Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.57 n.4 San José Dec. 2009
Phytoplankton assemblage of a small, shallow, tropical African reservoir
Moshood K. Mustapha
Department of Zoology, University of Ilorin, Ilorin, Nigeria; moonstapha@yahoo.com
Abstract: I measured physico-chemical properties and phytoplankton in the small, shallow tropical reservoir of Oyun (Offa, Nigeria) between January 2002 and December 2003. I identified 25 phytoplankton genera in three sampling stations. Bacillariophyceae dominated (75.3%), followed by Chlorophyceae (12.2%), Cyanobacteria (11.1%) and Desmidiaceae (0.73%). The high amount of nutrients (e.g. nitrate, phosphate, sulphate and silica) explain phytoplankton heterogeneity (p<0.05). Phytoplankton was abundant during the rainy season, but the transition period had the richest assemblage and abundance. Fluctuations in phytoplankton density were a result of seasonal changes in concentration of nutrients, grazing pressure and reservoir hydrology. The reservoir is eutrophic with excellent water quality and a diverse phytoplankton assemblage: fish production would be high. These conditions resulted from strategies such as watershed best management practices (BMPs) to control eutrophication and sedimentation, and priorities for water usage established through legislation. Additional measures are recommended to prevent oligotrophy, hypereutrophy, excessive phytoplankton bloom, toxic cyanobacteria, and run-off of organic waste and salts. Rev. Biol. Trop. 57 (4): 1009-1025. Epub 2009 December 01.
Key words: Freshwater reservoir, hydrology, phytoplankton assemblage, nutrients, water quality.
Phytoplankton are usually at the base of aquatic food web and are the most important factor for production of organic matter in aquatic ecosystem. Most reservoirs will require significant amount of phytoplankton to have productive and sustainable fisheries. The interplay of physical, chemical and biological properties of water most often lead to the production of phytoplankton, while their assemblage (composition, distribution, diversity and abundance) is also structured by these factors. Thus any perturbations in these factors may affect the phytoplankton assemblage which could have a significant impact on water quality and fisheries of reservoirs. The importance of phytoplankton in tropical reservoir ecosystems include its use in estimating potential fish yield (Hecky & Kling 1981), productivity (Park et al. 2003), water quality (Walsh et al. 2003), energy flow (Simciv 2005), trophic status (Reynolds 1999) and management (Beyruth 2000). Weiss & Stockner (1993) observed that energy in pelagic ecosystems flows from phytoplankton, the primary producers to zooplankton, the consumers through the classical food chains and microbial food webs. Models that use phytoplankton primary production as the main controlling variable for predicting fish yield in lakes and reservoirs have resulted in more successful predictions than many other methods (Knosche & Barthelmes 1998).
Phytoplankton studies of small, shallow, tropical African reservoirs such as Oyun reservoir could provide management guide to the reservoir water quality and its fisheries, since most of these reservoirs are constructed solely for drinking water supply, but, with fish production often superimposed on them. Such study could reveal information on phytoplankton that could impact unpleasant tastes and odours to the water (Muller et al. 1982), produce toxins that could affect fish when consumed (Codd 1995), and affect the physico-chemistry of the water. According to Bucka (1998), there is currently great interest in preventing or reducing the growth of planktonic algae and cyanobacteria in water supply reservoir.
The aim of this paper is to look at the species composition and relative abundance of the phytoplankton of Oyun reservoir, Offa, Nigeria to various physico-chemical variables; this is with a view to managing the reservoir for sustainable water and fish production.
Materials and methods
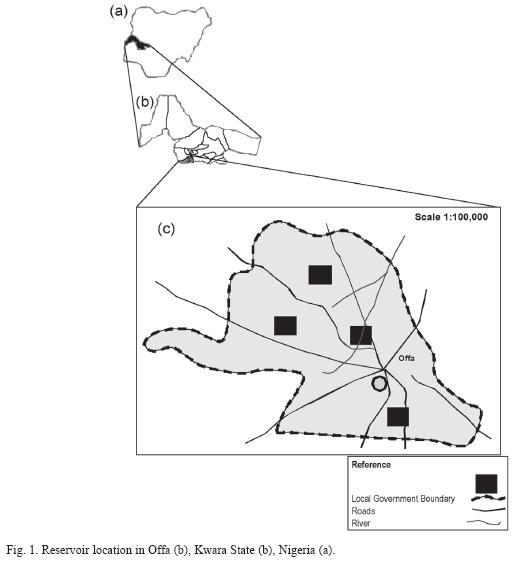
Study site description: Oyun reservoir is located in Offa, Kwara State, Nigeria (8°30’05" N and 8°15’55" E) (Fig. 1). It is a dam reservoir on Oyun River, created in 1964 (expanded in 1983 and 1995 with further expansions proposed) by damming the Oyun River. The reservoir is eutrophic (Mustapha 2008) with diverse species of littoral plant occupying the shoreline length.
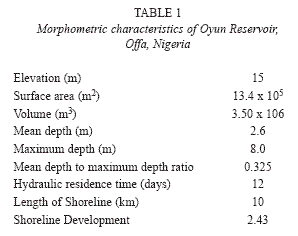
The reservoir was created to supply potable water for domestic and industrial uses to an estimated population of about 300 000 people. Subsistence and commercial fishing activities is also carried out on the reservoir. The reservoir has a maximum length of 128m, maximum width of 50m and maximum depth of 8.0m, mean depth of 2.6m. The surface area is 6.9x105m2 while the water volume is 3.50x106m3. The net storage capacity is 2.9x106m3. The reservoir is subjected to temporal fluctuations in water volume with high water volume in the rainy season and less water in the dry season due to high evaporation. The water retention time is between 4-5 months in the rainy season (April-October), with an average precipitation between 1 000mm and 1 200m, while the water residence time in the dry season (November-March) is between 1-2 months with average rainfall of about 100mm and 200mm. The morphometric characteristics of the reservoir are listed in Table 1.
Samplings: Physico-chemical variables of the water body selected as water quality parameters were sampled monthly from three stations between January 2002 and December 2003. Station 1 was at the dam axis where a lot of human activities such as washing, bathing, fish landing and cassava fermentation take place. It is the highest point of human and animal contact with the reservoir and also the deepest part close to run-offs from farm lands and outflow. Station 2 was at the mid-section of the reservoir which represented the area of lentic water, it is the transition zone between the riverine and lentic sections of the reservoir. Station 3 was at the head water of the reservoir which represented the lotic section and inflow of the reservoir. Duplicate surface water samples were collected from 10cm depth into 50ml plastic water bottles that have been acid-washed prior to water analyses. Surface water temperature, pH, electrical conductivity and total dissolved solids were measured in situ using Hanna portable pH/EC/TDS/Temperature combined water proof tester model HI 98129. Transparency was determined by extinction method using the secchi disc by measuring the mean of the depths at which the secchi disc disappears and at which it reappears, dissolved oxygen was determined by Azide modification of the Winkler method, chemical oxygen demand was measured using the dichromate reaction method (Hach 2003), carbon dioxide and alkalinity were determined by titration (APHA 1995). Nitrate-Nitrogen (NO3-N), Phosphate (PO4-P), Sulphate (SO4 2-), Calcium Ca2+, Magnesium Mg2+, total hardness and Silica (SiO2) were measured according to APHA (1995) standard procedures using Hach spectrophotometer model DR-EL/2. All the chemical analyses were done at the water quality laboratory of Kwara State Utility Board, Ilorin, Nigeria, immediately after the sample collection.
Phytoplankton sampling: Phytoplankton samples were collected with 1- litre plastic bottles (Wetzel 1999). The samples were fixed on the field with Lugol solution to arrest cell activity, for sedimentation and better staining (Sherr et al. 1989). The sample was stored in a dark compartment in the laboratory for overnight sedimentation. In the morning, the upper 900ml was siphoned out and the remaining 100ml homogenized samples were retained from which 1ml subsample was collected for phytoplankton identification and counting under a microscope (100X). The phytoplankton was identified to generic level using keys compiled by Edmonson (1959) and Withford & Schumacher (1973). Numerical estimations of the phytoplankton were done using the drop method of Margalef (1976). The relative abundance of the various taxa was then calculated. Data analyses were done using the GLM procedure of statistical analysis system 9.1.3 (SAS Institute 2003). Monthly mean differences in stations, seasons and years were assessed by LSD using two-way ANOVA with test of significance at p<0.05. Analysis of co-variance was used to investigate the correlation and level of dependence between the total phytoplankton and the phytoplankton classes with the physico- chemical variables. The test variables were stations, seasons and years. Statistical differences were considered at p<0.05
Results
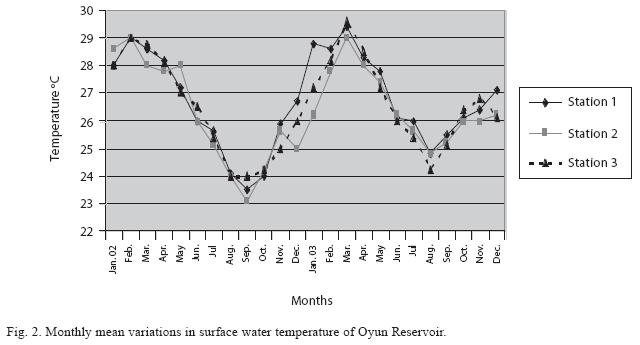
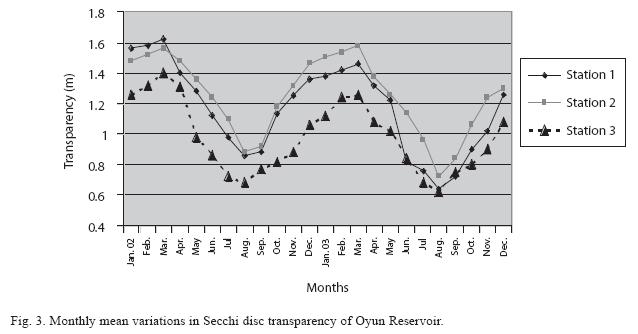
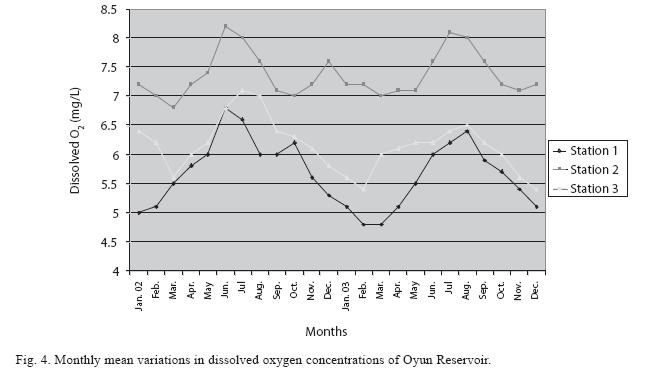
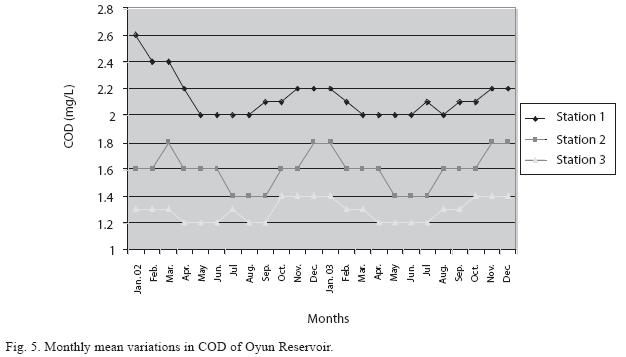
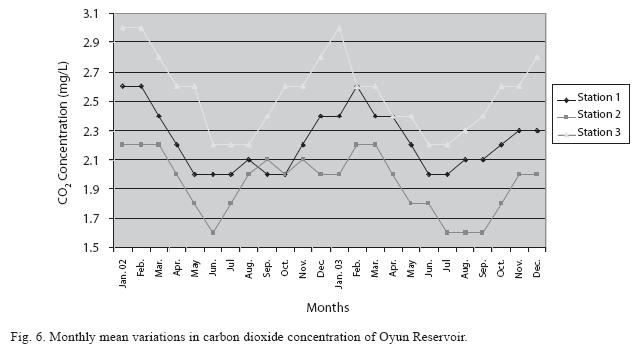
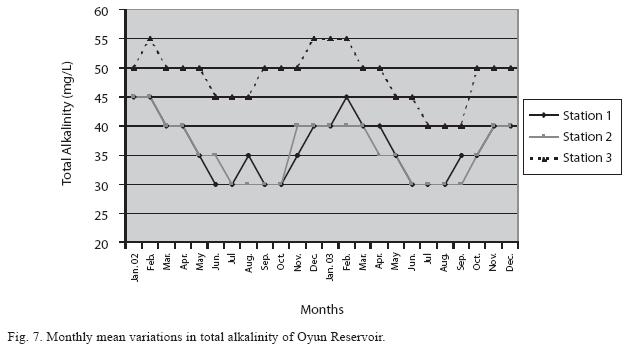
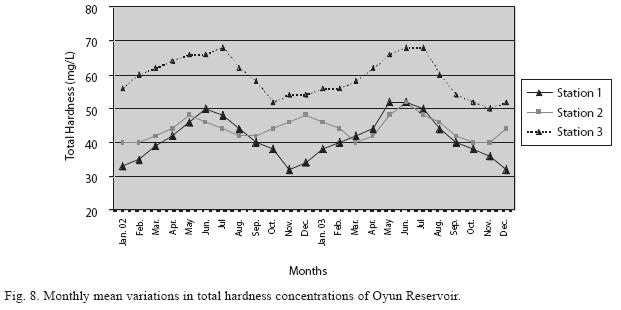
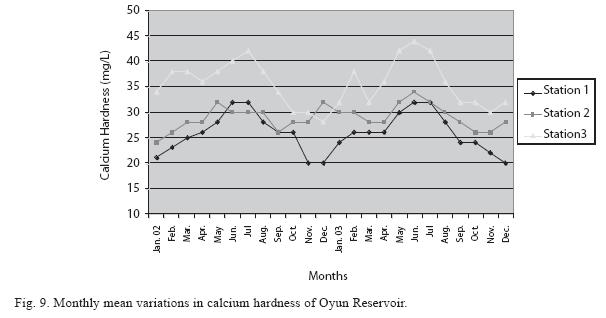
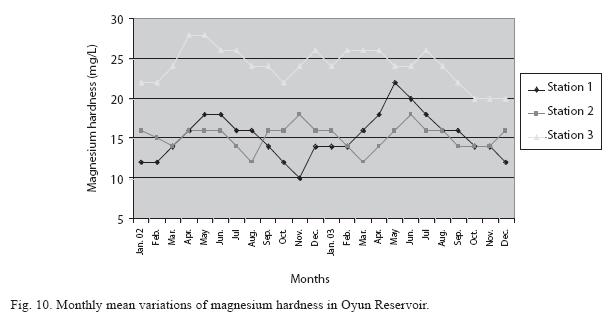
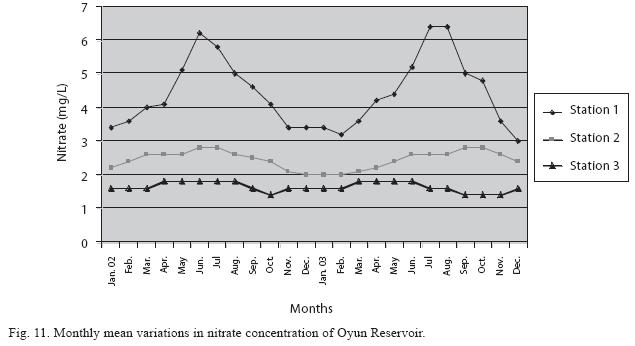
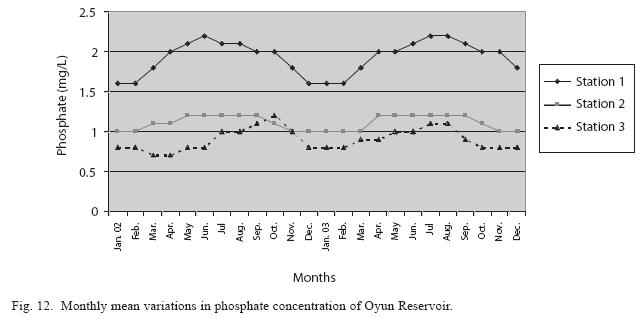
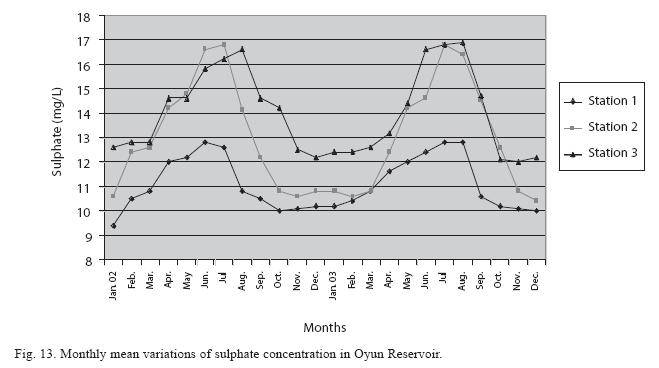
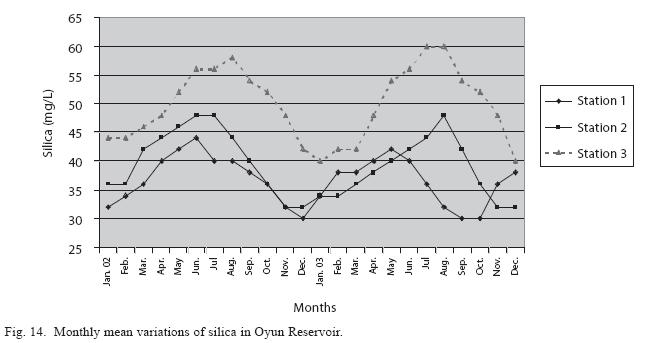
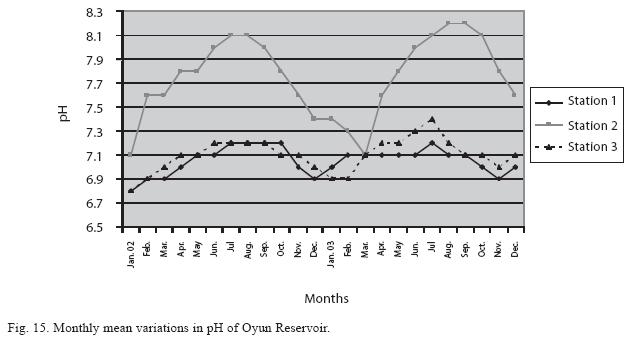
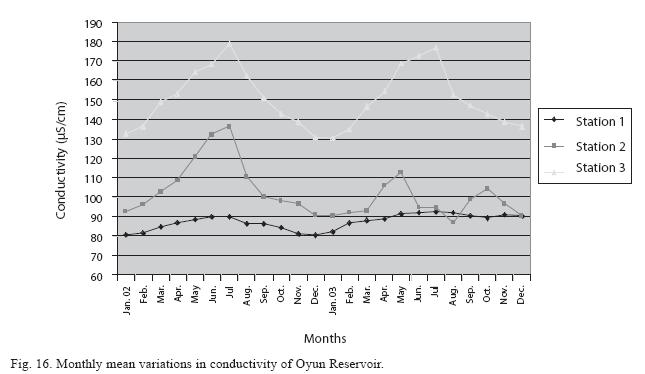
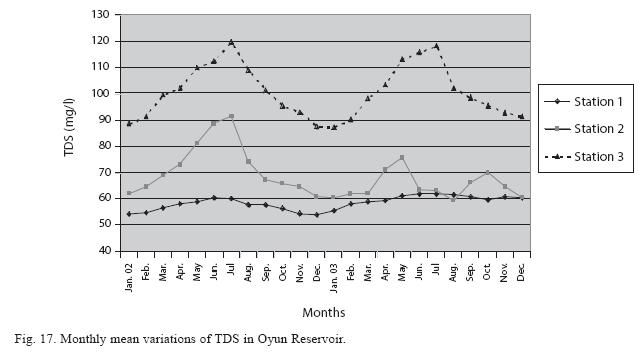
Physico-chemical factors: Seasonal fluctuations were recorded among the physicochemical factors. The driving forces for theses fluctuations were the rainy and dry seasons. The mean monthly variations in the surface water temperature of the three stations are presented in Fig. 2. The temperature ranged between the lowest value of 23.1±0.5°C obtained in September and highest of 29.6 ±0.1°C obtained in March, 2003. Dry season temperature was significantly higher (p<0.05) than the wet season. Secchi disc transparency was highest at 1.62±0.32m obtained in the dry season of 2002 and lowest at 0.62±0.8m in the rainy season of 2003 (Fig. 3). Significant higher transparency (p<0.05) were obtained in the dry season, Station 2 and year 2002. Dissolved oxygen fluctuated between lowest monthly mean of 4.8±0.25mg/L obtained in February and March 2003 and highest monthly mean of 8.2±0.31mg/L recorded in June 2002 (Fig. 4). Dissolved oxygen values in the wet season, Station 2 and year 2002 were significantly higher (p<0.05). Chemical Oxygen Demand (COD) varied between 1.2±0.1 mg/lL and 2.6±0.2mg/L COD was significantly higher in the dry season and in Station 1 (Fig. 5). Carbon dioxide and total alkalinity showed similar pattern with COD in their concentrations among the stations and in the seasons. Carbon dioxide ranged between monthly mean of 1.6±0.2mg/L to 3.0±0.6mg/L (Fig. 6) while total alkalinity fluctuated between monthly mean of 30 ±2.6mg/L and 55±3.4mg/L (Fig. 7). The total hardness value in the reservoir which is the sum of calcium and magnesium hardness concentrations was found to be significantly higher in the wet season. This was the same for calcium and magnesium ions. Station 3 showed significantly higher concentration of total, calcium and magnesium hardness. The mean monthly range of the total hardness (32±0.5-68±1.4mg/L), calcium hardness (20±0.1- 44±1.8mg/L) and magnesium hardness (10±0.4-28±0.6mg/L) are presented in figures 8, 9 and 10 respectively. The highest monthly mean concentration of nitrate recorded was 6.4mg/L ±0.3 obtained from Station 1 at the peak of the rains in August 2003. A decrease was observed in the dry season with the lowest concentration of 1.4±0.1mg/L recorded from Station 3 in October 2003 (Fig. 11). Nitrate was significantly higher in the raining season and the order of magnitude in the concentration among the stations was Stations 1>2>3. Phosphate had the least concentration among the ions. It ranged between 0.7±0.0mg/L to 2.2±0.2 mg/L (Fig. 12). Like Nitrate, phosphate concentration was significantly higher in raining season and in Station 1 (p<0.05). Sulphate concentration was lowest at 9±0.2mg/L; it gradually increased until a maximum concentration of 16.9±0.45mg/L (Fig. 13). Sulphate was significantly higher in the wet season while the order of higher concentration among the stations was Stations 3>2>1.The minimum and maximum monthly mean concentration of silica was from 30±0.2mg/L to 60±0.6mg/L (Fig. 14). Silica was significantly higher (p<0.05) in the rainy season and Station 3 recorded the highest concentration among the stations. The surface water pH fluctuated between slight acidity and moderate alkalinity. The lowest monthly mean pH was 6.8±0.05 obtained during the dry season in January 2002, while the highest was 8.2±0.2 obtained in the wet season of August and September of 2003 (Fig. 15). pH was significantly (p<0.05) higher during the wet season and in Station 2. The monthly mean variations in electrical conductivity and total dissolved solids (TDS) were similar. The lowest conductivity (80.4±0.8μs/cm) and TDS (53.9±0.8mg/L) values were recorded in December 2002 and the highest (178.8±2.0μs/cm) and TDS (119.8 ±2.0mg/L) were obtained in July 2002 (Figs. 16 and 17). The two factors were statistically higher (p<0.05) during the rainy season while the order of significant difference between the stations was Stations 3>2>1.
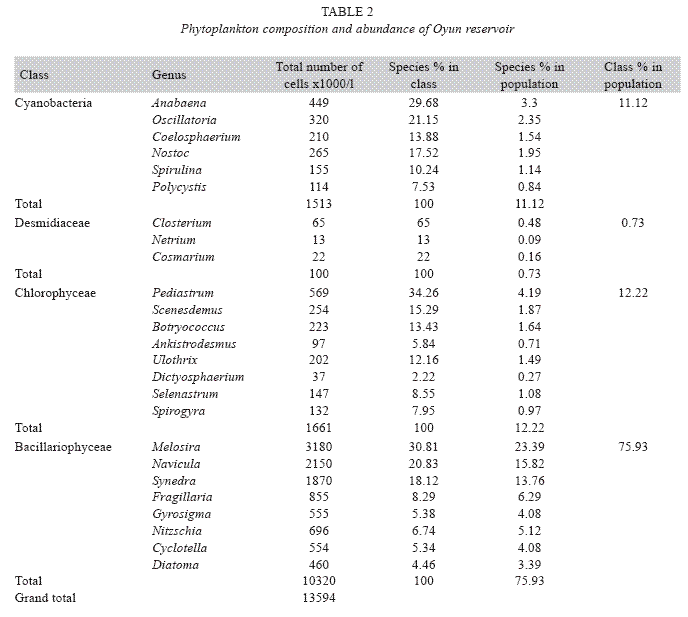
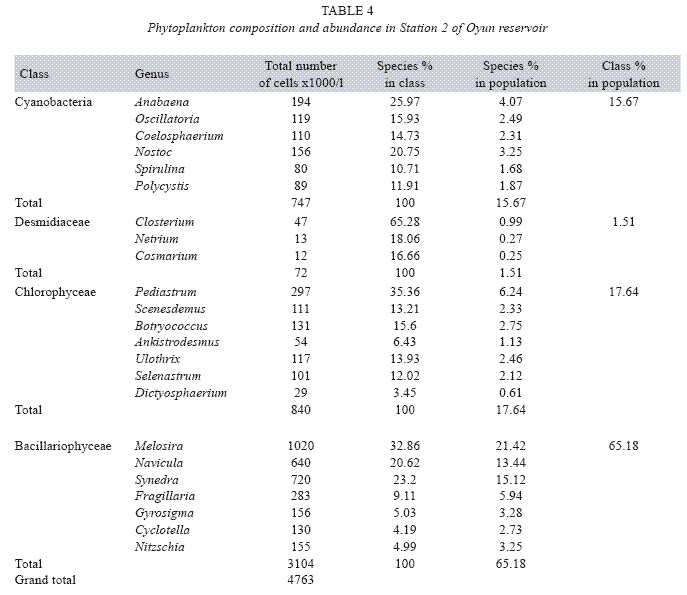
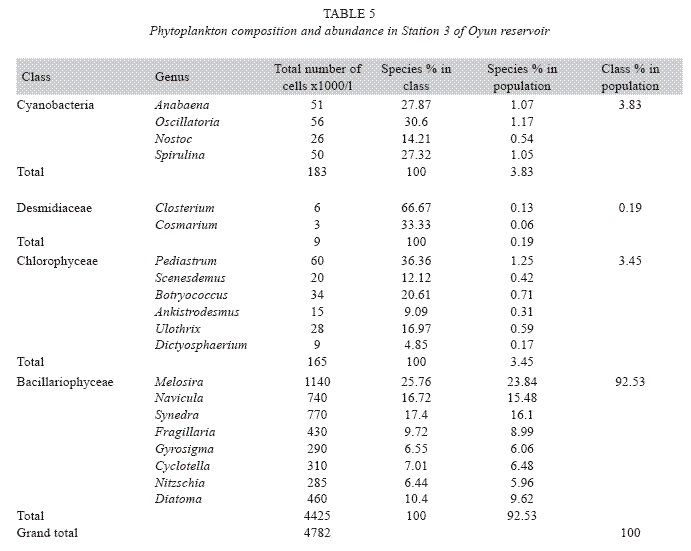
Phytoplankton species composition and abundance: Four classes of phytoplankton namely Bacillariophyceae (diatoms), Chlorophyceae (green algae), Cyanobacteria (bluegreen algae), and Desmidiaceae (desmids) were recorded in the reservoir. A total of 25 genera were found in the phytoplankton classes with Bacillariophyceae and Chlorophyceae having the highest of eight genera each, while Cyanobacteria and Desmidiaceae had six and three genera respectively (Table 2). A total phytoplankton biomass of 13 594x103cells per millilitre was recorded in the reservoir. Bacillariophyceae (180x103 to 460x103cells/ ml) dominated the phytoplankton classes in terms of individual cells/ml and abundance with a total of 10 320x103cells/ml constituting 75.93% of the phytoplankton. This was distantly followed by the Chlorophyceae (37 to 569x103cells/ml) dominated by Pediastrum, and making up 12.22% , while Cyanobacteria (114 to 449x103cells/ml) dominated by Anabaena and Desmidiaceae (13 to 65x103cells/ ml) dominated by Closterium made 11.12% and 0.73% respectively of the phytoplankton population. Among phytoplankton genera, Melosira was the most abundant constituting 25.81% of the total phytoplankton and was also the predominant in the class Bacillariophyceae while Netrium, a desmid had the least biomass of 13x103cells/ml which was 0.09% of the total phytoplankton population. ANOVA (p<0.05) showed significant difference in total phytoplankton count among the seasons, stations and the years. Phytoplankton was significantly abundant in the wet season than the dry season with Station 3 having higher phytoplankton cell number than Station 2 and Station 1 in that order, while year 2002 recorded a higher abundance of phytoplankton than 2003.
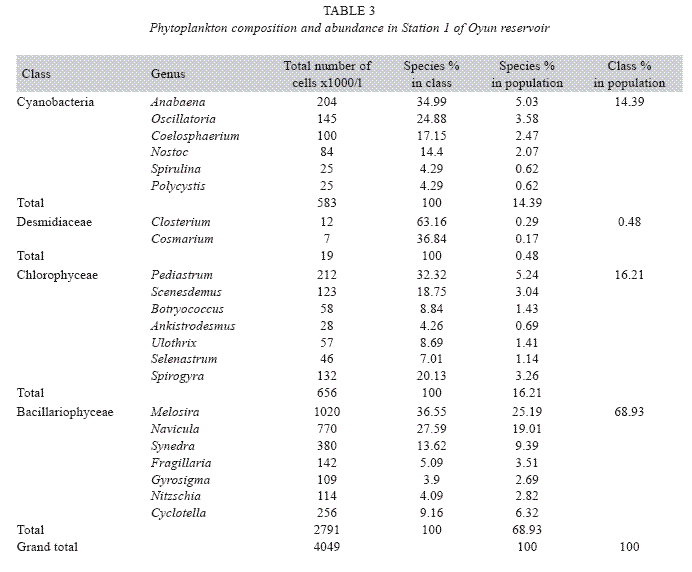
Phytoplankton community assemblage: The phytoplankton classes were present in all the three stations and highly abundant in the rains especially between May and October with peaks in June and July while in the dry season the phytoplankton density was significantly reduced. Station 2 had a higher genera diversity with 23 genera recorded, Station 1 recorded 22 genera, while 20 genera were observed from Station 3 (Tables 3, 4 and 5). ANOVA (p<0.05) also revealed that all the phytoplankton classes were significantly higher in the rains, Chlorophyceae, Cyanobacteria and Desmidiceae were significantly abundant (p<0.05) in Station 2, while Bacillariophyceae was more abundant in Station 3. Bacillariophyceae and Chlorophyceae had a higher cell count in 2002, while Cyanobacteria and Desmidiaceae recorded significant higher biomass in 2003.
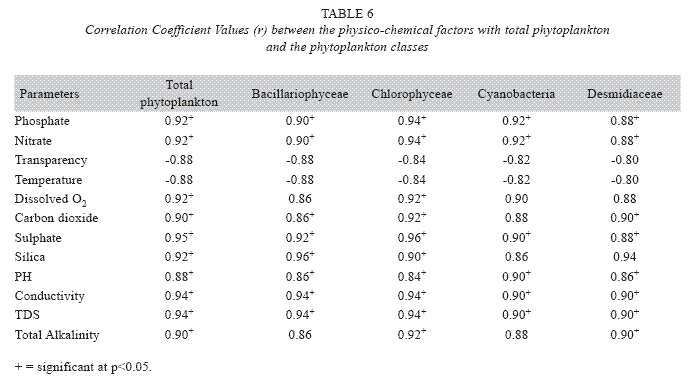
Correlations matrix: The correlation coefficient (r) values shows the total phytoplankton and all the classes to be positively correlated (p<0.05) with phosphate, nitrate, dissolved oxygen, sulphate, silica, carbon dioxide, total alkalinity, pH, conductivity and total dissolved solids. Only transparency and temperature showed negative correlation with the phytoplankton (Table 6).
Discussion
Phytoplankton assemblage: The phytoplankton assemblage of Oyun reservoir was highly diversed, heterogenous and abundant. This was due to the presence of large amount of nutrients especially nitrate, phosphate, silica and sulphate as well as the shallow nature of the reservoir, which greatly exposes the surface to light for algal productivity. This is reflected in the positive correlation of the nutrients with all the classes of phytoplankton found in the reservoir. Gasse et al. (1983) correlated phytoplankton species composition of lakes with a number of water quality parameters including temperature, alkalinity, concentration and pH. The phytoplankton were highly abundant during the rains which corresponded to the period when the ions were highly concentrated in the reservoir. Thomas et al. (2000) has reported that high primary productivity in tropical reservoir is usually rain induced.
The dominance of Bacilariophyceae was attributed to the high concentration of silica, while the high amount of nitrate phosphate and sulphate caused the abundance of Chlorophyceae, Cyanobacteria and Desmidiaceae. The nutrients have been known to be limiting in phytoplankton growth (Talling & Lemoalle 1998). This is evident from the reduction seen in concentration of these nutrients with the phytoplankton growth and abundance. Harris et al. (1989) described this scenario has bottle effect. The dominance of Bacillariophyceae is typical of tropical reservoirs (Wood & Talling 1988). Alfred-Ockiya & Otobo (1990) also reported the dominance of diatoms in Ofoniturobo Lake in Nigeria. The absence of many species of desmids could be due to their preference for brackish or high saline waters. Opute (2000) reported that Desmidiaceae are often not significantly abundant in Nigerian freshwater reservoir. The order of abundance and assemblage of the phytoplankton of Oyun reservoir Bacillariophyceae >Chlorophyceae>Cyanobacteria >Desmidiaceae was similar to the observations of Brooklemma (1995) on a tropical lake in Ethiopia. The dominace of Chlorophyceae, Cyanobacteria and Desmidiaceae in Station 2 (transition zone) was due to the station’s high nutrients; transparency and water residence time. Higher total phytoplankton biomass and density especially Bacillariophyceae in Station 3 was linked to higher inputs of nutrients in the station coming from rocks and sediments, with the station located in the riverine zone of the reservoir. The low density of the phytoplankton in Station 1 was attributed to factors such as high level of organic decomposition, low transparency, patchiness and low concentration of dissolved salts. The dominance of Melosira was due to the high concentration of silica and sulphate which were abundant in the rains. The preponderance of Anabaena, Pediastrum and Closterium in their respective classes might have occurred in response to the availability and utilization of nitrate and phosphate ions.
Correlation of physico-chemical factors with phytoplankton assemblage: The high species of phytoplankton diversity in the reservoir could also be due to its ‘old age’ since older reservoirs usually display higher species richness (Rocha et al. 1999) and its assemblages were similar to other tropical reservoirs. Factors such as high temperature in short water residence time, high flushing rate of nutrients in the dry season and grazing effects by zooplankton and fishes could have caused the reductions in numbers of the phytoplankton in the dry season. The negative correlation of temperature and transparency with all the classes of the phytoplankton was an indication that the phytoplankton were depleted during higher temperature and higher transparency (which occurs in the dry season). High transparency could have allowed grazers such as zooplankton and fishes to effectively predate on them.
Water quality parameters and phytoplankton assemblage: The phytoplankton assemblage of Oyun reservoir shows the reservoir to be eutrophic and its fluctuation is in response to seasonal concentration of nutrients, grazing pressure by biotic organisms and the reservoir hydrology. Nuisance conditions associated with high phytoplankton diversity and productivity were absent from the reservoir probably due to its shallow depth and high mixing volume. This made the water quality to be good in relation to phytoplankton composition. The water quality of Oyun reservoir as assessed from the physico-chemical properties compared well with the ranges found in other Nigerian reservoirs (Kemdirim 1990, Akinbuwa 1992). It also fell within the range of allowable limits recognized by WHO (1997) and NIS (2007).
Phytoplankton and fish yield: The heterogeneity of the phytoplankton of the reservoir will support a high fish production. The phytoplankton community composition diversity and abundance of this reservoir could be used as the main controlling variable for predicting fish yields in the reservoir. Knosche & Barthemes (1998) have used this model of phytoplankton primary production to predict fish yields in lakes and reservoirs and reported this model resulted in more successful predictions than many other methods. Cushing (1982) also observed a strong relationship between primary production and fish production and concluded that a long term fluctuation in fish stocks was due to this relationship.
Reservoir management: The diverse assemblage of the phytoplankton was due to the strategies being employed in the management of the reservoir. This include the use of watershed best management practices (BMPs) to control eutrophication and sedimentation, and the establishment of important priorities through legislations for water usage by space and time. Other measures that prevent oligotrophy, hypereutrophy, excessive phytoplankton bloom, establishment and proliferation of toxic cyanobacteria, run-off of organic wastes and high dissolved salts were also been used to maintain the water quality and this made the phytoplankton assemblage and productivity of the reservoir to be high.
Resumen
Investigué las propiedades físico-químicas y el fitoplancton del pequeño embalse tropical Oyun (Offa, Nigeria) entre enero de 2002 y diciembre de 2003, para determinar el efecto de las propiedades físico-químicas en el fitoplancton y la producción de peces. En las tres estaciones estudiadas identifiqué 25 géneros de fitoplancton pertenecientes a cuatro clases. Bacillariophyceae dominó (75.3%), seguida de Chlorophyceae (12.2%), Desmidiaceae (11.1%) y Cianobacteria (0.73%). La gran cantidad de nutrientes (e.g. nitrato, fosfato, sulfato y sílice) explica la heterogeneidad del fitoplancton (p<0.05), el cual fue abundante durante la época de lluvias. La zona de transición de la reserva tuvo el conjunto más rico y abundante. Las fluctuaciones en la densidad del fitoplancton fueron resultado de la concentración estacional de los nutrientes, la presión del forrajeo y la hidrología. El embalse es eutrófico con agua excelente y fitoplancton diverso, y por tanto su producción pesquera sería alta. Estas condiciones son consecuencia de estrategias como mejores prácticas de gestión de cuencas (BPM) para controlar la eutroficación y la sedimentación, y el establecimiento de prioridades legales para un buen uso del agua. Recomiendo medidas adicionales contra la oligotrofía, la hipereutrofia, el exceso de fitoplancton, las cianobacterias tóxicas, las escorrentías de residuos orgánicos y el exceso de sales disueltas.
Palabras clave: Embalse de agua dulce, hidrología, conjunto de fitoplancton, nutrientes, calidad de agua.
Received 15-XII-2008. Corrected 28-V-2009. Accepted 29-VI-2009.
References
Akinbuwa, O. 1992. A preliminary study of diurnal vertical distribution of rotifers in Opa Reservoir, Nigeria. J. Aquat. Sci. 7: 19-28. [ Links ]
Alfred-Ockiya, J.F. & A.J.T. Otobo. 1990. Biological studies of Ofonitorubuo Lake in the freshwater swamps of the Niger Delta, Rivers State, Nigeria. Journal of Aquatic Science 5: 77-82. [ Links ]
APHA (American Public Health Association). 1995. Standard methods for the examination of water and waste water. 20th edition. American Public Health Association, New York, New York, USA. [ Links ]
Beyruth, Z. 2000. Periodic disturbances, trophic gradient and phytoplankton characteristics related to cyanobacterial growth in Guarapiranga reservoir. Sao Paulo State, Brazil. Hydrobiologia 424: 51-60. [ Links ]
Bucka, H. 1998. The mass invasion of several blue-green algae in two drinking water supply reservoirs in Southern Poland, p. 145-151. In D.G. George, J.G. Jones, P.Puncochar, C.S. Reynolds & D.W. Sutcliffe (eds.). Management of lakes during global climate change. Kluwer, Dordrecht, Netherlands. [ Links ]
Brooklemma. 1995. Seasonal limnological studies on Lake Alemaya: a tropical African lake, Ethiopia. Arch. Hydrobiol. 107: 263-285. [ Links ]
Codd, G.A. 1995. Cyanobacterial toxins: occurrence, properties and biological significance. Wat. Sci & Tech. 32: 149-156. [ Links ]
Cushing, D.H. 1982. Climate and Fisheries. Cambridge,Cambridge, England. [ Links ]
Edmonson, W.T. 1959. Freshwater Biology. Wiley, New York, New York, USA. [ Links ]
Gasse, F., J.F Talling & P. Kilham. 1983. Diatoms assemblages in East Africa: classification, distribution and ecology. Rev. Hydrobiol. Trop. 16: 3-34. [ Links ]
Hach. 2003. Water analysis handbook (Fourth edition), Vol. 2: Hach Chemical Company, Loveland, Colorado, USA. [ Links ]
Harris, G.P., F.B. Griffiths & D.P. Thomas. 1989. Light and dark uptake of and loss of 14C: methodological problems with productivity measurements in oceanic waters. Hydrobiologia. 173: 95-105. [ Links ]
Hecky, R.E. & H.J. Kling. 1981. The phytoplankton and protozooplankton of the euphotic zone of Lake Tangayinka: species composition, biomass, chlorophyll content and spatio-temporal distribution. Limnol. Oceanogr. 26: 548-564. [ Links ]
Kemdrim, E.C. 1990. Periodicity and succession of phytoplankton in an upland and lowland impoundments in Plateau State, Nigeria in relation to nutrients levels and physical characteristics. J. Aquat. Sci. 5: 43-52. [ Links ]
Knosche, R. & D. Barthelmes. 1998. A new approach to estimate lake fisheries yield from limnological basic parameters and first results. Limnologica 28: 133-144. [ Links ]
Margalef, R. 1976. Limnology of the Spanish reservoirs. Publication No. 123, Centro de Estudios Hidrograficos, Madrid, Spain. [ Links ]
Muller, H., F. Juttner & U. De Harr. 1982. Schadstoffe im Wasser, Vol. III: Algenburtige Schadstoffe, Harald Boldt, Boppard. [ Links ]
Mustapha, M.K. 2008. Assessment of the water quality of Oyun Reservoir Offa, Nigeria, using selected physico-chemical parameters. Turk. J. Fish. Aquat. Sci. 8: 309-319. [ Links ]
NIS. 2007. Nigerian standard for drinking water quality. Standard organization of Nigeria. Lagos, Nigeria. [ Links ]
Opute, F.I. 2000. Contribution to the knowledge of algae of Nigeria. Desmids from Warri/Forcados estuaries. J. Limnol. 59: 131-155. [ Links ]
Park, S., M.T. Brett, E.T. Oshell & C.R. Goldman. 2003. Seston food quality and Daphnia production efficiencies in an oligo-mesotrophic Subalpine lake. Aquat. Ecol. 37: 123-136. [ Links ]
Reynolds, C.S. 1999. Non-determinism to probability, or N:P in the community ecology of phytoplankton: Nutrient ratios. Arch. Hydrobiol. 146: 23-35. [ Links ]
Rocha, O., T. Matsumura-Tundisi, E.L.G. Espindola, K.F. Roche, & A.C. Rietzler. 1999. Ecological theory applied to reservoir zooplankton, p. 29-51. In J.G Tundisi & M. Straskraba (eds.). Theoretical reservoir ecology and its applications. International Institute of Ecology, Brazilian Academy of Sciences. Backhuys, Leiden, Netherlands. [ Links ]
SAS Institute. 2003. SAS/Stat User’s Guide, Version 9.1.3. SAS Institute Inc., Carry, North Carolina, USA. [ Links ]
Sherr, B.F., E.B. Sherr & C. Pedro-Alio. 1989. Simultaneous measurement of bacterioplankton production and protozoan herbivory. Mar. Ecol. Progr. Ser. 54: 209-219. [ Links ]
Simciv, T. 2005. The role of plankton, zoobenthos, and sediment in organic matter degradation in oligotrophic and eutrophic mountain lakes. Hydrobiologia 532: 69-79. [ Links ]
Talling, J.F. & J. Lemoalle. 1998. Ecological dynamics of tropical inland waters, p. 82-117. In J.F. Talling & J. Lemoalle (eds.). Resource utilization and biological production- primary utilization: energy. Cambridge, Cambridge, England. [ Links ]
Thomas, S., P. Cecchi, D. Corbin & J. Lemoalle. 2000. The different primary producers in a small African tropical reservoir during a drought: temporal changes and interactions. Freshwater Biol. 45: 43-56. [ Links ]
Walsh, C.J., A.K. Sharpe, P.F. Breen & J.A. Sonneman. 2001. Effects of urbanization on streams of the Melbourne region, Victoria, Australia. I. Benthic macroinvertebrate communities. Freshwater Biol. 46: 535-551. [ Links ]
Weiss, T. & J.G. Stockner. 1993. Eutrophication: the role of microbial food webs. Mem. Ist. Ital. Idrobiol. 52: 133-150. [ Links ]
Wetzel, R.G. 1999. Biodiversity and shifting energetic stability within freshwater ecosystems. Arch. Hydrobiol. Spec. Issues Advances in Limnology 54: 19-32. [ Links ]
WHO (World Health Organization). 1997. Guidelines for drinking water quality. World Health Organization. Geneva, Switzerland. [ Links ]
Withford, L.A. & G.J. Schumacher. 1973. A manual of freshwater algae. Sparks, Raleigh, North Carolina, USA. [ Links ]
Wood, R.B. & J.F. Talling. 1988. Chemical and algal relationships in a salinity series of Ethiopian inland waters. Hydrobiologia 158: 29-67. [ Links ]