Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.59 n.1 San José Mar. 2011
Population structure and stock assessment of Hoplias malabaricus (Characiformes: Erythrinidae) caught by artisanal fishermen in river-reservoir transition area in Brazil
José Luís Costa Novaes1 & Edmir Daniel Carvalho2
1. Departamento de Ciências Animais-Universidade Federal Rural do Semi-Árido, BR 110, km 47 s/nº, Bairro: Costa e Silva, CEP 59.625-900, Mossoró/RN, Brasil; novaes@ufersa.edu.br
2. Departamento de Morfologia, Instituto de Biociências, Universidade Estadual Paulista, Distrito de Rubião Jr. s/nº, CEP 18.618-000, Botucatu/SP, Brasil; carvalho@ibb.unesp.br
Abstract
Hoplias malabaricus is a viable economic alternative for artisanal fishermen who used to live on the banks of rivers that now are dammed in Brazil. In order to preserve the species natural stock, the main objectives of this study were to obtain bio-ecological information on this species reproduction, feeding dynamics and the description of the extent of its exploitation in river-reservoir transition area in Brazil. This way, from January to December 2005, different fishery catches were made with gill nets by artisanal fishermen, and a total of 426 individual H. malabaricus were obtained and sampled. From each specimen, the following biometric and biological data were analyzed: standard length, total weight, stomach contents, reproductive data such as sex, weight and gonadal maturation stage; with these data we estimated sex ratio, reproduction period and feeding habits. To assess growth parameters and infer on the degree of exploitation, the standard length frequency data were analysed using the FISAT (ELEFAN I) computer program. Data revealed a sex ratio of 0.8:1 (M:F), that reproduction occurs all year around, being more intense during spring and summer; it also showed that H. malabaricus is piscivorous. The growth parameters and mortality were estimated at: L∞=35.18cm, k=0.32 year, longevity=9.3 years, Z=1.25 years, M=0.63 year and F=0.62 year. The weight-length relationship was: ln Wt=–3.1663+2.7456 ln Lp. The yield-per-recruit analysis produced the following values: E=0.496, Emax=0.916, E0.1=0.701 and E0.5=0.378. Overall, our data suggest that the stock of H. malabaricus is not yet overfished in the studied area. Rev. Biol. Trop. 59 (1): 71-83. Epub 2011 March 01.
Key words: mortality, reproduction, feeding, Hoplias malabaricus, Upper Paraná, yield-per-recruit, growth.
Resumen
Hoplias malabaricus representa una alternativa económica viable para los pescadores artesanales que viven a la orilla de ríos que actualmente cuentan con represas en Brasil. El objeto del presente estudio fue evaluar la dinámica de la reproducción, alimentación, crecimiento y nivel de explotación de la especie nativa Hoplias malabaricus, capturada por pescadores artesanales en la zona de transición entre el río Paranapanema y el Embalse de Jurumirim. En 2005, los especimenes de H. malabaricus se obtuvieron utilizando redes agalleras de forma mensual y se registraron las siguientes características y datos biológicos de cada uno: longitud estándar, peso corporal, sexo, estadio reproductivo, peso de las gónadas y contenido estomacal. Estos datos fueron utilizados para estimar la proporción sexual, el período reproductivo y los hábitos alimenticios. Para evaluar los parámetros de crecimiento y el grado de explotación, los datos de frecuencia de la longitud estándar fueron analizados utilizando el software FISAT a través de la rutina ELEFAN I. Según los resultados obtenidos, la proporción sexual fue de 0.8:1 (macho:hembra); la reproducción fue observada durante todo el año, pero sobre todo en la primavera y el verano. El hábito alimentario fue piscívoro. Los parámetros de crecimiento y mortalidad fueron estimados en: L∞=35.18cm, k=0.32/año; longevidad= 9.3, Z=1.25/año, M=0.63/año y F=0.62/año. La relación longitud-peso fue: Wt=-3.1663+2.7456lnLp. El análisis de rendimiento por recluta estimó los siguientes valores: E=0.496, Emax=0.916, E0.1=0.701 y E0.5=0.378. Puede concluirse que la población de H. malabaricus en la región estudiada todavía no está siendo altamente explotada.
Palabras claves: mortalidad, reproducción, alimentación, Hoplias malabaricus, Alto Paraná, producción-por-recluta, crecimiento.
The species Hoplias malabaricus (Bloch 1794), known as traíra, is a common predator at the top of the food chain during its adult. It lives in various aquatic microhabitats, such as river beds, lagoons, and flooded grasslands, which are principally shallow water environments that are near marginal submersed vegetation. H. malabaricus is present in almost all of Brazils hydrographic basins (Winemiller 1989, Carvalho et al. 2002). Because H. malabaricus sells for a good price due to the quality and taste of its flesh (Loureiro & Hahn 1996) and because it can easily adapt to semi-lentic environments (Suzuki & Agostinho 1997, Prado et al. 2006), this species is a viable economic alternative for artisanal fishermen who used to live on the banks of rivers that now are dammed. Moreover, the species is a good, ecologically responsible option compared to the alarming practice of introducing allochthonous and/or exotic species; in decades past, such species abounded along the entire main axis of the Paranapanema River without ever producing satisfactory results (Carvalho et al. 2005). Therefore, it is crucial that the institution that manages H. malabaricus develop a management and conservation program to i) preserve this economically viable and sustainable artisanal fishery in traditional riverine communities and ii) mitigate the damages occasioned on a local and regional scale by building large reservoirs.
At the Jurumirim Reservoir, which is located in the upper Paranapanema River Basin, artisanal fishing is practiced by a small group of fishermen (approximately 24 families) living in a small village on the banks of the Paranapanema River, at the point where it empties into the reservoir. Carvalho et al. (1998) reported that the aquatic resources in the reservoir consist mainly of native species, despite the unsuccessful and indiscriminate attempts made by fish stocking programs during the 1970/1980 by the former energy concessionaire in compliance with then prevailing legislation. A survey conducted in this small fishing community reported the productivity of the fishery to be 15.7 kg/ha/year, with a yield of 12.5 tons/year; the stretch of river surveyed is one of the most productive among the reservoirs of the Upper Paraná River Basin and H. malabaricus is one of its principal fish species (Novaes 2008). Therefore, bio-ecological studies and assessment of the population dynamics of H. malabaricus are necessary to estimate the current stock in this unique section of the reservoir. Results of such studies can be used to regulate fishery activities within the context of multiple uses of the open public water resources.
The aim of this survey was to characterize the population biology of H. malabaricus. In particular, we evaluated reproduction (e.g., breeding season and possible influences of environment and/or limnological variables on reproduction dynamics), feeding, growth parameters (L∞, k) and mortality (total, natural and fishery). We also estimated the present day stock of this species using Beverton & Holts yield-per-recruit analysis by model based on data from the artisanal fisheries catches.
Materials and methods
Data collection: H. malabaricus specimens were caught on a monthly basis with gill nets (with meshes with 10cm between opposing knots) by artisanal fishermen, from January to December 2005. The fish were landed at the fishing village of Bairro da Ponte, which is located at the mouth of the Paranapanema River at the point where it empties into the Jurumirim Reservoir (23º28'30" S - 48º37'59" W). This stretch of the Paranapanema River is characterized by a transition ecosystem between river and reservoir; it features various marginal oxbowtype lagoons and reasonably well-preserved segments of riparian woods (Carvalho et al. 2009). This study analyzed a total of 426 fish. For each specimen sampled, total weight (g), standard length (cm), sex, gonad weight (g) and maturation stage were determined. The stomachs that were partially full and full (repletion degree) were extracted, weighed and fixed in 5% formaldehyde for later analysis.
Each month the following limnological variables were measured in situ with a Horiba model U22 water quality analyzer: pH, water temperature, dissolved oxygen and electrical conductivity. The transparency of the water was also determined in situ using a Secchi disk. Water samples (~500ml) were collected in appropriate bottles for laboratory analysis of total alkalinity and chlorophyll a contents. In the laboratory, the total alkalinity was estimated by titration with 0.1 N of H2SO4 and chlorophyll a content was estimated using Millipore AP20 membranes and cold acetone (90%) extraction (Nogueira et al. 1999). The limnological variables and water samples were obtained at a distance of approximately 50m from the river bank. Information about monthly accumulated rainfall and the reservoirs water level benchmark was supplied by the concessionaire of the Jurumirim Hydroelectric Plant (Duke-Energy Generation Paranapanema).
Data analysis: To analyze reproduction dynamics and feeding data, the data were grouped into the following seasons: summer (January, February, March of 2005), autumn (April, May, June of 2005), winter (July, August and September of 2005) and spring (October, November and December of 2005).
Reproduction dynamics: The sex ratio was ascertained using the gender data from the 426 individuals sampled; the data were subjected to the x2 test (p<0.05) to test the null hypothesis that the sex ratio was 1:1.
Because the reproduction characteristics of females are better than those of males as an indicator of the reproduction period (Wootton 1995), only individuals of the female sex (240 individuals) were used to analyze reproduction dynamics. The following parameters were evaluated: i) the seasonal distribution of the macroscopic stages (immature, maturing, ripe, spent, and at rest) of development of the gonads; and ii) variation of the mean gonadosomatic index (GSI) (weight of gonads expressed as a percentage of body weight) as a function of time. A one-way ANOVA (p<0.05) was used to test the null hypothesis that the GSI mean values were the same during all seasons. The abiotic data did not meet the normality and homogeneity of variance criteria required for the use of parametric statistics, so we used the non-parametric Spearman correlation (p<0.05) to investigate the relationship between GSI and limnological and environmental variables.
Feeding: Only stomachs with contents were analyzed (n=101). Stomach contents were extracted, sorted, separated by item, counted and weighed. The food items were grouped into taxonomic categories (Family or Order) or in large groups. Occurrence frequency and group volumes (Hyslop 1980) were calculated and used to calculate the food index (IAi) proposed by Kawakami & Vazzoler (1980). The IAi results are shown as a percentage.
Length-weight relationships: For this analysis we used data from all 426 individuals. Exploratory data analysis (t test) showed no statistical difference in the value of b between females and males, thus we grouped males and females for further analysis. To examine the length-weight relationship, we used the following mathematical expression after logarithmic transformation of the data: ln Wt=a+blnLp, where Wt=total weight, Lp=standard length, a=a constant and b=inclination of slope. The parameters a and b were evaluated using the least squares method (Zar 1996). A t test (p<0.05) was performed to test the null hypotheses that b=0 and b=3 (isometric condition).
Length growth: Based on the monthly distributions of standard length frequency, the following growth parameters were obtained: L∞=asymptotic standard length (cm) and k=growth coefficient (years). These parameters were evaluated using the ELEFAN I routine within the computer program FiSAT II (Gayanilo Jr. et al. 2005), which is based on the Von Bertalanffy equation: Lt=L∞[1–exp(–k(t– t0))], where Lt=length in age t, L∞=asymptotic standard length; k=growth coefficient and t0=parameter referring to length of fish at birth.
Length and age structure: The lengthage structure was evaluated using the Von Bertalanffy equation (described above). We use the parameters L∞ and k calculated in this paper and replaced t with ages that ranged from 0 to 9.5 years. The parameter t0 was considered to be 0 because biologically speaking it is not significant (Sparre & Venema 1997).
Longevity potential: This parameter was evaluated using Paulys equation (1980): Tmax≈3/K.
Mortality rate: The natural mortality rate was evaluated using Pauly´s (1980) empirical model: lnM=– 0.0152–0.279lnL∞+0.6543lnK+0.4634lnT, where L∞ and k are the growth parameters obtained in this study from the Von Bertalanffy equation and T=mean water temperature (°C), which was 21.6°C. Total mortality rate (Z) was estimated using the following two techniques in an effort to diminish the degree of uncertainty associated with this evaluation: (i) Beverton & Holts model: Z=k*(L∞–Lm)/(Lm–Lc), where Lc=mean length of first capture (i.e., the time at which the fish are recruited for the fishery) and Lm=mean length starting from Lc. For the purpose of this study Lc was considered to be 19.0cm and Lm was 22.3cm. L∞ and k were obtained from the Von Bertalanffy equation; (ii) Ault & Ehrhardt´s model: [(L∞–Lmax)/(L∞–Lc)] z/k=Z(Lc–L)+k(L∞–Lm)/Z(Lmax–Lm)+k(L∞– Lm), wher Lmax=the largest individual sampled (33.3cm) and L∞, Lc, Lm and k are the same parameters described above. The mortality rate due to fishing (F) was calculated as the difference between the total mortality rate (Z) and the natural mortality rate (M) (i.e., F=Z–M).
Relative yield per recruit: (Y´/R): This parameter was calculated using Beverton & Holts method and the Knife Edges option in the program FiSAT II (Gayanilo Jr. et al. 2005) according to the following model: Y'/R=EUM/K[1–(3U/1+m)+(3U2/1+2m)– U3/1+3m)], where m=(1–E)/(M/k)=k/Z; U=1– (Lc/L∞) and E=F/Z (exploitation rate). Because the values Z and F calculated using Beverton & Holt's method and Ault & Ehrhardt´s method were similar, we opted to use the data obtained by the first technique for further analysis. FiSAT II also was used to evaluate the following: Emax (exploitation rate of maximum sustainable yield); E0.1 (exploitation rate at which the marginal increment of Y'/R is 10% of its virgin stock) and E0.5 (exploitation rate that will result in a 50% reduction of the non-exploited biomass).
Biomass per recruit (B´/R): This parameter is a measure of the mean survivor biomass as a function of the mortality caused by the fishery. It was evaluated using the mathematical expression: B´/R=(Y´/R)/F, where Y´/ R=relative yield per recruit and F=mortality caused by the fishery.
Results
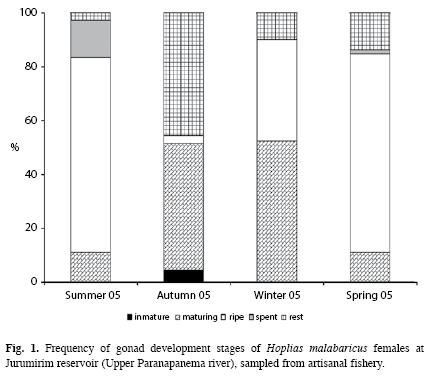
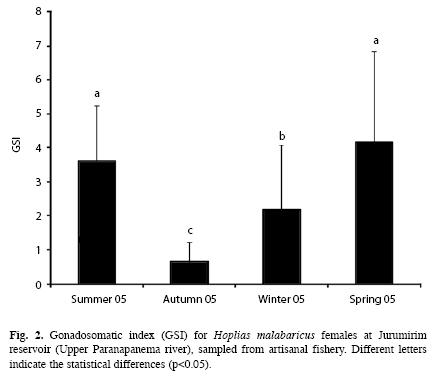
Reproduction dynamics: In this study, males represented 43.7% of the fish captured (n=186) and females represented 56.3% (n=240), which resulted in the following M:F ratio:0.8:1 (x2=6.594, p<0.0102). Seasonally, males predominated in summer [males=38% (51.4%), M:F=1:0.9; X2=0.014, p<0.9075] and females during the remaining seasons of the year: autumn [females=68% (57.1%), M:F=0.7:1, x2=2.151, p<0.1425]; winter [females=64% (52.0%), M:F=0.9:1, x2=0.23, p<0.7183] and spring [females=72% (65.5%), M:F=0.5:1, x2=9.90, p<0.0017]. Specimens of ripe females were sampled during all four seasons of the year but the relative frequencies (%) were higher in summer (72.2%) and spring (73.6%) (Fig. 1). The statistical analysis rejected the null hypothesis of equal mean values of the GSI (ANOVA, F (3.236)=43.979, p<0.0000) and revealed that values in spring and summer were statistically different (higher) than those in autumn and winter (Fig. 2). Spearman correlation analysis showed that the highest correlations occurred between GSI and temperature, precipitation, (positive) and the water level benchmark (negative) (Table 1).
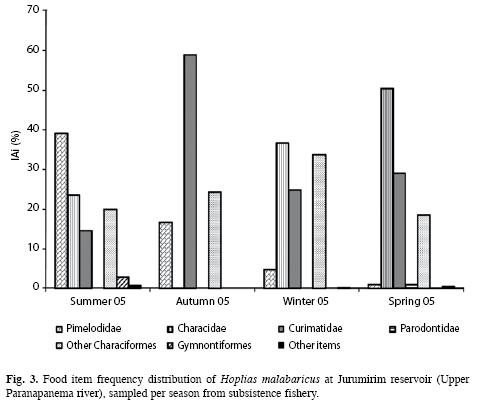
Feeding: The diet of H. malabaricus consisted almost exclusively of fish. Items such as Anuro, land insects, aquatic insects and vegetable material had little impact on the IAi frequency and totalled <1%. Among the fish items, a seasonal variation in the frequency of the IAi can be clearly observed; in summer the Pimelodidae had a higher frequency (39.12%); in autumn the Curimatidae predominated (with 58.92%) and in winter and spring the Characidae predominated (36.68% and 50.48%, respectively) (Fig. 3).
Length-weight relationships: The standard length and total weight of captured specimens ranged from 11.1 to 33.3cm (Lpaverage=22.0cm; SD=2.2) and from 29.59 to 722.01g (Wtaverage=209.7g, SD=67.4), respectively. The linear regression of the transformed data for the length-weight relationship can be described by the following equation: ln Wt=–3.1663+2.7456 lnLp and R2=0.9012. The t test result refuted the null hypothesis that b=zero(t(1.430)=62.62, p=0.00) and that b=3 (t(1.430)=5.90, p<0.01), which indicates that H. malabaricus exhibits negative allometric growth at the Jurumirim Reservoir.
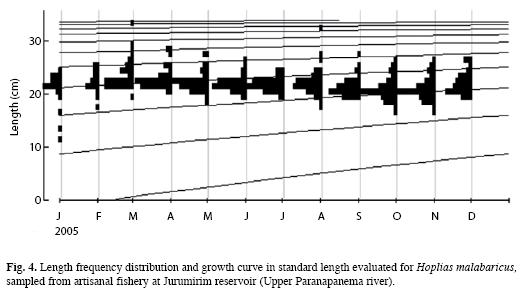
Length growth: The values of the parameters that describe the length growth of H. malabaricus were: L∞=35.18cm and k=0.32 year (Table 2). Therefore, the Von Bertalanffy growth equation can be described as follows: Lt=35.18[1–exp(–0.32(t–t0))]. The length growth curve suggests the presence of four cohorts in the captured sample, although this result is specific to the gear used by the artisanal fishermen in a particular section of the Jurumirim Reservoir (Fig. 4).
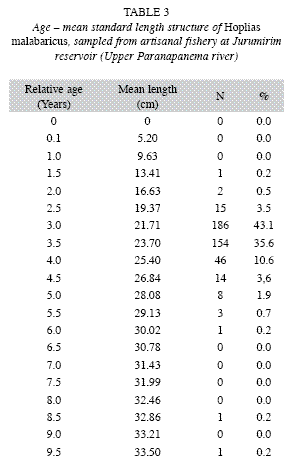
Length and age structure: The lengthage structure of the studied population (Table 3) demonstrated that capture of H. malabaricus by the artisanal fishery concentrated on individuals 2.5-4.5 years of age with a standard length of 19.37-26.84cm; these fish represented 96.4% of the population. Individuals between 3.0 and 3.5 years old were the most representative of this kind of fishery in the Jurumirim Reservoir, at 43.1% and 35.6% of the captures, respectively.
Longevity potential: The maximum life expectancy of H. malabaricus (tmax) was estimated to be 9.3 years (Table 2).
Mortality rates: The natural mortality rate (M) was estimated to be 0.71 year. The total mortality rates calculated using the methods of Beverton & Holt and Ault & Ehrhardt were 1.25 years and 1.24 years, respectively. The mortality rate due to fishing (F) was evaluated to be 0.54 year (Z=model Beverton & Holt) and 0.53 year (Z=model Ault & Ehrhardt) (Table 2). Thus, the rate of natural mortality was higher than the rate of mortality due to fishing (M>F).
Yield (Y´/R) and biomass (B´/Y) per recruit: The present exploitation level of the stock of H. malabaricus was E=0.496. This value was less than Emax=0.916 and E0.1=0.758 and higher than E0.5=0.381 (Table 2). Because E was <Emax, we infer that the stock of this species is not being overfished by this group of artisanal fishermen.
Discussion
Reproduction dynamics: The sex ratio of fish populations tends to be 1:1, although deviations may occur and seasonal variations are common (Nikolsky 1963). The sex ratio is governed by diverse factors, including mortality, longevity and growth rate, that lead to differences in capturing rates of the two sexes (King & Etim 2004). The sex ratio for H. malabaricus caught in the artisanal fishery at Bairro da Ponte deviated significantly from 1:1, with a predominance of females. Two possible explanations for this difference are as follows: (1) Behavioral differences between sexes leave females more vulnerable to capture. Males of the species are more territory conscious (Barbieri 1989, Prado et al. 2006) and also provide parental care by guarding the nest. The eggs are deposited at the bottom in small depressions, tree trunks, and under leaves or other surfaces. After spawning the females tend to leave the site, whereas the males remain to take care of the nest (Prado et al. 2006). For this reason, females tend to be more susceptible to be captured by fishing gears than males. (2) Differences between genders in growth and mortality rates affect capture. However, exploratory analyses of the growth data indicated no differences in these parameters between the sexes of H. malabaricus in Jurumirim Reservoir. Barbieri (1989) did not detect any growth difference between males and females of this species population inhabiting the Monjolinho Reservoir in Southeastern Brazil. Based on these results, we infer that the observed differences in sex ratio found in this study may be more related to reproductive behavior than to growth and mortality rates.
H. malabaricus exhibited a long reproduction period that encompassed spring and summer. The reproduction period also was positively correlated with water temperature and the monthly mean rainfall. Studies of the reproductive biology of H. malabaricus made in various environments, repeatedly gave similar results: greater reproductive intensity during the rainy season (Barbieri 1989, Suzuki & Agostinho 1997, Marques et al. 2001, Prado et al. 2006). Wootton (1995) and Lowe-McConnell (1987) reported that various exogenous factors can affect the reproduction process; some of these "triggers" for the reproduction of tropical fish include photoperiod, temperature, water flow, water level and rainfall. Our findings thus corroborate the results of previous studies that show that precipitation is one of the principal reproduction triggers for H. malabaricus.
Because H. malabaricus uses reproductive and behavioral tactics such as parental care, nest construction and reproduction in shallow environments and close to river margins, abrupt oscillations in the reservoir water level that cause retraction and expansion of the margins may increase the mortality rate of eggs and larvae, thereby may impact recruitment (Suzuki & Agostinho 1997) and ultimately affect the renewal capacity of the fish stock. Such changes may be accompanied by a drop in the fishery catch.
Feeding: In the studied area of the Jurumirim Reservoir, stomach contents of H. malabaricus consisted almost exclusively of fish; the presence of aquatic and land insects, vegetable matter, and Anuro was considered to be incidental. The piscivorous habit of adults is typical for this species (Winemiller 1989, Loureiro & Hahn 1996, Carvalho et al. 2002, Luz- Agostinho et al. 2006). A pattern of seasonality in feeding was clearly evident, with a predominance of Pimelodidae in summer, Curimatidae in autumn and Characidae (lambaris and piquira) in spring. The opportunistic choice of food seems to be one of the species' characteristics (Carvalho et al. 2002); which efficiently exploits the environment's available resources at a given time. Adaptability to a different diet is one life the tactics of a great number of tropical fish species (Lowe-McConnell 1987, Hahn & Fugi 2007) and is of great importance to H. malabaricus, as it allows adjustments to environmental conditions without alterations in population biomass.
The fact that H. malabaricus is a predator at the top of the chain (second and sometimes third order) at the Jurumirim Reservoir, may have a negative impact on the fishery. In long trophic chains, energy is lost after it moves from one level to the other and therefore the biomass sustained at the top of the chain tends to decrease. These long food chains, in tropical and particularly Brazilian reservoirs, are one of the reported causes for low fishery productivity (Fernando & Holcik 1982, Petrere Jr. 1996, Gomes & Miranda 2001).
Length-weight relationships: As a result of the capture gear employed (gill nets with meshes with more than 10cm between opposing knots), the catches of H. malabaricus by the artisanal fishery consist almost entirely of adult individuals, thus preserving juveniles. From this point of view, it seems that fishing pressure does not yet constitute a threat to the stock and the recruitment of the species. However, fishery pressure exerted only on larger individuals may lead to genetic degradation of the population, as reported by Conover & Munch (2002). In other words, the removal of larger individuals favours the survival of the smaller and slower growing individuals, which alters the genetic variability of the population by eliminating the genes that promote quick growth of the population (Conover & Munch 2002). Therefore, we agree with Agostinho et al. (2007, 2008) premise that the paradigms established in relation to the minimum size of first capture should be reviewed, as the preservation of longer individuals is of vital importance to maintain an economically viable fishery and for stock resilience.
In our study, H. malabaricus demonstrated a negative allometric growth pattern (i.e., it grew more quickly in length than in weight). This result differs from other findings for this species in different Brazilian reservoirs [Lobo, b=3.0 (Barbieri et al. 1982) and Monjolinho, b=3.1 (Barbieri 1989)], which both indicated isometric growth in those two environments. Even so, the value of b found in this study (2.7456) remained within the range of oscillation (2.5-3.5) reported by Le Cren (1951). The length-weight relationships provided a good model for the variables evaluation (R2=0.9012).
Various factors can influence fish growth, such as sex, age, season of the year, type of nutrition, physiological conditions, difference in availability of food, period of reproduction and growth increment (Lalèyè 2006). When studying Geophagus brasiliensis (Cichlidae) in the Lages Reservoir, Dos Santos et al. (2004) concluded that the oligotrophic characteristic and the resulting lack of food combined with stress produced by the variation of the reservoir´s water level, might contribute to negative allometric growth of the species. At the point where the Paranapanema River flows into the Jurumirim Reservoir, its trophic state varies between oligotrophic and mesotrophic depending on the season of the year (Nogueira et al. 1999). In addition, the water levels of the marginal lagoons and channel of that section of the river vary considerably (4m between low and high water) as a result of the operational system of the Jurumirim Hydroelectric Plant (Granado & Henry 2008). It is therefore reasonable to assume that availability of food in the aquatic environment may also vary seasonally and thus influence this (negative allometric) growth coefficient, despite the fact that the species efficiently uses the available resources.
Length growth and longevity potential: The growth parameters estimated for H. malabaricus (L∞=35.18cm, k=0.32 years and longevity of 9.3 years) indicated that this species grows slowly. According to some authors, species with specialized food and elevated trophic level have a tendency to slower growth, great longevity and retarded gonads maturation (Lowe-McConnel 1987, Winnemiller 1989, Wootton 1995). We thus include H. malabaricus from this part of the Jurumirim Reservoir in this category because of its specialized food consumption (piscivorous), high trophic level (consumer of second and/or third order) and great longevity.
Studies of growth parameters of H. malabaricus in other environments revealed different values of L∞, k and longevity (Barbieri 1989, Angelini & Agostinho 2005). Because various endogenous and exogenous factors affect fish growth and longevity (Wootton 1995), it is difficult to identify the causal factors for these different results.
Length and age structure: Evaluation of the age structure indicated that individuals between 3.0 and 3.5 years old made up the bulk of captures in the artisanal fishery; they represented 78.7% of landings and consisted of specimens with a standard length of >21.0cm. The individuals within these age classes were adults that may have reproduced once already, as the gonad analysis revealed that all individuals greater than >17.1cm long are mature.
Mortality rates: In this study, the natural mortality rate (M) exceeded the fishery mortality rate (F). Caddy & Mahon (1995) discussed the relationship between M and F and considered the maximum sustainable yield (MSY) to occur when F=M. Based on this concept, as the biological reference point (BRP), the exploitation of the stock of H. malabaricus in the study area does not suggest overfishing. However, scant evidence exists of such a relationship between M and F, so the maximum sustainable yield might be higher than M in certain cases (Caddy & Mahon 1995).
Yield and biomass per recruit: The relative yield-per-recruit analysis indicated that E (0.496) was bellow the values E0.1(0.758) and Emax(0.916). E0.1 has been adopted as the BRP for many fisheries and is defined as the exploitation rate at which the marginal increase of Y'/R is 10% of its virgin stock. In this study, the value of E remained below E0.1, which indicates that the stock of H. malabaricus is not yet overfished, which in turn would warrant a natural increase of fishing effort. On the other hand, the value of E exceeded the value of E0.5, which means that F diminished the stock's virgin biomass by 50%. Thus, a more conservative stance should be assumed and an increase in the level of fishery effort is not advisable.
Our study of the population dynamics of H. malabaricus based on samples derived from an artisanal fishery located in the Jurumirim Reservoir in the transition area between the Paranapanema River and the reservoir led to the following conclusions: i) H. malabaricus exhibited reproduction dynamics and diet similar to those determined in other ecosystems, such as a long reproduction period, more intense reproductive activity during the rainy season, and piscivorous and opportunistic habits; ii) the fishery stock did not show evidence of overfishing and thus may continue to be exploited in a sustainable way by the artisanal fishing community; iii) H. malabaricus continues to represent an important source of income for these fishermen, thus mitigating the negative effects of damming on migratory species; iv) we recommend emphatically that no increase in fishery effort should be tolerated, lest the stock of this species be threatened.
Acknowledgments
We wish to thank the fishermen of the fisher community Bairro da Ponte who answered willingly and spontaneously the production forms; Joel Marques who assisted us in establishing contacts with local fishermen; the laboratory technicians Ricardo André dos Santos Teixeira and Renato Devidé; the entire team of the Laboratory for Fish Ecology of UNESPBotucatu; the COORDENAÇÃO DE APERFEIÇOAMENTO DE PESSOAL DE NÍVEL SUPERIOR (CAPES), for concession of a scholarship and Armin Schmid for assistance in the English version.
References
Agostinho, A.A., L.C. Gomes & F.M. Pelicice. 2007. Ecologia e manejo de recursos pesqueiros em reservatórios do Brasil. Eduem, Maringá, Brasil. [ Links ]
Agostinho, A.A., F.M. Pelicice & L.C. Gomes. 2008. Dams and de fish fauna of the Neotropical region: impacts and management related to diversity and fisheries. Braz. J. Bio. 68: 1119-1132. [ Links ]
Angelini, R. & A.A. Agostinho. 2005. Parameter estimates for fishes of the Upper Paraná river floodplain and Iatipu reservoir (Brazil). NAGA 1-2: 53-57. [ Links ]
Barbieri, G., J. Verani & M.C. Barbieri. 1982. Dinâmica quantitativa da nutrição de Hoplias malabaricus (Bloch, 1974), na represa do Lobo (Brotas- Itirapinas/ SP.). (Pices, Erythrinidae). Rev. Bras. Biol. 42: 295-302. [ Links ]
Barbieri, G. 1989. Dinâmica da reprodução e crescimento de Hoplias malabaricus (Bloch, 1974) (Osteichthyes, erutrhynidae) da represa do Monjolinho, São Carlos/ SP. Rev. Bras. Zool. 6: 225-233. [ Links ]
Caddy, J.F. & R. Mahon. 1995. Reference points for fisheries management. FAO, Roma. [ Links ]
Carvalho, E.D., C.Y. Fujihara & R. Henry. 1998. A study of the ichthyofauna of the Jurumirim reservoir (Paranapanema River, São Paulo, Brzil): fish production and dominant species at three sites. Verh. Int. Verein. Limn. 26: 2199-2202. [ Links ]
Carvalho, L.N., C.H.V. Fernandes & V.S.S. Moreira. 2002. Alimentação de Hoplias malabaricus (Bloch, 1794) (Osteichthyes, Erytrinidae) no rio Vermelho, Pantanal Mato-grossense. Rev. Bras. Zoocie. 4: 227-236. [ Links ]
Carvalho, E.D., R.J. de Castro, V.F.B. da Silva & A.P. Vidotto. 2003. A estrutura das assembléias de peixes nas zonas de ecótono da represa de Jurumirim (Alto rio Paranapanema, SP), p. 249-278. In R. Henry (ed.). Ecótonos nas interfaces dos ecossistemas aquáticos. RiMA, São Carlos, Brasil. [ Links ]
Carvalho, E.D., S.G. de C. Britto & M.L. Orsi. 2005. O panorama das introduções de peixes na bacia hidrográfica do rio Paranapanema, Alto Paraná, Brasil, p. 253-274. In O. Rocha, E.L.G. Espíndola, N. Fenerich-Verani, J.R. Verani & A.C. Reitzler (eds.). Espécies invasoras em águas doces – estudo de caso e propostas de manejo. Edufscar, São Carlos, Brasil. [ Links ]
Conover, D.O. & S. Munch. 2002. Sustaining fisheries yields over evolutionary time scales. Science 297: 94-96. [ Links ]
Fernando, C.H. & J. Holčík. 1982. The nature of fish communities: A factor influencing the fishery potential and yields of tropical lakes and reservoirs. Hydrobiologia 97: 127-140. [ Links ]
Gayanilo Jr., F.C., P. Sparre & D. Pauly. 2005. FAOICLARM stock assessment tools II (FiSAT II). Revised version. User's guide. FAO Computerized Information Serie (Fisheries), Roma, Italia. [ Links ]
Gomes, L.C. & L.E. Miranda. 2001. Riverine characteristics dicate composition of fish assemblages and limit fisheries in reservoirs of the Upper Paraná River basin. Regulated River: Research & Management 17: 67-76. [ Links ]
Granado, D.C. & R. Henry. 2008. The influence of the hydrologic pulse on the water physical and chemical variables of lateral lakes with different connection levels to Paranapanema River in the mouth zone at Jurumirim Reservoir (São Paulo, Brazil). Acta Limnol. Bras. 20: 265-275. [ Links ]
Hahn, N. & R. Fugi. 2007. Alimentação de peixes em reservatórios brasileiros: alterações e conseqüências nos estágios iniciais do represamento. Oecol. Bras. 11: 469-480. [ Links ]
Hyslop, E.J. 1980. Stomach Contents Analysis – a Review of Methods and their Application. J. Fish. Biol. 17: 411-429. [ Links ]
Kawakami, E. & G. Vazzoler. 1980. Método gráfico e estimativa de índices alimentares aplicado no estudo de alimentação de peixes. Bol. Inst. Oceanogr. 29: 205-207. [ Links ]
King, R.P. & L. Etim. 2004. Reproduction, growth, mortality and yield of Tilapia mariae Boulenger 1899 (Cichlidae) in a Nigerian rainforest wetland stream. J. Appl. Ichthyol. 20: 502-510. [ Links ]
Lalèyè, P.A. 2006. Length-weight and length-length relationships of fish from the Ouémé River in Bénin (West Africa). J. Appl. Ichthyol. 22: 502-510. [ Links ]
Le Cren, E.D. 1951. The length-weight relationship and seasonal cycle in gonad weight and condition in Perch (Perca fluviatilis). J. Anim. Ecol. 20: 201- 219. [ Links ]
Loureiro, V.E. & N. Hahn. 1996. Dieta e atividade alimentar da traíra, Hoplias malabaricus (Bloch, 1794) (Osteichthyes: Erythrinidae), nos primeiros anos de formação do reservatório de Segredo – PR. Acta Limn. Bras. 8: 195-205. [ Links ]
Lowe-McConnell, R.H. 1987. Ecological studies in tropical fish communities. Cambridge, Cambridge, Great Britain. [ Links ]
Luz-Agostinho, K.D.G., L.M. Bini, R. Fugi, A.A. Agostinho & H.F. Júlio Jr. 2006. Food spectrum and trophic structure of the ichthyofauna of Corumbá reservoir, Paraná river basin, Brazil. Neotrop. Ichthyol. 4: 61-68. [ Links ]
Marques, D.K.S., H. de C.B. Gurgel & I. Lucena. 2001. Época de reprodução de Hoplias malabaricus Bloch, 1794 (Osteichthyes, Erythrinidae) da barragem do rio Gramame, Alhandra, Paraíba, Brasil. Rev. Bras. Zoocie. 3: 61-67. [ Links ]
Nikolsky, G.V. 1963. The Ecology of Fishes. Academic, London, Great Britain. [ Links ]
Nogueira, M.G., R. Henry & F.E. Maricatto. 1999. Spatial and temporal heterogeneity in the Jurumirim reservoir, São Paulo, Brazil. Lake and Reservoir Management 4: 107-120. [ Links ]
Novaes, J.L.C. 2008. Estudo comparativo da pesca artesanal em dois grandes reservatórios do alto Paraná: Barra Bonita (rio Tietê) e Jurumirim (rio Paranapanema). Ph.D. Thesis, Universidade Estadual Paulista, Botucatu, Brasil. [ Links ]
Pauly, D. 1980. On the interrelationships between natural mortality, growth parameters and mean environmental temperature in 175 stocks. J. Cons. CIEM. 39: 175-192. [ Links ]
Petrere Jr., M. 1996. Fisheries in large tropical reservoirs in South America. Lakes & Reservoirs: Research and Management 2: 111-133. [ Links ]
Prado, C.P.A., L.M. Gomeiro & O. Froehlich. 2006. Spawning and parental care in Hoplias malabaricus (Teleostei, Characiformes, Erytrinidae) in the Southern pantanal, Brazil. Braz. J. Biol. 66: 697-702. [ Links ]
Dos Santos, A.F.G.N., L.N dos Santos & F.G. Araújo. 2004. Water level influences on body condition of Geophagus brasiliensis (Perciformes: Cichlidae) in a Brazilian oligotrophic reservoir. Neotrop. Ichthyol. 2: 151-156. [ Links ]
Sparre, P. & S.C. Venama. 1997. Introdução à avaliação de mananciais de peixes tropicais. Parte 1: Manual. FAO, Roma, Italia. [ Links ]
Suzuki, H.I. & A.A Agostinho. 1997. Reprodução de peixes do reservatório de Segredo, p. 163-183. In A.A. Agostinho & L.C. Gomes (eds.). Reservatório de Segredo: bases ecológicas para o manejo. Eduem, Maringá, Brasil. [ Links ]
Winemiller, K.O. 1989. Ontogenetic diet shifts and resource portioning among piscivorous fishes in the Venezuela llanos. Environ. Biol. Fishes 26: 177-199. [ Links ]
Wootton, R.J. 1995. Ecology of teleost fishes. Chapman and Hall, London, Great Britain. [ Links ]
Zar, J.H. 1996. Biostatistical analysis. Prentice-Hall, New Jersey, USA. [ Links ]
Correspondencia a:José Luis Costa Novaes. Departamento de Ciências Animais-Universidade Federal Rural do Semi-Árido, BR 110, km 47 s/nº, Bairro: Costa e Silva, CEP 59.625-900, Mossoró/RN, Brasil; novaes@ufersa.edu.br
Edmir Daniel Carvalho Departamento de Morfologia, Instituto de Biociências, Universidade Estadual Paulista, Distrito de Rubião Jr. s/nº, CEP 18.618-000, Botucatu/SP, Brasil; carvalho@ibb.unesp.br
Received 22-IV-2010. Corrected 10-IX-2010. Accepted 12-X-2010.