Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.60 n.2 San José Jun. 2012
Reproduction, food dynamics and exploitation level of Oreochromis niloticus (Perciformes: Cichlidae) from artisanal fisheries in Barra Bonita Reservoir, Brazil
*Dirección para correspondencia:
Abstract
Nile tilapia (Oreochromis niloticus), which is exotic to South America, is the most common species caught in artisanal fisheries at the Barra Bonita Reservoir, Southeastern Brazil. This species is of great socioeconomic importance for the region and keeps active a population of about 500 fishers. In the present study we assess reproduction, food dynamics and level of exploitation of O. niloticus, caught by artisanal fisheries in the Barra Bonita Reservoir. Specimens were collected monthly, from July 2004-June 2005, and a total of 1 715 specimens were analyzed. Each specimen was examined to obtain biological and biometric data: standard length (cm), total weight (g), reproductive data (sex and stage of maturation), and stomach contents (empty, partly full, and full). We also estimated the sex ratio (by macroscopic observation of gonads), reproductive period (by ovarian development and seasonal average of gonadosomatic index in females), and feeding habits (by stomach contents). The possible relationship between abiotic factors and the reproductive period was statistically verified using Spearman’s Rank Correlation. The FiSAT (ELEFAN I) package was used to assess growth parameters, mortality rates and to infer exploitation rate from standard length frequencies. The O. niloticus population had a sex ratio of 1.3:1 (M:F). Results indicated that ripe females were captured throughout the year, with a higher frequency during the winter-2004 (with a frequency of 59%, at a mean temperature of 20.5°C), and in spring-2004 (with a frequency of 60.5% at a mean temperature of 21.18°C). The GSI mean values obtained by season were: winter-2004: 1.71; spring-2004: 1.72; summer-2005: 0.80, and autumn-2005: 1.19. The Spearman correlation indicated positive values with respect to pH, dissolved oxygen, electric conductivity, transparency and chlorophyll a, and negative values with respect to temperature, accumulated rainfall and altimetric benchmark. The main food items were phytoplankton and periphytic algae, observed in 99.6% of the analyzed stomachs. The estimated growth and mortality parameters were: L∞=33.60cm, k=0.63/year, longevity= 4.76years, Z=2.81/ year, M=1.20/year and F=1.61/year. The weight-length relationship was Ln Wt=–2.8532+2.8835 Ln Lp. The estimated yield per recruit values were as follows: E=0.570, Emax=0.776, E0.1=0.604 and E0.5=0.349. These results indicate that a well established population of O. niloticus is present at Barra Bonita Reservoir; with an active reproduction throughout the year, more intense during winter and spring, and that O. niloticus is a phytoplanktophagus species. There were no indications that this species is being overfished, we therefore recommend that, due to its exotic condition, no restrictions need to be taken on its fishing activities. Rev. Biol. Trop. 60 (2): 721-734. Epub 2012 June 01.
Key words: Tietê river, exotic species, Nile tilapia, Oreochromis niloticus, fisheries, stock.
Resumen
La exotica tilapia del Nilo (Oreochromis niloticus) es la especie mayormente capturada en la pesqueria artesanal del embalse de Barra Bonita, sudeste de Brasil, de gran importancia socioeconomica para la region y para una poblacion de unos 500 pescadores. El estudio analizo: la reproduccion, la dinamica alimentaria y la explotación de O. niloticus, la cual fue capturada en la pesqueria del embalse de Barra Bonita. Asimismo, se tomaron muestras mensuales y se analizo un total de 1 715 animales desde julio-2004 a junio-2005. De cada especimen, se obtuvieron los siguientes datos biologicos y biometricos: longitud estandar (cm), peso total (g), datos reproductivos (sexo y estadio de maduracion), contenido estomacal (vacio, parcialmente lleno y lleno). Estos datos fueron utilizados para estimar la proporcion de sexo (a traves de la observación macroscopica de las gonadas), el periodo reproductivo (a traves de la distribucion de la frecuencia de los estadios macroscopicos del desarrollo de los ovarios), y la media de la proporcion del indice gonadosomatico IGS de hembras, asi como los habitos alimentarios (a traves de la observacion del contenido estomacal). Para verificar las posibles relaciones entre los factores abioticos y el periodo reproductivo fue aplicada la correlacion de Spearman. El software FiSAT (ELEFAN I) fue utilizado para evaluar los parametros de crecimiento, tasa de mortalidad e inferir el grado de explotacion, en donde se usaron los datos de frecuencia de longitud estandar. La poblacion de O. niloticus presento una proporcion de sexo 1.3:1 (M:H). Los resultados indicaron que hembras fueron muestreadas durante todo el ano, pero ha sido mas frecuente en el invierno-2004-59.0% (temperatura media=20.05°C) y primavera-2004-60.5% (temperatura media=21.18°C). Los valores medios del IGS fueron: invierno-2004=1.71, primavera- 2004=1.72, verano-2005=0.80 y otoño-2005=1.19. La correlación de Spearman fue positiva para pH, oxigeno disuelto, conductividad electrica, transparencia y clorofila a, y negativo para temperatura, pluviosidad acumulada y variacion del nivel del agua en el embalse. Los principales items de alimentación fueron fitoplancton y algas perifiticas observadas en 99.6% de los estómagos analizados. Los parámetros estimados del crecimiento y mortalidad fueron los siguientes: L∞=33.60cm, k=0.63/año, longevidad= 4.76 años, Z= 2.81/año, M=1.20/año y F=1.61año. La relación peso-longitud fue Ln Wt=-2.8532+2.8835 Ln Lp. Los valores de produccion por reclutamiento estimado fueron=0.570, Emax=0.776, E0.1=0.604 y E0.5=0.349. Estos resultados indican que la poblacion de O. niloticus esta bien establecida en el embalse de Barra Bonita. Ademas, su reproduccion ocurre durante todo el ano, pero es mas intensa en el invierno y primavera; su dieta tiene como base el fitoplancton. Los resultados indican que no esta ocurriendo sobrepesca de O. niloticus, por tanto, recomendamos que, debido a su naturaleza exotica, no se tomen restricciones a la hora de su pesca.
Palabras clave: Rio Tietê, especie exotica, tilapia del Nilo, Oreochromis niloticus, poblaciones de peces.
Oreochromis niloticus (Linnaeus 1758) (Nile tilapia), with a known ability to adjust to various environmental conditions and its high potential for aquaculture, can now be found in many reservoirs in almost all continents (McCrary et al. 2007). In Brazil, O. niloticus was first introduced in the 1930s to increase fish production in the Northeastern dams (Lovshin et al. 1976), and into the upper Paraná River in the 1950s, also with the purpose of increasing fish production (Carvalho et al. 2005). In the 1970s, following legal requirements, the Energetic Company of Sấo Paulo (Companhia Energetica de Sấo Paulo-CESP) began a fish stocking program in the reservoirs under its grant, introducing a number of tilapia species, including O. niloticus and other exotic species, into ten reservoirs of the Tietê, Paranapanema, and Grande Rivers (CESP 1998).
Oreochromis niloticus is now well established in a number of Brazilian reservoirs that are of prime importance to artisanal fisheries: dams in the Northeast (Paiva et al. 1994), at the Billings Reservoir (Minte-Vera & Petrere Jr. 2000), in Pampulha Lagoon (Alvares et al. 2000), Paranoá Lake (Walter & Petrere Jr. 2007) and in the Barra Bonita Reservoir (Petesse et al. 2007). Oreochromis niloticus is the most important species of the artisanal fisheries in the Barra Bonita Reservoir, producing a yield estimated at 15tons per day. This supports the livelihoods of about 500 fishers and has great socioeconomic relevance for the region (Novaes 2008). It is thus important to understand the biology of this species and to evaluate its stock in the reservoir. The elimination of O. niloticus from the reservoir would be extremely difficult, if not impossible. Results from this study may, however, assist in the management and control the species in order to ameliorate environmental impacts.
The objective of this paper was to characterize the population dynamics of O. niloticus in the Barra Bonita Reservoir. The artisanal fishery landings, and the population reproduction (reproductive period and sex ratio), feeding, and growth were studied. Besides, parameters associated with mortality (total mortality, natural mortality, and fishing mortality) were analyzed and the current status of the stock was assessed, using Beverton & Holt’s model of analysis of yield per recruit.
Materials and methods
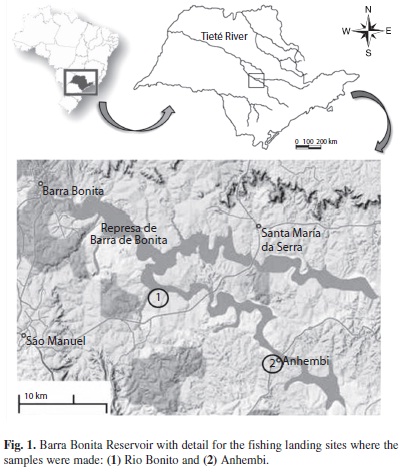
Data collection: Sampling was conducted at the Barra Bonita Reservoir (22°31’10’’ S -48°32’03’’ W), for which the Tietê and Piracicaba Rivers are the main tributaries (Fig. 1). Due to ongoing releases of high volumes of untreated domestic sewage, the reservoir is currently considered as hypertrophic (Straškraba & Tundisi 2000). Monthly samples were collected from July 2004 to June 2005 at two fish landing sites: in Rio Bonito (22°40’52.1’’ S- 48°18’16.2 W) and in Anhembi (22°47’10’’S- 48°07’27’’) (Fig. 1). After the arrival of fishers, a total of 20kg (about 80 individuals) of O. niloticus caught in lacustrine environment was randomly chosen for analysis and included in a box for transportation. For each specimen, total weight (g), standard length (cm), sex, and maturation status (according to the scale described in Helfman et al. 2007) were recorded. Besides, full stomachs were fixed in 5% formaldehyde for later analysis of stomach contents. A total of 1 715 specimens were analyzed. Data was grouped by to analyze reproduction and feeding by season: winter-2004 (July, August, September); spring-2004 (October, November, December); summer-2005 (January, February, March), and autumn-2005 (April, May, June).
Reproductive dynamics: The sex ratio and the sex frequency determined (N=1 687, immature specimens) were obtained by macroscopic observation of gonads. For the reproductive biology analysis only females (N=744) were used, because they are better indicators of the reproductive period (Wootton 1995). The reproductive period was evaluated using the following techniques: distribution and frequency of macroscopic stages (‘immature’, ‘mature’, ‘ripe’, ‘spent’ and ‘at rest’) of the ovarian development, and seasonal average of the gonadosomatic index in females (GSI=the gonad weight, expressed as a percentage of body weight).
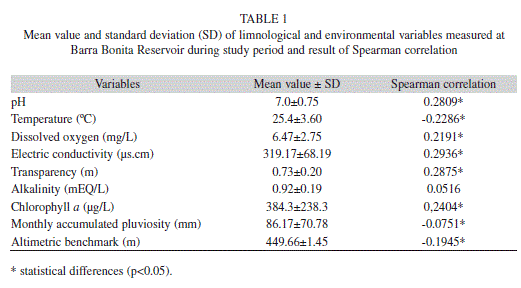
Physicochemical parameters: These were considered to determine possible influences of the environmental and/or limnological variables on the reproduction dynamics of this species. Limnological variables and water samples were obtained at a distance of approximately 50m from the river bank. A water quality analyzer (Horiba model U22) was used to measure in situ the following parameters: pH, water temperature, dissolved oxygen, and electrical conductivity. Water transparency was also determined with a Secchi disk. Additionally, water samples (of about 500mL) were collected in appropriate bottles for total alkalinity and chlorophyll a (Chl a) content determination. In the laboratory, total alkalinity was estimated by titration with 0.1 N H2SO4, and Chl a; was determined using Millipore AP20 membranes and cold acetone (90%) extraction (see Nogueira et al. 1999). Information about the monthly accumulated rainfall and the reservoir’s water level benchmark were obtained from the concessionaire of Barra Bonita Hydroelectric Plant (AES Tietê S/A). Results from the limnological and environmental monitoring programme were correlated with the GSI of females (N=744).
Feeding: The contents of 180 stomachs were analyzed using the Neubauer chamber, according to Tavares & Rocha (2001). To quantify food items we followed the method of frequency of occurrence by Hyslop (1980). The classification of microscopic algae followed Round’s system, using the classification key described in Bicudo & Menezes (2006). Algae were identified to the Division taxonomic level.
Length-weight relationship: For this and the following analysis, all available specimens (N=1 715) were used. The length-weight relationship was calculated with the next mathematical expression, after a logarithmic transformation of the data:
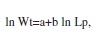
where Wt=total weight, Lp=standard length, a=a constant and b=inclination of slope.
Growth parameters and mortality estimates: Based on monthly distributions of standard length frequency, the following growth parameters were obtained: L¥=asymptotic standard length (cm), and k=growth coefficient (year). These parameters were evaluated using the ELEFAN I routine within the computer program FiSAT II (Gayanilo et al. 2005), which is based on the von Bertalanffy equation:
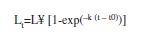
where Lt =length in age t, L¥=asymptotic standard length, k=growth coefficient and t0=fish length at birth.
The index of growth performance (Φ’) was quantified using the model proposed by Pauly & Munro (1984):
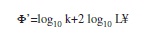
where k is a growth constant per year and L¥ is the asymptotic length in cm.
The longevity potential was estimated according to the equation of Pauly (1980): Tmax=3/K. The length-age structure was evaluated using the von Bertalanffy equation (described above). We used the parameters L∞ and K calculated in this paper, and replaced t with ages ranging from 0-5 years. The parameter t0 was considered as zero, because it is not biologically significant (Sparre & Venema 1997).
The natural mortality rate was evaluated using Pauly’s (1980) empirical model:
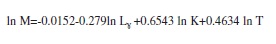
where L¥ and k are the growth parameters obtained from the von Bertalanffy equation and T=mean water temperature (°C), which was 24.6°C.
Total mortality rate (Z) was estimated using Beverton and Holt’s model:
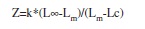
where Lc=mean length at first capture, and Lm=mean length starting from Lc.
For the purpose of this study, Lc was taken as 15.0cm and Lm was 18.7cm. The mortality rate due to fishing (F) was calculated as the difference between the total mortality rate (Z) and the natural mortality rate (M) (i.e., F=Z-M).
Relative yield per recruit (Y’/R) and relative biomass per recruit (B’/R): Y’/R was calculated using Beverton & Holt’s method, modified by Pauly & Soriano (1986), and the Knife Edges option of the program FiSAT II (Gayanilo et al. 2005), according to the following model:
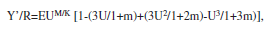
where m=(1-E)/(M/k)=k/Z, U=1-(Lc/L¥) and E=F/Z (exploitation rate).
We also calculated Emax (exploitation rate of maximum sustainable yield), E0.1 (exploitation rate at which the marginal increment of Y’/R is 10% of its virgin stock) and E0.5 (exploitation rate that will result in a 50% reduction of the non-exploited biomass). The relative biomass per recruit (B’/R) was estimated as B’/R=(Y’/R)/F. For these estimates we used the routine ELEFAN I in the program FiSAT II (Gaynilo et al. 2005).
The x2 test was used to evaluate if the male to female ratio of the population was 1:1. Before applying parametric tests, the data were transformed (ln) and their normality and homogeneity were analyzed using Kolmogorov-Smirnov’s and Bartlett’s tests, respectively. A one-way ANOVA was applied to test the null hypothesis of equal GSI values for different periods of the year and Tukey test was used to detect statistical differences. The t-test was applied to test whether the value of b of the length-weight relationship was equal to zero and whether b=3 (the condition of isometric growth). Because the data for the abiotic factors did not meet the assumption of variance normality and homogeneity, Spearman’s non-parametric correlation test was applied to examine possible relationships between abiotic factors and reproductive period, through values of GSI. For all statistical analyses p<0.05.
Results
Reproductive dynamics: Males comprised 56.1% (n=946) and females 43.9% (n=741) of the catches, with a sex ratio (M:F) of 1.3:1 (x2=24.911, d.f.=1, p<0.000). Seasonally, females predominated in winter-2004, comprising 251 (61.5%) and males 157 (38.5%) of the total; M:F=0.6:1, x2=21.657, d.f.=1, p<0.0000. Nevertheless, males predominated in the other seasons. In spring-2004 males comprised 246 (62.6%) and females 147 (37.4%) with M:F=1.7:1, x2=24.939, d.f.=1 and p< 0.0000. In summer-2005, males represented 316 (69.5%) and females 139 (30.5%), M:F=2.3:1, x2=68.855, d.f.=1 and p <0.0000. In autumn-2005, males were 227 (52.7%) and females 204 (47.3%), M:F=1.1:1, x=1.227, d.f.=1 and p<0.2893.
The distribution frequency of gonadal stages indicated that ripe females were present throughout the year, but relative frequencies of females were higher in the winter-2004 (N=148, 59.0%) and in spring-2004 (N=89, 60.5%) (Fig. 2a). Average values of GSI were as follows: winter-2004: 1.71, spring-2004: 1.72, summer-2005: 0.80 and autumn-2005: 1.19. The ANOVA showed statistical differences between GSI values among seasons (F(3:733)=30.754, p<0.0000) (Fig. 2b).
Physicochemical parameters: Spearman correlation analysis indicated that average values of GSI were positively related to pH, dissolved oxygen, conductivity, water transparency and Chl a; and negatively correlated to temperature, precipitation, and water level benchmark altimetry (Table 1). When wáter level was plotted against GSI, it was observed that GSI values presented a response to the hydrologic cycle at Barra Bonita Reservoir, with a two month delay for both increases and decreases of values (Fig. 3).
Feeding: Dietary items found in the stomachs of O. niloticus consisted almost exclusively of phytoplankton and periphytic algae (99.6%) with a low presence of zooplankton (0.4%). The algae group Crysophyta was predominant in winter-2004 (64.9%) and moderately high in spring-2004 (39.1%). The Cyanophyta, particularly the filamentous types, predominated in summer-2005 (41.3%) and autumn-2005 (62.9%) (Fig. 4). A large amount of sediment was present in fish stomachs, suggesting that O. niloticus may search for food along the bottom of the reservoir.
Length-weight relationship: The standard length of O. niloticus varied from 11.0cm-31.2cm (Lpmaen=18.3cm, SD=2.1), but most of the specimens were between 17.0cm-17.9cm. The length-weight relationship can be described by the formula: ln Wt=-2.8532+2.8835 ln Lp, where R2=0.8941. The t-test indicated a value of b that differed from zero (t(1:1713) =119.28, p=0.00) and three (the condition of isometric growth) (t (1:1713)=75.76, p=0.00), thus indicating negative allometric growth.
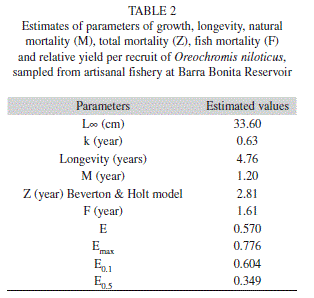
Parameters of growth and mortality estimates: Growth parameters were as follows: L∞=33.60cm, k=0.63/year (Table 2). The length growth equation was as follows: Lt=33.60[1exp(0.63 (t–t0))]. The length growth curve suggested the presence of two welldefined cohorts (Fig. 5). The index of growth performance was estimated to be Φ’=2.85 (Table 2).
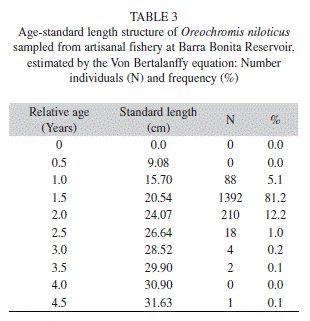
Longevity potential was estimated to be tmax=4.76 years (Table 2). The length-age structure analysis indicated that most O. niloticus caught in this fishery were 1-2 years old (98.5%), of which 81.2% were about 1.5 years old (Table 3).
The values for instantaneous total mortality rate (Z), natural mortality rate (M) and fishing mortality rate (F) were estimated to be 2.81/year, 1.20/year and 1.61/year, respectively (Table 2). Mortality due to fishing was more important than the other causes of mortality (F>M). Table 4 provides a comparison, based on parameters for growth and mortality, between this study and other studies in O. niloticus.
Relative yield per recruit (Y’/R) and relative biomass per recruit (B’/R): The values of E (0.570) and Emax (0.776) (Table 2) indicate that overfishing of O. niloticus is not occurring in the Barra Bonita Reservoir (i.e., E <Emax). It was, however, noted that the value of E exceeded the value of E0.5 (0.349) and was very close to E0.1 (0.604) (Table 2).
Discussion
Reproductive dynamics: The sex ratio of most fish species in the wild tends to be 1:1, but deviations can occur and seasonal variations are common (Helfman et al. 2007). The sex ratio is influenced by several factors, including mortality, longevity and growth rate; these in turn lead to differences in the catch rate (King & Etim 2004). Differences in sex ratio for tilapia species have been verified in introduced populations such as O. niloticus in Mexico (Peña-Mendoza et al. 2005) and in Lake Victoria, Kenya (Njiru et al. 2006) and in natural populations, such as Tilapia mariae in Nigeria (King & Etim 2004) and O. esculentus in Lake Victoria and Lake Kyoga (Nagayi-Yawe et al. 2006). Two possible reasons may account for the sex ratio observed in our study. Firstly, it may relate to the reproductive strategies of the population and to differences in the dispersal of males and females after completion of the reproductive process. Lowe-McConnell (1987) reported that males make nests in shallow water during the reproductive process. Females release the eggs immediately after spawning (the eggs are usually fertilized in their mouth) and remain in the beds, or move to safer places for incubation and protection of the spawn. The males are, however, more likely to be caught because they disperse when searching for food. This hypothesis might explain the differences in sex ratio of O. niloticus in Lake Victoria (Njiru et al. 2006) and in the Emiliano Zapata Reservoir in Mexico (Peña-Mendoza et al. 2005). Secondly, differential growth rates in males and females (Wootton 1995, King & Etim 2004), with higher rates being found in males, may mean that males are more susceptible than females to capture by fishing gears (10cm mesh with opposite knots). We performed exploratory data analysis, which did not reveal significant differences in the growth rates of O. niloticus males and females, in the Barra Bonita Reservoir. Thus, the differences in the sex ratio observed in this study are likely to relate more to reproductive behavior than to growth rates.
An analysis of average values of GSI and the frequency distribution of maturation stages indicated that O. niloticus reproduced throughout the year in the Barra Bonita Reservoir, with reproduction peaks in winter and spring. A long reproductive period for this species was reported in other ecosystems in which it was introduced, although reproductive peaks varied among localities (Cala & Bernal 1997, Barbieri et al. 2000a, Duponchelle et al. 2000, Gomez-Marquez et al. 2003, Peterson et al. 2004, Pena-Mendonza et al. 2005, Njiru et al. 2006, Komolafe & Arawono 2007, Kwarfo-Apegyah & Ofori-Danson 2010).
Physicochemical parameters: Exogenous factors sometimes influence endogenous reproductive processes. Some of these, such as temperature, water flow, water level, and precipitation, can serve as triggers for tropical fish reproduction (Lowe-McConnell 1987, Wootton 1995). In reservoirs in the Ivory Coast (Duponchelle et al. 2000) and Ghana (Kwarfo-Apegyah & Ofori-Danson 2010), the reproductive period of O. niloticus is associated with high temperature, precipitation, water level, and higher photoperiod during summer. In the rivers of the state of Mississippi in the USA, the reproductive peak occurs in spring, when the temperature is over 22°C (Peterson et al. 2004). In the Guarapiranga Reservoir (Brazil), greater reproductive activity is associated with the rainy season and higher temperatures in summer (Barbieri et al. 2000a). Our results did not identify a positive relationship between average values of GSI in females and the water level, we believe that this variable is one of the main triggers of the increase in reproductive activity of O. niloticus in the Barra Bonita Reservoir during winter and spring. Because, of its position, the Barra Bonita Reservoir controls water levels in other reservoirs of the cascade system, resulting in a reversal of water flow regime compared to that found in natural environments, i.e. the lowest water levels in the reservoir occur between September and December. The water level of the reservoir began to rise in January 2005 and reached máximum capacity from June to July, 2005. The values of GSI were a response to this hydrologic cycle, with a two-month delay in the case of both increasing and decreasing values.
Feeding: Our analyses revealed that O. niloticus in the Barra Bonita Reservoir is phytoplanktophagus, which is in agreement with results from African (Bwanika et al. 2004, Njiru et al. 2004) and Asian reservoirs and lakes (Weliange & Amarasinghe 2003). The presence of large quantities of sediment in the stomach contents suggests, however, that O. niloticus may also be taking food from the bottom of the reservoir, where accumulated organic matter is available, often in large quantities (Hahn & Fugi, 2007, Vidotto & Carvalho 2009). Thus in addition to phytoplankton and periphytic algae, bacteria, protozoa and fungi from the sediment may also be a source of food for O. niloticus.
Dietary analysis indicated seasonal differences in diet, with a predominance of chrysophytes during winter and spring-2004 and cyanophytes in summer and autumn-2005. Seasonality in diet is a result of variation in the availability of food in the environment. These variations can occur for many reasons, including life cycle changes of prey, changes in the predators preying on the food item, and changes in the foraging habitat (Wootton 1995). In Sri Lankan reservoirs, the diet of O. niloticus varied from phytoplankton to zooplankton to detritus, depending on the availability of these items in the environment (Weliange & Amarasinghe 2003). In Lake Victoria, phytoplankton and zooplankton, insects, vegetation, and fish were also part of the diet of this species (Njiru et al. 2004). The phytoplankton community in the Barra Bonita Reservoir consisted mainly of chrysophytes, cyanophytes, and chlorophytes and the availability of these algal groups varied seasonally (Matsumura-Tundisi & Tundisi 2005). Although O. niloticus feeds mostly on phytoplankton throughout the year, it is also well adapted to seasonal variations in the availability of various food items. Thus, we infered that the population of O. niloticus was able to survive on food resources available at Barra Bonita Reservoir along the year.
Length-weight relationship: Samples caught in the fishery were mainly adult and they had probably already reproduced at least once; few young individuals were caught. The O. niloticus fishery does not focus on Young fish. Accordingly, fishery pressure does not yet seem to be a threat to the stock. On the other hand, fishery pressure on larger individuals may result in genetic degradation in the population. The removal of larger individuals favors the survival of smaller individuals and those that grow more slowly. This changes the genetic variability of the population, because it eliminates genes that promote rapid growth (Conover & Munch 2002). Furthermore, older and larger females tend to spawn earlier and lay more eggs. Larvae from these eggs grow three times faster, and are more resistant to periods of low food availability, than those born from young females (Berkeley et al. 2004). Thus the conservation of larger individuals, which are currently the target of the fishery due to fisheries legislation, is crucial for the maintenance of fish stocks.
The b value of O. niloticus (2.8838) was statistically different from three. This is an indicative of negative allometric growth (i.e. the species exhibited faster gains in lengthgrowth than in weight). In contrast, Njiru et al. (2006) reported positive allometric growth in this species in Lake Victoria, and in the Betania Reservoir in Colombia (Cala & Bernal 1997), and the Guarapiranga Reservoir in Brazil (Barbieri et al. 2000b), isometric growths were reported. Values of b often oscillate between 2.5 and 3.5, depending on the species. Natural intraspecific variation, in terms of sex and age and the influence of season, type and availability of food and reproductive period, has been observed (Laleye 2006). For these reasons research workers need to be cautious when interpreting the meaning of b, because temporal variations in the value may occur naturally. Santos et al. (2004) studied Geophagus brasiliensis (a Brazilian cichlid) in reservoirs and concluded that stress caused by variations in the reservoir level, may have contributed to the negative allometric growth observed for this species. Water level fluctuations in the Barra Bonita Reservoir were estimated at about five meter. The hypothesis that stress, induced by variation in the fluctuation of the reservoir’s water level, affected O. niloticus growth is thus feasible. This could be due to more energy being allocated to reproduction than to growth, causing a decrease in the weight of individuals.
Parameters of growth and mortality estimates: Growth parameters for O. niloticus, estimated using FiSAT, were biologically feasible, because the growth performance rate (Φ’=2.85) was within the range estimated for other cichlid populations (Moreau et al. 1986, De Silva et al. 1988, Amarasinghe & De Silva 1992, Amarasinghe 2002, King & Etim 2004). The growth parameters estimated for the species (L∞=33.6cm, k=0.63/year and longevity 4.7 years) indicated rapid growth. According to some authors (Lowe-McConnell 1987, Winemiller 1989) species at a low trophic level, such as O. niloticus, tend to have rapid growth, short longevity, and early sexual maturation, that applies to the present results. When we compared the growth parameters (L∞ and k) and the mortality rates (Z, M and F) estimated for O. niloticus in our study, with those estimated in other studies, several differences were apparent. Various endogenous and exogenous factors influence fish growth (Helfman et al. 2007) and these may be responsible for the observed differences. The level of fishing effort is a variable that clearly affects growth and mortality rates (Sparre & Venema 1997). This factor was identified as the cause of differences (in terms of growth and mortality rates) between species of Oreochromis in two Asian lakes (Amarasinghe 2002). It is likely that high fishing efforts (Maruyama et al. 2009) will affect the growth and mortality rates of O. niloticus in the Barra Bonita Reservoir.
Relative yield per recruit (Y’/R) and relative biomass per recruit (B’/R): The value of Y/R’ in our study indicates that stock is not being over-fished. The observations that E is greater than E0,5, that E is very close to E0,1, and that F is greater than M, all suggest that the current fishing effort is very close to the maximum sustainable level. The artisanal fishing effort in the Barra Bonita Reservoir currently includes a contingent of about 500 fishers and 200 boats (Maruyama et al. 2009), which represents the maximum effort for the artisanal fisheries in this reservoir. It is necessary to take appropriate management steps so that the artisanal fishery does not decline in the reservoir.
Based on present results, we came to a number of conclusions: (i) O. niloticus is well established in the studied areas of the reservoir; (ii) The long reproductive period (more intense in winter and spring) and the phytoplankton feeding habit characterized in this study agree with results from other studies on O. niloticus; (iii) The fish stock is not currently over-fished, but the level of exploitation by the artisanal fisheries is close to the maximum sustainable level. Since O. niloticus is exotic to the Barra Bonita Reservoir, conservation measures to protect this species, such as temporary prohibition of fishing, prohibition of fishing gear, controls of fish size caught and control of fishing effort, are not recommended for this locality.
Acknowledgments
We wish to thank the fishers at Barra Bonita Reservoir; the entire team of the Laboratory for Fish Ecology of UNESP-Botucatu; and the Coordenacao de Aperfeicoamento de Pessoal de Nivel Superior (CAPES) for a grant.
References
Alvares, E.S.S., M.A.S. Canelas, R.G. Costa, L.G. Dias, H.N. Ribeiro-Mendes & R.M. Pinto-Coelho. 2000. A pesca na represa da Pampulha, p. 375-382. In Anais do V Simposio de Ecossistemas Brasileiros: Conservacao Vol. 3. Belo Horizonte, Minas Gerais, Brasil. [ Links ]
Amarasinghe, U.S. & S.S. De Silva. 1992. Population dynamics of Oreochromis mossambicus and O. niloticus (Cichlidae) in two reservoirs in Sri Lanka. Asian Fish. Sci. 5: 37-61. [ Links ]
Amarasinghe, U.S. 2002. The fishery and population dynamics of Oreochromis mossambicus and Oreochromis niloticus (Oesteichthyes, Cichlidae) in a shallow irrigation in Sri Lanka. Asian Fish. Sci. 15: 7-20. [ Links ]
Barbieri, G., A.R. Teixeira Filho, E.C. Campos, H. Vermulm Jr. & M.T.D. Giamas. 2000a. Biologia populacional da tilapia, Oreochromis niloticus, represa de Gurapiranga, Sao Paulo–II. Dinamica da reproducao. B. Inst. Pesca 26: 9-13. [ Links ]
Barbieri, G., A.R. Teixeira Filho, E.C. Campos, H. Vermulm Jr. & M.T.D. Giamas. 2000b. Biologia populacional da tilapia, Oreochromis niloticus, represa de Gurapiranga, Sao Paulo–I. Estrutura populacional, idade e crescimento. B. Inst. Pesca 26: 1-7. [ Links ]
Berkeley, S.A., C. Chapman & S.M. Soagard. 2004. Maternal age as a determinant of larval growth and survival in a marine fish, Sabastes melanops. Ecology 85: 1258-1264. [ Links ]
Bicudo, C.E. de M. & M. Menezes. 2006. Gêneros de algas de água continental do Brasil. Chave para identificação e descrições. RiMA, São Carlos, São Paulo, Brasil. [ Links ]
Bwanika, G.N., B. Makanga, Y. Kizito, L.J. Chapman & J. Balirwa. 2004. Observations on the biology of Nile tilapia, Oreochromis niloticus L., in two Ugandan crater lakes. Afr. J. Ecol. 42: 93-111. [ Links ]
Cala, P. & G. Bernal. 1997. Ecologia y adaptaciones de la tilapia nilotica (Oreochromis niloticus) en ambientes naturales–Caso embalse de Betania y cienaga de chilloa, sistema del rio Magdalena, Colombia. Dahlia 2: 3-29. [ Links ]
Carvalho, E.D., S.G de C. Britto & M.L Orsi. 2005. O panorama das introduções de peixes na bacia hidrografica do rio Paranapanema, Alto Paraná, Brasil, p. 253-274. In O. Rocha, E.L.G. Espíndola, N. Fenerich-Verani, J.R. Verani & A.C. Reitzler (eds.). Especies invasoras em águas doces–estudo de caso e propostas de manejo. Editora da Universidade São Carlos, São Carlos, São Paulo, Brasil. [ Links ]
CESP 1998. Conservação e manejo nos reservatorios: limnologia, ictiologia e pesca. Serie Divulgação e Informação, 220. Companhia Eletrica de São Paulo, Departamento de Estudos e Planejamento Ambiental. São Paulo, São Paulo, Brasil. [ Links ]
Conover, D.O. & S. Munch. 2002: Sustaining fisheries yields over evolutionary time scales. Science 297: 94-96. [ Links ]
De Silva, S.S., J. Moreau & K.A.D.W. Senaratne 1988. Growth of Oreochromis mossambicus (Pisces, Cichlidae) as evidence of its adaptability to Sri Lankan reservoirs. Asian Fish. Sci. 1: 47-156. [ Links ]
Duponchelle, F., P. Cecchi, D. Corbin, J. Nunez & M. Legendre. 2000. Variations in fecundity and eggs size of female Nile tilapia, Oreochromis niloticus, from man-made lakes of Cote d’Ivoire. Environ. Biol. Fish. 57: 155-170. [ Links ]
Gayanilo, F.C. Jr., P. Sparre & D. Pauly, D. 2005. FAOICLARM stock assessment tools II (FiSAT II). Revised version. User’s guide. FAO Computerized Information Serie (Fisheries). No 8, FAO, Roma, Italia. [ Links ]
Gomez-Marquez, J.L., B. Peña-Mendoza, I.H. Salgado-Urgate ; M. Guzman-Arroyo. 2003. Reproductive aspects of Oreochromis niloticus (Perciformes: Cichlidae) at Coatetelco lake, Morelos, Mexico. Rev. Biol. Trop. 51: 221-228. [ Links ]
Hahn, N.S. & R. Fugi. 2007. Alimentação de peixes em reservatórios brasileiros: alterações e conseqüências nos estágios iniciais de colonização. Oecolg. Bras. 11: 469-480. [ Links ]
Helfman, G.S., B.B. Collette, D.E. Facey & B.W. Bowen. 2007. The diversity of fish: Biology, Evolution and Ecology. Wiley-Blackwell, Oxford, England. [ Links ]
Hyslop, E.J. 1980. Stomach Contents Analysis – a Review of Methods and their Application. J. Fish. Biol. 17: 411-429. [ Links ]
King, R.P. & L. Etim. 2004. Reproduction, growth, mortality and yield of Tilapia mariae Boulenger 1899 (Cichlidae) in a Nigerian rainforest wetland stream. J. Appl. Ichthyol. 20: 502-510. [ Links ]
Komolafe, O.O. & G.A.O. Arawono. 2007. Reproductive strategy of Oreochromis niloticus (Pisces: Cichlidade) in Opa Reservoir, Ile-Ife, Nigeria. Rev. Biol. Trop. 55: 595-602. [ Links ]
Kwarfo-Apegyah, K., P.K. Ofori-Danson & F.K.E. Nunoo. 2008. Exploitation rates and management implications for the fisheries of Bontanga Reservoir in the Northern region of Ghana. W. Afr. J. Appl. Ecol. 14: 1-7. [ Links ]
Kwarfo-Apegyah, K. & P.K. Ofori-Danson. 2010. Spawning and recruitment patterns of major fish species in Bontanga Reservoir, Ghana, West Africa. Lake Reserv. Manage. 15: 1-14. [ Links ]
Laleye, P.A. 2006. Length-weight and length-length relationships of fish from the Oueme River in Benin (West Africa). J. Appl. Ichthyol. 22: 502-510. [ Links ]
Lovshin, L., J.T. Peixoto & E.A. Vasconcelos. 1976. Considerações ecológicas e econômicas sobre Tilápia sp. no Nordeste do Brasil, p. 227-237. In J.I. Vargas, C.G.C. Loureiro & R.M. de Andrade (eds.). Anais do I encontro nacional sobre limnologia, piscicultura e pesca continental. Fundacao Joao Pinheiro, Centro de Recursos Naturais. Belo Horizonte, Brasil. [ Links ]
Lowe-McConnell, R.H. 1987. Ecological studies in tropical fish communities. Cambridge University, Cambridge, England. [ Links ]
Maruyama, L.S., P.M.G. Castro & P. Paiva. 2009. Pesca artesanal no medio e baixo Tietê, São Paulo, Brasil: aspectos estruturais e socioeconomicos. B. Inst. Pesca 35: 61-81. [ Links ]
Matsumura-Tundisi, T. & J.G. Tundisi. 2005. Plankton richness in a eutrophic reservoir (Barra Bonita, reservoir, SP, Brazil). Hydrobiologia 542: 367-378. [ Links ]
McCrary, J.K., B.R. Murphy, J.R. Stauffer Jr. & S.S. Hendrix. 2007. Tilapia (Teleostei: Cichlidae) status in Nicaraguan natural waters. Environ. Biol. Fish. 78: 78-107. [ Links ]
Minte-Vera, C.V. & M. Petrere Jr. 2000. Artisanal fisheries in urban reservoirs: a case study from Brazil (Billings Reservoir, São Paulo metropolitan region). Fish. Manag. Ecol. 7: 537-549. [ Links ]
Moreau, J., C. Bamboni & D. Pauly. 1986. Indices of overall growth performance of 100 tilapia (Cichlidae) population, p. 201-206. In J.L. Maclean, L.B. Dizon & L.V. Hosillo (eds.). The First Asian Fisheries Forum. Asian Fisheries Society, Manila, Philippines. [ Links ]
Nagayi-Yawe, K.J., R. Ogutu-Ohwayo, Y.S. Kizito & J.S. Balirwa 2006. Population characteristics of Oreochromis esculentus in the Victoria and Kyoga lake basins. Implications for conservation and improvement of the stocks. Afr. J. Ecol. 44: 423-430. [ Links ]
Njiru, M., J.B. Okeyo-Owuor, M. Muchiri & I.G. Cowx. 2004. Shifts in the food of Nile tilapia, Oreochromis niloticus (L.) in Lake Victoria, Kenya. Afr. J. Ecol. 42: 163-170. [ Links ]
Njiru, M., J.E. Ojuok, J.B. Okeyo-Owuor, M. Muchiri, M.J. Ntiba & I.G. Cowx. 2006. Some biological aspects and life history strategies of Nile tilapia Oreochromis niloticus (L.) in Lake Victoria, Kenya. Afr. J. Ecol. 44: 30-37. [ Links ]
Nogueira, M.G., R. Henry & F.E. Maricatto. 1999. Spatial and temporal heterogeneity in the Jurumirim reservoir, São Paulo, Brazil. Lake Reserv. Manage. 4: 107-120. [ Links ]
Novaes, J.L.C. 2008. Estudo comparativo da pesca artesanal em dois grandes reservatorios do alto Paraná: Barra Bonita (rio Tietê) e Jurumirim (rio Paranapanema). Ph.D. Thesis, Instituto de Biociências, Universidade Estadual Paulista, Botucatu, Sao Paulo, Brasil. [ Links ]
Ofori-Danson, P.K. & K. Kwarfo-Apegyah. 2008. An assessment of the Cichlid fishery of Bontanga Reservoir, Northern Ghana. W. Afr. J. Appl. Ecol. 14: 8-17. [ Links ]
Paiva, M.P., M. Petrere Jr., A.J. Petenate & F.H. Nepomuceno. 1994. Relationship between the number of predatory of species and fish yield in large North-eastern Brazilian reservoirs, p. 120-129. In I.G. Cowx (ed.). Rehabiliation of Freshwater Fisheries. Fishing News Books, London, England. [ Links ]
Pauly, D. 1980. On the interrelationships between natural mortality, growth parameters and mean environmental temperature in 175 stocks. J. Cons. Int. Expl. Mer. 39: 175-192. [ Links ]
Pauly, D. & J.L. Munro. 1984. Once more on the comparison of growth in fish and invertebrates. Fishbyte 2: 21. [ Links ]
Pauly, D. & M.L. Soriano. 1986. Some pratical extensions to Beverton and Holt’s relative yield-per-recruit model, p. 491-496. In J.L. Maclean, L.B. Dizon & L.V. Hosillo (eds.). The First Asian Fisheries Forum. Asian Fisheries Society, Manila, Philippines. [ Links ]
Peña-Mendoza, B., J.L. Gomez-Marquez, I.H. Salgado-Ugarte & D. Ramirez-Noguera. 2005. Reproductive biology of Oreochromis niloticus (Perciformes: Cichlidae) at Emiliano Zapata dam, Morelos, Mexico. Rev. Biol. Trop. 53: 515-522. [ Links ]
Peterson, M.S., W.T. Slack, N.J. Brown-Peterson & J.L. McDonald. 2004. Reproductive in nonnative environments: Establishment of Nile tilapia, Oreochromis niloticus, in Coastal Mississippi watersheds. Copeia 4: 842-849. [ Links ]
Petesse, M.L., M. Petrere Jr. & R.J. Spigolon. 2007. The hydraulic management of the Barra Bonita Reservoir (SP, Brazil) as a factor influencing the temporal secession of its fish community. Braz. J. Biol. 67: 433-445. [ Links ]
Santos, A.F.G.N., L.N. Santos & F.G. Araujo. 2004. Water level influences on body condition of Geophagus brasiliensis (Perciformes: Cichlidae) in a Brazilian oligotrophic reservoir. Neotrop. Ichthyol. 2: 151-156. [ Links ]
Sparre, P. & S.C. Venema. 1997. Introduction to tropical fish stock assessment - Parte 1: Manual. FAO Fisheries Technical Paper 1: Roma, Italia. [ Links ]
Straškraba, M. & J.G. Tundisi. 2000. Diretrizes para o gerenciamento de lagos: Gerenciamento da qualidade da água de represas. Serie Gerenciamento da qualidade da água de represas. Instituto Internacional de Ecologia, São Carlos, São Paulo, Brasil. [ Links ]
Tavares, L.H.S. & O. Rocha. 2001. Produção de plâncton (fitoplâncton e zooplâncton) para a alimentação de organismos aquáticos. RiMa, São Carlos, São Paulo, Brasil. [ Links ]
Vidotto-Magnoni, A.P. & E.D. Carvalho. 2009. Aquatic insects as the main food resource of fish the community in a Neotropical reservoir. Neotrop. Ichthyol. 7: 701-709. [ Links ]
Walter, T. & M. Petrere Jr. 2007. The small-scale urban reservoir fisheries of Lago Paranoa, Brasilia, DF, Brazil. Braz. J. Biol. 67: 9-21. [ Links ]
Weliange, W.S. & U.S. Amarasinghe. 2003. Seasonality in dietary shifts in size-structured freshwater fish assemblages in three reservoirs of Sri Lanka. Environ. Biol. Fishes 68: 269-282. [ Links ]
Winemiller, K.O. 1989. Patterns of variation in life history among South American fishes in seasonal environmental. Oecologia 81: 225-241. [ Links ]
Wootton, R.J. 1995. Ecology of teleost fishes. Chapman & Hall, London, England. [ Links ]
*Correspondencia a: Jose Luis Costa Novaes: Universidade Federal Rural do Semiárido, Departamento de Ciências Animais, BR 110, km 47, s/n, Costa e Silva, Mossoró, Rio Grande do Norte, Brazil; novaes@ufersa.edu.br
Edmir Daniel Carvalho: Universidade Estadual Paulista, Departamento de Morfologia, Distrito de Rubião Jr. s/n, Botucatu, São Paulo, Brazil; carvalho@ibb.unesp.br
1. Universidade Federal Rural do Semiárido, Departamento de Ciências Animais, BR 110, km 47, s/n, Costa e Silva, Mossoró, Rio Grande do Norte, Brazil; novaes@ufersa.edu.br
2. Universidade Estadual Paulista, Departamento de Morfologia, Distrito de Rubião Jr. s/n, Botucatu, São Paulo, Brazil; carvalho@ibb.unesp.br
Received 03-V-2011. Corrected 15-XII-2011. Accepted 24-I-2012.