Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.58 suppl.3 San José Oct. 2010
Temporal and Spatial Distribution Patterns of Echinoderm Larvae in La Parguera, Puerto Rico
Stacey M. Williams & Jorge García-Sais
Abstract
This study describes temporal and spatial abundance patterns of echinoderm larvae in La Parguera,Puerto Rico. For the temporal study, larvae were sampled by a series of monthly tows taken with a 64μm mesh net between the new and full moon from April 2005 to July 2006, September 2006 and August 2007. In order to measure spatial variation of echinoderm larval bundances, oblique tows were taken with 64 and 202μm mesh nets at seven different sites within the shelf, at the shelf-edge, and at a nearby oceanic stations during August 2007. Overall, Echinoidea (sea urchin) exhibited the highest abundance with a total of 11 921 larvae, representing 52.5% of the total collection. Ophiuroidea (brittle star) ranked second in abundance with 45.6% of the total larvae. Holothuroidea (sea cucumber) and Asteroidea larvae (sea star) accounted for less than 2% of the total echinoderm larval collection. Early larval stages (2-8 day old) of Diadema antillarum represented 20% of the total Echinoidea larvae. There was no marked seasonal trend of echinoderm larval abundance; Echinoidea and Ophiuroidea larvae were present in all monthly samples indicating that reproduction occurs year-round. Peak abundances of later-stage Echinoidea larvae were observed during January, July and October and of later-stage Ophiuroidea larvae during June, August and October. The observed peaks of later-stage larval abundances may be indicative of higher recruitment activity during these months. There was a significant difference of echinoderm larval abundance between spatial stations, with higher abundances collected at the shelf-edge. Later-stage (~24 day old) D. antillarum larvae were mostly collected at shelf-edge and oceanic locations. In addition, the 64mm mesh net was more efficient for collection of echinoderm larvae than the 202mm mesh net. Rev. Biol. Trop. 58 (Suppl. 3): 81-88. Epub 2010 October 01.
Key words: Echinodermata, larvae, Diadema antillarum, Puerto Rico, Caribbean.
Resumen
Este estudio describe patrones de abundancia temporal y espacial de larvas de equinodermos en La Parguera, Puerto Rico. La distribución temporal de equinodermos se caracterizó mediante una serie de arrastres mensuales entre la luna nueva y luna llena, entre abril y julio 2005, en septiembre 2006 y agosto 2007. Para evaluar la distribución espacial de las larvas de equinodermos se llevaron a cabo arrastres oblicuos con mallas de 64 y 202mm en siete estaciones dentro de beríl, en el beríl y mar afuera (oceánica) durante agosto 2007. En total, los equinoideos exhibieron la mayor abundancia con un total de 11 921 larvas colectadas, de las cuales 52.5% eran de Echinoidea (erizos). Las de Ophiuroidea (estrellas quebradizas) representaron el 45.6% del total de larvas colectadas. Larvas de Holothuroidea (pepino del mar) y Asteroidea (estrella del mar) constituyeron menos del 2% del total de larvas. Estadíos larvales tempranos (2-8 días) de Diadema antillarum fueron el 20% del total de larvas de equinoideos. No se observaron patrones estacional es de larvas de equinodermos, larvas de equinoideos y ofiuroideos se observaron en todos los muestreos mensuales, indicando reproducción continua durante todo del año. Se observó una alta abundancia de larvas de equinoideos en estado avanzado durante los meses de julio, octubre y enero, y en los meses de junio, octubre y agosto para ofiuroideos. El incremento observado de abundancia de larvas en estos meses puede ser indicativo de una alta actividad de reclutamiento durante este periodo. Hubo una diferencia significativa de abundancia larval entre estaciones a través del gradiente costero-oceánico, resultando ser el beril donde la abundancia fue más alta. Larvas de D. antillarum en estado avanzado (~24 días) se colectaron mayormente en el beril y localidades oceánicas. Estos patrones de distribución mar afuera pueden estar inhibiendo una recuperación de las poblaciones de D. antillarum mar adentro. La malla de 64mm fue más eficiente para colectar larvas de equinodermos en este estudio.
Palabras claves: equinodermos, larvas, Diadema antillarum, Puerto Rico, Caribbean.
Assessments of temporal and spatial abundance patterns of pelagic larval stages are relevant for understanding dispersal strategies, spatial connectivity and recruitment dynamics. Larval distribution and aggregation can result from responses to physical factors such as temperature and salinity gradients, water motion and/or biological factors such as predation and food availability (Omori & Hamner 1982). Zooplankton patchiness influences the variability in the estimates of distribution and abundances of zooplankton communities (Cassie 1968). There are several zooplankton community studies of coastal Caribbean systems (Reeve 1970, Youngbluth 1980, McWilliam et al. 1981, Yoshioka et al. 1985) that do not report on the occurrence of echinoderm larvae from plankton sampling.
There have been a few plankton studies carried out in La Parguera, which have focused on sea urchin larval abundance and distribution. Cameron (1986) reported the larval occurrence of four species of sea urchins, Lytechinus variegates, Tripneustes ventricosus, Echinometra viridis and E. lucunter on the shallow insular shelf of La Parguera, Puerto Rico. In a more recent study at La Parguera, Puerto Rico, Williams et al. (2009) measured the year-round abundance of Diadema antillarum larvae, evidencing the continuous reproduction of this population. However, up until now there have been no reports on the larval composition and abundance of ophiuroid, asteroid and holothuroid in Puerto Rico. The present study describes the general taxonomic composition and temporal (monthly) and spatial variations of echinoderm larvae within the coral reef system and adjacent oceanic waters in La Parguera, Puerto Rico. Also, this study provides a comparative analysis of echinoderm larval collections using two different mesh nets.
Material and methods
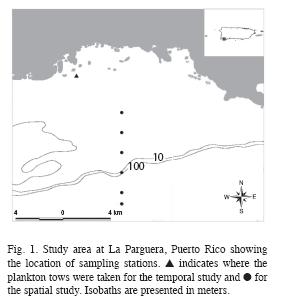
Study Site: La Parguera is located on the southwest coast of Puerto Rico (Fig. 1). Two lines of emergent reefs run roughly parallel to the mangrove shoreline and a submerged spur and groove coral reef formation fringes the shelf edge. The coral reef habitat has an extension of ~193km2. The insular shelf extends approximately 11km offshore.
Spatial Plankton Surveys: In order to provide an assessment of the larval spatial distribution a series of tows were performed across a neritic-oceanic gradient during August 13, 2007 (Fig. 1) in order to coincide with the new and full moon. The distance from one station to another was ~1.6km and the farthest station (sta.1) was approximately 15km offshore from the coastline and ~3.7km from the shelf-edge. There were 7 stations in total; this includes the data collected at the station for temporal surveys for this particular month. Conical 64μm and a 202 μm mesh plankton nets with a 0.5m mouth diameter were used. At each station three oblique two minute tows were taken with a 64μm mesh net and three oblique five minute tows were taken with the 202μm mesh net. All oblique tows were integrated at depths between the surface and near benthos. The mean water volume filtered by tows with the 64 and 202μm nets was approximately 20m3 and 66m3, respectively.
All plankton tows (temporal and spatial study) were taken during the mid-morning to early afternoon and samples were preserved in 3% formalin. The given time duration of the plankton tows using the 64μm net was due to prevent clogging of the net. Echinoderm larvae were identified and counted at class level, with the exception of D. antillarum larvae. Metamorphosing larvae (noted as later-stage larvae) were also identified.
Data Analysis: Since the larval abundance (ind/100m3) data of the temporal and spatial study did not follow a normal distribution, a Kruskal-Wallis ANOVA test by ranks was performed to determine if there were differences of echinoderm larval abundances between 1) sampling events during the temporal survey, 2) spatial stations and 3) mesh net sizes. The Statistica 7 package was used for these analyses.
In addition, Cassie´s (1963) index of patchiness, P, was calculated to evaluate dispersion patterns of echinoderm larvae within replicate samplings. Cassie´s index was calculated as
P = (s -x)/ x2,
Results
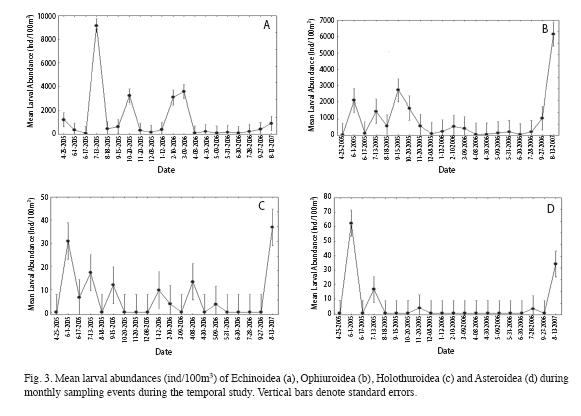
A pulse of Ophiuroidea larvae was observed during the summer (June, July and September) of 2005 (Fig. 3b). Peak abundances of laterstage Echinoidea larvae were observed during July 2005 (70.55 Ind/100m3), October 2005 (79.36 Ind/100m3) and January 06 (133.12 Ind/100m3). The later-stage Ophiuroidea larval abundances peaked during June 2005 (73.57 Ind/100m3), October 2005 (90.70 Ind/100m3) and August 2006 (286.01 Ind/100m3).
The inconsistent inter-annual abundance pattern observed may be the result of high within station variability associated with patchiness.
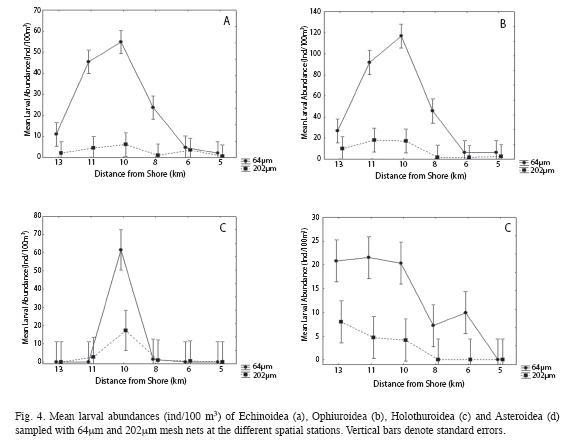
Spatial Study: The distribution of larvae collected by both nets at the different stations was similar in that Echinoidea, Ophiuroidea and Holothuroidea larval abundance all peaked at the shelf-edge (Fig. 4a-c). Asteroidea larvae displayed an increasing pattern of abundance from inshore to offshore, with peak abundance at the oceanic station 1 (sta. 1, Fig. 4d). Also, older larval stages (~24 day old) of D. antillarum were mostly collected from the shelfedge station and oceanic stations. Echinoderm larvae were collected in higher mean abundance with the 64 mm mesh net (24.11±35.02, Ind/100m3±SD) than with the 202mm mesh net (4.52±6.71, Ind/100m3±SD) (Kruskal-Wallis ANOVA, H=14.35, df=1, p=0.0002).
Discussion
Larval composition of the different classes found in this study is similar to the adult population structure of echinoderms found in La Parguera reported in Benavides (2006) study. Greenwood et al. (2000) stated that the composition of echinoderm larvae collected in samples might reflect the populations of adult echinoderms in a given sampling site. Benavides´s (2006) benthic echinoderm inventory in La Parguera consisted of 15 different species of sea urchin, 44 species of brittle star, 14 species of sea star and 29 species of sea cucumber. Adult Echinoidea were the numerically dominant class measured at inshore reefs and seagrass beds; with a relative abundance of 51.3% (Benavides 2006). In this study, sea urchin larvae also represented the numerically dominant assemblage among echinoderm larvae (relative abundance 52.46%) followed by the brittle star larvae (relative abundance 45.61%). In Benavides´s (2006) study the relative abundance of brittle star adults was 46.8% and relative abundance of sea star and sea cucumber adults combined was 2%.Echinoidea and Ophiuroidea larvae were collected in all 20 monthly samplings. Mladenov (1983) studied the reproductive periodicity of three species of brittle stars by examining their gonad index and observed that these species reproduce continuously. It was also stated that prolonged breeding seasons and continuous reproduction are common in tropical brittle stars. Continuous presence of larvae was also observed for D. antillarum (Williams et al. 2009). Holothuroidea and Asteroidea larvae were absent on approximately two-thirds or more of the samples collected in this study. This may be due to under-sampling associated with short pelagic larval duration and relatively low abundance of adults compared to Echinoidea and Ophiuroidea (Nievales & Junio-Meñez 2007). In this study, Echinoidea and Ophiuroidea larvae presented monthly abundance peaks but there were no clear seasonal abundance trends. Lack of seasonal abundance trend has been reported in other echinoderm larval studies from the Caribbean (Morales & Murillo 1996, Williams et al. 2009). However, higher abundance of later-stage larvae of Echinoidea and Ophiuroidea peaked during the summer and fall. The observed peaks of later-stage larval abundances may be indicative of higher recruitment activity during these months.
The spatial distribution of echinoderm larvae resulting from this study does not reflect the distributions of adult benthic populations in La Parguera. Echinoid, ophiuroid and holothuroid larval abundance peaked at the shelf-edge and declined at oceanic and inshore stations. Benavides (2006) found a trend of adult echinoderm benthic populations decreasing offshore, with only a few adult echinoderms identified from the shelf-edge. However, Williams et al. (2010) recorded higher recruitment of D. antillarum at shelf-edge locations than at inshore sites in La Parguera and in this study later-stage (~24 day old) D. antillarum larvae were mainly collected from the shelf-edge and oceanic locations. So why is there a peak in larval abundance at the shelf-edge?
The shelf-edge in La Parguera, Puerto Rico is a transition zone for coral reef fish communities with more planktivore fishes found inshore than at offshore waters (Ramírez-Mella & García-Sais 2003). The depth of the reef substrate at the shelf-edge and lack of benthic and planktonic predators inshore may account for the higher abundance of echinoderm larvae at this location. Lasker (1981) argued that competition and predation might play an important role in controlling the distribution and abundance of zooplankton. Johannes (1978) observed reef fishe spawning offshore as a behavioral adaptation in order to avoid predation by planktivorous fishes. Also, the flow dynamics at the shelfedge is unique, possibly influencing the presence of larvae. For example, Esteves (2005) witnessed an internal wave that broke at the shelf-edge of La Parguera, Puerto Rico. This internal wave brought colder waters with presumably higher nutrient concentrations inshore and displaced warm waters offshore. The potential nutrient increase can influence higher zooplankton abundance.
Additional factors that appear to have influenced the abundance and distribution of echinoderm larvae in this study were mesh size and patchiness in their distribution. More echinoderm larvae were collected in La Parguera, Puerto Rico than in Morales and Murillo (1996) study in Costa Rica. Morales & Murillo (1996) used a 280mm mesh size when collecting zooplankton at the Cahuita coral reef in Costa Rica. In this study it was found that mesh size influenced the amount of echinoderm larvae collected. The 64mm mesh net was more efficient for collecting echinoderm larvae, even though there was less volume of filtered seawater. Therefore, echinoderm larval abundance may be underestimated when a 202mm and/or greater mesh net sizes are used.
The overall variations and inconsistent peaks of larval abundances between years of the corresponding months during this study are influenced by the patchy distribution of the larvae. The patchiness index of echinoderm larvae was found to be greater using the 202mm mesh net thus suggesting an increasingly aggregated larval behavior with increasing size and age of the larvae collected. Patchiness has been reported in other echinoderm larval studies (Greenwood et al. 2000, Lavitra et al. 2006, Williams et al. 2009) and is a responsive behavior to chemical and physical parameters (Levinton 1982), light intensity, food availability and predators (Omori & Hamner 1982). Unfortunately, the high sampling variability associated with patchiness in this study constrains our ability to detect abundance patterns in larger spatial scales.
Acknowledgments
We would like to thank the Sea Grant Program at UPRM for funding part of the project (SEED money, PD-265). We would also like to thank K. Flynn, B. Todd, W. Rovira, R. Esteves, M. Carlo and G. Lopez for their assistance in collecting the data. In addition, we like to thank R. Emlet for identification of the larvae.
Referencias
Benavides, M. 2006. Taxonomic list of the shallow water of echinoderms of Puerto Rico with new information for La Parguera. MS Thesis, University of Puerto Rico, Mayagüez, Puerto Rico. [ Links ]
Cameron, A.C. 1986. Reproduction, larval occurrence and recruitment in Caribbean sea urchins. Bull. Mar. Sci. 39: 332-346. [ Links ]
Caso, M.E. 1984. Contribución al conocimiento de los equinoideos de México, II , Algunas especies de equinoideos litorales. Inst. Biol. Mexicana Anales19: 183-231. [ Links ]
Cassie, R.M. 1963. Microdistribution of plankton. Oceanogr. Mar. Biol. Ann. Rev.1: 223-252. [ Links ]
Cassie, R.M. 1968. Sample designs, p. 105-122. In D.J. Transfer (ed.). Zooplankton Sampling. Monographson Oceanographic Methodology, Paris, UNESCO. [ Links ]
Esteves, R. 2005. Dispersal of reef fish larvae from known spawning sites in La Parguera, Puerto Rico. MS Thesis, University of Puerto Rico, Mayagüez, Puerto Rico. [ Links ]
Greenwood, A., D.K.A. Barnes & R.M. O´Riordan. 2000. Seasonality of echinoderm plankton in Lough Hyne Marine Nature Reserve. Biology and environment: Proc. Royal Irish Acad. 100B: 171-180. [ Links ]
Guzmán, H.M., C.A. Guevara & I.C. Hernández. 2002. Reproductive cycle of two commercial species of sea cucumber (Echinodermata: Holothuroidea) from Caribbean Panama. Mar. Biol. 142: 271-279. [ Links ]
Johannes, R.E. 1978. Reproductive strategies of coastal marine fishes in the tropics. Environ. Biol. Fish. 3: 65-84. [ Links ]
Lasker. L. 1981. Marine Fish Larvae: Morphology, Ecology and Relation to Fisheries. Washington Uniersity, Seatle, Washington, USA. [ Links ]
Lavitra, T., D. Vaïtilington, R. Rasolofonirina & I. Eeckhaut. 2006. Seasonal abundance of sea cucumber larvae at Toliara Great Reef, Madagascar. SPC Beche-de-mer, July 2006, Information Bulletin # 24. [ Links ]
Lessios, H.A., M.J. Garrido & B.D. Kessing. 2001. Demographic history of Diadema antillarum, a keystone herbivore on Caribbean reefs. Proc. R. Soc. Lond. B. 268: 1-7. [ Links ]
Levinton, J.S. 1982. Marine Ecology. Prentice Hall, New Jersey, USA. [ Links ]
Lugo, M.A. 2004. Population status of the black sea urchin Diadema antillarum (Phillipi) in La Parguera, Puerto Rico, 20 years after the mass mortality event. MS Thesis, University of Puerto Rico, Mayagüez, Puerto R. [ Links ]
McWilliam, P.S., P.F. Sale & D.T. Anderson. 1981. Seasonal changes in resident zooplankton sampled by emergence traps in One Tree Lagoon, Great Barrier Reef. J. Exp. Mar. Biol. Ecol. 52: 185-204. [ Links ]
Mladenov, P.V. 1983. Breeding patterns of three species of Caribbean brittle stars (Echinodermata: Ophiuroidea). Bull. Mar. Sci. 33: 363-372. [ Links ]
Morales-Ramírez, A. & M.M. Murillo. 1996. Distribution, abundance and composition of coral reef zooplankton, Cahuita National Park, Limon, Costa Rica. Rev. Biol. Trop. 44: 619-630. [ Links ]
Nievales, M.F.J. & M.A. Junio-Meñez. 2007. Effects of varying temperature-salinity larval food level during rearing on larval survival, development and post larval yield in Holothuria scabra. February 2007, SPC Beche-de-mer Information Bulletin #25 [ Links ]
Omori, M. & W.M. Hamner. 1982. Patchy distribution of zooplankton: behavior, population assessment and sampling problems. Mar. Biol. 72: 193-200. [ Links ]
Ramírez-Mella, J.T. & J.R. García-Sais. 2003. Offshore dispersal of Caribbean reef fish larvae: How far is it? Bull. Mar. Sci. 72: 997-1017. [ Links ]
Reeve, M.R. 1970. Seasonal changes in the zooplankton of south Biscayne Bay and some problems of assessing the effects on the zooplankton of natural and artificial thermal and other fluctuations. Bull. Mar. Sci. 20: 894-921. [ Links ]
Williams, S.M., J. García-Sais & J. Capella. 2009. Temporal variation of early larval stages of the long-spined sea urchin Diadema antillarum in La Parguera, Puerto Rico. Carib. J. Sci. 45: 110-117. [ Links ]
Williams, S.M., P.M. Yoshioka & J.R. García Sais. 2010. Recruitment patterns of Diadema antillarum in La Parguera, Puerto Rico. Coral Reefs 29: 809-812. [ Links ]
Yoshioka, P.M., G.P. Owen & D. Pesante. 1985. Spatial and temporal variations in Caribbean zooplankton near Puerto Rico. J. Plank. Res. 7: 733-752. [ Links ]
Youngbluth, M.J. 1980. Daily, seasonal, and annual fluctuations among zooplankton populations in an unpolluted tropical embayment. Est. Coast. Shelf Sci. 10: 265-287. [ Links ]
Correspondencia: Stacey M. Williams & Jorge García-Sais. Department of Marine Sciences, University of Puerto Rico, P.O. Box 9013, University of Puerto Rico, Mayagüez, PR 00681; e-mailsReceived 16-VII-2009. Corrected 07-VI-2010. Accepted 12-VII-2010.