Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.60 n.2 San José Jun. 2012
Factors affecting establishment success of the endangered Caribbean cactus Harrisia portoricensis (Cactaceae)
*Dirección para correspondencia:
Abstract
Early plant stages may be the most vulnerable within the life cycle of plants especially in arid ecosystems. Interference from exotic species may exacerbate this condition. We evaluated germination, seedling survival and growth in the endangered Caribbean cactus Harrisia portoricensis, as a function of sunlight exposure (i.e., growing under open and shaded areas), different shade providers (i.e., growing under two native shrubs and one exotic grass species), two levels of predation (i.e., exclusion and non-exclusion) and variable microenvironmental conditions (i.e., temperature, PAR, humidity). Field experiments demonstrated that suitable conditions for germination and establishment of H. portoricensis seedling are optimal in shaded areas beneath the canopy of established species, but experiments also demonstrated that the identity of the shade provider can have a significant influence on the outcome of these processes. Harrisia portoricensis seedlings had higher probabilities of survival and grew better (i.e., larger diameters) when they were transplanted beneath the canopy of native shrubs, than beneath the exotic grass species, where temperature and solar radiation values were on average much higher than those obtained under the canopies of native shrubs. We also detected that exclusión from potential predators did not increase seedling survival. Our combined results for H. portoricensis suggested that the modification of microenvironmental conditions by the exotic grass may lower the probability of recruitment and establishment of this endangered cactus species. Rev. Biol. Trop. 60 (2): 867-879. Epub 2012 June 01.
Key words: columnar cactus, Harrisia portoricensis, Megathrysus maximus, Mona Island, nurse plants, seed germination, seedling survival.
Resumen
Las etapas iniciales de las plantas parecen ser las más vulnerables de su ciclo de vida, especialmente en ecosistemas áridos. Interferencias de especies exóticas pueden exacerbar esta condición. Evaluamos la germinación, supervivencia y crecimiento de plántulas del cactus en peligro de extinción Harrisia portoricensis, en función de la exposición a la luz solar, diferentes proveedores de sombra, exclusión de potenciales depredadores y condiciones micro-ambientales variables. Los experimentos demostraron que las condiciones adecuadas para la germinación y el establecimiento de plántulas de H. portoricensis son óptimas en áreas de sombra, bajo el dosel de especies establecidas, pero también demostraron que la identidad de los proveedores de sombra puede tener una influencia significativa sobre los resultados de estos procesos y que la exclusión de depredadores no aumentó la probabilidad de supervivencia de las plántulas. Las plántulas de H. portoricensis tuvieron mayores probabilidades de supervivencia y crecieron mejor cuando fueron transplantadas bajo la sombra de los arbustos nativos que bajo la sombra del pasto exótico, donde la temperatura y la radiación solar fueron en promedio mucho mayores que las obtenidas bajo la sombra de los arbustos nativos. Nuestros resultados sugieren que las modificaciones de las condiciones micro-ambientales por este pasto exótico pueden disminuir la probabilidad de reclutamiento y establecimiento de esta especie en peligro.
Palabras clave: cactus columnares, Harrisia portoricensis, Megathrysus maximus, Isla de Mona, plantas nodriza, germinación de semillas, supervivencia de plántulas.
Germination, seedling establishment, and survival of young plants appear to be the most critical phase in the life cycle of cacti (Bowers 1997, Pimienta & Del Castillo 2002, Valiente et al. 2002, Godínez et al. 2003, Valverde et al. 2004). In arid and semi-arid environments, the combined effects of temperature, light, and water availability regulate germination, establishment, and survival of plants. Unpredictable rainfall, very high temperatures, and high solar radiation in those environments may influence these processes negatively leading to rare and sporadic germination and survival of early plant stages (Franco & Nobel 1989, Nolasco et al. 1997, Fleming & Valiente 2002, Nobel & Bobich 2002, De la Barrera & Nobel 2003, Suzán & Sosa 2006). Understanding, how abiotic factors ultimately influence plant recruitment processes is particularly important to endangered plant species where population persistence is at risk and development of plant management strategies is expected.
Cacti may be buffered from the harsh environmental conditions that characterize arid and semi-arid habitats when growing under the foliage of perennial plants functionally known as “nurse plants” (Steenbergh & Lowe 1977, Pierson & Turner 1998, Sosa & Fleming 2002, Godínez et al. 2003, Larrea & Soriano 2006). Trees, shrubs, grasses, agaves and other cacti may play the role of nurse plants. Evidence demonstrates that these nurse plants may protect early plant stages from intense sunlight, reduce soil temperatures, increase soil moisture, provide a shady microhabitat in which evapotranspiration is reduced, and protect seeds and seedlings from predation (Steenbergh & Lowe 1977, Jordan & Nobel 1981, Godínez & Valiente 1998, Valiente et al. 1991, Sosa & Fleming 2002, Valiente et al. 2002, Godínez et al. 2005). This phenomenon has been reported in about 36 cacti species representing different life-forms within the family (Steenbergh & Lowe 1977, Jordan & Nobel 1981, Valiente & Ezcurra 1991, Suzán et al. 1996, Sosa & Fleming 2002, Godínez et al. 2003).
Harrisia portoricensis Britton is a columnar cactus endemic to four Caribbean islands of the Puerto Rican bank. At present, this species is extinct on the island of Puerto Rico and is geographically restricted to the small islands of Mona, Monito and Desecheo (US Fish and Wildlife Service 1990, Liogier 1994). Harrisia portoricensis is listed as a threatened species under US Federal Regulation (US Fish and Wildlife Service 1990). This threatened status has been primarily attributed to habitat loss and vegetation changes from feral goats and pigs (US Fish and Wildlife Service 1990). The population on Mona Island is the largest remnant population of H. portoricensis to date and is distributed primarily across two vegetation types: lowland dry cactus shrubland and lowland dry limestone shrubland (Martinuzzi et al. 2008). At these sites, Croton discolor and Reynosia uncinata are xerophytic shrubs that dominate the landscape and are often found spatially associated with H. portoricensis (Rojas-Sandoval 2010). One hypothesis is that both species may serve as nurse plants of H. portoricensis. The grass Megathyrsus maximus, is a perennial African grass that was introduced on Mona Island to support prior cattle activities at this reserve (Cintrón & Rogers 1991) and seems to be spreading into prime H. portoricensis hábitat (Rojas-Sandoval 2010). Literature suggests that grass invasions may have important negative effects on native biodiversity because they can compete effectively with native species (Hughes et al. 1991, D’Antonio & Vitousek 1992, Cabin et al. 2002, Levine et al. 2003, Seabloom et al. 2003). At present, no data exist on whether or not the presence of M. maximus has negative impacts on H. portoricensis populations. While M. maximus may compete for space and resources, positive effects on germination and establishment may also result if this species also functions as a nurse plant. Data on the role of native shrub species versus that of M. maximus on germination and establishment in H. portoricensis is one way to understand the potential interactions between this endemic cactus and the exotic grass that is invading its current habitat.
In this study we evaluated whether or not germination and seedlings survival of H. portoricensis was influenced by micro-environmental variation and by extension whether or not these processes were influenced by the type of species under which they were occurring (i.e., exotic grass: M. maximus or native shrubs: C. discolor and R. uncinata). We used this information to understand on how vulnerable are H. portoricensis populations to the potential effects of an exotic grass species, how important is the presence of shade providers for germination and establishment, and how susceptible are seedlings and young individuals to changes in micro-environmental factors. We hypothesized that germination, survival and growth of seedlings would be higher in shaded areas than in open areas as it occurs in many columnar cacti species. We also hypothesized that micro-environmental conditions (i.e., temperature, PAR, humidity) would be most favorable for H. portoricensis seedlings beneath the canopies of native shrubs than beneath the leaf cover of the exotic grass, and as a result seedling survival and growth would also be higher under native shrubs.
Materials and methods
Study species: Harrisia portoricensis Britton is a slender columnar cactus where juveniles are usually unbranched and adults may reach heights of more than 2m and present an extensive branching pattern (Liogier 1994). The density of H. portoricensis on Mona Island is 0.001plants/m2 and a population size estímate of 59 857 (SE=1058) with a demographic profile that included plants in the adult (59%), juvenile (34%) and seedling stages (7%), indicating that at least some recruitment is occurring at this locality (Rojas-Sandoval 2010). This species is a night flowering cactus and plants produce flowers multiple times within a year (Rojas-Sandoval 2010). Reproductive plants produced large hermaphroditic flowers (mean length: 21.16±0.98cm; mean width: 5.25±0.27cm) that open only for one night. Flowers are nectariferous (mean volumen 0.26±0.04mL/night) but visits by animals are highly infrequent, and pollination is mainly wind-facilitated with most of the fruit production resulting from self-pollination (Rojas-Sandoval & Meléndez-Ackerman 2009, 2011). Fruit production under natural conditions is high (88% fruit-set) and fruits mature in approximately 56 day. Fruits are typically ornithochorous with yellow, spineless and fleshy berries with more than 1 500 small black seeds enclosed in a white pulp (Rojas-Sandoval & Meléndez-Ackerman 2009). Propagation in H. portoricensis is mainly through seeds and spread by vegetative growth has not been detected (Rojas-Sandoval 2010).
Study site: The study was conducted at Mona Island Reserve, a semi-arid island located in the Caribbean Sea between Puerto Rico and Hispaniola (18°05’ N - 67°54’ W). The island itself is a raised platform of dolomite (magnesium-rich limestone rock) reaching up to 100m at its highest point (Frank et al. 1998, Cintrón 1991), and covering an area of 5 517ha. Mona has an annual mean temperature of 25°C, and receives an average annual rainfall of 810mm (Ewel & Whitmore 1973). The rainy season last from August to November with peaks in May and October, and the dry season run from December to April (Murphy & Lugo 1995). During the time of the experiments (May 2007-May 2009), annual mean rainfall was 986mm with peaks occurring always in May and between September-October. Mona is characterized by an irregular distribution of plants, exposed rocks, crevasses, cracks, and sinkholes with accumulated soil. Water availability is limited by an excessive wáter percolation through shallow soils and a limestone parent rock (Martinuzzi et al. 2008). The island’s vegetation is classified as a subtropical dry forest. On the island, H. portoriscensis is most common at two plant communities: the lowland dry cactus shrubland and the lowland dry limestone shrubland (Martinuzzi et al. 2008). The lowland dry cactus shrubland is an area where exposed limestone rocks surround small shrubs and cactus species. The lowland dry limestone shrubland is an area impacted continuously by strong winds, salt spray and poor soil accumulation. The shrubs Croton discolor (Euphorbiaceae) and Reynosia uncinata (Rhamnaceae) are dominants species of both plant communities and frequently can be found in association with H. portoricensis. Croton discolor is a short semi-deciduous shrub (less than 1.5m) that only sheds its leaves under extreme drought conditions while R. uncinata is a taller (up to 4m) woody evergreen shrub that gradually replaces its leaves throughout the year. Approximately a 25% of both forests have been invaded by the African grass Megathyrsus maximus (Rojas-Sandoval 2010).
Seeds collection: Fifteen mature fruits of H. portoricensis were randomly collected from plants growing in the study site in August 2007. Each fruit was collected from a different plant and their seeds were extracted and washed with tap and distilled water to remove the pulp. Seeds were dried using absorbent paper, pooled and placed in paper bags for storage at room temperature until required. These seeds were used to perform seedlings survival and seedlings performance field experiments. In August 2008, we repeated the same procedure and collected 15 mature fruits from 15 different plants to obtain a new pool of seeds that were later used in the germination experiment.
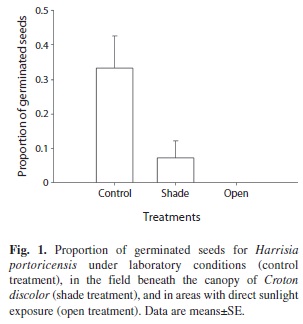
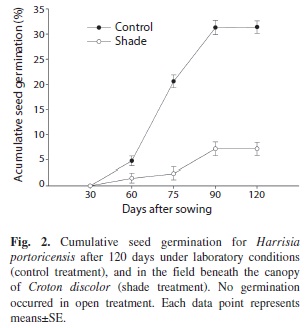
Seed germination experiment: To estimate the rate of seed germination under natural and laboratory conditions, and to identify conditions required for seed germination, we performed a germination experiment in September 2008 that included the following treatments: (1) open treatment: seeds were germinated in open areas receiving direct sunlight at any time during the day, (2) shade treatment: seeds were germinated in shaded areas beneath the canopy of Croton discolor, the most common shrub species in the study site, and (3) control treatment: seeds were germinated in the laboratory under controlled conditions of light (15h light: 9h dark), humidity (90% relative humidity) and temperature (minimum 24ºC: maximum 30ºC) (Sosa & Fleming 2002). For open and shade treatments, 100 seeds were sown in the field and placed directly on the soil in a 25×25cm plot subdivided into 25 grids (four seeds were placed each 5cm). These plots were located in areas fully exposed to the sun (open treatment) and in areas fully shaded in which shade was provided by the surrounding canopy of C. discolor and seedlings received no direct sunlight at any time during the day even with changes in sun direction (shade treatment). Ten replicates were performed per treatment for a total of 1 000 seeds per treatment. For the control treatment, 1 000 seeds were sown among ten 12×22cm plastic-trays (100 seeds per trays) filled with non-sterilized soil collected from the study site. For all treatments, the proportion of germinated seeds was recorded for 120 days. The criterion used to determine seed germination was radical emergence. Differences in seed germination among treatments were compared using Kruskal-Wallis because data did not fulfill assumptions to carry out parametric analysis.
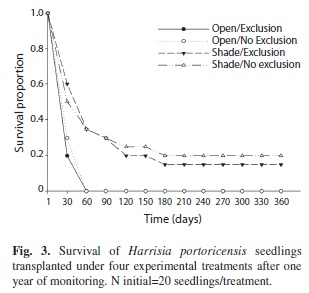
Seedling survival experiment: To estímate the survival and growth of H. portoricensis seedlings under natural conditions, we designed an experiment that evaluated two contrasting sunlight exposure conditions, and ruled out the potential effects of natural predators on the outcomes by performing the experiments under conditions of exclusion and non-exclusion of potential predators. Five hundred seeds from 15 different fruits were sown in November 2007 in plastic nursery traits with multiple cavities filled with soil collected from the study site. We sowed one seed per cavity under greenhouse conditions on Mona Island. In February 2008, one month after seedling emergence, we randomly selected and transplanted 80 seedlings to the field. Forty seedlings were transplanted to 20 different open sites (two seedlings per site) with direct sunlight at any time during the day, and another 40 seedlings were transplanted to 20 shaded sites (two seedlings per site) under the canopy of Croton discolor. Open and shaded sites were distributed on Mona Island across an area of approximately 10ha that included the two vegetation types where H. portoricensis is most common: lowland dry cactus shrubland and lowland dry limestone shrubland. At each site, half of the seedlings were covered with a cage (7×7×7cm) made of 2mm metal screen mesh to exclude potential predators. Once in the field, seedlings were not watered artificially. The height and diameter of each seedling at the moment that they were placed on the field were measured with a caliper. Seedlings were checked for survival and measured for growth (height and diameter) monthly for one year. Differences in seedling survivorship at the end of the experiment were analyzed following the Kaplan-Meier method and statistical differences among the survival curves of each treatment were tested with Log-Rank and Wilcoxon tests (Pepe & Fleming 1989, Kleinbaum & Klein 2005). Seedling growth parameters for each treatment were analyzed separately because all seedlings transplanted to open sites had died before the third month.
Seedling performance experiment: To compare the performance of H. portoricensis, we used a field experiment where seedlings were placed beneath the canopies of three shade provider species: Croton discolor, Reynosia uncinata and Megathyrsus maximus. Ten focal plants were randomly selected in February 2008 for each shade provider species, and two one-month-old seedlings were transplanted directly on soil beneath the canopy of each plant. Following the same methodology that we described in the previous experiment, seedling survival and growth parameters (height and diameter) were recorded monthly from February 2008 until May 2009 (15 months). Differences in seedling growth and survivorship at the end of the experiments were compared among the three shade provider species using one-way ANOVA and the Kaplan-Meir method respectively. All analyses were performed using JMP 7.0 (SAS Institute Inc. 2007). We recorded temperature and relative humidity beneath the focal plants and in nearby open sites with direct sunlight exposure with HOBO® Prov2 data-loggers. Additionally, in March 28, 2008 (dry season) and September 30, 2008 (rainy season) at 0 700, 1 300 and 1 700hr we quantified photosynthetic active radiation (PAR) at the projected shade beneath the canopy of different types of focal plants using a Ceptometer-AccuPAR LP80 (Decagon Devices, USA). Specifically we measured PAR at five plants for each shade provider species and for each focal plant there was an adjacent open site with direct sunlight where PAR was recorded as well. The combined information on temperature, relative humidity and PAR was used to characterize the ‘typical’ microenvironmental conditions beneath each shade provider species as well as in open sites.
Results
Seed germination experiment: The number of seeds germinated after 120 days differed among treatments (Kruskal-Wallis χ2=24.34, df=2, p<0.001, Fig. 1). The proportion of germinated seeds under laboratory conditions was 4.3 times higher than the proportion of germinated seeds placed in field shade treatments in the field. None of the seeds sown in open sites with direct sunlight exposure germinated (Fig. 1). On average, seeds began to germínate 54 days after sowing for both the laboratory and the field shade treatments (Fig. 2). The end of the experiment obtained the highest germination percentage under laboratory conditions with 31.4% of germination. In the shade treatment the percentage of germinated seeds was much lower and scarcely reached values of 7.2%. Seed germination halted for both treatments after 90 days (Fig. 2).
Seedling survival experiment: The survival of transplanted seedlings was low and upon completion of the experiment only seven individuals were alive at shaded sites in the field (Table 1). Analyses of the survivorship curves indicated significant differences among experimental treatments (Log-Rank test: χ2=34.2, p<0.0001, Wilcoxon test: χ2=30.4, p<0.0001, Fig. 3). All seedlings transplanted to open sites died by the second month and only seedlings that were placed at shaded sites survived after one year in the field. Contrary to our expectations, seedling survival in shaded sites was not higher when seedlings were excluded from potential predators (G test=3.06, df=1, p=0.2, Fig. 3). Finally, for the two growth parameters analyzed, surviving seedlings growing under exclusion and non-exclusion conditions did not differ neither in height (t=-0.69, df=5, p=0.52) nor in diameter (t=0.73, df=5, p=0.49) by the end of the experiment (Table 1).
Seedling performance experiment: In the field, all three shade provider species had a fraction of surviving H. portoricensis seedlings underneath them 15 months after seedlings were transplanted. However, analysis of the survivorship curves indicated significant differences among shade providers (Log-Rank test: χ2=31.2, p<0.0001, Wilcoxon test: χ2=48.2, p<0.0001, Fig. 4). By the end of the experiment, a higher percentage of seedlings survived beneath the canopy of R. uncinata (40%) and C. discolor (30%) than beneath the shade of M. maximus (15%). For the two growth parameters analyzed, we detected differences among shade providers in seedling diameter (F2,14=3.21, p=0.04, Fig. 5A) but not in seedling height (F2,14=1.87, p=0.17, Fig. 5B). At the end of the experiment, the average diameters of seedlings growing under C. discolor and R. uncinata were twice the average diameter of seedlings growing under M. maximus (Fig. 5A).
Monthly mean temperatures were significantly higher in open sites than beneath the canopies of the shade providers (F3,61=3.32, p=0.02, Table 2), but contrary to our expectations did not differ among shade provider species (Table 2). During the experiment, mean temperatures varying from 29.09ºC in open sites to 27.62ºC under shade providers (Table 2). In contrast, monthly mean maximum temperatures were significantly different among shade providers (F3,61=4.33, p=0.03, Table 2). We found that máximum temperatures in open areas and beneath M. maximus were significantly higher than beneath the canopies of the native shrubs. Maximum temperatures were beneath the canopy of M. maximus and in open areas were 1.4ºC and 3.8ºC respectively, and higher than under the canopies of native shrubs (Table 2). We did not find differences in the percentages of relative humidity recorded beneath the shade providers and in open sites over the length of the experiment (F3,61=0.07, p=0.92, Table 2). As we expected, PAR values were significantly higher in open sites than underneath the shade providers, but they were also highest beneath M. maximus foliage than under the canopies of C. discolor and R. uncinata in March and September (p<0.0001 in all cases, Table 2). On average PAR values were 2.8 times higher beneath the leaf cover of M. maximus than beneath the canopy of the native shrubs (Table 2).
Discusion
We predicted that germination, survival and growth of H. portoricensis seedlings on Mona Island would be higher in shaded areas than in open areas due to more benign microclimatic conditions. In general, our results for field and laboratory experiments support this hypothesis and demonstrate the importance of “shaded” environments for the early life cycle stages of this cactus species. Our results also suggest that temperature and solar radiation are abiotic factors that could influence the occurrence of recruitments events into the populations of H. portoricensis. Studies have shown that shade created by perennial nurse plants can decrease solar radiation, temperatures, and water evaporation, and as a result enhance the germination, growth and establishment of young cacti beneath their canopies (Jordan & Nobel 1981, Valiente & Ezcurra 1991, Valiente et al. 1991, Nolasco et al. 1997, Leira & Parra 1999, Sosa & Fleming 2002). For many species of cacti, in addition to nurse plants, rocks and other surface irregularities such as holes and cracks may act as potential facilitators of germination and seedling establishment (Godínez et al., 2003, Mungía & Sosa 2007, Peters et al. 2008). However, for H. portoricensis in the study site we have not observed nurse rock syndrome, but we do not rule out the existence of this phenomenon.
Our combined results for H. portoricensis also demonstrated that suitable conditions for germination and seedling establishment are tied to type of shade provided by the different species in our study. Although the number of seedlings used in the experiments were low, our data showed that H. portoricensis seedlings on Mona Island have higher probabilities of survival and grew better (i.e., higher diameters) if they were transplanted beneath the canopies of two native shrubs species C. discolor and R. uncinata than beneath the exotic grass M. maximus. It is likely that these differences are related to differences in microclimatic conditions under the different shade providers because both temperature and incident solar radiation were much higher underneath this exotic grass than underneath the native shrubs. Results strongly suggest that the exotic grass does not provide the same degree of environmental shelter that native shrubs do.
Seedling survival and seedling growth were higher under the canopy of C. discolor and R. uncinata which in contrast to M. maximus do not suffer from leaf loss during the dry season and thus provide a more consistent foliage cover and less solar radiation underneath their canopies throughout the year (Rojas-Sandoval 2010). Areas with high solar radiation can lead to high temperatures, high evapotranspiration and low soil moisture availability, and as a result lower the probability of survival and growth for cactus seedlings (Godínez & Valiente 1998, Valiente & Ezcurra 1991, Nolasco et al. 1997, Suzán & Sosa 2006). Another alternative explanation, although not mutually exclusive, for the observed lower seedling survival and performance under M. maximus canopies relative to the native shrub species is that this grass is indeed a better competitor for water and/or nutrients than H. portoricensis seedlings (Williams & Baruch 2000, Rojas-Sandoval 2010). When they become invasive, alien grasses have been shown to influence soil’s hydrology, rates of mineralization, litter structure and may compete more efficiently for water and nutrients than native plant species (Hughes et al. 1991, D’Antonio & Vitousek 1992, Cabin et al. 2002, Levine et al. 2003, Seabloom et al. 2003). A recent study reports that regeneration of the columnar cactus species Pachycereus pectenaboriginum is inhibited at areas dominated by buffelgrass (Pennisetum ciliare) in the Sonoran Desert. For those areas, the authors suggest that interference with buffelgrass and the extraction of water from the upper soil surface by this grass may be responsible for the high levels of mortality in this cactus species (Morales-Romero & Molina-Freaner 2008).
Our results indicated that the percentages of seeds germinated and the probabilities of seedling survival for H. portoricensis under natural conditions were very low. For field experiments, seed germination was nil in open areas but even under the shade only 7.2% seeds germinated. Because temperature and soil moisture are often inversely related in the field (as temperature increases soil moisture decreases), one possibility is that in open areas direct solar radiation warms the soil and reduces soil moisture to levels below those required for germination (Valiente & Ezcurra 1991, Laksmi et al. 2003). On Mona Island, soil temperatures can reach 70ºC in open areas (Rojas-Sandoval 2010), and these elevated temperatures may also directly inhibit germination or damage seeds placed in the field. Nevertheless, experiments that control air temperatures independent from surface moisture are needed to decide on the mechanisms by which temperature influences seed germination on this species. On the other hand, even under the most optimal conditions, germination dynamics for H. portoricensis were noticeably different than what is reported for other cactus species. Literature on seed germination shows that for most cactus species, seeds germinate within a week (Rojas & Vázquez 2000). For H. portoricensis, seed germination, on average, occurred after 54 days. It is possible that germination may require ample hydration periods as a way to keep germination from occurring until environmental conditions are suitable. Dudrovsky (1996) demonstrated that seeds for five cacti species in the Sonoran Desert have a hydration memory which can allow them to tolérate dehydration and hydration events and germínate in accordance with the previous hydration experience. An alternative is that this species are naturally animal dispersed and may need scarification to trigger germination. This possibility has not been shown in columnar cacti like H. portoricensis but has been shown for cacti in the subfamily Opuntiodeae, in which germination may take few months if seeds do not pass by digestive tract of birds (Rojas & Vázquez 2000). Further studies evaluating seed longevity, dormancy capacity, and the specific conditions (i.e., light, temperature and scarification) necessary to enhance germination rates are essential for the management and conservation of this species.
Surprisingly, we found that predator exclusions did not improve the probability of H. portoricensis seedling survival but our study only evaluated this possibility at one point in time. Previous studies comparing predation on seedlings have indicated that predation intensity may vary depending on environmental conditions, intensity of the drought, latitude, and predator abundance (Valiente & Ezcurra 1991, Sosa & Fleming 2002). In addition, predation intensity on columnar cactus seedlings could also vary depending upon whether seedlings are located beneath shrubs species or in open areas (Sosa & Fleming 2002). Temporal studies that look at potential cactus-predator interactions at times where environmental conditions are more benign might be required to determine whether or not predation can be ruled out completely as a factor limiting the survivorship and growth of H. portoricensis at Mona Island.
Overall, our data suggested that for H. portoricensis, microenvironmental conditions provided by native shrubs species from the understory may play a primary role in the germination, establishments, and growth of this cactus species. During the study period rainfall was above the average annual rainfall value reported for Mona Island, thus water limitation appears not to be a factor affecting the establishment and survivorship of H. portoricensis seedlings. The presence and expansión of M. maximus may diminish the availability of environmental conditions that are optimal (i.e., mild temperatures and low solar radiation) for germination and establishment in this species (Rojas-Sandoval & Meléndez-Ackerman in preparation). African grasses are often fireadapted and are able to thrive under conditions of low water availability (Williams & Baruch 2000). The observed trends for temperature increases and the expected reductions in rainfall events for the Caribbean region as a result of the climate change (Singh 1997, Neelin et al. 2006) may be conditions that could facilitate further spread and invasion of M. maximus. Further spread of this African grass may reduce the amount of optimal environments for germination and establishment of H. portoricensis and if so present and added vulnerability beyond those listed thus far to this native endemic and threaten cactus. From a management perspective,efforts to protect H. portoricensis would also need to consider increasing in situ management of this exotic grass and the use of native shrub species as a way to enhance germination and seedling establishment.
Acknowledgments
The authors thank José Fumero-Cabán, Ricardo Rodríguez and Daniel Anglés-Alcázar for support and field assistance. We also thank Jafet Nassar and TPEE Lab members for helpful comments that improved this manuscript. This research was funded by NSF-CREST (HRD-0206200 and HRD 0734826) through the Center for Applied Tropical Ecology and Conservation (CATEC) of the University of Puerto Rico. The Department of Natural Resources and Environment of the Commonwealth of Puerto Rico provided the respective permits.
References
Bowers, J.E. 1997. Demographic patterns of Ferocactus cylindraceus in relation to substrate age and grazing history. Plant Ecol. 133: 37-48. [ Links ]
Cabin, R.J., S.G. Weller, D.H. Lorence, S. Cordell, L.J. Hadway, R. Montgomery, D. Goo & A. Urakami. 2002. Effects of light, alien grass, and native species additions on Hawaiian Dry Forest restoration. Ecol. Appl. 12: 1595-1610. [ Links ]
Cintrón, B. 1991. Introduction to Mona Island. Acta Científica 5: 6-9. [ Links ]
Cintrón, B. & L. Rogers. 1991. Plant communities of Mona Island. Acta Científica 5: 10-64. [ Links ]
D’Antonio, C.M. & P.M. Vitousek. 1992. Biological invasions by exotic grasses, the grass/fire cycle, and global chance. Ann. Rev. Ecol. Syst. 23: 63-87. [ Links ]
De la Barrera, E. & P.S. Nobel. 2003. Physiological eology of seed germination for the columnar cactus Stenocereus queretaroensis. J. Arid Environ. 53: 297-306. [ Links ]
Dubrovsky, J.G. 1996. Seed hydration memory in Sonoran Desert cacti and its ecological implication. Am. J. Bot. 83: 624-632. [ Links ]
Ewel, J.J. & J.L. Whitmore. 1973. The ecological life zones of Puerto Rico and U.S. Virgin Islands. USDA Forest Service Research Report. ITF-18, Río Piedras, Puerto Rico. [ Links ]
Franco, A.C. & P.S. Nobel. 1989. Effect of nurse plants on the microhabitat and growth of cacti. J. Ecol. 77: 870-886. [ Links ]
Frank, E.F., C. Wicks, J. Mylroie, J. Troester, E. Calvin Alexander & J.L. Carew. 1998. Geology of Isla de Mona, Puerto Rico. J. Cave Karst. Stud. 60: 68-72. [ Links ]
Fleming, T.H. 2002. Pollination biology of four species of Sonoran Desert columnar cacti, p. 207-224. In T.H. Fleming & A. Valiente-Banuet (eds.). Columnar Cacti and their Mutualists. The University of Arizona, Tucson, Arizona, USA. [ Links ]
Godínez, H. & A. Valiente. 1998. Germination and early seedling growth of the Tehuacan Valley cacti species: the role of soils and seed ingestion by dispersers on seedling growth. J. Arid Environ. 39: 21-31. [ Links ]
Godínez, H., T. Valverde & P. Ortega. 2003. Demographic trends in the Cactaceae. Bot. Rev. 69: 173-203. [ Links ]
Godínez, H., L. Ríos & F. Pérez. 2005. Characteristics of seedling establishment of Stenocereus stellatus (Cactaceae) in the Tehuacán Valley, Mexico. The Southwest. Nat. 50: 375-407. [ Links ]
Hughes, F., P.M. Vitousek & T. Tunison. 1991. Alien grass invasion and fire in the seasonal submontane zone of Hawaii. Ecology 72: 743-746. [ Links ]
Jordan, P.W. & P.S. Nobel. 1981. Seedling establishment of Ferocactus acanthodes in relation to drought. Ecology 62: 901-906. [ Links ]
Kleinbaum, D.G. & M. Klein. 2005. Survival Analysis: A self-learning text. Springer Science Media, Inc. New York, USA. [ Links ]
Lakshmi, V., T.J. Jackson & D. Zehrfuhs. 2003. Soil moisture-temperature relationship: results from two field experiments. Hydrol. Process. 17: 3041-3057. [ Links ]
Larrea, D.M. & P.J. Soriano. 2006. Spatial associations, size-distance relationship and population structure of two dominant life forms in a semiarid enclave of the Venezuelan Andes. Plant Ecol. 186: 137-149. [ Links ]
Leira, J. & V. Parra. 1999. Factors affecting the distribution, abundance and seedling survival of Mammillaria gaumeri, an endemic cactus of coastal Yucatán, México. J. Arid Environ. 41: 421-428. [ Links ]
Levine, J.L, M. Vilà, C.M. D’Antonio, J.S. Dukes, K. Grigulis & S. Lavorel. 2003. Mechanisms underlying the impact of the exotic plant invasions. Proc. R. Soc. Lon. B. 270: 775-781. [ Links ]
Liogier, H.A. 1994. Descriptive flora of Puerto Rico and adjacent islands. Editorial de la Universidad de Puerto Rico, San Juan, Puerto Rico. [ Links ]
Martinuzzi, S., W.A. Gould, O.M. Ramos, A. Martínez, P. Calle, N. Pérez-Buitrago & J.J. Fumero-Cabán. 2008. Mapping tropical dry forest habitats integrating Landsat NDVI, Ikonos imagery, and topographic information in the Caribbean Island of Mona. Rev. Biol. Trop. 56: 625-639. [ Links ]
Morales, D. & F. Molina. 2008. Influence of buffelgrass pasture conversion on the regeneration and reproduction of the columnar cactus Pachycereus pectenaboriginum, in the northwestern Mexico. J. Arid Environ. 72: 228-237. [ Links ]
Mungías, M.A. & V.J. Sosa. 2007. Nurse plants vs. nurse objects: effects of woody plants and rocks cavities on the recruitment of the Pilosocereus leucocephalus columnar cactus. Ann. Bot. 101: 175-185. [ Links ]
Murphy, P.G. & A.E. Lugo. 1995. Dry forests of Central America and the Caribbean, p. 9-34. In S.H. Bullock, H.A. Mooney & E. Medina (eds.). Seasonally Dry Tropical Forests. Cambridge University, Cambridge, United Kingdom. [ Links ]
Neeling, J.D., M. Münnich, H. Su, J.E. Meyerson & C.E. Holloway. 2006. Tropical drying trends in global warming models and observations. Proc. Natl. Acad. Sci. 103: 6110-6115. [ Links ]
Nobel, P.S. & E.G. Bobich. 2002. Environmental Biology, p. 57-74. In P.S. Nobel (ed.). Cacti: Biology and Uses. University of California, Berkeley, California, USA. [ Links ]
Nolasco, F., F. Vega-Villasante & A. Diaz. 1997. Seed germination of Stenocereus thurberi (Cactaceae) under different solar irradiation levels. J. Arid Environ. 36: 123-132. [ Links ]
Pepe, M.S. & T.R. Fleming. 1989. Weighted Kaplan-Meier statistics: A class of distance tests for censored survival data. Biometrics 45: 497-507. [ Links ]
Peters, E.M., C. Martorell & E. Ezcurra. 2008. Nurse rocks are more important than nurse plants in determining the distribution and establishment of globose cacti (Mammillaria) in the Tehuacan Valley, Mexico. J. Arid Environ. 72: 593-601. [ Links ]
Pierson, E.A. & R.M. Turner. 1998. An 85-years study of saguaro (Carnegiea gigantea) demography. Ecology 79: 2676-2693. [ Links ]
Pimienta-Barrios, E. & R.F. Del Castillo. 2002. Reproductive Biology, p. 75-90. In P.S. Nobel (ed.). Cacti: Biology and Uses. University of California, Berkeley, California, USA. [ Links ]
Rojas, M. & C.Vázquez. 2000. Cactus seed germination: a review. J. Arid Environ. 44: 85-104. [ Links ]
Rojas-Sandoval, J. 2010. Identification and evaluation of vulnerability factors affecting the Caribbean cactus species Harrisia portoricensis. Ph.D. Thesis, University of Puerto Rico, San Juan, Puerto Rico. [ Links ]
Rojas-Sandoval, J. & E. Meléndez-Ackerman. 2009. Pollination biology of Harrisia portoricensis (Cactaceae), an endangered Caribbean species. Am. J. Bot. 96: 2270-2278. [ Links ]
Rojas-Sandoval, J. & E. Meléndez-Ackerman. 2011. Windfacilitated self-pollination in Harrisia portoricensis (Cactaceae): A mechanism for reproductive assurance. Int. J. Plant Reprod. Biol. 3: 1-5. [ Links ]
SAS Institute. 2007. JMP user´s guide, version 7.0, SAS Institute, Cary, North Carolina, USA. [ Links ] Singh, B. 1997. Climate changes in the greater and southern Caribbean. Int. J. Climatol. 17: 1093-1114. [ Links ]
Seabloom, E.W., W.S. Harpole, O.J. Reichman & D. Tilman. 2003. Invasion, competitive dominance, and resource use by exotic and native California grassland species. Proc. Natl. Acad. Sci. 100: 13384-13389. [ Links ]
Steenbergh, W.F. & C.H. Lowe. 1977. Ecology of the saguaro: II. Reproduction, germination, establishment, growth, and survival of the young plants. National Park Service, Washington DC, USA. [ Links ]
Suzán, H., G.P. Nabhan & D.T. Patten. 1996. The importance of Olneya tesota as a nurse plant in the Sonoran Desert. J. Veg. Sci. 7: 635-644. [ Links ]
Suzán, H. & V.J. Sosa. 2006. Comparative performance of the giant cardon cactus (Pachycereus pringlei) seedlings under two leguminous nurse plant species. J. Arid Environ. 65: 351-362. [ Links ]
Sosa, V.J. & T.H. Fleming. 2002. Why are columnar cacti associated with nurse plants?, p. 306-323. In T.H. Fleming & A. Valiente-Banuet (eds.). Columnar Cacti and their Mutualists. The University of Arizona, Tucson, Arizona, USA. [ Links ]
US Fish and Wildlife Service (USFWS). 1990. Endangered and threatened wildlife and plant: Rules and Regulations. US Fish and Wildlife Service Report 50 CRF: 32252-32255. Federal Register, Atlanta, Georgia, USA. [ Links ]
Valiente, A., A. Bolongaro, O. Briones, E. Ezcurra, M. Rosas, H. Nuñez, G. Barnard & E. Vázquez. 1991. Spatial relationship between cacti and nurse shrubs in a semiarid environment in Central Mexico. J. Veg. Sci. 2: 15-20. [ Links ]
Valiente, A. & E. Ezcurra. 1991. Shade as a cause of the association between the cactus Neobuxbaumia tetetzo and the nurse plant Mimosa luisana in the Tehuacán Valley, Mexico. J. Ecol. 79: 961-971. [ Links ]
Valiente, A., M. Arizmendi, A. Rojas, A. Casas, H. Godínez, C. Silva & P. Dávila. 2002. Biotic Interactions and Population Dynamics of Columnar Cacti, p. 225-240. In T.H. Fleming & A. Valiente-Banuet (eds.). Columnar Cacti and their Mutualists. The University of Arizona, Tucson, Arizona, USA. [ Links ]
Valverde, T., S. Quijas, M. López & S. Castillo. 2004. Population dynamics of Mammillaria magnimamma Haworth (Cactaceae) in a Mexican lava-field. Plant Ecol. 170: 167-184. [ Links ]
Williams, D.G. & Z. Baruch. 2000. African grass invasión in the Americas: ecosystem consequences and the role of ecophysiology. Biol. Invasions 2: 123-140. [ Links ]
*Correspondencia a: Julissa Rojas-Sandoval: Department of Biology, University of Puerto Rico, Río Piedras Campus, P.O. Box 23360, San Juan, Puerto Rico 00931-3360 USA; julirs07@gmail.com
Julissa Rojas-Sandoval & Elvia Meléndez-Ackerman: Center for Applied Tropical Ecology and Conservation, University of Puerto Rico, Río Piedras Campus, P.O. Box 23341, San Juan, Puerto Rico 00931-3341 USA. Institute for Tropical Ecosystems Studies, University of Puerto Rico, Río Piedras Campus, P.O. Box 21910, San Juan, Puerto Rico 00931-1910 USA; elmend@gmail.com
1. Department of Biology, University of Puerto Rico, Río Piedras Campus, P.O. Box 23360, San Juan, Puerto Rico 00931-3360 USA; julirs07@gmail.com
2. Center for Applied Tropical Ecology and Conservation, University of Puerto Rico, Río Piedras Campus, P.O. Box 23341, San Juan, Puerto Rico 00931-3341 USA.
3. Institute for Tropical Ecosystems Studies, University of Puerto Rico, Río Piedras Campus, P.O. Box 21910, San Juan, Puerto Rico 00931-1910 USA; elmend@gmail.com
Received 27-V-2011. Corrected 20-X-2011. Accepted 23-XI-2011.