma is lignified towards the pith, where little accumulation of starch is detected. The leaves are glabrous and hypostomatic, with the ordinary epidermal cells of the adaxial surface rectangular in FV, parallel to each other, with straight anticlinal walls (Fig. 3a). Tetracytic or anisocytic stomata are located at the same level of the ordinary epidermal cells, and randomly distributed. Small cell wall projections above the stomata pore form suprastomatic chambers. The epidermis is one-layered, with thin cuticle. The chlorophyllous parenchyma is five-seven cell layered, and homogeneous (Fig. 3b). The vascular bundles are collateral, with parenchymatic sheath cells parallel to each other, and with phenolic derivatives (Fig. 3b, c, Table 1). Idioblasts with raphids and lipidic droplets are interspersed to the cells of cortical parenchyma (Fig.3d, e).
Epidendrum secundum: The roots are cylindrical, with 58.4mm2 in perimeter (Fig. 4a), four-five layered velamen with dead cells and conspicuous wall thickenings (Fig. 4b, c). There is a distinct epivelamen with root hairs just in the portions in contact to substratum surface. The cells of the exodermis are dead in maturity, hexagonal in TS, rectangular in LS, with O-thickened lignification (Fig. 4b, c, Table 1). There are some pentagonal and alive passage cells with dense cytoplasm, usually smaller than the other exodermal cells. Some covering cuneiform cells with peculiar wall thickening (Fig. 4b) are placed externally to the passage cells. The cortical parenchyma is six-12 layered with small cells next to exodermis and endodermis. Some mycorrhizal groups are mainly located in the contact surface with the substratum. Pelotons intercommunicate through thin hyphae and are degraded in some cells (Fig. 4d). Idioblasts with conspicuous wall thickening, starry-shaped in TS (Fig. 4e), and with positively reaction to lipids occur (Table 1). Few starch grains and raphids are immersed in pectic matrix inside some cortical cells (Table 1). The endodermis is O-thickened, with square-shaped cells in TS (Fig. 4e, g) and rectangular in LS. Solitary passage cells are interspersed to ordinary endodermal cells (Fig. 4g). The pericycle is lignified with twothree thin-walled cells with dense cytoplasm (Fig. 4g, Table 1) facing the nine protoxylem poles (Fig. 4f). Vascular parenchyma cells are lignified towards the pith, where starch grains accumulate (Table 1).
The leaves are glabrous and succulent (Fig. 5a). The epidermis in adaxial surface has polyedric ordinary cells parallel to each other. On the abaxial surface, tetracytic stomata are placed in straight strips at the same level of the ordinary square-shaped epidermal cells which present straight anticlinal walls. The cuticle projections above the stomata pore form suprastomatic chambers (Fig. 5b). Interspersed to the strips of stomata, two-three layers of longer rectangular cells can occur. The epidermis is one-layered, with thick cuticle. The chlorophyllous parenchyma is homogeneous, and 12-layered. Fiber bundles are disposed in subepidermal position, interpolated to collateral vascular bunches (Fig. 5a, c), and surrounded by lignin-poor walled cells. Below the phloematic portion, a cap of fibers is observed. Cells with reticular-thickened walls form the bundle sheath (Fig. 5d). Adjacent to the abaxial epidermal surface, there are five subepidermal layers with discontinuous bundles of secondary spiral-thickened cells. The mesophyll presents cells with slightly lignified walls, and idioblasts. The idioblasts have slight or strong thickened walls and raphids immersed in pectic matrix (Fig. 5e, Table 1). Starch grains and lipidic droplets occur in the chlorophyllian parenchyma (Fig. 5f, Table 1).
Neither alkaloids nor flavonoidic derivatives were detected in D. cougniauxiana and in E. secundum (Table 1).
Discussion
The epiphytic environment is considered arid for the difficulty in acquisition of water by the plants (Fahn & Cutler 1992). So, within the set of features registered for the roots of D. cogniauxiana and E. secundum, we can depict developed velamen, distinct exodermis and endodermis, and specialized thick walled cortical cells as characteristic of epiphytic plants. The velamen is observed in both species, but it is differentiated in endovelamen and epivelamen just in E. secundum. As well as in many Orchidaceae species described by Sanford & Adanlawo (1973), the epivelamen cells are pentagonal in TS, and more uniform in size and shape than the other cells of the velamen. The velamen in E. secundum is two-three cell layers thicker than that of D. cogniauxiana. Dycus & Knudson (1957) and Sanford & Adanlawo (1973) believed that this tissue is just capable to absorbing a small amount of water, and that its main function would be the mechanical protection and prevention against the loss of water. Moreover, Benzing et al. (1982) affirm that the velamen reduces root transpiration, and acts as a sponge, allowing the root to mobilize water and mineral salts. The highest number of velamen layers in E. secundum can, then, be related either to the effectiveness in water absorption or to the reduction in transpiration, which is in agreement with its exposition to high light intensities and humidity fluctuations, and may confer adaptability to its microenvironment. Therefore, the smallest number of velamen layers in D. cogniauxiana could permit the larger entrance of light in root cortex, exciting the chlorophyll in cortex chloroplasts, and making photosynthesis. Although it has been demonstrated that orchid root photosynthesis does not guarantee the formation of new tissues (Dycus & Knudson 1957, Erickson 1957), the photosynthetic electron flow may increase oxygen availability, avoiding hypoxia conditions in tissues with low gas exchange (Pfanz et al. 2002, Lemos-Filho & Isaias 2004, Moreira et al. 2009).
In both species, root hairs are exclusively differentiated in the portion of root surface in contact to the host tree trunk, whereas the flattened velamen cells occupy the whole organ perimeter. Oliveira & Sajo (1999a) attribute to the root hairs associated to the last layer of the velamen, the function of increasing the surface of the root for water absorption, which seems reasonable for the plants herein studied. Moreover, the cells of the velamen can develop irregular secondary thickenings capable to promoting mechanical support (Noel 1974). These thicknesses are considered an important variation of velamen, with relevant taxonomic value (Porembski & Barthlott 1988).
The covering cells associated to passage cells of the exodermis in E. secundum had already been described by Oliveira & Sajo (1999a), and previously observed in other orchids by Engard (1944). These authors attribute to the covering cells the function of aiding condensation of steam of water, and other gases. The covering cells and the properties of the prominent wall thickenings of velamen in E. secundum may represent an important gain for the epiphytic colonization of areas with more intense light.
Thickened exodermis seems to have an important role for the conquest of the epiphytic habit, and as so it commonly occurs in epiphytic Orchidaceae, Bromeliaceae and Araceae (Fahn 1990, Oliveira & Sajo 1999a, Ma & Peterson 2003, Moreira & Isaias 2008). The cell wall thickenings of the exodermis are conspicuous in E. secundum and inconspicuous in D. cogniauxiana, but both are impregnated with lipidic substances. Similarly to the velamen, the exodermal thickenings aid in reduction of water loss by root transpiration (Benzing et al. 1983), turning into an important apoplastic barrier (Hose et al. 2001, Ma & Peterson 2003). This way, the largest cell wall thickening of E. secundum exodermis can confer more efficient protection against the excess of transpiration at the border of the nebular forest where this species occur.
Some authors believe that passage cells correspond to the main entrance of water and mineral salts in the cortex (Benzing et al. 1982, 1983). However, a secondary function has been attributed to them, namely, the emission of chemical signals for attraction of endomycorrhizal fungi, allowing them to cross root cortex (Senthilkumar et al. 2000a, Peterson & Enstone 1996). The hyphae reach and stay in parenchymatic cells, where pelotons formation begins. The interaction between orchids and endocellular fungi suggests that this may be a secondary mechanism of nutrition (Lesica & Antibus 1990, Senthilkumar & Krishnamurthy 1998, Senthilkumar et al. 2000b) and acquisition of carbon (Benzing et al. 1983) by the host plant. The mycorrhizal colonization can vary with endophytic specificity, seasonal changes and mechanisms of defense by the host organ. Abiotic factors such as water and availability of nutrients can also affect the infection (Senthilkumar et al. 2000b). However, no differences were observed in mycorrhizal colonization between both studied species, even with the peculiar microclimates that they are inserted.
Crystalliferous idioblasts are common in monocotyledons (Prychid & Rudall 1999) and can assume several functions, such as storage of calcium and oxalic acid, plant defense, mechanical support, ionic changing, osmotic control and regulation of the levels of calcium in cells of the phloem (Franceschi & Horner 1980, Paiva & Machado 2005). In both species, the raphids are submerged in a mucilaginous matrix with pectic droplets. According to Smith (1923), the mucilage helps to preventing cell lesions caused by their sharp shape.
The peculiarities of the exodermis and the endodermis are important environmentaldependent features of the roots. After acquiring water by the velamen, the exodermal cells do contribute to their channeling through cortex. However, when cell water contents fall down, the thickenings of the parenchyma cell walls prevent the organ to collapse. Reticulate thickenings in cells adjacent to the endodermis were observed in roots and leaves of D. cogniauxiana and E. secundum, and also help in substances translocation for the vascular cylinder (Stern & Judd 2001). The endodermis presents secondary U-thickening in D. cogniauxiana and O-thickening in E. secundum. These two thickenings may begin with suberin deposition, followed by cellulose and lignin impregnation (Ma & Peterson 2003). Passage cells are usually aligned with protoxylem poles (Fleet 1961, Peterson & Enstone 1996), so the flow of water through them is more intense and efficient than through the other cells.
In the studied specimens of E. secundum, nine protoxylem poles were observed, different from the description of Oliveira & Sajo (1999a), that registered 17 protoxylem poles for this species. The possible variation in the number of protoxylem poles in roots of the same species is pointed by Fahn (1990), and may depend on the environmental conditions.
The leaves of E. secundum showed a thicker cuticle and a higher number of stomata per area than those of D. cogniauxiana. These characteristics are coherent with the distinct pattern of exhibition to sun light of both species. Leaves exposed to highest luminous incidence tend to exhibit thicker cuticle, whereas many stomata increases gas exchanges efficiency (Fahn 1990, Valladares & Niinemets 2008). The cuticle is a mechanically important structure, whose properties are dynamically modified by the plant in response to internal and external stimuli (Bagel et al. 2006). In the studied Orchidaceae, the cutin deposition forms suprastomatic chambers, which probably maintain a small compartment of humidity above stomata ostiole, reducing the transpiration. These chambers are common in epiphytic orchids that are subjected to high temperatures and scarce water availability (Rosso 1966). Moreover, the leaves of E. secundum are succulent and show CAM, presenting stomata that are kept open during the night period when transpiration is smaller (Winter et al. 1983).
The many types of wall thickening in mesophyll cells, such as the spiral wall thickenings along the mesophyll cells, and the groups of lignified cells adjacent to the epidermis, are typical features of plants capable to surviving in arid atmospheres. They may be related to the retention of water, mechanical support, and avoidance of cell collapse during desiccation, as pointed out by several authors (Olatunji & Nengim 1980, Pridgeon 1982, Koller & Rost 1988).
Differently of E. secundum, the secondary walls of the cells of the bundle sheath in D. cogniauxiana react positively to phenolic derivatives and negatively to lignin. It can be attributed to the polymeric phenolic constituents of lignin deposited in walls of the perivascular fibers, as described by Campbell & Sederoff (1996). Besides, lignin in monocotyledons, as well as in ferns and gymnosperms, maintain certain proportion of p-hidroxifenil groups, a proportion that falls at despicable levels in dicotyledons (Gottlieb et al. 1996). Therefore, the different lignin constitution of some monocotyledons can reduce their detection for the acidified phloroglucinol (Johansen 1940).
The positive reaction to Sudan tests observed in cell walls of endodermis and exodermis is probably suberin, commonly found in other species of orchids. Lipidic droplets in parenchyma cells of the mesophyll can serve as energy reserve or even as precursors of important components for plant metabolism (Buchanan et al. 2000). Both localized reactions can mean important features either for selective transport in epiphytic environment or storage to be used after inhospitable conditions. In the same way, the accumulation of starch grains in cortical and pith cells of the roots in both species, as well as in the parenchyma cells of the mesophyll of E. secundum, is coherent with the pattern of accumulation described for roots and leaves of Orchidaceae (Zotz 1999). Moreover, in leaves of CAM plants, the dynamics of carbohydrates is essential. The substrate of three carbons for PEP-carboxylase, the phosphenolpiruvate, is formed from glucose, polysaccharides and soluble sugars stored in this organ (Dodd et al. 2002).
The epiphytic environment implies in some structural adaptive convergences for nutrient and water acquisition. In D. cogniauxiana and E. secundum, we depict developed velamen, distinct exodermis and endodermis, and specialized thick walled cortical cells as characteristic of epiphytic plants. After acquiring water by the velamen, the exodermis and endodermal cells do contribute to their channeling through cortex. When the cell water contents fall down, the wall thickenings of the parenchymatic cells prevent the organ to collapse. In D. congniauxiana, the two-layered velamen permits light entrance and photosynthetic process, helping to prevent hypoxia of the roots which are under shade conditions. On the other hand, the leaves of E. secundum showed all structural apparatus that propitiate CAM metabolism.
Acknowledgments
J.P. Lemos-Filho and R.M.S. Isaias thank CNPq for scholarships and grants.
References
Alves, R.J.V. 1990. The Orchidaceae of Itacolomi State Park in Minas Gerais, Brazil. Acta Bot. Bras. 4: 65-72. [ Links ]
Bargel, H., K. Koch, Z. Cerman & C. Neinhuis. 2006. Structure-function relationships of the plant cuticle and cuticular waxes-a smart material? Func. Plant Biol. 33: 893-910. [ Links ]
Barros, F. 2004. Distribuição geográfica de orquídeas do Planalto Central do Brasil. In F. Barros & G.B. Kerbauy (eds.). Orquidologia sul-americana: uma compilação científica. Secretaria do Meio Ambiente, São Paulo, São Paulo, Brazil. [ Links ]
Benzing, D.H., D.W. Ott & W.E. Friedman. 1982. Roots of Sobralia macrantha (Orchidaceae): structure and function of the velanmen-exodermis complex. Am. J. Bot. 4: 608-614. [ Links ]
Benzing, D.H., W.E. Friedman, G. Peterson & A. Renfrow. 1983. Shootlessness, velamentous roots, and the pre-eminence of Orchidaceae in the epiphytic biotipe. Am. J. Bot. 70: 121-133. [ Links ]
Benzing, G.P. 1987. Vascular epiphytism: taxonomic participation and adaptive diversity. Ann. Miss. Bot. Gard. 74: 183-204. [ Links ]
Berlyn, G.P. & J.P. Miksche. 1976. Botanical microtechnique and cytochemistry. The Iowa State University, Ames, Iowa, USA. [ Links ]
Buchanan, B.B., W. Gruisúem & R.L. Jones. 2000. Biochemistry and molecular biology of plants. American Society of Plant Physiologist, Rockville, USA. [ Links ]
Campbell, M.M. & R.R. Sederoff. 1996. Variation in lignin content and composition. Plant Physiol. 110: 3-13. [ Links ]
Cockburn, W., C.J. Goh & P.N. Avadhani. 1985. Photosynthetic carbon assimilation in a shootless orchid, Chiloschista usneoides. Plant Physiol. 77: 83-86. [ Links ]
Cushman, J.C. 2001. Crassulacean Acid Metabolism. A plastic photosynthetic adaptation to arid environments. Plant Physiol. 127: 1439-1448. [ Links ]
Dodd, A.N., A.M. Morland, R.P. Haslam, H. Griffiths & K. Maxwell. 2002. Crassulacean acid metabolism: plastic, fantastic. J. Exp. Bot. 53: 569-580. [ Links ]
Dycus, A.M. & L. Knudson. 1957. The role of the velamen of the aerial roots of orchids. Bot. Gaz. 119: 78-87. [ Links ]
Engard, C.J. 1944. Morphological identity of the velamen and exodermis in orchids. Bot. Gaz. 105: 457-462. [ Links ]
Erickson, L.C. 1957. Respiration and photosynthesis in Cattleya roots. Am. Orch. Soc. Bull. 26: 401-402. [ Links ]
Fahn, A. 1990. Plant Anatomy. Pergamon, Oxford, England. [ Links ]
Fahn, A. & D.F. Cutler. 1992. Xerophytes. Gebrüder Borntraeger, Berlin, Germany. [ Links ]
Feucht, W., P.P.S. Schmidt & E. Christ. 1986. Distribution of flavonols in meristematic and mature tissues of Prunus avium shoots. J. Plant Physiol. 125: 1-8. [ Links ]
Franceschi, V.R. & H.T. Horner. 1980. Calcium oxalate crystals in plants. Bot. Rev. 46: 361-427. [ Links ]
Fleet, D.S.V. 1961. Histochemical and function of the endodermis. Bot. Rev. 27: 165-220. [ Links ]
Gahan, P.B. 1984. Plant histochemistry and cytochemistry: an introduction. Academic, New York, USA. [ Links ]
Gottlieb, O.R., M.A.C. Kaplan & M.R.M.B. Borin. 1996. Biodiversidade: um enfoque químico-biológico. Editora da Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brazil. [ Links ]
Griffiths, H., Ü. Lüttge, K.H. Stimmel, C.E. Crook, N.M. Griffiths & J.A.C. Smith. 1986. Comparative ecophysiology of CAM and C3 bromeliads. III. Environmental influences on CO2 assimilation and transpiration. Plant Cell Environ. 9: 385-393. [ Links ]
Holtzmeier, M.A., W.L. Stern & W.S. Judd. 1998. Comparative anatomy and systematics of Senghas’s cushion species of Maxillaria (Orchidaceae). Bot. J. Lin. Soc. 127: 43-82. [ Links ]
Hose, E., D.T. Clarkson, E. Steudle, L. Schreiber & W. Hartung. 2001. The exodermis: a variable apoplastic barrier. J. Exp. Bot. 52: 2245-2264. [ Links ]
Jensen, W.A. 1962. Botanical Histochemistry, Principles and Practice. W. H. Freeman, San Francisco, USA. [ Links ]
Jiménez, M.S. & D. Morales. 1987. Kleinia neriifolia –a facultative CAM plant. Photosynthetica 21: 329-332. [ Links ]
Johansen, D.A. 1940. Plant microtechnique. McGraw Hill Book Co, New York, USA. [ Links ]
Koller, A.L. & T.L. Rost. 1988. Structural analysis of water-storage tissue in leaves of Sansevieria (Agavaceae). Bot. Gaz. 149: 260-274. [ Links ]
Lemos-Filho, J.P. & R.M.S. Isaias 2004. Comparative stomatal conductance and chlorophyll a fluorescence in leaves vs. fruits of the Cerrado legume tree, Dalbergia miscolobium. Braz. J. Plant Physiol. 16: 89-93. [ Links ]
Lesica, P. & R.K. Antibus. 1990. The occurrence of mycorrhizae in vascular epiphytes of two Costan Rican rain forests. Biotropica 22: 250-258. [ Links ]
Lüttge, U. 2004. Ecophysiology of crassulacean acid metabolism (CAM). Ann. Bot. 93: 629-652 [ Links ]
Ma, F. & C.A. Peterson. 2003. Current insights into the development, structure and chemistry of the endodermis and exodermis of roots. Can. J. Bot. 81: 405-421. [ Links ]
Moreira, A.S.F.P. & R.M.S. Isaias. 2008. Comparative anatomy of the absorption roots of terrestrial and epiphytic orchids. Braz. Archiv. Biol. Technol. 5: 83-93. [ Links ]
Moreira, A.S.F.P., J.P. Lemos-Filho, G. Zotz & R.M.S. Isaias. 2009. Anatomy and photosynthetic parameters of roots and leaves of two shade-adapted orchids, Dichaea cogniauxiana Shltr. and Epidendrum secundum Jacq. Flora 204: 604-611. [ Links ]
Noel, A.R.A. 1974. Aspects of cell wall structure and the development of the velamen in Ansellia gigantean Reichb. f. Ann. Bot. 38: 495-504. [ Links ]
O’Brien, T.P. & M.E. McCully. 1981. The study of plant. Structure: principles and selected methods. Termarcarphi Pty, Melbourne, Australia. [ Links ]
Olatunji, O.A. & R.O. Nengim. 1980. Occurrence and distribution of tracheoidal elements in the Orchidaceae. Bot. J. Linn. Soc. 80: 357-370. [ Links ]
Oliveira, V.D.C. & M.G. Sajo. 1999a. Root anatomy of nine Orchidaceae species. Braz. Arch. Biol. Techn. 42: 405-413. [ Links ]
Oliveira, V.D.C. & M.S. Sajo. 1999b. Anatomía foliar de espécies epífitas de Orchidaceae. Rev. Bras. Bot. 22: 365-374. [ Links ]
Paiva, E.A.S. & S.R. Machado. 2005. Role of intermediary cells in Peltodon radicans (Lamiaceae) in the transfer of calcium and formation of calcium oxalate crystals. Braz. Arch. Biol. Techn. 48: 147-153. [ Links ]
Peterson, C.A. & D.E. Enstone. 1996. Functions of passage cells in the endodermis and exodermis of roots. Physiol. Plantarum 97: 592-598. [ Links ]
Pfanz, H., G. Aschan, R. Langenfeld-Heyser, C. Wittmann & M. Loose. 2002. Ecology and ecophysiology of tree stems: corticular and wood photosynthesis. Naturwissenschaften 89: 147-162. [ Links ]
Porembski, S. & W. Barthlott 1988. Velamen radicum micromorphology and classification of Orchidaceae. Nord. J. Bot. 8: 117-137. [ Links ]
Pridgeon, A.M. 1982. Diagnostic anatomical characters in the Pleurothallidinae (Orchidaceae). Am. J. Bot. 69: 921-938. [ Links ]
Prychid, C.A. & P.J. Rudall. 1999. Calcium oxalate crystals in monocotyledons: a review of their structure and systematics. Ann. Bot. 84: 725-739. [ Links ]
Rosso, S.W. 1966. The vegetative anatomy of the Cypripedioideae (Orchidaceae). Bot. J. Linn. Soc. 59: 309-341. [ Links ]
Sanford, W.W. & I. Adanlawo. 1973. Velamen and exodermis characters of West African epiphytic orchids in relation to taxonomic grouping and habit tolerance. Bot. J. Lin. Soc. 66: 307-321. [ Links ]
Senthilkumar, S. & K.V. Krishnamurthy. 1998. A cytochemical study on the mycorrhizae of Spathoglotttis plicata. Biol. Plantarum 41: 111-119. [ Links ]
Senthilkumar, S., K.V. Krishnamurthy, S.J. Britto & D.I. Arockiasamy. 2000a. Visualization of orchid mycorrhizal fungal structures with fluorescence dye using epifluorescence microscopy. Curr. Scienc. 76: 1527-1528. [ Links ]
Senthilkumar, S., S.J. Britto, K.V. Krishnamurthy & C. Hariharam. 2000b. Biochemical analysis of mycorrhizal roots of Aerides maculosum. Phytomorphol. 50: 273-279. [ Links ]
Smith, E.L. 1923. The histology of certain orchids with reference to mucilage secretion and crystal formation. Bull. Torr. Bot. Club 50: 1-17. [ Links ]
Stern, W.L. & W.M. Whitten. 1999. Comparative vegetative anatomy of Stanhopeinae (Orchidaceae). Bot. J. Linn. Soc. 129: 87-103. [ Links ]
Stern, W.L. & W.S. Judd. 2001. Comparative anatomy and systematics of Catasetinae (Orchidaceae). Bot. J. Linn. Soc. 136: 153-178. [ Links ]
Valladares, F. & Ü. Niinemets. 2008. Shade tolerance, a key plant trait of complex nature and consequences. Ann. Rev. Ecol. Syst. Evol. 39: 237-257. [ Links ]
Winter, K., B.J. Wallace, G.C. Atocker & Z. Roksandic. 1983. Crassulacean Acid Metabolism in Australian vascular epiphytes and some related species. Oecologia 57: 129-141. [ Links ]
Zotz, G. 1999. What are Backshoots good for? Seasonal changes in mineral, carbohydrate and water content of different organs of the epiphytic orchid, Dimerandra emarginata. Ann. Bot. 84: 791-798. [ Links ]
Zotz, G. 2004. How prevalent is crassulacean acid metabolism among vascular epiphytes? Oecologia 138: 184-192. [ Links ]
Bargel, H., K. Koch, Z. Cerman & C. Neinhuis. 2006. Structure-function relationships of the plant cuticle and cuticular waxes-a smart material? Func. Plant Biol. 33: 893-910. [ Links ]
Barros, F. 2004. Distribuição geográfica de orquídeas do Planalto Central do Brasil. In F. Barros & G.B. Kerbauy (eds.). Orquidologia sul-americana: uma compilação científica. Secretaria do Meio Ambiente, São Paulo, São Paulo, Brazil. [ Links ]
Benzing, D.H., D.W. Ott & W.E. Friedman. 1982. Roots of Sobralia macrantha (Orchidaceae): structure and function of the velanmen-exodermis complex. Am. J. Bot. 4: 608-614. [ Links ]
Benzing, D.H., W.E. Friedman, G. Peterson & A. Renfrow. 1983. Shootlessness, velamentous roots, and the pre-eminence of Orchidaceae in the epiphytic biotipe. Am. J. Bot. 70: 121-133. [ Links ]
Benzing, G.P. 1987. Vascular epiphytism: taxonomic participation and adaptive diversity. Ann. Miss. Bot. Gard. 74: 183-204. [ Links ]
Berlyn, G.P. & J.P. Miksche. 1976. Botanical microtechnique and cytochemistry. The Iowa State University, Ames, Iowa, USA. [ Links ]
Buchanan, B.B., W. Gruisúem & R.L. Jones. 2000. Biochemistry and molecular biology of plants. American Society of Plant Physiologist, Rockville, USA. [ Links ]
Campbell, M.M. & R.R. Sederoff. 1996. Variation in lignin content and composition. Plant Physiol. 110: 3-13. [ Links ]
Cockburn, W., C.J. Goh & P.N. Avadhani. 1985. Photosynthetic carbon assimilation in a shootless orchid, Chiloschista usneoides. Plant Physiol. 77: 83-86. [ Links ]
Cushman, J.C. 2001. Crassulacean Acid Metabolism. A plastic photosynthetic adaptation to arid environments. Plant Physiol. 127: 1439-1448. [ Links ]
Dodd, A.N., A.M. Morland, R.P. Haslam, H. Griffiths & K. Maxwell. 2002. Crassulacean acid metabolism: plastic, fantastic. J. Exp. Bot. 53: 569-580. [ Links ]
Dycus, A.M. & L. Knudson. 1957. The role of the velamen of the aerial roots of orchids. Bot. Gaz. 119: 78-87. [ Links ]
Engard, C.J. 1944. Morphological identity of the velamen and exodermis in orchids. Bot. Gaz. 105: 457-462. [ Links ]
Erickson, L.C. 1957. Respiration and photosynthesis in Cattleya roots. Am. Orch. Soc. Bull. 26: 401-402. [ Links ]
Fahn, A. 1990. Plant Anatomy. Pergamon, Oxford, England. [ Links ]
Fahn, A. & D.F. Cutler. 1992. Xerophytes. Gebrüder Borntraeger, Berlin, Germany. [ Links ]
Feucht, W., P.P.S. Schmidt & E. Christ. 1986. Distribution of flavonols in meristematic and mature tissues of Prunus avium shoots. J. Plant Physiol. 125: 1-8. [ Links ]
Franceschi, V.R. & H.T. Horner. 1980. Calcium oxalate crystals in plants. Bot. Rev. 46: 361-427. [ Links ]
Fleet, D.S.V. 1961. Histochemical and function of the endodermis. Bot. Rev. 27: 165-220. [ Links ]
Gahan, P.B. 1984. Plant histochemistry and cytochemistry: an introduction. Academic, New York, USA. [ Links ]
Gottlieb, O.R., M.A.C. Kaplan & M.R.M.B. Borin. 1996. Biodiversidade: um enfoque químico-biológico. Editora da Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brazil. [ Links ]
Griffiths, H., Ü. Lüttge, K.H. Stimmel, C.E. Crook, N.M. Griffiths & J.A.C. Smith. 1986. Comparative ecophysiology of CAM and C3 bromeliads. III. Environmental influences on CO2 assimilation and transpiration. Plant Cell Environ. 9: 385-393. [ Links ]
Holtzmeier, M.A., W.L. Stern & W.S. Judd. 1998. Comparative anatomy and systematics of Senghas’s cushion species of Maxillaria (Orchidaceae). Bot. J. Lin. Soc. 127: 43-82. [ Links ]
Hose, E., D.T. Clarkson, E. Steudle, L. Schreiber & W. Hartung. 2001. The exodermis: a variable apoplastic barrier. J. Exp. Bot. 52: 2245-2264. [ Links ]
Jensen, W.A. 1962. Botanical Histochemistry, Principles and Practice. W. H. Freeman, San Francisco, USA. [ Links ]
Jiménez, M.S. & D. Morales. 1987. Kleinia neriifolia –a facultative CAM plant. Photosynthetica 21: 329-332. [ Links ]
Johansen, D.A. 1940. Plant microtechnique. McGraw Hill Book Co, New York, USA. [ Links ]
Koller, A.L. & T.L. Rost. 1988. Structural analysis of water-storage tissue in leaves of Sansevieria (Agavaceae). Bot. Gaz. 149: 260-274. [ Links ]
Lemos-Filho, J.P. & R.M.S. Isaias 2004. Comparative stomatal conductance and chlorophyll a fluorescence in leaves vs. fruits of the Cerrado legume tree, Dalbergia miscolobium. Braz. J. Plant Physiol. 16: 89-93. [ Links ]
Lesica, P. & R.K. Antibus. 1990. The occurrence of mycorrhizae in vascular epiphytes of two Costan Rican rain forests. Biotropica 22: 250-258. [ Links ]
Lüttge, U. 2004. Ecophysiology of crassulacean acid metabolism (CAM). Ann. Bot. 93: 629-652 [ Links ]
Ma, F. & C.A. Peterson. 2003. Current insights into the development, structure and chemistry of the endodermis and exodermis of roots. Can. J. Bot. 81: 405-421. [ Links ]
Moreira, A.S.F.P. & R.M.S. Isaias. 2008. Comparative anatomy of the absorption roots of terrestrial and epiphytic orchids. Braz. Archiv. Biol. Technol. 5: 83-93. [ Links ]
Moreira, A.S.F.P., J.P. Lemos-Filho, G. Zotz & R.M.S. Isaias. 2009. Anatomy and photosynthetic parameters of roots and leaves of two shade-adapted orchids, Dichaea cogniauxiana Shltr. and Epidendrum secundum Jacq. Flora 204: 604-611. [ Links ]
Noel, A.R.A. 1974. Aspects of cell wall structure and the development of the velamen in Ansellia gigantean Reichb. f. Ann. Bot. 38: 495-504. [ Links ]
O’Brien, T.P. & M.E. McCully. 1981. The study of plant. Structure: principles and selected methods. Termarcarphi Pty, Melbourne, Australia. [ Links ]
Olatunji, O.A. & R.O. Nengim. 1980. Occurrence and distribution of tracheoidal elements in the Orchidaceae. Bot. J. Linn. Soc. 80: 357-370. [ Links ]
Oliveira, V.D.C. & M.G. Sajo. 1999a. Root anatomy of nine Orchidaceae species. Braz. Arch. Biol. Techn. 42: 405-413. [ Links ]
Oliveira, V.D.C. & M.S. Sajo. 1999b. Anatomía foliar de espécies epífitas de Orchidaceae. Rev. Bras. Bot. 22: 365-374. [ Links ]
Paiva, E.A.S. & S.R. Machado. 2005. Role of intermediary cells in Peltodon radicans (Lamiaceae) in the transfer of calcium and formation of calcium oxalate crystals. Braz. Arch. Biol. Techn. 48: 147-153. [ Links ]
Peterson, C.A. & D.E. Enstone. 1996. Functions of passage cells in the endodermis and exodermis of roots. Physiol. Plantarum 97: 592-598. [ Links ]
Pfanz, H., G. Aschan, R. Langenfeld-Heyser, C. Wittmann & M. Loose. 2002. Ecology and ecophysiology of tree stems: corticular and wood photosynthesis. Naturwissenschaften 89: 147-162. [ Links ]
Porembski, S. & W. Barthlott 1988. Velamen radicum micromorphology and classification of Orchidaceae. Nord. J. Bot. 8: 117-137. [ Links ]
Pridgeon, A.M. 1982. Diagnostic anatomical characters in the Pleurothallidinae (Orchidaceae). Am. J. Bot. 69: 921-938. [ Links ]
Prychid, C.A. & P.J. Rudall. 1999. Calcium oxalate crystals in monocotyledons: a review of their structure and systematics. Ann. Bot. 84: 725-739. [ Links ]
Rosso, S.W. 1966. The vegetative anatomy of the Cypripedioideae (Orchidaceae). Bot. J. Linn. Soc. 59: 309-341. [ Links ]
Sanford, W.W. & I. Adanlawo. 1973. Velamen and exodermis characters of West African epiphytic orchids in relation to taxonomic grouping and habit tolerance. Bot. J. Lin. Soc. 66: 307-321. [ Links ]
Senthilkumar, S. & K.V. Krishnamurthy. 1998. A cytochemical study on the mycorrhizae of Spathoglotttis plicata. Biol. Plantarum 41: 111-119. [ Links ]
Senthilkumar, S., K.V. Krishnamurthy, S.J. Britto & D.I. Arockiasamy. 2000a. Visualization of orchid mycorrhizal fungal structures with fluorescence dye using epifluorescence microscopy. Curr. Scienc. 76: 1527-1528. [ Links ]
Senthilkumar, S., S.J. Britto, K.V. Krishnamurthy & C. Hariharam. 2000b. Biochemical analysis of mycorrhizal roots of Aerides maculosum. Phytomorphol. 50: 273-279. [ Links ]
Smith, E.L. 1923. The histology of certain orchids with reference to mucilage secretion and crystal formation. Bull. Torr. Bot. Club 50: 1-17. [ Links ]
Stern, W.L. & W.M. Whitten. 1999. Comparative vegetative anatomy of Stanhopeinae (Orchidaceae). Bot. J. Linn. Soc. 129: 87-103. [ Links ]
Stern, W.L. & W.S. Judd. 2001. Comparative anatomy and systematics of Catasetinae (Orchidaceae). Bot. J. Linn. Soc. 136: 153-178. [ Links ]
Valladares, F. & Ü. Niinemets. 2008. Shade tolerance, a key plant trait of complex nature and consequences. Ann. Rev. Ecol. Syst. Evol. 39: 237-257. [ Links ]
Winter, K., B.J. Wallace, G.C. Atocker & Z. Roksandic. 1983. Crassulacean Acid Metabolism in Australian vascular epiphytes and some related species. Oecologia 57: 129-141. [ Links ]
Zotz, G. 1999. What are Backshoots good for? Seasonal changes in mineral, carbohydrate and water content of different organs of the epiphytic orchid, Dimerandra emarginata. Ann. Bot. 84: 791-798. [ Links ]
Zotz, G. 2004. How prevalent is crassulacean acid metabolism among vascular epiphytes? Oecologia 138: 184-192. [ Links ]
*Correspondencia a:
Ana Sílvia Franco Pinheiro Moreira. Universidade Federal de Uberlândia, Instituto de Biologia. Av. João Naves de Ávila, 2121, CP 593, cep 38400-902. Uberlândia, Minas Gerais, Brazil; anasilviamoreira@gmail.com, anasilvia@inbio.ufu.br
José Pires de Lemos Filho.Universidade Federal de Minas Gerais, Instituto de Ciências Biológicas, Departamento de Botânica. Av. Presidente Carlos Luz, 6627. Pampulha, cep. 31270-901. Belo Horizonte, Minas Gerais, Brazil; lemos@icb.ufmg.br
Rosy Mary dos Santos Isaias. Universidade Federal de Minas Gerais, Instituto de Ciências Biológicas, Departamento de Botânica. Av. Presidente Carlos Luz, 6627. Pampulha, cep. 31270-901. Belo Horizonte, Minas Gerais, Brazil; rosy@icb.ufmg.br
1. Universidade Federal de Uberlândia, Instituto de Biologia. Av. João Naves de Ávila, 2121, CP 593, cep 38400-902. Uberlândia, Minas Gerais, Brazil; anasilviamoreira@gmail.com, anasilvia@inbio.ufu.br
2. Universidade Federal de Minas Gerais, Instituto de Ciências Biológicas, Departamento de Botânica. Av. Presidente Carlos Luz, 6627. Pampulha, cep. 31270-901. Belo Horizonte, Minas Gerais, Brazil; lemos@icb.ufmg.br
3. Universidade Federal de Minas Gerais, Instituto de Ciências Biológicas, Departamento de Botânica. Av. Presidente Carlos Luz, 6627. Pampulha, cep. 31270-901. Belo Horizonte, Minas Gerais, Brazil; rosy@icb.ufmg.br
Received 13-VI-2012. Corrected 06-X-2012. Accepted 06-XI-2012














