Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.60 n.3 San José Sep. 2012
Fishery of the Green Jack Caranx caballus (Osteichytes: Carangidae) in Las Perlas Archipelago, Pacific Panama
*Direccción para correspondencia
Abstract
Green Jacks, Caranx caballus, are distributed along the Eastern Pacific coast. In Panama, C. caballus was heavily fished around Las Perlas Archipelago to sustain local markets until 2007, when the archipelago was declared a marine protected area. This first study in Panama, analyzed a sample of 4 990 individuals from Las Perlas, obtained monthly from June 2005 to June 2006, from landings at the central fish market. Average total length was 36.1±6.4cm and optimum length 38.9cm. Approximately 68% of fish lengths were within ±10% of the optimal length but only six (15%) were considered mega-spawners. The von Bertalanffy parameters describe a long-lived and fast growing species, while mortality rates revealed that C. caballus is under high fishing pressure. Standard length at which half of the population is mature was 38.8cm, and the size at which individuals matured massively, 33cm. Only 10-13% of the fish were immature. C. caballus reproduces two to three times per year, with peaks in December, April, and probably September, and recruits to the population at least twice per year. Catch per unit effort (CPUE) was best predicted by minimum annual values of the Multivariate ENSO/ LNSO Index (MEI) (R2=0.54). Results show that C. caballus in Pacific Panama is overfished. We recommend the raising of the minimum capture/landing size of this species in order to increase the proportion of megaspawners in the population and guarantee the sustainability of this resource.
Key words: Caranx caballus, cojinua, green jack, fishery assessment, Eastern Pacific, Panama.
Resumen
El jurel verde o “cojinúa”, Caranx caballus, se distribuye a lo largo de la costa del Pacífico Oriental. En el Archipiélago de Las Perlas, Panamá, la cojinúa fue pescada sin control para sostener mercados locales hasta el 2007, fecha en la que esta región fue declarada área marina protegida. En este primer estudio para Panamá, se analizaron desde junio de 2005 a junio de 2006, 4 990 individuos en las descargas del Mercado Central de Mariscos provenientes de Las Perlas. La longitud media total de esta especie fue de 36.1±6.4cm y la longitud óptima, 38.9cm. Aproximadamente, 68% de los individuos muestreados poseen longitudes dentro de ±10% de la longitud óptima, pero sólo seis (15%) eran mega-reproductores. Los parámetros de la ecuación de von Bertalanffy describen la especie como de larga vida y de rápido crecimiento, mientras que las tasas de mortalidad indican que la cojinúa está bajo una alta presión de pesca. La longitud estándar a la que la mitad de la población es sexualmente madura es 38.8cm y el tamaño de maduración masiva, 33cm. Sólo entre el 10-13% de los individuos muestreados son inmaduros. La cojinúa se reproduce 2-3 veces al año, con picos en diciembre, abril y probablemente septiembre, y recluta a la población por lo menos dos veces al año. Los valores mínimos anuales del Índice ENSO/LNSO multivariado (MEI) son los mejores predictores de la CPUE de esta especie (R2=0.54). Nuestros resultados demuestran que la cojinúa en el Pacífico Panameño está sobreexplotada por lo que recomendamos elevar la talla mínima de captura para permitir que aumente la proporción de mega-reproductores en la población con el fin de garantizar la sostenibilidad del recurso.
Palabras clave: Caranx caballus, cojinua, jurel verde, evaluación pesquera, Pacífico oriental, Panamá.
Caranx caballus, Günther 1868, is a pelagic species inhabiting coastal waters from Southern California to Chile, the Galapagos, Cocos, Malpelo, the Revillagigedo Islands in the Eastern Pacific Ocean, and Hawaii in the Central Pacific Ocean (Robertson & Allen 2008, Froese & Pauly 2010). Juveniles are found in coastal waters, estuaries (Smith-Vaniz 1995), and swimming close to floating objects in the open ocean, while adults live 30 to 100m deep forming schools over soft bottoms (Robertson & Allen 2008, Froese & Pauly 2010). They feed upon crustaceans, mollusks, chaetognaths, cnidarians and pelagic fish such as engraulids and clupeids, and are prey of sharks, marlins and other large predators (Bernal-Ornelas et al. 2005, Robertson & Allen 2008).
Most catches of Green Jack in the Eastern Pacific result from unregulated, artisanal, and subsistence fisheries. Equipment commonly used includes gill and cast nets, lines, hooks, and spinning tackles deployed from small boats or from land (Gallardo-Cabello et al. 2007). As these catches are consumed locally, fishing records are generally non-existent or incomplete. In small towns, Green Jacks are marketed in salted, dried, and smoked forms, and are often used to produce fishmeal and fish oil (Froese & Pauly 2010).
In Las Perlas Archipelago, Gulf of Panama, Caranx caballus, Cojinúas, or Green Jacks, are fished using seine nets, operated by winches from boats up to 25m long, either known as bolicheros, or deployed by hand from smaller boats during fishing campaigns lasting from 4 to 5 days, and very occasionally 8-9 days. After capture, Green Jacks are kept in ice and transported to Mercado del Marisco, the Fish Market in Panama City, where they are sold fresh to the public.
Their relative high abundance, meat of poor to average quality, and lack of intermediaries between fishermen and consumers, have kept the Green Jack sale prices accessible to people with low incomes, varying from US$0.5-1.1 in 2006 to US$3.1-3.7 in 2010 (ACODECO 2010). Green Jacks are also one of the food items of the “basic shopping basket” (canasta básica, in Spanish), which is used to calculate the overall cost of household and food essentials for a family in Panama.
In recent years, the annual production of Green Jacks in Panama has become a topic of concern to scientists and authorities, as it decreased from 1 436t in 2003 to 477t in 2006. As the total number of fishing boats had increased slowly in the region since 1998 (Anonymous 2010b), these abrupt changes in production were feared to be early signs of overexploitation (Anonymous 2010a). On May 31 2007, the Assembly of Panama approved Law N°18, banning fisheries operating with mechanical gears, such as those carried out on bolicheros, from the waters surrounding Las Perlas Archipelago, and declaring these waters as a Special Management Marine and Coastal Zone.
In 2008, the annual production of Green Jacks recovered to 1 640t. Despite the introduction of Law N°18, which aimed to create management plans for marine natural resources in the archipelago, using the best scientific evidence available, there was in 2010 still very scarce information in the region’s technical literature on Green Jacks, such as growth, reproduction, ecology, and population dynamics. The few studies available were on the populations fished along the shores of Colima, Pacific Mexico (e.g. Cruz-Romero et al. 1993, Gallardo-Cabello et al. 2007).
In this study, analyses were carried out on the length frequency distributions of Green Jacks or C. caballus obtained from the Panama Fish Market from June 2005-June 2006, and these were then used to estimate stable age groups, parameters of the corresponding von Bertalanffy’s model, longevity, and mortality rates. In addition, descriptions are given of their reproductive stages using both external gonadic features and a weight-based gonadic index, and this information is combined with growth data to determine size at the onset of maturity. Finally, the association between the fishing productivity and the effects of the yearly upwelling event and the ENSO/ LNSO in Central and South Eastern Pacific was assessed.
Materials and methods
Study area and samples: Las Perlas Archipelago, Roca Trollope, and Isla Galera, are important fishing grounds in the Gulf of Panama (Cipriani et al. 2008a, b, Guzman et al. 2008). Enclosed within a Marine Special Management Zone, extending 168 771ha, the archipelago has 255 islands (33 153ha) of which Del Rey (8° 22’54.64’’ N-78° 54’20.16’’ W) is the largest (Guzman et al. 2008). During the dry season, between January and April, Northeast trade winds create upwelling along the Gulf of Panama. The warm, nutrient depleted water carried away from the coast by the winds is replaced by cold, nutrient-rich water (15º-20ºC) from the deep, triggering large-scale phytoplankton blooms that boost the archipelago’s productivity (D’Croz et al. 1991). The region also experiences the effects of El Niño (ENSO) and La Niña Southern Oscillation (LNSO) (McNiven 2003, D’Croz & O’Dea 2007).
Between June 2005 and June 2006, 4 990 C. caballus from Mercado del Marisco were obtained: 400 fish were haphazardly selected and bought every month, apart from in June 2005 (200 fish) and May 2006 (790 fish). No fish were available in November 2005 as bad weather grounded the fishing fleet. Samples were analyzed, as explained in next paragraphs, in the Coastal Ecology and Conservation Biology Laboratory at the STRI facility of Naos. Samples from May and June 2006 were strongly biased in size and were excluded from growth, mortality, age structure, and maturity analyses.
Biometrics, growth and mortality: Measurements taken were the standard (LS) and total length (LT) of all individuals using an ichthyometer, and their total wet weight (WT) to the nearest ounce (transformed to grams in our analyses) using a weighing scale (BAICO, 40lb max, Colombia). The relationship between LT and LS was described using a reduced major axis model (RMA) in the form:
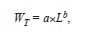
where a and b are regression parameters.
The power law equation
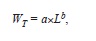
where a and b are regression parameters and L is length, was used to describe the relation between LS and WT and that between LT and WT. The model was fitted to the data using non-linear weighted least-squares using the statistical package R (R Development Core Team 2011). Intervals of 95% confidence (CI) of parameters a and b were obtained by bootstrapping each data set 4 999 times. Finally, we estimated LOPT following Beddington & Cooke (1983) and (1992), 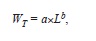
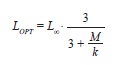
where M is the average natural mortality (0.5 in the current samples), L∞ is the infinite length, k is the curvature of von Bertalanffy’s model (eq. 4). Also estimated was LOPT using the approximation of Froese & Binohlan (2000).
Optimal fisheries should exploit fish within the approximate range LOPT ±10%: smaller fish are too young or immature and larger fish are mega-spawners (Froese 2004).
Length frequency histograms with bins of 1cm were prepared. von Bertalanffy’s models (Sparre & Venema 1998, Essington et al. 2001) in the form:
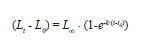
Green Jacks were assumed to start growing at lengths of 0cm (LO=0) at year 0 (tO=0), but rounding-off errors produced small values of tO. Models were fitted using the ELEFAN I procedure implemented in the software program FAO-ICLARM Fish Assessment Tools (FiSAT II, ver. 1.2.2) (Gayanilo et al. 2005). The best value of k was reported from the scan method, L∞ as the ratio of the instantaneous rate of mortality Z and k (Powell 1979, Wetherall 1986), and the two best estimates of L∞ and k from the surface response analysis (Sparre & Venema 1998).
The growth performance index
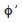 was calculated as:
was calculated as: 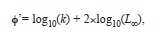
implemented in FiSAT II, ver. 1.2.2 (Gayanilo et al. 2005), and longevity, the time required to achieve 95% of L∞, following:
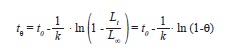
where θ=0.95 (Taylor 1958). All chronological ages were obtained from eq. 6 using parameters from pooled data of both sexes.
The age distribution and growth rate of Green Jacks were assumed to be stable, so Z was approximated using length-converted catch curves and the methods of Jones and van Zalinge (1981), Hoenig (1982), Hoenig & Lawing (1982), Beverton & Holt (1956), and Ault & Erhardt (1991), and Erhardt & Ault (1992). The natural mortality coefficient M was estimated using the Rikhter & Efanov’s (1976) and Taylor’s method (1958, 1960), and Pauly´s empirical formula (1980, 1983, 1984), using 27.4°C as the average surface temperature of the Gulf of Panama. All these methods, apart from Taylor’s, were implemented in FiSAT II, ver. 1.2.2 (Gayanilo et al. 2005). Pauly’s M was multiplied by 0.8 (Pauly 1983) as Green Jacks are a schooling species (Allen & Robertson 1994). The fishing mortality coefficient F, was calculated as F=Z-M and the exploitation ratio as E=F/Z (Sparre & Venema 1998).
Age structure and maturity: The monthly age structure of the stock was estimated using Bhattacharya’s method on histograms of LS. Average values of LS transformed to LT were used to estimate mean age groups using NORMSEP. Both methods were also implemented in FiSAT II, ver. 1.2.2 (Gayanilo et al. 2005).
Green Jacks were sexed by the macroscopic appearance of their gonads. Monthly, assessment was made of the ratio males: females and the length of the smallest mature male and female in the samples, MLS. Fully mature individuals were recognized by the presence of gametes, enlarged gonads, and tissue rich in blood vessels. Testes ready to spawn were whitish and triangular in cross-section and oviducts were enlarged and full of eggs.
Cumulative frequency distributions were used to estimate the LS at the 50th percentile (LS50), average standard length at which half of males and females in the population were mature. Intervals of 95% confidence of LS50 were estimated by randomly sampling 2000 times each dataset. Only the largest CI is reported.
Gonads were weighed to the nearest 0.01g using an ACCULAB weighing scale, model VI-200. The ratio between the gonadic weight (oviducts in females) WG, and WT, times 100, was used as the gonadic index IG. The size at first massive spawning or at first maturity Lm, was that at which the maximum values per month of IG increased abruptly.
The associations between the monthly arcsine- transformed proportions of fully mature stages and IG monthly maxima, and the monthly averages of sea surface temperature (SST, Celsius degrees) and chlorophyll concentrations (mg/m3) from the Gulf of Panama, recorded between 1999 and 2003 (Agujetas 2005), were tested using the Pearson’s product-moment correlation coefficient (Sokal & Rohlf 1995).
Green Jack’s fisheries: Values of the annual production of Green Jack in Panama from 1994-2006 were obtained from Autoridad Marítima de Panamá (Anonymous 1999) and Dirección de Estadística y Censo (Anonymous 2010a). Catch per unit effort (CPUE) was estimated using two different proxies of effort: a) the number of boats of the semi-industrial and artisanal fleets from 1994-2007 (CPUE1), and b) the number of boats of the semi-industrial and artisanal fleets in Panamanian coastal provinces on the Pacific Ocean (i.e. Panamá, Coclé, Herrera, Darién, Veraguas y Chiriquí) from 2000-2004 (CPUE2) (Anonymous 2010b). The scarce data available suggest an increasing number of fishing boats per year, between 1994 and 2007. Missing values within this interval were approximated using linear regression models (CPUE1: boats=391·year+776198, R2=0.98, CPUE2: boats=584·year, R2=0.98).
RMA linear models, such as those in eq.1, were used to describe the association between the annual minima, maxima, and average of the Multivariate ENSO/LNSO Index (MEI) and the CPUE, from December 1994 to December 2007. Only results from the models that best fitted the data were reported. Positive values of MEI were associated with the occurrence of ENSO events, while negative values with the occurrence of LNSO events (Wolter & Timlin 1993, 1998). CPUE was standardized to standardized values, CPUEs, and to their natural logarithms plus a constant 1.5. Bimonthly MEI values were aligned to those of CPUEs by matching month 1-month 2 in the former to month 2 in the latter.
Results
Biometrics, growth and mortality: A total of 2 370 male and 2 620 female C. caballus were examined. The smallest fish in the sample measured LS=12.7cm (LT=17.0cm; 0.9-1.0 years) and the largest LS=45.0cm (56.5cm; 7.8–8.5 years). The average LS in males was 28.5±5.2cm (LT 36.4±6.3cm) and in females 28.1±5.3cm (35.8±6.5cm), while that of the full sample (±1 standard deviation) was LS=28.3±5.3cm (LT =36.1±6.4cm, 2.5-2.8 years, 495g). The average wet weight of males was 548±244g (13.4-1 588g) and that of females was 525±250g (85.0-1 644g).
Regression parameters within sexes and within the pooled sample of both sexes were positive and significant. Models describing the linear associations of LS and LT (Fig. 1A) explained a large proportion of the data variance (R2=97%, Table 1). Parameter differences between sexes in models describing the non-linear associations of LS and WT (Fig. 1B, Table 1) were significant. However, those obtained from LT and WT (Fig. 1C, Table 1), were not.
Values of LOPT±10% from eq. 3 were 31.8±3.2cm for males and 32.6±3.3cm for females. Estimates of LOPT±10% in the pooled data of both sexes were 32.2±3.2cm in the LS data set, and 38.9±3.9cm in the LT data set (Fig. 2A,B, Table 2). Values of LOPT from Froese & Binholan (2000) were 29.1cm for males, 30.8cm for females, 29.8cm for the pooled sample of the LS data set, and 37.4cm for the pooled sample of the LT data set. Ranges of LOPT of male and female data were similar to that of the pooled sample, so only the latter were considered for further analyses. According to the LS data set, 68% of all the fish in the sample had lengths within the corresponding optimal ranges, and 6% were mega-spawners (Fig. 2A). In the LT data set, corresponding estimates were 68% and 15% (Fig. 2B).
Parameters of the von Bertalanffy models, growth performance indexes, and estimates of longevity are shown in table 2, while plots of the growth curves are shown in figure 3. Three independent estimations of L∞ within sex groups were all similar, and 2.6 to 4.0cm larger in females than in males. Similarities were also found between the first two best estimates of k, 0.33 to 0.36/year from LS data, and 0.32/year from LT data. Differences between tO and 0 were almost negligible in the model fitted to values of LS, as they represented intervals not larger than 13 days. However, differences between tO and 0 were as high as 23.3 days in the model fitted to values of LT.
Corresponding estimates of LO were 0.55 to 1.22cm (Table 2).
The growth performance index, a linear function of the logarithmic transformations of L∞ and k (eq. 5), was similar between all sex groups within the LS data set (2.89-2.92/year) and slightly smaller than the performance index obtained from the LT data set (3.05/year). Estimates of longevity were also very similar between data sets and sex groups, and ranged between 8.3-9.4 years (Table 2).
Estimates of Z from length-converted catch curves were 2.0-.9/year in LS and LT data sets, respectively. Those obtained from the Jones and van Zalinge’s method (1981), 2.0 and 1.8/year, closely matched them. However, the Hoenig’s method (1982, Hoenig & Lawing 1982) produced values of Z equal to 1.0 and 1.1/year, respectively, as low as half the previous estimates. Those from the Beverton & Holt’s (1956), and Ault & Erhardt’s methods (1991, Erhardt & Ault 1992) were between 0.1-0.2 units off from Hoenig’s (1982, Hoenig & Lawing 1982) estimates (Table 3).
The Rikhter & Efanov’s (1976) method produced estimates of M=0.7/year in the LS data set (Lm=26.5cm; 2.3 years) and M = 0.6/ year in the LT data set (33cm, 2.5 years). Taylor’s (1958, 1960) estimates of M were 0.3/year in all data sets, and Pauly’s (1980, 1983, 1984) were 0.6/year (at 27.4°C) (Table 3).
In the LS data set, estimates of F obtained from the catch curve mortality ranged between 1.3-1.7/year, while those from Hoening’s method (1982, Hoenig & Lawing 1982) ranged between 0.5-0.8/year. These values of F were similar to those in the LT data set, as they ranged from 1.3-1.6/year and from 0.4-0.7/year,-respectively. High values of F matched high values of E and ranged between 0.4-0.8 (Table 3).
Age structure and maturity: Bhattacharya’s method suggests the presence of eight age groups from the Green Jack sample (A through H, in LS) (Table 4). Groups A - E were also found by NORMSEP (in LT), but NORMSEP pooled Bhattacahrya’s F and G into a single group, and was unable to discriminate group H. Chronological ages in groups A through E in both analyses were different by no more than 2.5 months (i.e. maximum difference found in Group E) (Table 4).
Male to female ratios ranged from 1:1.64 in July to 1:0.77 in May, averaging 1:1.28±0.98 per month during the sampling period. The sex ratio of the full sample was 1:1.1. The smallest mature male found was MLS=18.5cm long (1.4 years, 146g) and the smallest mature female MLS=16.8cm (1.2 years, 111g). The average standard lengths at which half of the population was mature in males and females were very similar, LS50=30.5±0.1cm (LT50=38.8cm, 2.9-3.2 years) and LS50=30.2±0.1cm (38.7cm, 2.8-3.2 years) respectively (Fig. 4A,B).
The distribution of IG maxima in relation to LS was similar in both sexes. Low values of IG maxima (1 or 2) were common among fish as large as LS=26cm. IG maxima increased abruptly, up to values of 9 and 10 in males and females attaining LS=Lm=26.5cm and larger, and LT=Lm=33.0cm and larger (2.3–2.5 years).
In fish larger than LS=32.0cm (LT=43.0cm), IG maxima decreased gradually to four in fish of LS=41cm (LT=50.0cm) and larger (Fig. 4C,D). The age at first maturity of the pooled sample was Lm=26.8cm in the LS data set (20.0-35.9cm), and Lm=32.6cm in the LT data set (24.3-43.7cm). Of all the fish in the sample, 10-13% were immature or smaller than the estimates of Lm (Fig. 4C,D).
The reproduction of Green Jacks is associated with the upwelling events that annually occur in the Gulf of Panama. Monthly SST averages were inversely correlated to monthly IG maxima (Pearson’s r=-0.67, p≤0.018 males; -0.80, p≤0.002 females) and to proportions of mature individuals (0.74, p≤0.006 males; -0.72, p≤0.009 females). Monthly chlorophyll surface concentrations were directly correlated to IG maxima (0.77, p≤0.001 males; 0.918, p≤0.003 females) and to the proportion of mature individuals (0.91, p≤0.001 males; 0.88, p≤0.001 females). The largest values of IG and mature individuals occurred approximately three months before the local upwelling reached its maximum, a time when the lowest values of SST and the highest concentrations of chlorophyll occurred (Fig. 5A,B,C).
IG maxima and the proportion of mature individuals changed synchronously in relation to environmental variables (Fig. 5A,B,C). Male and female annual values in IG maxima peaked in December for males (10.5), in January for females (10.6), in April (6.9 males: 5.4 females), and in September (5.0 males: 4.3 females). Notable values of IG maxima (>0.25) also occurred in January, February, August, and October (Fig. 5B). The highest proportion of individuals with fully mature gonads occurred in December 2005 (0.98 males: 0.88 females) and April 2006 (0.42:0.36) and other high values (>0.30) occurred also in January and February. Mature individuals (1-25% of the current sample) were found every month, and the monthly proportion of mature males and females was approximately constant during the sampling period (Fig. 5C).
Green Jack’s fisheries: The CPUE of Green Jacks was significantly associated to MEI. Standardized values of CPUE1s and their natural logarithms were best described by the minimum annual values of MEI (CPUE1s= [1.17±0.85] + MEI ∙[2.34±1.16], R2=0.54; Ln [ CPUE1s + 1.5] = [1.02±0.28] + MEI ∙ [1.65±0.62], R2=0.54) (Fig. 5D,E,F). Models using MEI minimum annual values to predict CPUE2s and those using MEI annual averages to predict CPUE1s, explained poorly the variability of the data sets (R2<0.31).
Discussion
Within its range of distribution, the Green Jack is one of the most abundant pelagic fish of commercial interest in the Central Eastern Pacific. From 1994-2002, its yearly average production in Panama ranged between 159- 850t/year (446±216t/year) and based on the available underestimated local fishing statistics. Despite the sudden reduction of catches in 1994-95 (850-341t), in 1998-99 (469-159t), and in 2003-04 (1 436-737t), Panama increased its yearly production 3.4 times in 2006-08 (477-1 640t), boosting its average production to 1 234±656t/year during the same period (Anonymous 1999, 2010a).
The Las Perlas Archipelago was the most important fishing ground of Green Jacks until 2007. By the time Law Nº18 was approved by the Assembly of Panama in May of that year, semi-industrial fisheries operating in the Archipelago were responsible for approximately 70% of the catches.
Even although the production of Green Jack in the state of Colima, Pacific Mexico, is of one order of magnitude smaller than that of Panama it has been monitored since the 1980’s (Cruz-Romero et al. 1993, Espino-Barr 2000, Godínez-Domínguez et al. 2000, Navarro- Rodríguez 2000, Rojo-Vázquez et al. 2001, Gallardo-Cabello et al. 2006a, b, 2007). From 1980-2002, Green Jack catches ranged between 9-250t/year (93t on average) and in 2007 they represented 6% of all the production in this Mexican state, only surpassed by that of the highly profitable snappers (Lutjanidae) (Gallardo- Cabello et al. 2007). The average body length of Green Jacks from Colima, LS=23.0cm (LT=29cm), and their L∞ (47.0-68.0cm) (estimated using LS), did not show any significant trend between 1982-1997 (Espino-Barr 2000).
The average length remained slightly smaller during this period of time than the average LS of Green Jacks from Las Perlas recorded in this study, LS=28.3±5.3cm (LT=36.1±6.4).
Old fish play a very important role in the long-term survival of populations, as they are reservoirs of desirable genes and a safeguard to recruitment failure. They are more fecund than younger individuals, produce larger eggs with better chances of survival, and their presence is a measure of the population’s resilience (Froese 2004). The largest Green Jacks from Las Perlas measured LS=45.0cm (LT=56.0cm, 8.2-7.8 years, 1 644g) and were also larger than those from Colima, LS=44.0cm (LT=53cm, 1 689g) (Espino-Barr 2000), but both were smaller than the record sizes attributed to the species; 70cm total length (Robertson & Allen 2008) and 55.0cm fork length (Froese & Pauly 2010). Stocks with a percentage of mega-spawners lower than 20% are very susceptible to the effects of random events and massive recruitment failures (Froese 2004). Only 6-15% of Green Jacks in Las Perlas sample were mega-spawners.
The smallest Green Jacks from Las Perlas, measuring LS=12.7cm (LT=17.0cm, 0.9-1.0 years, 50g), were smaller than the smallest from Colima collected between 1982-1998 (LS=14cm, LT=19cm) (Espino-Barr 2000). Fish measuring between 16.8-18.5cm (MLS) were the youngest mature Green Jacks from the archipelago, but most individuals matured simultaneously and massively after attaining Lm=26.5cm (in LS data set) and Lm=33.0cm (in the LT data set) (2.3-2.5 years). All estimates of Lm from Las Perlas were also similar to those from Colima. Typically, Lm is lower than the size at which half of the population is mature or LS50, and lower than LOPT ± 10%. In Las Perlas samples, 10-13% of all fish were smaller than Lm meaning that between 2005 and 2006, one in every 10 Green Jacks caught in fisheries from Las Perlas never had a chance to reproduce.
Gallardo-Cabello et al. (2007) also estimated parameters for the power law relationship between LT and WT of Green Jacks from Colima, a=0.012 and b=2.972 (N=747). In samples from Las Perlas, parameters a=0.025 and b=2.77, were highly significant (p<0) and significantly different from those reported in Gallardo-Cabello et al. (2007). Other authors, studying the length-weight relationship of Green Jacks, used weights from eviscerated fish (Gallardo-Cabello et al. 2006a, 2007) or used fork lengths as independent variables (e.g. Froese & Pauly 2010), making further comparisons difficult with our current results.
Significant differences in equation parameters between sexes in non-linear models of LS and WT indicated that the body weight of males increased faster per unit length than that of females. All parameter estimations were well within the range of those reported in other species of the family Carangidae (Froese & Pauly 2010).
Average sex ratios and maximum values of LS, LT, L∞, k, and longevity, in samples from Las Perlas, indicate that Green Jack females were somewhat more numerous and perhaps longer living than males. Females also grew slightly faster and larger than the opposite sex.
However, all these differences were very small, and in general, males and females were indistinguishable from their external morphology. For these reasons, only growth models fitted to the pooled data were used for further comparisons.
All growth curves of Green Jacks from Colima plotted within those of models fitted to the LS and the LT data sets from Las Perlas.
Most values of k were very similar between data sets, localities, and sources, ranging from 0.32-0.35/year. In contrast, L∞ estimates were all dissimilar, starting at 59.2cm (LT data set) in Las Perlas, 55.4-51.5cm in Colima, and finishing at 47.6cm in Las Perlas (LS data set). Similar values of L∞ in samples from Las Perlas and Colima suggest comparable environmental and biological conditions for this species in both localities, while similar values of k, equivalent life history traits. Values of k >0.3 are commonly associated with highly productive stocks (e.g. Dulvy et al. 2004).
However, not only were the range of values of L∞ and
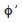 (2.62-3.08) the largest found in this study, but also those of longevity (7.7-20 years) and k (0.15-0.39/year). In samples from Las Perlas, the first best estimate of k obtained, from the surface of response method from the LT data set, was as low as 0.28/year. The second best estimate, 0.33/year, was closer to the value of k obtained from the Powell-Wetherall’s method (Powell, 1979; Wetherall, 1986), 0.32/year. If the growth signal in the data set is robust, approximation methods are expected to produce congruent parameter values for the same data sets. In contrast, if the growth signal is poor, several different combinations of parameter values may explain equally well the same data set. The lack of growth signal is probably responsible for growth parameters from the Espino-Barr (2000) study to be so variable, as she had access to only 1 320 Green Jacks to describe the 16-year long behavior of fisheries in Colima (on average, only 83 fish/year).
(2.62-3.08) the largest found in this study, but also those of longevity (7.7-20 years) and k (0.15-0.39/year). In samples from Las Perlas, the first best estimate of k obtained, from the surface of response method from the LT data set, was as low as 0.28/year. The second best estimate, 0.33/year, was closer to the value of k obtained from the Powell-Wetherall’s method (Powell, 1979; Wetherall, 1986), 0.32/year. If the growth signal in the data set is robust, approximation methods are expected to produce congruent parameter values for the same data sets. In contrast, if the growth signal is poor, several different combinations of parameter values may explain equally well the same data set. The lack of growth signal is probably responsible for growth parameters from the Espino-Barr (2000) study to be so variable, as she had access to only 1 320 Green Jacks to describe the 16-year long behavior of fisheries in Colima (on average, only 83 fish/year). In samples from Las Perlas, Z ranged between 0.9-2.0/year. Estimates from the length-converted catch curves and from the Jones and van Zalinge’s (1981) method, were similar, as expected, as both use comparable assumptions (Pauly 1984). All mortality rates from Las Perlas were high, but they were still lower than the 16-year long average of Z, 2.2 ±1.31 year-1, estimated from yearly catch curve values from Colima.
Instantaneous mortality rates from Hoenig’s (1982; Hoenig & Lawing 1982), Beverton and Holt’s (1956), and Ault and Erhardt’s (1991; Erhardt & Ault 1992) methods produced estimates that were approximately one-half of those obtained from length-converted catch curves and from the Jones and van Zalinge’s (1981) method. Both types were expected to differ, as estimates from the first three models are based on longevity, asymptotic length, mean length, and k, while those from the emaining models depend on the distribution of catches, expressed as lengths or counts, and ordered according to a proxy of age (Sparre & Venema 1998). As length parameters are used in a number of these models, estimates of Z are obviously strongly (and inversely) correlated with them, as shown by McGurk (1986) and the current results. For example, in Green Jacks from Las Perlas Z=1.9/year and L∞=59.2cm, while in those from Colima; Z=2.2/year and L∞=51.5-55.4cm. In Caranx caninus, values of Z, also from Colima, decreased on average from 0.79-0.65/year in a decade, while their asymptotic lengths increased from 74.4- 104.1cm during the same period (Cruz-Romero et al. 1993, Espino-Barr et al. 2008).
Nothing is known about natural mortality in Green Jacks from the Central Eastern Pacific other than approximations from methods that are based on regression models, such as those used in this study. Estimates of M from Las Perlas ranged between 0.3-0.6/year and those from Colima, between 0.3/year, from Taylor’s method (1958, 1960) and 0.4/year, from Pauly’s method (1980, 1983, 1984). Values from Rikhter & Efanov’s (1976) and from Pauly’s methods were similar in Green Jacks from Las Perlas (0.5 - 0.7/year) while those from Taylor’s method (1958, 1960) were the smallest (0.3/year) in all data sets. In Colima, values of M were estimated (using Pauly’s equation with 27.5º C) at 0.6/year, from data published by Cruz-Romero et al. (1993) and Gallardo- Cabello (2006a, 2007). Despite these differences, all estimates are well within the range of M reported in other genera of carangids (Froese & Pauly 2010): Carangoides (0.15-1.40/year), Caranx (0.34-0.58/year), Decapterus (1.40- 2.02/year), Megalaspis (0.39-1.36/year), Selarioides (1.46/year), Seriola (0.50-0.94/year), and Trachurus (0.49-1.36/year).
Values of E ranging from 0.4-0.8/year in Green Jacks from Las Perlas, and 0.8/year in fish from Colima, suggest that high exploitation rates occur in both localities.
Subtle evidence of the high intensity of fishing in Colima is observed in the low values of L∞ and LOPT and the high values of k of its samples, compared to those from Las Perlas, while M (from Pauly’s equation) remains comparable in both localities (but not according to Espino-Barr 2000).
Six separate age groups were found by NORMSEP, clearly representing individuals between 2-5 years old. Green Jacks recruit to the population in Las Perlas before they reach 0.9-1.0 year old (LS=12.7cm, LT=16.5cm). The bias against small sizes in Las Perlas samples (<LS=23cm) is attributed in part to the selectivity of the fishing methods. Habitat preferences also play an important role in this bias, as most juveniles of Green Jacks grow in coastal and estuarine waters before recruiting to populations (Smith-Vaniz 1995). Migration to these areas does not occur before they attain 2.4cm in total length, as individuals up to this size and smaller larvae are commonly collected by plankton tows all along the Central Eastern Pacific coast (Sumida et al. 1985, Rojo-Vázquez et al. 2001, Escarria et al. 2006).
Groups of old individuals were not identified by NORMSEP, consistent with the fact that older fish and mega-spawners are not abundant in Las Perlas samples. In addition to this, individuals grow slower the older they become, and the size classes of old individuals tend to overlap and decrease in relative frequency, becoming harder to separate from classes of younger adults.
The observation that two age groups are included in every single age suggests that at least two recruitment events to the population occur every year. The percentage of fully mature individuals only increasing in November-February and April also suggests the existence of at least two main reproductive events. However, the IG index indicates that there are at least three reproductive events every year: in August-September, December-February, and April. Under both scenarios, the evidence points to December as the strongest reproductive month. Information that could help corroborate these patterns, such as recruitment and temporal distribution of larvae in Las Perlas, is not available.
Two to three months was the delay between the yearly maximum of IG and the top percentage of fully mature individuals in the samples (occurring in December), and the highest chlorophyll concentration and the lowest SST. Spawning two to three months in advance of the moment of highest productivity guarantees a continuous food supply to the growing larvae. The growth models presented here suggest that approximately 1.5 months are required for Green Jack larvae to reach 2.4cm of total length, the size of the largest juveniles sampled in the Central Eastern Pacific by Sumida et al. (1985). To achieve this size, larvae must grow 0.5mm/day, a rate well within the range reported in other carangids, 0.2-0.8mm/day (Leis et al. 2006). This result suggests that abundant food may be available in Las Perlas for Green Jacks larvae, hatched between November and February, to become juveniles two months later.
The abundance of Green Jacks in the region increases during the warmest years, especially in those in which ENSO occurs (Espino-Barr 2000, Godínez-Domínguez et al. 2000, Rojo-Vázquez et al. 2001). Espino-Barr (2000) measured the cross-correlation between the Multivariate ENSO Index (MEI) and the monthly catch per unit effort (CPUE) of Green Jacks from Colima, in a 16-year long time series with 192 data points (one for each month). The best correlation between these two time series (r=0.412) was obtained when both data sets were shifted by 16 months. No clear explanation was given for such a difference. Las Perlas time series had only 14 data points, one for each month, the reason why the temporal structure of Las Perlas data was dismissed. However, a significant direct association between the CPUEs and the corresponding yearly minimum values of MEI was found. Given the time scale of the current analysis, shifting MEI values in relation to the CPUEs time series did not improved the fitting scores. The results suggest that the minimum values of MEI could be used as an additional tool to evaluate a posteriori and very roughly, the productivity of Green Jacks during a given year, by comparing current and previous annual records with the oceanographic conditions of the region.
It is also apparent that the current status of Green Jacks is far better than that of other marine commercial resources in Las Perlas, such as the severely overexploited pearl oysters, lobsters, and conchs (Cipriani et al. 2008a, b, Guzman et al. 2008). However, the results demonstrate that 10-13% of the current captures of Green Jacks are on average immature individuals, 68% of catches are within the LOPT range, and only 6-15% of fish in the population are mega-spawners. According to Froese (2004), this is enough evidence to assert that Green Jacks from Las Perlas are overfished. At the same time, results also describe a population with fast growing individuals, with rates of natural mortality similar to those of other carangid species, and under strong fishing pressure. It is speculated that the production of Green Jacks in Las Perlas is probably maintained by the enormous availability of larvae resulting from two or three reproductive events each year, fueled by one strong annual upwelling that boosts the primary productivity of the region, and by the capacity of these larvae to successfully recruit to the local population.
Under this scenario, if recruitment fails due to an unfortunate combination of environmental and man-made conditions, and the fisheries of Green Jacks collapse, the low proportion of mega-spawners will hamper their recovery. Implementing strategies to increase the proportion of mega-spawners in an exploited population is not a simple task in fisheries where the upper limits of size are usually not regulated, and larger fish are targeted from the very onset of the fishing activities.
Acknowledgments
The authors thank Carlos Guevara and Carlos Vega for processing samples and Julio Agujetas for providing access to satellite data and all fishermen who provided information about the fisheries in Las Perlas. The Government of Panama provided permits to work in the area. This research was partially sponsored by DEFRA’s Darwin Initiative Fund (UK), Heriot-Watt University, Smithsonian Tropical Research Institute, Panama, and Universidad Simon Bolivar (GID-67), Caracas, Venezuela.
References
ACODECO. 2010. Autoridad de Protección al Consumidor y Defensa a la Competencia. (Downloaded: October 30, 2010, http://www.acodeco.gob.pa). [ Links ]
Agujetas, J. 2005. The seasonal upwelling and primary production of the Panama Bight estimated from satellite data: ENSO implications. M.Sc. Thesis, University of Wales, Bangor, United Kingdom. [ Links ]
Allen, G.R. & D.R. Robertson. 1994. Fishes of the Tropical Eastern Pacific. University of Hawaii, Hawaii, USA. [ Links ]
Anonymous. 1999. Compendio Estadístico 1998. Departamento de Estadísticas Generales, Autoridad Marítima de Panamá, Panama, Panama. [ Links ]
Anonymous. 2010a. Publicacions: Panamá en Cifras 1999- 2003 and Panamá en Cifras 2003-2007. Contraloría General de la República de Panamá. Dirección de Estadística y Censo. (Downloaded: November 1, 2010, http://www.contraloria.gob.pa/dec/). [ Links ]
Anonymous. 2010b. Publicacions: Estadísticas Ambientales 1998-2000 and Estadísticas Ambientales 2002-2004. Contraloría General de la República de Panamá. Dirección de Estadística y Censo. (Downloaded: November 1, 2010, http://www.contraloria.gob.pa/dec/). [ Links ]
Ault, J.S. & N.M. Ehrhardt. 1991. Correction to the Beverton and Holt Z-estimator for truncated catch lengthfrequency distributions. ICLARM Fishbyte 9: 37-39. [ Links ]
Beddington, J.R. & J.G. Cooke. 1983. On the potential yield of fish stocks. FAO Fish. Tech. Pap. 242, Rome, Italia. [ Links ]
Bernal-Ornelas, I.H., M. Saucedo-Lozano, E. Espino-Barr, A. García-Boa, E.G. Cabral-Solis & M. Puente--Gómez. 2005. Análisis de la alimentación de Caranx caballus Günther 1868, en la costa de Manzanillo, Colima, México. Abs. Mem. XVIII Cong. Nac. Zool., Monterrey, Mexico. [ Links ]
Beverton, R.J.H. & S.J. Holt. 1956. A review of methods for estimating mortality rates in exploited fish populations, with special reference to sources of bias in catch sampling. Rapp.P.-V.Réun. CIEM, 140: 67-83. [ Links ]
Cipriani, R., H.M. Guzman & M. Lopez. 2008a. Harvest history and current densities of the pearl oyster Pinctada mazatlanica (Bivalvia: Pteriidae) in Las Perlas and Coiba Archipelagos, Panama. J. Shellfish Res. 27: 691-700. [ Links ]
Cipriani, R., H.M. Guzman, A.J. Vega & M. Lopez. 2008b. Population assessment of the conch Strombus galeatus (Gastropoda, Strombidae) in Pacific Panama. J. Shellfish Res. 27: 889-896. [ Links ]
Cruz-Romero, M., E. Espino-Barr & A. Garcia-Boa. 1993. Carángidos: Aspectos biológico-pesqueros en el litoral colimense. Cuad. Mex. Zool. 1: 81-88. [ Links ]
D’Croz, L., J. Del Rosario & J. Gomez. 1991. Upwelling and phytoplankton in the Bay of Panama. Rev. Biol. Trop. 39: 233-241. [ Links ]
D’Croz, L. & A. O’Dea. 2007. Variability in upwelling along the Pacific shelf of Panama and implications for the distribution of nutrients and chlorophyll. Est. Coast. Shelf. Sci. 73:325-340. [ Links ]
Dulvy, N.K., J.R. Elvis, N.B. Goodwin, A. Grant, J.D. Reynolds & S. Jennings. 2004. Methods of assessing extinction risk in marine fishes. Fish Fish. 5: 255-276. [ Links ]
Ehrhardt, N.M. & J.S. Ault. 1992. Analysis of two lengthbased mortality models applied to bounded catch length frequencies. Trans. Am. Fish. Soc. 121: 115-122. [ Links ]
Escarria, E., B.S. Beltrán-León & A. Giraldo. 2006. Ictioplancton superficial de la Cuenca del Océano Pacífico Colombiano (Septiembre 2003). Invest. Mar. 34: 169-173. [ Links ]
Espino-Barr, E. 2000. Criterios biológicos para la administración de la pesca multiespecífica artisanal en la costa de Colima, México. Tesis de Doctorado, Universidad de Colima, Colima, México. [ Links ]
Espino-Barr, E., M. Gallardo-Cabello, E.G. Cabral-Solís, A. García-Boa & M. Puente-Gómez. 2008. Growth of the Pacific jack Caranx caninus (Pisces: Carangidae) from the coast of Colima, México. Rev. Biol. Trop. 56: 171-179. [ Links ]
Essington, T.E., J.F. Kitchel & C.J. Walters. 2001. The von Bertalanffy growth function, bioenergetics, and the consumption rates of fish. Can. J. Fish. Aqua. Sci. 58: 2129-2138. [ Links ]
Froese, R. 2004. Keep it simple: three indicators to deal with overfishing. Fish Fish. 5: 86-91. [ Links ]
Froese, R. & C. Binohlan. 2000. Empirical relationships to estimate asymptotic length, length at first maturity and length at maximum yield per recruit in fishes, with a simple method to evaluate length frequency data. J. Fish Biol. 56: 758-773. [ Links ]
Froese, R. & D. Pauly. 2010. FishBase. World Wide Web Electronic Publication. www.fishbase.org. version (08/2010). [ Links ]
Gallardo-Cabello, M., E. Espino-Barr, A. García-Boa, E.G. Cabral-Solís & M. Puente-Gómez. 2006a. Algunos parámetros biológicos del cocinero Caranx caballus en la costa de Colima, México, p. 485-493. In M.C. Jiménez-Quiroz & E. Espino-Barr (eds.). Los recursos pesqueros de Colima, Jalisco y Michoacán. Secretaría de Agricultura, Ganadería, Desarrollo Rural, Pesca y Alimentación. Instituto Nacional de la Pesca, México. [ Links ]
Gallardo-Cabello, M., E. Espino-Barr, A. García-Boa, E.G. Cabral-Solís & M. Puente-Gómez. 2006b. Morphologic and morphometric analysis and growth rings identification of otholiths: sagitta, asteriscus and lapillus of Caranx caballus (Pisces: Carangidae) in the coast of Colima, Mexico. Int. J. Zool. Res. 2: 34-47. [ Links ]
Gallardo-Cabello, M., E. Espino-Barr, A. García-Boa, E.G. Cabral-Solís & M. Puente-Gómez. 2007. Study of the Growth of the Green Jack Caranx caballus Günther 1868, in the coast of Colima, Mexico. J. Fish. Aquat. Sci. 2: 131-139. [ Links ]
Gayanilo, F.C. Jr., P. Sparre & D. Pauly. 2005. The FAOICLARM Stock Assessment Tools (FiSAT II) User’s Guide. FAO Comput. Inf. Ser. Fish. No. 8, Rome, Italia. [ Links ]
Godínez-Domínguez, E., J. Rojo-Vázquez, V. Galván- Piña & B. Aguilar-Palomino. 2000. Changes in the Structure of a Coastal Fish Assemblage Exploited by a Small Scale Gillnet Fishery During an El Niño-La Niña Event. Estuar. Coast. Shelf. Sci. 51: 773-787. [ Links ]
Guzman, H.M., R. Cipriani, A.J. Vega, M. Lopez & J.M. Mair. 2008. Population assessment of the Pacific green spiny lobster Panulirus gracilis in Pacific Panama. J. Shellfish Res. 27: 907-915. [ Links ]
Hoenig, J.M. 1982. Estimating mortality rate from the maximum observed age. ICES. C.M./1982/D:5, Copenhagen, Denmark. [ Links ]
Hoenig, J.M. & W.D. Lawing. 1982. Estimating the total mortality rate using the maximum-order statistic for age. ICES C.M./1982/D: 7, Copenhagen, Denmark. [ Links ]
Jones, R. & N.P. van Zalinge. 1981. Estimations of mortality rate and population size for shrimp in Kuwait waters. Kuwait Bull. Mar. Sci. 2: 273-288. [ Links ]
Leis, J.M., A.C. Hay, D.A. Clark, I-S. Chen & K-T. Shao. 2006. Behavioral ontogeny in larvae and early juveniles of the giant trevally, Caranx ignobilis (Pisces: Carangidae). US. Fish. Bull. 104: 401-414. [ Links ]
McGurk, M.D. 1986. Natural mortality of marine pelagic fish eggs and larvae: role of spatial patchiness. Mar. Ecol. Prog. Ser. 34: 227-242. [ Links ]
McNiven, G. 2003. An Assessment of the Pearl Islands Archipelago, Pacific Panama. M. Sc. Thesis, Heriot-Watt University, Edinburgh, United Kingdom. [ Links ]
Navarro-Rodríguez, M.C. 2000. Variación anual de la distribución y abundancia de larvas de peces de las familias Serranidae, Haemulidae, Sciaenidae y Carangidae (Perciformes: Actinopterygii) de la plataforma continental de Jalisco y Colima, México. Tesis de Maestría, Universidad de Colima, Colima, Mexico. [ Links ]
Pauly, D. 1980. On the interrelationships between natural mortality, growth parameters and mean environmental temperature in 175 fish stocks. J. Cons. CIEM 39: 175-192. [ Links ]
Pauly, D. 1983. Some simple methods for the assessment of tropical fish stocks. FAO Fish. Tech. Pap. 234, Rome, Italia. [ Links ]
Pauly, D. 1984. Fish population dynamics in tropical waters: a manual for use with programmable calculators. ICLARM Stud. Rev. Manila, Philippines. [ Links ]
Powell, D.G. 1979. Estimation of mortality and growth parameters from the length-frequency in the catch. Rapp. P.-V. Réun. CIEM. 175: 167-169. [ Links ]
R Development Core Team. 2011. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. [ Links ]
Rikhter, V.A. & V.N. Efanov. 1976. On one of the approaches to estimation of natural mortality of fish populations. ICNAF Res. Doc., 79/VI/8, Dartmouth, Nova Scotia, Canada. [ Links ]
Robertson, D.R. & G.R. Allen. 2008. Shorefishes of the Tropical Eastern Pacific online information system. Version 1.0 (2008). Smithsonian Tropical Research Institute, Panamá. (http:ww.neotropicalfishes.org/sftep, ww.stri.org/sftep). [ Links ]
Rojo-Vázquez, J.A., B. Aguilar-Palomino, V. Galvan-Pina, H.E. Godínez-Domínguez, S. Hernández-Vázquez, S. Ruiz-Ramírez & G. Lucano-Ramírez. 2001. Ictiofauna de la pesquería ribereña en Bahía de Navidad, Jalisco, México, asociada al evento El Niño 1997-1998. Rev .Biol. Trop. 49: 915-929. [ Links ]
Smith-Vaniz, W.F. 1995. Carangidae. Jureles, pámpanos, cojinúas, zapateros, cocineros, casabes, macarelas, chicharros, jorobados, medregales, pez pilota, p.940- 986. In W. Fischer, F. Krupp, W. Schneider, C. Sommer, K.E. Carpenter & V. Niem (eds.). Guía FAO para la Identificación de Especies para los fines de la Pesca. Pacífico Centro-Oriental. FAO, Rome, Italia. [ Links ]
Sokal, R.R. & F.J. Rohlf. 1995. Biometry: the principles and practice of statistics in biological research. W.H. Freeman, New York, USA. [ Links ]
Sparre, P. & S.C. Venema. 1998. Introduction to tropical fish stock assessment. Part 1. Manual. FAO Fish. Tech. Pap. 306/1 Rev. 2, Rome, Italia. [ Links ]
Sumida, B.Y., H.G. Moser & E.H. Ahlstrom. 1985. Descriptions of larvae of California Yellowtail, lalandi, and three other carangids from the eastern Tropical Pacific: Chloroscombrus orqueta, Caranx caballus, and Caranx sexfasclatus. CalCOFI Rep. 26: 139-159. [ Links ]
Taylor, C.C. 1958. Cod growth and temperature. J. Cons. Int. Explor. Mer. 23: 366-370. [ Links ]
Taylor, C.C. 1960. Temperature, growth and mortality – The Pacific Cockle. J. Cons. Int. Explor. Mer. 26:117-124. [ Links ]
Wetherall, J.A. 1986. A new method for estimating growth and mortality parameters from length-frequency data. ICLARM, Fishbyte 4: 12-14. [ Links ]
Wolter, K. & M.S. Timlin. 1993. Monitoring ENSO in COADS with a seasonally adjusted principal component index. Proc. of the 17th Climate Diagnostics Workshop, University of Oklahoma, Oklahoma, USA. [ Links ]
Wolter, K. & M.S. Timlin. 1998. Measuring the strength of ENSO events - how does 1997/98 rank? Weather 53: 315-324. [ Links ]
*Correspondencia:
James M. Mair: Centre for Marine Biodiversity and Biotechnology, School of Life Sciences, Heriot-Watt University, Edinburgh EH14 4AS, Scotland, UK. j.m.mair@hw.ac.uk
Roberto Cipriani: Departament of Biological Science, California State University, Fullerton, US. rcipriani@fullerton.edu
Hector M. Guzman: Smithsonian Tropical Research Institute, Apartado 0843-03092, Balboa, Ancon, Panama; guzmanh@si.edu.
David Usan: Centre for Marine Biodiversity and Biotechnology, School of Life Sciences, Heriot-Watt University, Edinburgh EH14 4AS, Scotland, UK. dausan@hotmail.com
1. Centre for Marine Biodiversity and Biotechnology, School of Life Sciences, Heriot-Watt University, Edinburgh EH14 4AS, Scotland, UK; j.m.mair@hw.ac.uk, dausan@hotmail.com
2. Departament of Biological Science, California State University, Fullerton, US; rcipriani@fullerton.edu
3. Smithsonian Tropical Research Institute, Apartado 0843-03092, Balboa, Ancon, Panama; guzmanh@si.edu
Received 29-vIII-2011.Corrected 10-III-2012. Accepted 12-Iv-2012














