Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.60 n.2 San José Jun. 2012
Reproduction of Omalonyx matheroni (Gastropoda: Succineidae) under laboratory conditions
*Dirección para correspondencia:
Abstract
The life histories of succineids have received relatively little attention. To evaluate life history characteristics of Omalonyx matheroni, we studied a Brazilian population (Reserva Particular do Patrimônio Natural Feliciano Miguel Abdala, in Caratinga, Minas Gerais, Brazil) under laboratory conditions. The aims of the present study were (1) to describe in detail an appropriate rearing method; (2) to investigate the effects of different temperature and photoperiod conditions; and (3) to assess the effects of self and cross-fertilization on the reproductive biology of these mollusks. We studied the oviposition site, the time to sexual maturity and the influences of photoperiod and temperature on reproductive parameters of O. matheroni reared under laboratory conditions. We tested three combinations of temperature and photoperiod, designated A, B and C (A: 25ºC, 24 hours of light; B: environmental conditions of temperature and photoperiod, characterized as follows: average máximum temperature=27.1ºC, average minimum temperature=18.3ºC, average day length=12.06 hours; and C: 25ºC, zero hours of light) and two rearing densities (I: isolated and G: grouped) on reproductive parameters (number of eggs per egg mass, number of unviable eggs per mass, egg mass incubation period, and duration of the hatching period). A total of 186 individuals and 565 egg masses were studied. Data were analyzed by Student’s t-test, two-way ANOVA and Chi-Square test. Eight generations were produced (March/2004-March/2006), from 35 field specimens, 91% of 3 197 eggs hatched. The time to sexual maturity was approximately three months for individuals reared in groups or in isolation (Student’s t-test: t=1.41, df=31, p=0.16); however, they differed significantly in weight (Student’s t-test: t=3.6, df=31, p<0.001). Regarding the influences of temperatura and photoperiod on reproductive parameters, under natural environmental conditions, individuals produced a greater number of eggs per mass (ANOVA: F2,573=84.15, p<0.001), with a longer incubation period (ANOVA: F2;559=170.05, p<0.001). The extreme photoperiod conditions of 24 hours of light or zero hours of light likely imposed stress and could be related to the significant reductions in the number of eggs per mass, and egg incubation period as well as the increased synchrony in egg hatching. No correlations were observed between the number of unviable eggs per mass and the temperature, photoperiod (ANOVA: F2,573=0.87, p=0.92) or rearing density (ANOVA: F1,573=0.21, p=0.64). Individuals reared in isolation under natural conditions produced more eggs per mass and did not presented any disadvantage with respect to the variables analyzed as compared to the animals reared in groups. These results indicate that O. matheroni can successfully reproduce by selfing. Rev. Biol. Trop. 60 (2): 553-566. Epub 2012 June 01.
Key words: reproduction, life history, mollusk, temperature, photoperiod, laboratory maintenance.
Resumen
En vista de que las historias de vida de los Succineidae han recibido relativamente poca atención, se estudio una población de Omalonyx matheroni, de Brasil (Reserva Privada y Patrimonio Natural Feliciano Miguel Abdala, en Caratinga, Minas Gerais, Brazil), en condiciones de laboratorio. Los objetivos de este trabajo fueron: (1) describir una metodología para el cultivo en laboratorio de Omalonyx matheroni; (2) investigar los efectos de diferentes condiciones de temperatura y fotoperíodo en su cultivo y, (3) verificar los efectos de la autofecundación y de la fecundación cruzada en la biología reproductiva de estos moluscos. Se estudiaron 186 individuos y 565 masas de huevos. La edad de madurez sexual fue aproximadamente de tres meses, igual para los animales aislados que para los que estuvieron en grupo (Student’s t-test: t=1.41, df=31,p=0.16). En condiciones más cercanas a la naturaleza se presentaron más huevos por masa (ANOVA: F2,573=84.15,p<0.001) y un tiempo más largo de eclosión (ANOVA: F2,559=170.05, p<0.001). En condiciones más extremas, debido probablemente al estrés producido por las mismas, se observó una reducción en el número de huevos por masa, en el tiempo de incubación y una eclosión más sincrónica. No se encontró una correlación entre el número de huevos inviables y las condiciones de temperatura y fotoperíodo (ANOVA: F2,573=0.87, p=0.92) y la densidad de cultivo (ANOVA: F1,573=0.21, p=0.64). Animales en aislamiento se reprodujeron con éxito lo que muestra la capacidad que tiene O. matheroni para la autofecundación.
Palabras clave: reproducción, historia de vida, molusco, fotoperíodo, temperatura, mantenimiento en laboratorio.
The genus Omalonyx d’Orbigny 1837, belongs to the family Succineidae and represents one of the lineages with a reduced flat shell and slug-like body shape (Patterson 1971, Tillier 1981, Barker 2001). Species of this genus are distributed throughout South America; in Brazil, they are widely distributed in all regions of the country (Lange de Morretes 1949, Tiller 1981, Dutra-Clarke et al. 2001, Salgado & Coelho 2003, Simone 2006, Arruda et al. 2009, Coscarelli & Vidigal 2011).
In spite of their abundance and widespread distribution, research on the life history of succineids, including Omalonyx, has received relatively little attention (Hermann & Dundee 1967, Patterson 1970, Villalobos et al. 1995, Rundell & Cowie 2003, Villalobos & Monge- Nájera 2004, Brown et al. 2006, Dillen et al. 2009). Laboratory studies on the life history of pulmonates are considered to be extremelyimportant for developing an understanding of their complex reproductive processes and determining the environmental factors that influence them (Gomot de Vaufleury 2001, Heller 2001). Influences of temperature, photoperiod and rearing density (individuals raised in isolation or in a group) on reproduction have been demonstrated in several pulmonate species, including succineids (Enee et al. 1982, South 1992, Gomot de Vaufleury 2001, Hommay et al. 2001, Meireles et al. 2008, Dillen et al. 2009).
According to Parodiz (1963), species of the Omalonyx genus are found in humid sites and are never far from water. They are often found submerged among aquatic plants or in the mud at the bottom of freshwater systems. Information concerning the natural habitat and lifestyle of Omalonyx is somewhat controversial (Parodiz 1963, Hermann & Dundee 1967, Patterson 1971, Poi de Neiff et al. 1977, Olazarri 1979, Tillier 1981, Cazzaniga 1985); however, all of the authors agree that this genus is strongly associated with freshwater systems such as swamps, lakes and river margins, as well as the surrounding vegetation.
Hermann & Dundee (1967) studied the habitat of Omalonyx matheroni (Pontiez & Michaud 1835) in Ecuador and found that individuals in freshwater systems could be reared in plastic containers with water and vegetation from the lake, where they had been collected. Patterson (1970, 1971) described a generalized rearing methodology for succineids that involved separating egg masses and young and adult individuals into different containers and providing a diet based on lettuce and chopped cereal grasses. Sterilized sand with clay and calcium carbonate was added to the bottom of the containers. A different diet was fed to juveniles. Employing this methodology for two years, Patterson (1971) reared 16 generations of Omalonyx. However, further details regarding the success of this rearing methodology and other life history data were not mentioned.
To evaluate life history characteristics of O. matheroni, we studied a Brazilian population under laboratory conditions. The aims of the present study were (1) to describe in detail an appropriate rearing method; (2) to investigate the effects of different temperature and photoperiod conditions; and (3) to assess the effects of self and cross-fertilization on the reproductive biology of these mollusks.
Materials and methods
Field collection and laboratory rearing of mollusks: A laboratory population of Omalonyx was established from 35 individuals collected in March 2004 at the Reserva Particular do Patrimônio Natural Feliciano Miguel Abdala, a private nature preserve in Caratinga, Minas Gerais, Brazil (19o43’55’’ S - 41°49’03’’W). The snails were identified as O. matheroni by means of comparative morphology based on their reproductive organs, according to the criteria defined by Tillier (1981). Voucher specimens were deposited in the malacology collection of the Laboratório de Malacologia e Sistemática Molecular (LMSM), Departamento de Zoologia, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil, under the catalog number LMSM 1 128. The individuals were manually collected from wet soil and vegetation in the vicinity of freshwater systems. The mollusks were maintained at room temperature in two plastic containers (28.2L) with perforated lids containing 17-18 individuals each. The mollusks were reared based on the procedures described by Hermann & Dundee (1967) and Patterson (1970, 1971) with some modifications as described below. The rearing containers were prepared with a thin layer of water at the bottom (approximately 600mL) to maintain a high-humidity environment. Specimens of Pistia stratiotes (Araceae), popularly known as water lettuce, were used as a substrate for egglaying and shelter. Water lettuce obtained from the field was washed in tap water and placed in the rearing containers. Mollusks were fed daily with lettuce and supplied with powdered rodent chow to which ten percent calcium carbonate (CaCO3) was added; this mixture was renewed every two days. The chow was placed in a round plastic container (diameter: 30mm; height: 7mm) to avoid contact with the water. As the eggs were laid, they were transferred to a separate container, which was called the nursery (Nr) and is described below.
Rearing was divided into stages according to the ages of the specimens. The cleaning procedure was performed every two days. Chow and fecal residues were removed with tap wáter and a thin paint brush. We avoided touching the animals in order to reduce the number of stress factors. Each day, the egg masses were individually transferred from the main container (Mn) to the nursery (Nr) and cleaned with tap water and a thin paint brush.
Nursery (Nr): Hatching-day five. Glass Petri dishes (diameter: 80mm; height: 17mm) with moistened filter paper at the bottom were used to create the moist chambers. Petri dishes were labeled with the container from which the egg mass was taken and the date of oviposition. The moist chambers were visually inspected every day. The filter papers were kept permanently wet. After hatching, the newly hatched young were fed twice a week with macerated lettuce and approximately 10mg of chow.
Growth (Gr): Day six-30. Individuals were kept in transparent plastic containers (diameter: 75mm, height: 55mm) with perforated lids. The bottoms of the containers were covered with a thin layer of water (approximately 3mL) and one P. stratiotes specimen. Up to five six-dayold individuals were placed in each container and fed macerated lettuce three times a week and 20mg of chow diet twice a week. The containers were visually inspected every day, and dead specimens were removed when necessary.
Maintenance (Mn): More than 30 days old. Mollusks were placed in transparent plastic containers (diameter: 145mm, height: 95mm) with perforated lids. The bottom of each container was covered with a thin layer of wáter (approximately 10mL) and one P. stratiotes specimen. Up to four 30-day-old individuals were placed in each container and fed lettuce three times a week and chow diet twice a week (25mg per individual). Isolated individuals were kept in the smaller plastic container (diameter: 75mm, height: 55mm).
Oviposition in the laboratory: The mollusks collected in the field (35 individuals) were kept in the laboratory under natural conditions (condition B, according to table 1) in two containers with perforated lids (17-18 individuals in each container). These mollusks were reared as described above for stage Mn. The containers were checked every day for new egg masses. The egg masses were cleaned and kept as described above for stage Nr. The number of viable and unviable eggs was determined. Egg diameter was measured using an ocularmicrometer (Leica, Germany, MZ12).
To determine the preferred egg-laying site, we analyzed 100 egg masses from ten individuals reared in groups (condition B). The containers were checked every day or new egg masses, which were removed daily, counted and identified according to the site at which they were laid (water lettuce or container wall).
Time to sexual maturity in the laboratory: Mollusks were considered sexually mature at their first oviposition. Data on the age, weight and size of specimens (F1 generation) reared under natural conditions (condition B) in isolation (16 specimens) or in groups (17 specimens in groups of three or four individuals) at first egg-laying were analyzed by Student’s t-test.
Measurement of specimens from each rearing stage: From each rearing tage, 20 individuals were randomly selected (Nr: five days old; Gr: 30 days old; Mn: more than 100 days old), measured using an ocular micrometer (length of the body and shell and width of the shell) (stereoscopic microscope: Leica, Germany, MZ12) and weighed with an analytical balance (sensitivity 0.001g, Marte, AL 500).
Influence of the rearing density and conditions on reproductive parameters: The F1 generation was subjected to three different temperature and photoperiod conditions (A: 25ºC, 24 hours of light; B: environmental conditions of temperature and photoperiod, characterized as follows: average maximum temperature=27.1ºC, average minimum temperature=18.3ºC, average day length=12.06 hours; and C: 25ºC, zero hours of light) and two rearing densities (I: isolated and G: grouped). These conditions are described in table 1 along with the numbers of individuals in each condition. Individuals were divided into different rearing densities (I or G) when they were six days old and maintained in isolation or in groups until the end of the experiment. For treatment I, after being transferred from the Nr, six-day-old specimens were individually maintained in 75mm containers until the end of the experiment. For treatment G, two to four specimens were initially maintained in 75mm containers (Gr) and then transferred to 145mm containers (Mn) until the end of the experiment.
The following parameters were analyzed for all of the egg masses laid under the different conditions: number of eggs per egg mass, number of unviable eggs per mass, egg mass incubation period and duration of the hatching period, i.e., the time elapsed between the hatching of the first egg and the hatching of the last egg in a given egg mass.
Two-way ANOVA (Zar 2009) was used to test the effects of the temperature and photoperiod (A, B, and C) and rearing density (I and G) conditions on the following log-transformed variables: number of eggs per egg mass, number of unviable eggs per egg mass, and egg mass incubation period. The duration of the egg mass hatching period was categorized as synchronous (hatching period=zero days) or nonsynchronous (hatching period=one or more days), and the association of this variable with the temperature and photoperiod conditions (A, B and C) and rearing densities (I and G) was tested using the chi-square test (Zar 2009).
Results
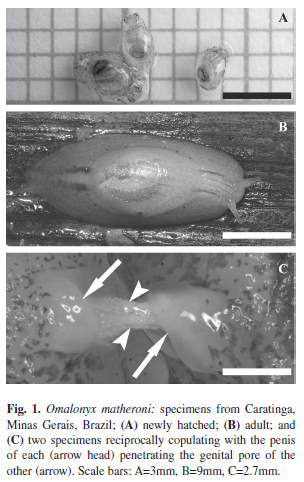
Field collection and laboratory rearing of mollusks: Eight generations were reared in 24 months with the methodology described above. Aspects of the rearing procedure considered important for the success of laboratory maintenance include the following: (1) Defining rearing stages according to age. This technique improved the laboratory maintenance of O. matheroni and allowed us to achieve the specific demands of each developmental phase (newly-hatched and adult specimens are shown in Fig. 1A and 1B, respectively). (2) Maintaining the humidity in the rearing containers (moist chambers were used for Nr, and wáter was placed at the bottom of the container for Gr and Mn). (3) Separating egg masses into individual moist chambers (in the nursery). This procedure facilitated observations of hatching and the development of the egg. (4) Improving the diet with chow. Providing a source of calcium carbonate is an essential component for any gastropod rearing strategy. Mollusks demonstrated a clear preference for the chow diet when they were simultaneously presented with chow and lettuce.
As observed in field populations, laboratory-reared individuals did not stay in the water. Exceptions occurred when they were laying eggs at the roots, under the leaves or on the leaf axis of the water lettuce or when they were moving to another site. Most of the time, the individuals remained on the substrate (container wall or water lettuce leaves). P. stratiotes was not consumed as food, but it was an excellent egg-laying site.
Oviposition in the laboratory: From the 35 field specimens kept in the laboratory under condition B, 218 egg masses were obtained after 125 days of observation, and 91% of the 3 197 eggs hatched. Each individual laid a mean of 91.34 eggs during this period. The egg diameter was 2.1±0.3mm (mean±SD). Only 4% of the egg masses were laid on the plastic container wall; 96% were found on P. stratiotes. In addition to being readily available, P. stratiotes is very manageable and can be kept alive in the laboratory for over a month. Reciprocal copulation was observed when mollusks were housed in groups (Fig. 1C).
Time to sexual maturity in the laboratory: No significant differences in the time to sexual maturity were observed between specimens reared in groups or in isolation under condition B (Grouped=91.7±7.6 days; Isolated=86.2±14.1 days) (Student’s t-test: t=1.41, df=31, p=0.16). However, they significantly differed in weight (Grouped=181±80mg; Isolated=320±133mg) (Student’s t-test: t=3.6, df=31, p<0.001), and the p-value obtained for size was very close to significance (Grouped=14±0.44mm; Isolated=16.6±0.28mm) (Student’s t-test:t=1.84, df=31, p=0.072).
Measurement of specimens from each rearing stage: The mean body length, body weight, and shell size of specimens reared in groups under natural environmental conditions (B/G) are shown in table 2.
Influence of the rearing density and conditions on reproductive parameters: Each egg mass was analyzed in relation to four reproductive parameters and these results are presented separately below.
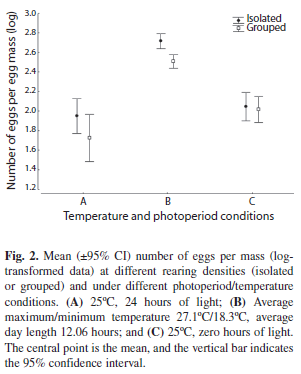
Number of eggs per egg mass: Significant effects of rearing density (ANOVA: F1,573=5.81, p=0.016) and temperature and photoperiod conditions (ANOVA: F2,573=84.15, p<0.001) on the number of eggs per mass were observed. At both rearing densities (I and G), the number of eggs per mass was higher under natural environmental conditions (B); however, isolated individuals laid more eggs per mass as compared to grouped individuals (Fig. 2 and Table 3). The effect of the interaction between rearing density and the temperature and photoperiod conditions was not significant (ANOVA: F2, 573=1.12, p=0.28).
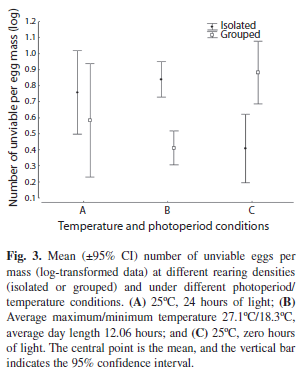
Number of unviable eggs per egg mass: There were no significant differences in the number of unviable eggs, neither among the different temperature/photoperiod conditions (A, B and C) (ANOVA: F2,573=0.87, p=0.92) nor between the rearing densities (G and I) (ANOVA: F1,573=0.21, p=0.64). However, the interaction between these factors presented a significant effect (ANOVA: F2,573=14.62, p<0.001). Isolated individuals produced more unviable eggs per egg mass than grouped individuals under condition B, but under condition C, they produced fewer unviable eggs per egg mass (Fig. 3 and Table 3).
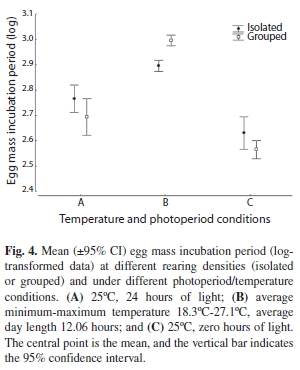
Egg mass incubation period: There was a significant difference in egg mass incubation period among the temperature and photoperiod conditions (A, B and C) (ANOVA: F2,559=170.05, p<0.001) but not between the two rearing densities (ANOVA: F2,559=0.41, p=0.52). The interaction between rearing density and temperature and photoperiod conditions was significant (ANOVA: F2,559=13.03, p<0.001). The egg mass incubation period was longer in condition B than in either of the extreme conditions (A and C). Under condition B, the egg mass incubation period was longer for grouped specimens than for isolated individuals. In conditions A and C, no significant differences were observed between isolated and grouped individuals (Fig. 4 and Table 3).
Duration of egg mass hatching period: There was a significant association between the rearing condition and the synchrony of hatching (χ2=377.6, df=2, p<0.001). Egg hatching was not synchronous under condition A or B; however, under condition C, 71% of hatching events were synchronous (Table 4). We also found an association between rearing density and the duration of the egg mass hatching period (χ2=23.9, df=1, p<0.001). The egg masses from isolated individuals were synchronous in 5.1% of the hatching events, compared to 18.8% of egg masses from grouped specimens (Tables 3 and 4).
Discussion
Pulmonates are simultaneous hermaphrodites, well-known for their broad variation in life history characteristics. Snails of the genus Omalonyx are capable of reproduction by both outcrossing and apparent self-fertilization (Patterson 1970). The ability to rear stable populations in the laboratory allows for controlled laboratory trials and consequently informative studies on life history. Studying life history aspects such as rearing density, fecundity and fertility rates allows for a better understanding of pulmonate population structure, and contributes to the management and conservation plans for these mollusks. Data of this sort also enable further studies on snail parasitology (e.g., studies on snail-fluke interactions) and physiology (e.g., regulation of growth and reproduction) (South 1992, Lam 1994, Villalobos et al. 1995, Gomot de Vaufleury 2001, Hommay et al. 2001, Daoust et al. 2009, Dillen et al. 2009).
The use of local vegetation as shelter and substrate for egg-laying was first reported by Hermann & Dundee (1967). According to these authors, in the laboratory setting, eggs were preferentially laid on the walls of the plastic containers, with plants serving as a second choice. In the present study, P. stratiotes was not consumed as food but was the first choice for an egg-laying site, contrary to the results of Hermann & Dundee (1967).
In the absence of a partner, some hermaphrodites, including Omalonyx, do not cease reproduction, and apparent self-fertilization occurs (Patterson 1970). Thus, by rearing O. matheroni in isolation or in groups, we favored selfing (parthenogenesis or self-fertilization) or cross-fertilization, respectively. Diverse hermaphroditic mollusks, including Omalonyx, copulate reciprocally. The first observation of reciprocal copulation in Omalonyx was recorded by Hermann & Dundee (1967). During our routine observations of the grouped individuals, we verified the occurrence of reciprocal copulation.
Information on the life history of a given species allows inferences about its ecology. Oviposition comprises an important life history characteristic. Laboratory trials are needed to determinate the range of this variable, due to the difficulty in studying life history characteristics in the field. The hatching rate of Succinea costaricana von Martens 1898 (Stylommatophora: Succineidae), is close to 100% when proper laboratory conditions are available (Villalobos et al. 1995). However, this rate is likely very different from that which occurs in the wild, where several factors that cannot be controlled, such as dryness, predators and parasites, likely destroy most of the eggs (Villalobos et al. 1995). In the present work, oviposition data for O. matheroni revealed a hatching rate of 91% and a mean production of 91.34 eggs per individual over a period of 125 days. An 80% hatching success rate and a mean production of 23.4 eggs per snail in the laboratory over the same period were observed for the succineid Catinella rotundata (Gould 1846) (Stylommatophora: Succineidae) (Rundell & Cowie 2003). Likewise, in the same period of time, a production of 71.44 eggs per individual and an 81.22% hatching rate were reported for Bradybaena similaris (Férussac 1821) (Stylommatophora: Bradybaenidae) (Almeida & Bessa 2001). The authors also commented about the large intraspecific and interspecific variation in these variables among several different mollusks. These reproductive variables are related to the species’ success in their natural habitat and are important for laboratory rearing trials and for the development of pest control methods (Villalobos et al. 1995, Hommay et al. 2001). The egg size of stylommatophoran mollusks varies under different ecological and environmental conditions (Peake 1978). Villalobos et al. (1995) reported an egg diameter of 1.367±0.163mm for the succineid S. costaricana. The egg diameter of Omalonyx measured in the present work (2.1±0.3mm) fell in the range expected for eggs lacking a calcium carbonate cover (one-eight mm) (Peake 1978).
The time to sexual maturity is an important life history characteristic that can present large intraspecific and interspecific variation and be influenced by several factors (Roff 1992, Stearns 1992). The time to sexual maturity observed in this work did not vary among groups; despite the fact that they were larger, specimens reared in isolation reached sexual maturity at the same time as grouped individuals. This result is in accordance with the observations of Succinea thaanumi (Ancey 1899) performed by Rundell & Cowie (2003) and with the period estimated by Hermann & Dundee (1967) for Omalonyx. The time to sexual maturity in all of these cases was approximately three months.
Variation in weight was substantial among individuals of the same age at all of the rearing stages studied herein. Hommay et al. (2001) observed a significant variation in the weight of Limax valentianus Férrusac 1823 (Stylommatophora: Limacidae), particularly in the first days after hatching. Peake (1978) suggested that the nutritional content in individual eggs may be an important factor determining future growth patterns in pulmonates. Several Works have also demonstrated that increased size is related to increased fecundity in many animals, including mollusks (Stearns & Koella 1986, Heller 2001, D’Ávila & Bessa 2005). Considering that isolated individuals were larger and produced more eggs per egg mass, further studies might investigate the relation between size and fecundity in O. matheroni.
We expected to observe a clear disadvantage in the reproductive parameters of isolated individuals relative to those of grouped individuals. However, our results indicate that selfing may be a successful reproductive strategy for O. matheroni. This supposition is based on the fact that under natural environmental conditions (B), isolated individuals (I) laid egg masses containing more eggs than the individuals maintained in groups (G). The two rearing densities presented similar results for the time of sexual maturity, the number of eggs per egg mass under extreme conditions (A and C) and the number of fertile eggs under condition A. However, the influence of self-fertilization on the life history characters of succineids seems to vary. A study performed by Dillen et al. (2009) demonstrated that the succineid Succiena putris (Linnaeus 1758) is predominantly an outcrosser. These authors observed that mated individuals laid clutches with more eggs and demonstrated a higher fertility rate than unmated individuals. On the other hand, Silva et al. (2009) verified an increase in the reproductive success of Habroconus semenlini (Moricand 1846) (Stylommatophora: Euconulidae) reared in isolation. To verify the impact of cross-fertilization and selfing on life history traits of O. matheroni, further studies should determine the number of descendants that reach the adult phase.
Temperature and photoperiod are important factors that influence life history. Animals adapt to photoperiod to allow efficient timing relative to the favorable part of the season. Several aspects of development are determined by environmental cues, of which the photoperiod is perhaps the most important (Beck 1980). In turn, temperature can initiate responses or affect rates of development, growth and reproduction (Atkinson 1996). The effect of temperature depends on both mean temperatura and the frequency and magnitude of any fluctuations (Ratte 1985, Cossins & Bowler 1987).
In the present study, the extreme photoperiod conditions (A: 24 hours of light and C: zero hours of light, both at 25°C) resulted in reductions in the number of eggs per egg mass, the incubation period and the egg mass hatchingperiod. However, the consequences of these extreme conditions on the growth and survival of the snails were not evaluated. Despite the longer egg incubation period, a greater number of eggs per egg mass were produced under natural conditions (B), indicating that in the absence of a day-night cycle (in A and C), individuals produced fewer eggs per egg mass. Indeed, temperature and light incidence vary daily and over time under natural conditions (B), which was not the case in condition A or C.
Variations in the number of eggs per egg mass have been reported by several authors. Dimitrieva (1975) and Heller (2001) noted the influence of factors such as age, size, environmental conditions and seasonality on the clutch size (number of eggs per clutch) for several terrestrial mollusks. Lam (1994) mentioned interpopulational differences in clutch size that were likely environmentally determined, and Heller (2001) discussed interpopulational differences in clutch size among mollusks of the same size maintained under the same conditions.
The average numbers of eggs per egg mass for the succineids S. costaricana, S. thaanumi and C. rotundata were 6.8±4.5, 9 (range 1-16, n=21) and 12.0 (range 3-19, n=4), respectively (Villalobos et al. 1995, Rundell & Cowie 2003). Hermann & Dundee (1967) observed a range of four to 20 eggs per egg mass in Omalonyx populations, with occasional drops to one to three eggs per egg mass. The mean number of eggs per egg mass found in the present investigation varied from 6.4±3.5 (A/G) to 16.5±7.2 (B/I), which is approximately the same range observed by Hermann & Dundee (1967), Villalobos et al. (1995) and Rundell & Cowie (2003).
Despite the higher number of eggs per egg mass produced by isolated individuals under natural conditions (B), these snails also yielded more unviable eggs and shorter incubation periods as compared to the grouped individuals. Tompa (1984) and Hommay et al. (2001) mentioned that slug eggs are highly temperature-sensitive. Thus, further studies that include more temperature variations are necessary to identify the factors that may influence the number of unviable eggs.
Development is highly influenced by photoperiod (Beck 1980). Several works have demonstrated that continuous light accelerates development and induces egg hatching in avians and crustaceans, while continuous darkness inhibits hatching (Jones 1970, Horiguchi et al. 2009). In this work, the extreme photoperiods in conditions A and C (continuous light and continuous darkness, respectively) were associated with shorter egg incubation periods relative to that observed under natural conditions (B). This finding suggests that continuous light and continuous dark both resulted in the acceleration of development. These conditions probably represent stress factors that led to the production of fewer eggs per egg mass and reductions in the egg incubation period.
The mean incubation period in our study varied from 13.0±1.1-20.5±4.5 days. Hermann & Dundee (1967) reported that the median incubation period for Omalonyx populations from the Lesser Antilles was 11 days. They did not report methodological details regarding the number of specimens studied, the statistical analysis or the environmental conditions under which these observations were conducted. Different species of the same genus can show variations in the incubation period, as demonstrated in Succinea (Rundell & Cowie 2003, Dillen et al. 2009). Variations in incubation period were previously reported for slugs and snails, and temperature was identified as an influential factor (Carrick 1942, Almeida & Bessa 2001). Carrick (1942) mentioned that the incubation period decreases with increasing temperature. Hommay et al. (2001) reported that small differences between temperatures did not produce substantial differences in incubation period for L. valentianus. In our study, variation in temperature occurred only in the natural conditions (B), in contrast to the constant temperature maintained in conditions A and C. Further investigations into the life history characteristics of O. matheroni should include diverse daily variations in temperature.
Concerning the duration of the egg hatching period, shorter hatching periods were observed in the absence of light, indicating that this condition stimulated synchronous egg hatching. In contrast, Hommay et al. (2001) observed that temperature, but not photoperiod, influenced the egg hatching period of L. valentianus. These authors also showed that variation in the egg hatching period was lower for the animals reared in isolation compared to those maintained in groups. In the present work, the duration of the egg mass hatching period was not influenced by rearing density.
Rearing density is known to affect several life history characters in mollusks, including growth and reproduction (South 1982). Several studies demonstrated that crowding increases mortality and decreases the growth rate of snails (Charrier 1981, Lazaridou-Dimitriadou & Daguzan 1981, Dan & Bailey 1982, Lucarz & Gomot 1985, Jess & Marks 1995) and that snails reared in isolated conditions presented less variation in weight, a higher growth rate and were generally heavier than those raised in groups (South 1982, Gutiérrez et al. 2001). However, Hommay et al. (2001) commented that contrary to Deroceras reticulatum (Müller 1774) (Stylommatophora: Agriolimacidae) and Limax pseudoflavus Evans, L. valentianus did not reach higher weights when raised in isolation vs. groups. Our results indicate that rearing density did affect the analyzed variables when individuals were maintained under natural environmental conditions (B). Isolated individuals were larger at the time of sexual maturity, produced more eggs per egg mass and more unviable eggs and presented shorter incubation periods as compared to individuals housed in groups (condition B). Under the extreme temperature and photoperiod conditions (A and C), the differences among isolated and grouped individuals were attenuated, probably due to the greater influence of the stressful photoperiod conditions. The only exception was the number of unviable eggs per egg mass under condition C, which was significantly smaller for isolated individuals.
Life history characters are known to be influenced by diverse factors, present wide intraspecific and interspecific variation and be related to life history evolution (Roff 1992, Stearns 1992, Silva et al. 2009). Research on mollusks reaffirms this statement (South 1982, Dillen et al. 2009). Efforts to better understand the life histories of mollusks are essential to understanding the biology of these animals and to the improvement of conservation efforts.
Acknowledgments
This work was supported by grants from the foundation for research from the State of Minas Gerais (Fapemig). We thank Daniel Coscarelli, Henry Lima, Eliane Silveira (Laboratório de Malacologia e Sistemática Molecular, UFMG) for the field collection of Omalonyx and for laboratory assistance. Silvia Chiarello, Alfredo Wieloch and Marcelo Lorenzo provided valuable comments on an earlier version of this manuscript. We also thank Queila Garcia in whose laboratory part of this work was performed. Almir Pepato, Diego Zorio and Mario Cozzuol helped to write the Spanish abstract. Photography was by Lângia Montresor (Fig. 1A and 1C) and André Fernandes (Fig. 1B).
Referencias
Almeida, M.N. & E.C.A. Bessa. 2001. Estudo do crescimento e da reprodução de Bradybaena similares (Mollusca, Xanthonychidae) em laboratório. Rev. Bras. Zool. 18: 1115-1122. [ Links ]
Arruda, J.O., D. Pereira, P.E.A Bergonci, C.P. Santos & M.C.D. Mansur. 2009. Novos registros de Omalonyx matheroni (Potiez & Michaud, 1835) (Mollusca, Gastropoda, Succineidae) para os Estados de São Paulo e Paraná, Brasil. Biotemas 22: 187-190. [ Links ]
Atkinson, D. 1996. Ectotherm life history responses to developmental temperature, p. 183-204. In I.A. Johnston & A.F. Bennett (eds.). Animals and temperature: Phenotypic and evolutionary adaptation. Cambridge University, Cambridge, Massachusetts, USA. [ Links ]
Barker, M.G. 2001. Gastropods on land: phylogeny, diversity and adaptative morphology, p. 01-146. In M.G. Barker (ed.). The biology of terrestrial mollusks. CABI, New York, New York, USA. [ Links ]
Beck, S.D. 1980. Insect photoperiodism. Academic, New York, New York, USA. [ Links ]
Brown, S.G., J.M. Spain & M. Arizumi. 2006. A field study of the life history of the endemic hawaiian snail Succinea newcombiana. Malacologia 48: 295-298. [ Links ]
Carrick, R. 1942. The grey field slug Agriolimax agrestis L. and its environment. Ann. Appl. Biol. 29: 43-55. [ Links ]
Cazzaniga, N.J. 1985. Anotaciones sobre algunos gasterópodos no marinos de la Argentina. Comun. Soc. Malacol. Uruguay 6: 329-331. [ Links ]
Charrier, M. 1981. Contribution à l’étude des effets du groupement sur la croissance de l’escargot petit-gris Helix aspersa Müller (Gastéropode: Pulmoné: Stylommatophore). Arch. Zool. Exp. Gén. 122: 29-38. [ Links ]
Coscarelli, D. & T.H.D.A. Vidigal. 2011. Mollusca, Gastropoda, Succineidae, Omalonyx unguis (d’Orbigny, 1835): Distribution extension and new records for Brazil. Check List 7: 400-403. [ Links ]
Cossins, A.R. & K. Bowler. 1987. Temperature biology of animals. Chapman & Hall, London, London, United Kingdom. [ Links ]
Dan, N. & S.E.R. Bailey. 1982. Growth, mortality, and feeding rates of the snail Helix aspersa at different population densities in the laboratory, and the depression of activity of helicid snails by other individuals, or their mucus. J. Mollus. Stud. 48: 257-265. [ Links ]
Daoust, S.P., B.J. Mader, J.D. McLaughlin & M.E. Rau. 2009. Plagiorchis elegans (Trematoda) and incompatible snail hosts: implications for snail life history traits and biocontrol of human schistosomiasis. J. Parasitol. 95: 1383-1386. [ Links ]
D’Ávila, S. & E.C.A. Bessa. 2005. Influência do substrato sobre o crescimento de Subulina octona (Brugüière) (Mollusca, Subulinidae), sob condições de laboratório. Rev. Bras. Zool. 22: 205-211. [ Links ]
Dillen, L., K. Jordaens & T. Backeljau. 2009. Life-history variation and breeding system in the hermaphroditic land snail Succinea putris (Pulmonata: Succineidae). J. Mollus. Stud. 75: 311-313. [ Links ]
Dimitrieva, E.F. 1975. The influence of temperature and moisture of the upper soil layer on the hatching intensity of the slug Deroceras reticulatum Müller. Malacol. Rev. 10: 32-45. [ Links ]
Dutra-Clarke, A.V.C., C. Williams, R. Dickstein, N. Kaufer & J.R. Spotila. 2001. Inferences on the phylogenetic relationships of Succineidae (Mollusca, Pulmonata) based on 18S rRNA gene. Malacologia 43: 223-236. [ Links ]
Enee, J., R. Bonnefoy-Claudet & L. Gomot. 1982. Effets de la photoperiode artificielle sur la reproduction de l’escargot Helix aspersa Muller. Compt. Rend. Acad. Sci. 294: 357-360. [ Links ]
Gomot de Vaufleury, A. 2001. Regulation of growth and reproduction, p. 331-355. In M.G. Barker (ed.). The biology of terrestrial mollusks. CABI, New York, New York, USA. [ Links ]
Gutiérrez, A., G. Perera, M. Young & L. Wong. 2001. The effect of isolation on the life-history traits of Pseudosuccinea columella (Pulmonata: Lymnaeidae). Mem. Inst. Oswaldo Cruz 96: 577-581. [ Links ]
Heller, J. 2001. Life history strategies, p. 413-445. In M.G. Barker (ed.). The biology of terrestrial mollusks. CABI, New York, New York, USA. [ Links ]
Hermann, P.W. & D.S. Dundee. 1967. Notes on Omalonyx. Sterkiana 28: 1-6. [ Links ]
Hommay, G., J.C. Kienlen, C. Gertz & A. Hill. 2001. Growth and reproduction of the slug Limax valentianus Fèrussac in experimental conditions. J. Mollus. Stud. 67: 191-207. [ Links ]
Horiguchi, T., C. Ito & H. Numata. 2009. Regulation of embryogenesis by light and its ecological significance in the Asian tadpole shrimp Triops granarius. Zool. Sci. 26: 483-490. [ Links ]
Jess, S. & R.J. Marks. 1995. Population density effects on growth in culture of the edible snail Helix aspersa var. maxima. J. Mollus. Stud. 61: 313-323. [ Links ]
Jones, P.C.T. 1970. Temperature and light-mediated alterations of ATP level in the chick embryo in egg; and their role in morphogenesis. Comp. Biochem. Physiol. 36: 87-92. [ Links ]
Lam, P.K.S. 1994. Intraspecific life-history variation in Radix plicatulus (Gastropoda: Pulmonata: Lymnaeidae). J. Zool. Lond. 232: 435-446. [ Links ]
Lange de Morretes, F. 1949. Ensaio de catálogo dos moluscos do Brasil. Arq. Mus. Paranaense 7: 5-216. [ Links ]
Lazaridou-Dimitriadou, M. & J. Daguzan. 1981. Etude de l’effet du “groupement” des individus chez Theba pisana (Mollusque: Gasteropode: Pulmone: Stylommatophore). Malacologia 20: 195-204. [ Links ]
Lucarz, A. & L. Gomot. 1985. Influence de la densite de population sur la croissance diametrale et ponderale de l’escargot Helix Aspersa Müller dans diferentes conditions d’elevage. J. Mollus. Stud. 51: 105-115. [ Links ]
Meireles, L.M.O., L.C. Silva, F.O. Junqueira & E.C.A. Bessa. 2008. The influence of diet and isolation on growth and survival in the land snail Bulimulus tenuissimus (Mollusca: Bulimulidae) in laboratory. Rev. Bras. Zool. 25: 224-227. [ Links ]
Olazarri, J. 1979. Los Moluscos plaga de los cultivos de “berro” en Salto, Uruguay. Comun. Soc. Malacol. Uruguay 5: 63-69. [ Links ]
Parodiz, J.J. 1963. Observaciones anatomicas sobre Omalonyx patera Doer., com una nota biografica acerca de Adolfo Doering (1848-1926). Sterkiana 12: 1-7. [ Links ]
Patterson, C.M. 1970. Self-fertilization in the land snail family Succineidae. J. Conch. 108: 61-62. [ Links ]
Patterson, C.M. 1971. Taxonomic studies of the land snail family Succineidae. Mal. Rev. 4: 131-202. [ Links ]
Peake, J. 1978. Distribution and ecology of the Stylommatophora, p. 429-526. In V. Fretter & J. Peake (eds.). Pulmonates. Academic, New York, New York, USA. [ Links ]
Poi de Neiff, A., J.J. Neiff & A. Bonetto. 1977. Enemigos naturales de Eichornia crassipes en el nordeste argentino y posibilidades de su aplicación el control biológico. Ecosur 4: 137-156. [ Links ]
Ratte, H.T. 1985. Temperature and insect development, p. 33-66. In K.H. Hoffmann (ed.). Environmental physiology and biochemistry of insects. Springer Verlag, Berlin, Berlin, Germany. [ Links ]
Roff, D.A. 1992. The evolution of life histories: theory and analysis. Chapman & Hall, New York, New York, USA. [ Links ]
Rundell, R.J. & R.H. Cowie. 2003. Growth and reproduction in Hawaiian succineid land snails. J. Mollus. Stud. 69: 288-289. [ Links ]
Salgado, N.C. & A.C.S. Coelho. 2003. Moluscos terrestres do Brasil (Gastrópodes operculados ou não, exclusive Veronicellidae, Milacidae e Limacidae). Rev. Biol. Trop. 51: 149-189. [ Links ]
Silva, L., L. Meireles, T. Vargas, F.O. Junqueira & E.C.A. Bessa. 2009. Life history of the land snail Habroconus semenlini (Stylommatophora: Euconulidae) under laboratory conditions. Rev. Biol. Trop. 57: 1217-1222. [ Links ]
Simone, L.R.L. 2006. Land and freshwater mollusks of Brazil. Fapesp, São Paulo, São Paulo, Brazil. [ Links ]
South, A. 1982. A comparison of the life cycles of the slugs Deroceras reticulatum (Müller) and Arion intermedius Normand on permanent pasture. J. Mollus. Stud. 55: 9-22. [ Links ]
South, A. 1992. Terrestrial Slugs: Biology, ecology and control. Chapman & Hall, London, London, United Kingdom. [ Links ]
Stearns, S.C. & J.C. Koella. 1986. The evolution of phenotypic plasticity in life-history traits: predictions of reaction norms for age and size at maturity. Evolution
40: 893-913. [ Links ]
Stearns, S.C. 1992. The evolution of life histories. Oxford University, Oxford, Oxfordshire, United Kingdom. [ Links ]
Tillier, S. 1981. South American and Juan Fernandez succineid slugs (Pulmonata). J. Mollus. Stud. 47: 125-146. [ Links ]
Tompa, A.S. 1984. Land snails (Stylommatophora), p. 47-139. In A.S. Tompa, N.H. Verdonk & J.A.M. Van Den Biggelaar (eds.). The Mollusca, vol. 7. Reproduction. Academic, New York, New York, USA. [ Links ]
Villalobos, C.M., J. Monge-Nájera, Z. Barrientos & J. Franco. 1995. Life-cycle and field abundance of the snail Succinea costaricana (Stylommatophora: Succineidae), a tropical agricultural pest. Rev. Biol. Trop. 43: 181-188. [ Links ]
Villalobos, C.M. & J. Monge-Nájera. 2004. Yearly body size distribution in the terrestrial snail Succinea costaricana (Stylommatophora: Succineidae). Brenesia 62: 47-50. [ Links ]
Zar, J.H. 2009. Biostatistical analysis. Prentice Hall, Upper Saddle River, New Jersey, USA. [ Links ]
*Correspondencia a: Lângia Montresor, Ana Teixeira & Teofânia Vidigal: 1. Departamento de Zoologia, Universidade Federal de Minas Gerais, Av. Presidente Antônio Carlos 6627, Belo Horizonte, MG, Brazil, CEP 31270-901; lcmontresor@gmail.com, annabio@pop.com.br, teofania.vidigal@gmail.com
Lângia Montresor: Laboratório de Malacologia, Instituto Oswaldo Cruz, FIOCRUZ, Av. Brasil 4365, Rio de Janeiro, RJ, Brazil, CEP 21.040-900; lcmontresor@gmail.com
Adriano Paglia:Departamento de Biologia Geral, Universidade Federal de Minas Gerais, Av. Presidente Antônio Carlos 6627, Belo Horizonte, MG, Brazil, CEP 31270-901; apaglia@icb.ufmg.br
1. Departamento de Zoologia, Universidade Federal de Minas Gerais, Av. Presidente Antônio Carlos 6627, Belo Horizonte, MG, Brazil, CEP 31270-901; lcmontresor@gmail.com, annabio@pop.com.br, teofania.vidigal@gmail.com
2. Laboratório de Malacologia, Instituto Oswaldo Cruz, FIOCRUZ, Av. Brasil 4365, Rio de Janeiro, RJ, Brazil, CEP 21.040-900; lcmontresor@gmail.com
3. Departamento de Biologia Geral, Universidade Federal de Minas Gerais, Av. Presidente Antônio Carlos 6627, Belo Horizonte, MG, Brazil, CEP 31270-901; apaglia@icb.ufmg.br
Received 01-VI-2011. Corrected 20-X-2011. Accepted 18-XI-2011.