Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.58 suppl.3 San José Oct. 2010
Environmental state and tendencies of the Puerto Morelos
CARICOMP site, Mexico
Rosa E. Rodríguez-Martínez, Francisco Ruíz-Rentería, Brigitta van Tussenbroek, Guadalupe Barba-Santos, Edgar Escalante-Mancera, Guillermo Jordán-Garza & Eric Jordán-Dahlgren
Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México. Ap. Postal 1152, 77500 Cancún, Q. Roo, México; rosaer@cmarl.unam.mx, renteria@cmarl.unam.mx, vantuss@cmarl.unam.mx, isis@cmarl. unam.mx, escalant@cmarl.ail.com, nadroj_ome@hotmail.com, jordan@cmarl.unam.mx
Abstract
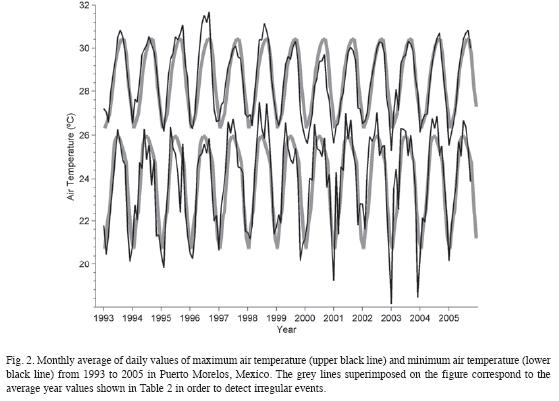
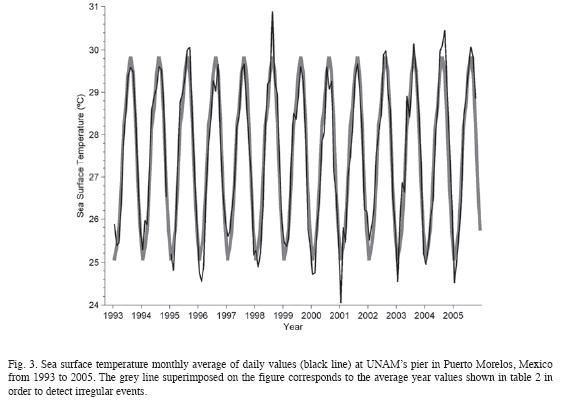
The CARICOMP site at Puerto Morelos, Mexico was monitored from 1993 to 2005. No significant changes in air temperature, wind patterns, periodicity and quantity of rainfall, sea-surface temperature and water transparency were observed between sampling years. During the study four hurricane impacts were registered. At the coral reef site overall mean cover of fleshy algae (47%) and turf algae (36%) were high, whereas cover of corals (2%) and sponges (3%), and abundance of sea-urchins (0.04 org m-2) were consistently low. Gorgonians were dominant and showed changes in their community structure; the number of species increased from 1993 to 1995, their abundance decreased after Hurricane Roxanne (1995) and recovered by 2001. At four seagrass sites total community biomass remained constant (707.1-929.6 g dry m-2) but the above-ground biomass of the seagrass Syringodium filiforme and fleshy algae increased gradually. Total biomass (531-699 g dry m-2) and leaf productivity (0.89-1.56 g dry m-2 d-1) of the seagrass Thalassia testudinum remained constant, but the species invested proportionally more biomass in above-ground leaf tissues at the end of the study. The minor hurricanes from 1993 until 2005 had no detectable impacts on the seagrass beds, however, the major Hurricane Wilma (October 2005) changed the community composition at three stations and caused complete burial of the vegetation at a coastal station. The gradual changes in the seagrass and reef communities recorded in the 12 years of continuous monitoring of the CARICOMP site may reflect the increased pollution caused by the rapid augment in urban and tourist developments along the coasts and inland from Puerto Morelos, coupled with poor water management practices. Rev. Biol. Trop. 58 (Suppl. 3): 23-43. Epub 2010 October 01.
Key words: environmental monitoring, CARICOMP, Mexico, coral reef, seagrass.
El sitio CARICOMP en Puerto Morelos, México, se monitoreo de 1993 a 2005. No se registraron cambios significativos en la temperatura del aire, los patrones de viento, la periodicidad y cantidad de lluvia, la temperatura superficial del mar y la transparencia del agua entre los años de muestreo. Durante el estudio se registraron cuatro huracanes menores. En el sitio del arrecife coralino la cobertura global promedio de algas carnosas (47%) y de algas tipo tapete (36%) fue elevada, mientras que la cobertura de corales (2%), esponjas (3%) y la abundancia de erizos (0.04 m-2) fue consistentemente baja. Los gorgonáceos fueron dominantes y mostraron cambios en su estructura comunitaria; el número de especies aumentó de 1993 a 1995, su abundancia disminuyó después del Huracán Roxanne (1995) y se recuperó en el 2001. En cuatro estaciones de pastos marinos la biomasa total de la comunidad permaneció constante (707.1-929.6 gps m-2) pero la biomasa sobre el sustrato del pasto Syringodium filiforme y de algas carnosas aumentó gradualmente. La biomasa total (531-699 gps m-2) y productividad de hojas (0.89-1.56 gps m-2 d-1) del pasto Thalassia testudinum permaneció constante, pero la especie invirtió proporcionalmente más biomasa en el tejido de hojas sobre el sustrato al final del estudio. Los huracanes menores entre 1993 y 2005 no produjeron impactos detectables en las camas de pastos, sin embargo, el huracán mayor Wilma (Octubre 2005) cambió la composición de la comunidad en tres estaciones y causó el enterramiento completo de la vegetación en la estación costera. El cambio gradual en las comunidades de pastos marinos y arrecife registrado en 12 años de monitoreo continuo en el sitio CARICOMP puede reflejar un aumento en la contaminación causada por el rápido incremento en desarrollo urbano y turístico a lo largo de la costa y tierra adentro de Puerto Morelos, acompañado por malas prácticas de manejo del agua.
Palabras clave: monitoreo ambiental, CARICOMP, México, arrecife coralino, pastos marino.
In this study we report on the variations recorded in environmental variables and changes at the level of reef and seagrass communities between 1993 and 2005 at the Puerto Morelos CARICOMP site. Possible correlations between environmental parameters and changes at the reef and seagrass communities were assessed. The results obtained in this study provide a baseline that will allow for the evaluation of present and future potential impacts of the continuous increasing human developments along the Caribbean coasts of Mexico and global natural and anthropogenic disturbances.
Materials and methods
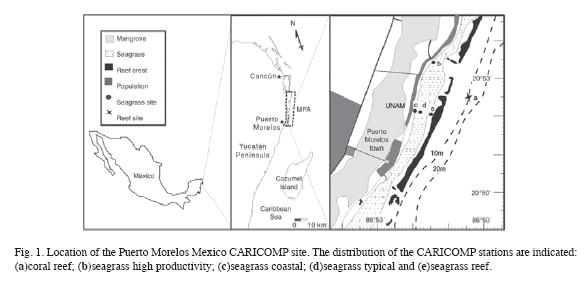
Monitoring of environmental parameters. From 1993 to 2005, sea-surface temperature, maximum-minimum air temperature and accumulated precipitation were sampled on near-daily basis following the CARICOMP level I protocol (CARICOMP 1994). Sea surface temperature was measured at the pier of the Universidad Nacional Autonóma de México (UNAM) research facilities at Puerto Morelos, Mexico where the depth is about 2.5m. Seasurface temperature and refractometer readings of salinity were measured weekly at the Reef, Typical and High-productivity seagrass stations (Fig. 1). Weekly Secchi disk (30cm diameter) measurements were carried at the Typical and High-productivity seagrass stations (horizontal measurements) and 2-3km east from the reef crest (vertical measurement). On some occasions measurements were taken at different times than the ones established by the protocol and usually no measurements were conducted on weekends and vacation periods.
Monitoring of the coral reef site: The reef site was established in 1993, approximately 3km offshore (20°53.11´ N-86°50.28" W) on a typical low-relief fore-reef as indicated by the CARICOMP protocol (Fig. 1). Five permanent 10m transects were deployed randomly perpendicular to the slope at a depth of 10m. The end-points of each permanent transect were fixed with stainless steel stakes. Line intercept measurements were taken using a brass chain (link length=1.4cm) deployed between the fixed stakes. The number of chain-links covering a given benthic category/species provided an estimate of its proportion of relative cover along the chain, the main benthic categories where coral, gorgonian, algae (fleshy, alcareous, turf, coralline), sponges, rock and sediment. Identification of corals and gorgonians was carried out to species level. Gorgonians were counted when their branches or fronds crossed the transect line under the normal surge conditions, and in order to follow the standardization of the CARICOMP database were assigned to the following growth form categories: rod, feather and, fan. Sea urchin density was estimated by a visual inspection of 2m wide belt transect, whose center was the brass chain. Surveys were conducted between May and September, except those in 1994 and 1996 which were realized in October, and those of 2000 and 2002 that were omitted. The last survey was conducted in 2004. Some of the information reported here on coral bleaching and diseases was obtained using different protocols and surveys on other reef habitats (back-reef and scarce high relief sites on the fore-reef) of Puerto Morelos.
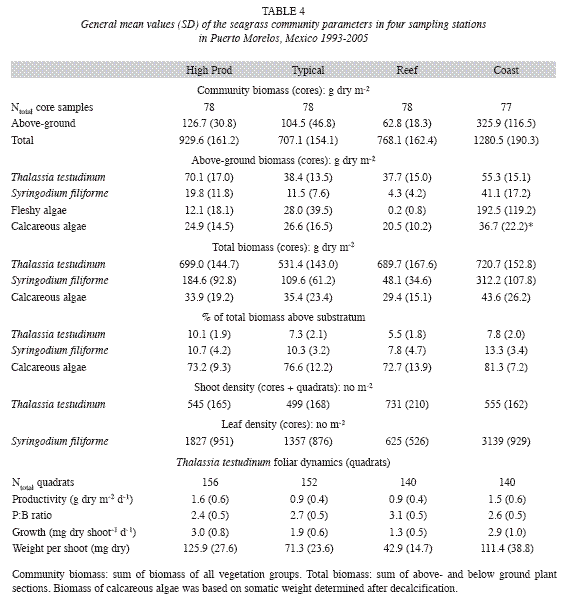
Monitoring of the seagrass community: From 1993 to 2005, four stations were sampled within the Puerto Morelos reef lagoon representing the local seagrass range of environments (Fig. 1): High Productivity (20°53.76´ N, 86°51.07´ W), (2) Typical (20º52.063´ N, 86º52.913´ W), (3) Reef (20º51.851´ N, 8º51.487´ W) and (4) Coast (20º52.038´ N, 86º52.025´W). The first two stations were located in the middle of the reef lagoon; the High Productivity station (High Prod) was in the vicinity of marine springs venting water from the mangrove wetlands, whereas the Typical station was away from obvious point sources of groundwater. The Reef station was ~200m shoreward from the reef tract, in an area with a relative high hydrodynamic regime, and the Coast station was ~30m from the coastline and represented the dense fringe vegetation. Depths at these stations varied between 2.5-3.5 m. Further descriptions of the study sites can be found in Van Tussenbroek (1995, 1998). Biomass and community composition of the seagrass communities were determined following the CARICOMP Level I protocol with the following modifications: a metal corer of 22cm diameter and depth of 30-35cm was employed instead of a PVC pipe (diameter 15cm) and three (rarely two), instead of four, haphazardly chosen replicates were collected at each sampling time per station. In addition to standard procedures, the number of foliar shoots of Thalassia testudinum and the number of leaves of Syringodium filiforme were determined for each sample. Foliar dynamics of T. testudinum were determined according the standard CARICOMP methods employing 6 randomly placed quadrats of 10x20cm per site per sampling time. Weight per shoot was calculated by dividing the leaf biomass by the number of shoots per quadrat and the time interval between the beginning of two successive leaves or leaf plastochron (PIleaf) following Van Tussenbroek (1998). On occasions, one or two quadrats were lost reducing the number of samples. Sampling times were winter (December of the previous year until February of the sampling year) and summer (June until August). In addition to the above-mentioned CARICOMP study period of 1993-2005, sampling was conducted in 2006 and 2009 to evaluate the impact of Hurricane Wilma which passed over the area in October 2005.
Statistical analyses: Parametric statistical tests (e.g. repeated measures ANOVA, t-test, Tukeys Honestly Significant Difference) were employed whenever assumptions of normality and homoscedasticity were satisfied; otherwise non-parametric procedures (e.g. Friedman ANOVA, Wilcoxon) were employed. The coral reef database had missing data, as two stakes were lost on occasions and two years were not sampled, and missing values were estimated using multiple linear regression and adding a random component from a t distribution to the regression estimates. To discern seasonal differences in the parameters of community biomas and foliar dynamics of T. testudinum, summer data of all sampling years were pooled and compared with a Student t-test with the pooled data collected during the winter. To obtain an annual value of all sampled parameters the winter and summer values were pooled per sampling year. A two-way ANOVA was applied to latter parameters as independent factors, and station and year as fixed factors because they were specifically chosen as specified by the CARICOMP protocol. If necessary, the data were log transformed to comply with the prerequisites of the analysis of variance. Interannual fluctuations were of special interest; the interaction factor of the two-factor ANOVA indicated whether temporal tendencies were similar or distinct at the four sampling sites. If a station were to be responsible for significant interaction it will be eliminated from the twoway ANOVA analyses because general temporal patterns were of interest for the study. For all analyses alpha was set at 0.05. Statistical analyses were completed using Statistica version 6 software.
Results
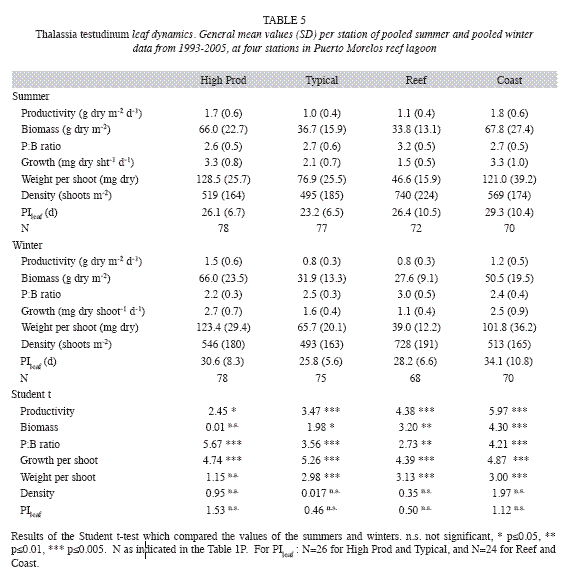
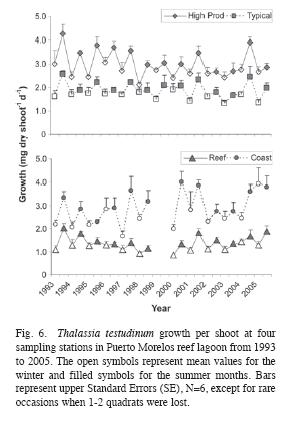
Seasonal variations were appreciated in the foliar dynamics of T. testudinum, presenting, in general, higher leaf growth (Fig. 6) and productivity, and lower PIleaf during the summer than in the winter (Table 5). Shoot density, in contrast, remained constant throughout the year (Table 5), and leaf biomass did not vary significantly between summer and winter in the High Prod site. The pooled summer values of the above-ground- and total biomass of all other vegetation groups did not vary from the pooled winter data, and the p of the Student t-test exceeded 0.1 in all cases.
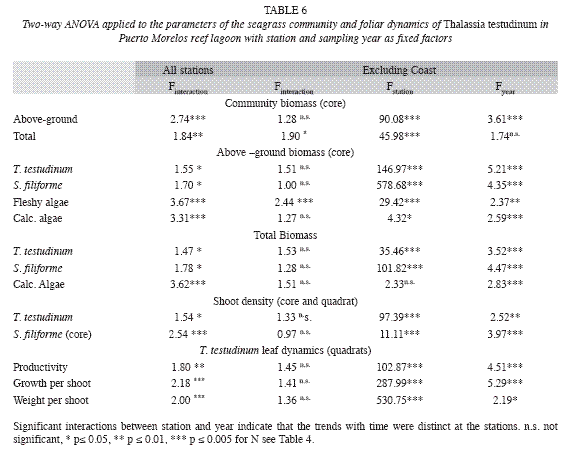
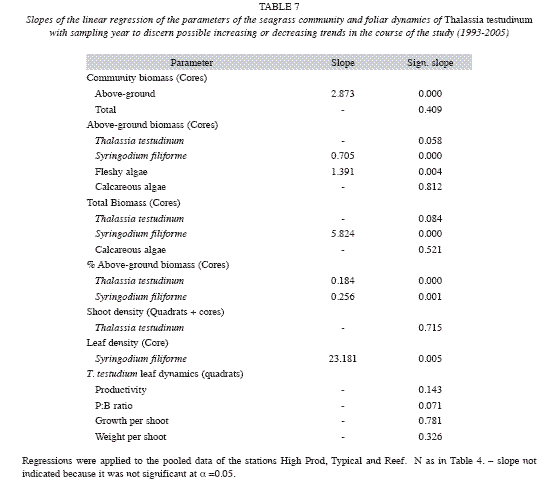
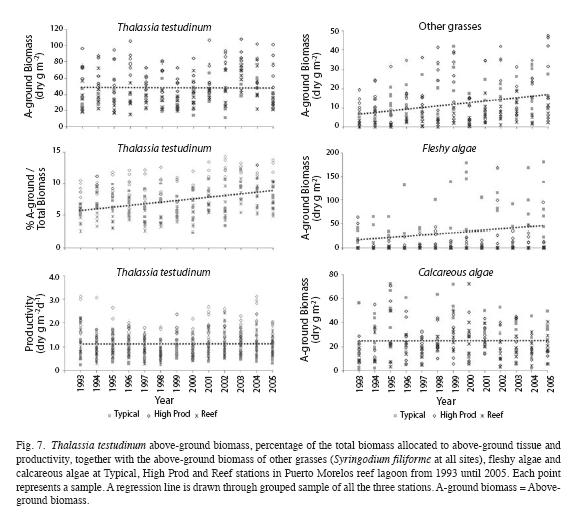
Interannual fluctuations of the studied parameters of the seagrass community did not show the same trends at all sampling stations, as indicated by their significant interaction between station and year of the two-way ANOVA (Table 6). However, excluding the Coast station resulted in, interactions becoming insignificant for most parameters (except for total community biomass and for the biomass of the fleshy algae), and thus this station was omitted from further analysis. Differences between years were then significant for all parameters (Table 6), but interannual variations of the parameters of foliar dynamics of T. Testudinum (Fig. 7) and the biomass of the other eagrasses (S. filiforme), calcareous and fleshy algae were erratic, without an obvious pattern. The clearest signals of changes in time, from 1993 until 2005, were ascending tendencies in the total biomass of the seagrass Syringodium filiforme (corresponding to the group "other seagrasses") and in the fleshy algal group (Table 7). S. filiforme also showed increased leaf density and both seagrass species (T. testudinum and S. filiforme) showed an increment in the proportion of the biomass allocated to the above-ground leaves (% aboveground biomass) in the course of the study (Table 7, Fig. 7).
The impact of the major Hurricane Wilma (October 2005) on the sampling stations in the seagrass beds of Puerto Morelos reef lagoon was not severe, with exception of the vegetation at the Coast site, which was buried by a ~1-1.5m thick sand layer. In the persistent beds, above-ground biomass, shoot density, % of total biomass allocated to above-ground tissue and productivity of T. testudinum were often slightly lower, but within the range of "normal" fluctuations at these sites (Fig. 8). In comparison with 2005, the abundance of S. filiforme, and fleshy and calcareous algae, decreased slightly after passage of the hurricane at the High Prod and Typical sites (Fig. 8), but at the Reef site declines in these vegetation groups were less notable or absent. In early 2009, three years after Hurricane Wilma, the composition of the vegetation in the High Prod, Typical and Reef sites were similar to those recorded during the CARICOMP observation period, although not always comparable with the pre-hurricane values recorded in 2005 (Fig. 8). In 2009, the previously bare sandy area at the Coast station was sparsely colonized by calcareous and fleshy algae (Caulerpa spp.) and the seagrasses Halodule wrightii and Syringodium filiforme (Fig. 8).
Discussion
At the reef site, the benthic community remained relatively stable throughout the study. Hard coral cover was consistently low (<2.5%) and represented the lowest extreme of the coral cover values recorded in all of the CARICOMP sites. Species richness, cover and density values recorded were considerably lower than those reported for the same reef area in 1978 (Jordán-Dahlgren 1979), before the reef was impacted by major Hurricane Gilbert (1988, class V). That single event resulted in a reduction in species richness from 16 to 12, in coral cover from 8.4% (±3.1%) to 3.1% ±1.2%) and in coral colony density from 0.9 col m-1 (±0.2) to 0.3 col m-1 (±0.2), at a depth of 10m (Rodríguez-Martínez 1993). Fifteen years later, the reef community showed little recovery, which could be attributed to the locally low rugosity fore-reef. Although relatively frequent recruitment events could account for the relatively abundant small coral colonies, only a few of these recruits reached larger sizes (over 75% of the colonies had diameters <10cm). Suggested forcing functions responsible for this type of community include high re-suspension of sediments, due to the gentle slope (<5°), that drive the selection of resistant species such as Montastraea cavernosa, Diploria strigosa, Dichocoenia stokesiis and Siderastrea siderea (Loya 1976, Jordán-Dahlgren 1979). Further sediment abrasion and colony detachment during storms and hurricanes also accounted for dominance of juvenile stages in the population (Jordán-Garza 2004).
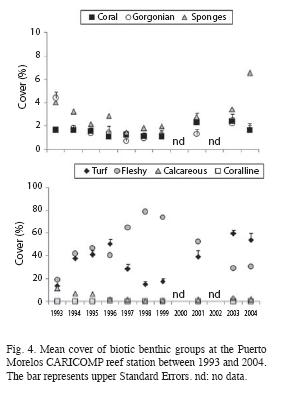
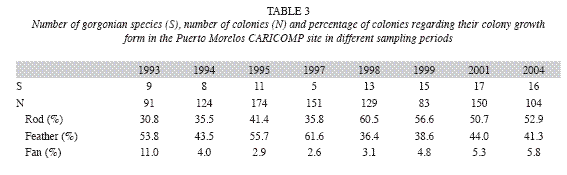
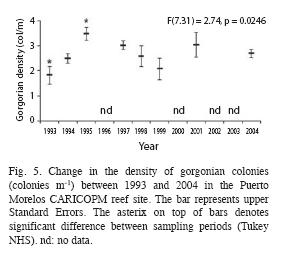
During the study period the major forcing factors that affected the coral community where hurricanes, coral diseases and bleaching events. The effects of the minor hurricanes were small, considering that the community suffered a major impact by Hurricane Gilbert in 1988. In 1993, when the CARICOMP project started, gorgonians hadn´t fully recovered from the impact of Hurricane Gilbert, as their density (1.8±0.7 org m-1) was lower than that recorded in the same area in 1978 (2.8 org m-1±0.5; Jordán-Dahlgren 1979) but by 1995 gorgonian density (3.5±0.6 org m-1) surpassed pre-hurricane values. In 1995, Hurricane Roxanne (class III) made land 100km south of Puerto Morelos and did not cause major damage to the reef, but in the next three years gorgonian density showed a down-trend pattern coupled with a change in the dominant gorgonian growth forms, from feather to rod, which could be the result of differential recruitment, different growth rates or competitive outcomes (Connell et al. 2004). By 2004, gorgonian composition and density (2.7±0.4 org m-1) was very similar to that of 1978. Fluctuations through time in the cover of fleshy algae and turf algae were more likely the result of the time of the year when the surveys were conducted and the life history characteristics of the component species; when the surveys were done in the summer the abundance of fleshy algae, mainly Sargassum spp., increased and overgrew the turf algae. Coverage by fleshy and turf algae (84%) was higher than that found in other CARICOMP sites like Panamá (21%; Guzmán et al. 2005), Puerto Rico (maximum 38%, Linton & Fisher 2004), Costa Rica (63%; Fonseca et al. 2006) and Jamaica (73%; Linton & Fisher 2004). Density of sea urchins throughout the study (<0.2 org m-²) was low and comparable to that reported for other CARICOMP sites (Linton & Fisher 2004). The population of Diadema antillarum increased slightly in our last survey but we can´t determine if this was the beginning of the recovery process. Density of D. antillarum, however, has increased in other Mexican reefs, such as Mahahual (7.3±4.2 org m-²; Jordán-Garza et al. 2007) and Akumal (Rodríguez-Martínez pers. obs.) but mostly in shallow back-reef areas.
Disease outbreaks were recorded and their effects measured throughout the study period in many areas of Puerto Morelos reef. The data set from the CARICOMP reef site showed no ignificant increase in the prevalence of coral diseases. However, the increment in the number and prevalence of coral diseases elsewhere in Puerto Morelos (Rodríguez-Martínez et al. 2001, Jordán-Dahlgren et al. 2005) suggest that higher sea surface temperature trends observed in present study may be a causative factor, either by thermal stress on the host reducing coral resistance to infections or thermally induced increase in pathogens´ virulence (Bruno et al. 2007, Harvell et al. 2007). In 1998, only two diseases, white-band and white-pox, were recorded affecting colonies of Acropora palmata, in the back-reef of Puerto Morelos. In 2000, the number of recorded diseases had increased to five and by 2004 to six, reflecting the same situation that has been observed throughout the Caribbean. The first thermal bleaching event of corals and other reef organisms recorded at Puerto Morelos was in 1995 (CARICOMP 1997) and, since then, bleaching occurred in all of the years when the maximum sea surface temperature exceeded 30°C in the summer, except in the year of 1997. During the study period, however, no mass mortality of scleractinian corals associated with thermal coral bleaching was recorded on Puerto Morelos reef. The most severe bleaching events were recorded in 1995 and 2005 (Table 1), when it was estimated that over 50% of the coral colonies were bleached at different intensities. The bleaching event of 1998, which caused catastrophic mortalities of corals around the world (Aronson et al. 2000), was recorded as moderate in Puerto Morelos reef (Table 1). At the CARICOMP reef site, no significant bleaching was recorded possibly because surveys were carried out at months that coincided with the onset of the bleaching events.
Variations in the seagrass community were in many aspects distinct from those in the reef community and subject to different forcing factors. For example, the possible rise in seawater temperature most likely did cause detectable changes in the seagrass community structure during the study period. The temporal dynamics of the seagrass community occurred at three levels: (1) season, (2) gradual changes from 1993 until 2005, probably forced by increased nutrient inputs, and (3) perturbations due to the major Hurricanes Gilbert (1988) and Wilma (2005). In general, biomass, growth rates, and productivity, were lower in the winter than in the summer. Such seasonal fluctuations in the foliar dynamics of Thalassia testudinum have already been reported by Van Tussenbroek (1995) and were most likely related to variations in the solar cycle. In the present study, in some of the years the contrast between the summer highs and the winter lows in the growth rates and productivity were less obvious, more likely because the 7-10 days lasting observation periods were not always representative of the prevailing conditions during that season. Seasonal trends were not observed in the biomass of the other vegetation groups, but at this point it can´t be discerned whether this is a real trend or a sampling effect. The general-purpose and simple sampling design for the study of the composition seagrass communities of the CARICOMP project allowed for comparison between sites within a wide geographical area but possibly did not have the necessary resolution to discern small changes in the seagrass community. Sampled areas were limited to the small area covered by the cores and there was a very wide spread of the individual sampling points, as is demonstrated in figures 7 and 8. Therefore, a signal of change must be exceptionally strong to be discerned and it is possible that the seasonal changes in biomass of the different vegetation groups remained undetected.
Conclusion
The CARICOMP research in Puerto Morelos, Mexico provided a baseline data set to detect environmental changes which will allow for the evaluation of present and future potential impacts of the continuous coastal development and global rise in temperature. Between 1993 and 2005, the benthic coral and seagrass communities at Puerto Morelos CARICOMP appeared stable, although sometrends of potential permanent changes were discerned. Gorgonian and seagrass communities recovered relatively fast from the impact of major hurricanes suggesting high ecological resilience (Nyström et al. 2008). Coral cover at the reef site was expected to recover from Hurricane Gilbert (1988) but remained low. The coral community seems to be on a semipermanent initial phase of development where recruitment is high but mortality is also high, seeming in a sense resilient to present conditions. However, algal outbreaks may change this situation as there is an ongoing increase in coastal development and there is no control of residual waters. Gradual changes in the seagrass community structure suggest an increasing nutrient load into the reef lagoon. We recommend the implementation of regulation measurements for the adjacent coastal zone, mainly for sewage and garbage treatment, in order to prevent deterioration of the coral reef and seagrass communities.Acknowledgments
This project was financed by the National University of Mexico and CARICOMP. We thank Miguel Angel Maldonado, Leonardo Vázquez, Daniel Gasca and all of the students that collaborated with the field work. Paul Blanchon and three anonymous reviewers made valuable comments to improve the manuscript.
References
Aronson, R.B., W.F. Precht, I.G. Macintyre & T.J.T. Murdoch. 2000. Coral bleach-out in Belize. Nature 405: 36. [ Links ]
Bruno, J.F. & E.R. Selig. 2007. Regional decline of coral cover in the Indo-Pacific: timing, extent, and subregional comparisons. PLoS ONE: 2: e711. [ Links ]
Cabaço, S., R. Santos & C.M. Duarte. 2008. The impact of sediment burial and erosion on seagrasses: A review. Est. Coast. Shelf Sci. 79: 354-366. [ Links ]
CARICOMP. 1994. CARICOMP Methods Manual-Level I: Manuals of methods for Mapping and monitoring of physical and biological parameters in the coastal zone of the Caribbean. Kingston, Jamaica: CARICOMP Data Management Centre, University of the West Indies. [ Links ]
CARICOMP. 1997. Studies on Caribbean coral bleaching 1995-96. Proc. 8th Int. Coral Reef Symp, Panamá 1: 673-678. [ Links ]
Carruthers, T.J.B., B.I. van Tussenbroek & W.C. Dennison. 2005. Influence of submarine springs and wastewater on nutrient dynamics of Caribbean seagrass meadows. Est. Coast. Shelf Sci. 64: 191-199. [ Links ]
Connell, J.H., T.E. Hughes, C.C. Wallace, J.E. Tanner. K.E. Harms & A.M. Kerr. 2004. A long-term study of competition and diversity of corals. Ecol. Monogr. 74: 179-210. [ Links ]
Cruz-Palacios, V. & B.I. van Tussenbroek. 2005. Simulation of hurricane-like disturbances on a Caribbean seagrass bed. J. Exp. Mar. Biol. Ecol. 324: 44-60. [ Links ]
Done, T.J., J.C. Ogden, W.J. Wiebe & B.R.Rosen. 1996. Biodiversity and ecosystem function of coral reefs, 393-429. In H.A. Mooney, J.H. Cushman, E. Medina, O.E. Sala & E.D. Schulze (eds.). Functional Roles of Biodiversity: A Global Perspective. SCOPE 1996, John Wiley and Sons, New York. USA. [ Links ]
Duarte, C.M. 1992. Nutrient concentrations of aquatic plants: patterns across species. Limnol. Oceanogr. 37: 882-889. [ Links ]
Farnsworth, E.J. & A.M. Ellison. 1997. The global conservation status of mangroves. Ambio 26: 328-334. [ Links ]
Fonseca A.C., E. Salas & J. Cortés. 2006. Monitoreo del arrecife coralino Meager Shoal, Parque Nacional Cahuita, Costa Rica (sitio CARICOMP). Rev. Biol. Trop. 54: 755-763. [ Links ]
Fourqurean, J.W., A. Willsie, C.D. Rose & L.M. Rutten. 2001. Spatial and temporal pattern in seagrass community composition and productivity in south Florida. Mar. Biol. 138: 341-354. [ Links ]
Fourqurean, J.W. & L.M. Rutten.2004. The impact of hurricane Georges on soft-bottom, back reef communities: site- and species-specific effects in South Florida seagrass beds. Bull. Mar. Sci. 75: 239-257. [ Links ]
Goreau, T.F. 1959. The Ecology of Jamaican Coral Reefs. I. Species composition and zonation. Ecology 40: 67-90. [ Links ]
Guzmán, H.M., P.A.G. Barnes, C.E. Lovelock & I.C. Feller. 2005. A site description of the CARICOMP mangrove, seagrass and coral reef sites in Bocas del Toro, Panama. Carib. J. Sci. 4: 430-440. [ Links ]
Hughes, T.P. 1994. Catastrophes, phase shifts, and largescale degradation of a Caribbean coral reef. Science 265: 1547-1551. [ Links ]
Hughes, T.P., D. Ayer & J.H. Connell. 1992. The evolutionary ecology of corals. Trends Ecol. Evol. 7: 292-295. [ Links ]
Hughes, T.P. & J.H. Connell. 1999. Multiple Stressors on Coral Reefs: A Long-Term Perspective. Limnol. Oceanogr. 44: 932-940. [ Links ]
Jordán-Dahlgren, E. & R.E. Rodríguez-Martínez. 2003. The Atlantic coral reefs of México, p. 131-158. In J. Cortés (ed.). Latin American Coral Reefs. Elsevier, Amsterdam, Holland. [ Links ]
Jordán-Dahlgren, E., M.A. Maldonado & R.E. Rodríguez- Martínez. 2005. Incidence of coral diseases and colony mortality in the Montastraea annularis complex in reefs on the Southeastern Gulf of Mexico and Northwestern Mexican Caribbean. Dis. Aqua. Org. 63: 3-12. [ Links ]
Jordán-Garza, A.G. 2004. Efecto de la rugosidad del substrato sobre Siderastrea siderea, Diploria strigosa y Montastraea cavernosa en la costa Noreste de la Península de Yucatán, México. Bachelor Thesis, Facultad de Ciencias, Universidad Nacional Autónoma México, México D.F. [ Links ]
Jordán-Garza, A.G., Maldonado M.A., Baker D.M. & Rodríguez-Martínez R.E. 2008. High abundance of Diadema antillarum on a Mexican reef. Coral Reefs 27: 295. [ Links ]
Linton, D. & T. Fisher. 2004. Caribbean Coastal Marine Productivity Program, 1993-2004. CARICOMP, Kingston, Jamaica. [ Links ]
Loya, Y. 1976. Effects of water turbidity and sedimentation on the community structure of Puerto Rican corals. Bull. Mar. Sci. 26: 450-466. [ Links ]
Moberg, F. & C. Folke 1999. Ecological goods and services of coral reef ecosystems. Ecol. Econ. 29: 215-233. [ Links ]
Patriquin, D.G. 1975. "Migration" of blowouts in seagrass beds at Barbados and Carriacou, West Indies, and its ecological and geological implications. Aquat. Bot. 1: 163-189. [ Links ]
Powell, G.V.N., W.J. Kenworthy & J.W. Fourqurean. 1989. Experimental evidence for nutrient limitation of seagrass growth in a tropical estuary with restricted circulation. Bull. Mar. Sci. 44: 324-340. [ Links ]
Rodríguez-Martínez, R.E. 1993. Efectos de un ciclón en la estructura comunitaria de corales escleractinios. Bachelor Thesis. E.N.E.P. Iztacala. Universidad Nacional Autónoma México, México. [ Links ]
Rodríguez-Martínez, R.E. 2008. Community involvement in marine protected areas: the case of Puerto Morelos reef, México. J. Environ. Manag. 88: 1151-1160. [ Links ]
Rose, C.D. & C.J. Dawes. 1999. Effects of community structure on the seagrass Thalassia testudinum. Mar.Ecol. Prog. Ser. 184: 83-95. [ Links ]
Ruíz-Rentería, F., Van Tussenbroek B.I. & E. Jordán-Dahlgren. 1998. Puerto Morelos, Quintana Roo, Mexico, p 57-66. In B.J. Kjerfve (ed.). CARICOMP-Caribbean Coral Reef, Seagrass and Mangrove Sites. UNESCO, Paris. [ Links ]
Short, F.T. & S.W. Echeverria. 1996. Natural and humaninduced disturbance of seagrasses. Environm. Conserv. 23: 17-27. [ Links ]
Van Tussenbroek, B.I. 1994a. Spatial and seasonal variability in biomass and leaf morphology of the manatee grass Syringodium filiforme in a tropical coral reef lagoon, Mexico. An. Inst. Cienc. del Mar y Limnol. UNAM 21: 15-22. [ Links ]
Van Tussenbroek, B.I. 1995. Thalassia testudinum leaf dynamics in a Mexican Caribbean reef lagoon. Mar. Biol. 122: 33-40. [ Links ]
Van Tussenbroek, B.I. 1998. Above- and below-ground biomass and production of Thalassia testudinum in a tropical reef lagoon. Aquat. Bot. 61: 69-82. [ Links ]
Van Tussenbroek, B.I., K. Hermus & T. Tahey. 1996. Thalassia testudinum (Banks ex König) biomass and growth in a shallow tropical lagoon system, in relation to tourist development. Carib. J. Sci. 32: 357-364. [ Links ]
Van Tussenbroek, B.I., J.A. Vonk, J. Stapel, P.L.A. Erftemeijer, J.J. Middelburg & J.C. Zieman. 2006. The biology of Thalassia, p. 409-439. In A.W.D. Larkum, R.J. Orth & C.M. Duarte (eds.). Paradigms and Recent Advances in Research Seagrass Biology. Springer, The Netherlands. [ Links ]
Van Tussenbroek, B.I., M.G. Barba Santos, J.K. van Dijk, S.N.M. Sanabria Alcaraz & M.L. Téllez Calderón. 2008. Selective elimination of rooted plants from a tropical seagrass bed in a back-reef lagoon: a hypothesis tested by Hurricane Wilma (2005). J. Coast. Res. 24: 278-281. [ Links ]
Williams, S.L. 1990. Experimental studies of Caribbean seagrass bed development. Ecol. Monogr. 60: 449- 469. [ Links ]
Zieman, J.C. & R.G. Wetzel. 1980. Productivity in seagrasses: methods and rates, p. 87-116. In R.C. Phillips & C.P. McRoy (eds.). Handbook of Seagrass Biology: An Ecosystem Perspective. Garland STPM, New York, USA. [ Links ]
Correspondencia: Rosa E. Rodríguez-Martínez. Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México. Ap. Postal 1152, 77500 Cancún, Q. Roo, México; rosaer@cmarl.unam.mxFrancisco Ruíz-Rentería;Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México. Ap. Postal 1152, 77500 Cancún, Q. Roo, México;renteria@cmarl.unam.mx
Brigitta van Tussenbroek. Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México. Ap. Postal 1152, 77500 Cancún, Q. Roo, México; vantuss@cmarl.unam.mx
Guadalupe Barba-Santos. Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México. Ap. Postal 1152, 77500 Cancún, Q. Roo, México; isis@cmarl. unam.mx
Edgar Escalante-Mancera. Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México. Ap. Postal 1152, 77500 Cancún, Q. Roo, México; escalant@cmarl.ail.com
Guillermo Jordán-Garza. Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México. Ap. Postal 1152, 77500 Cancún, Q. Roo, México; nadroj_ome@hotmail.com,
Eric Jordán-Dahlgren. Instituto de Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México. Ap. Postal 1152, 77500 Cancún, Q. Roo, México; jordan@cmarl.unam.mx
Received 10-VII -2009. Corrected 22-II -2010. Accepted 12-VII -2010.