Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.56 n.4 San José Dec. 2008
Antifeedant activity of botanical crude extracts and their fractions on Bemisia tabaci (Homoptera: Aleyrodidae) adults: II. Sechium pittieri (Cucurbitaceae)
Guillermo Flores1, Luko Hilje2, Gerardo A. Mora3 & Manuel Carballo2
1. Ministry of Agriculture and Animal Husbandry (MAG). Cartago, Costa Rica.
2. Department of Agriculture and Agroforestry. Tropical Agricultural Research and Higher Education Center (CATIE). Turrialba, Costa Rica. Fax (506) 558-2043; lhilje@catie.ac.cr
3. Associate Researcher. Natural Products Research Center (CIPRONA). Universidad de Costa Rica. San José, Costa Rica.
Abstract: Bemisia tabaci is a key pest of vegetables and other crops worldwide, but it is a particularly serious problem in the tropics, due to its ability to transmit several types of viruses, especially begomoviruses (Geminiviridae). Therefore, a preventive approach to deal with viral epidemics may be the deployment of repellents or phagodeterrents at earlier stages of plant development (critical period). Thus, the crude extract and four fractions thereof (water, water: methanol, methanol, and diethyl ether) of wild "tacaco" (Sechium pittieri, Cucurbitaceae), were tested for phagodeterrence to B. tabaci adults under greenhouse conditions, on tomato plants, in Costa Rica. Both restricted-choice and unrestricted-choice experiments showed that the crude extract as well as some fractions exert such effect on the insect. In the former (in sleeve cages), fractions caused deterrence at doses as low as 0.1% (ether) and 0.5% (water and water: methanol), with the methanol fraction showing no activity. However, in the latter (plants exposed in a greenhouse) no one of the fractions performed well, suggesting that the deterrent principles somehow decomposed under the experimental conditions. Rev. Biol. Trop. 56 (4): 2115-2129. Epub 2008 December 12.
Key words: Bemisia tabaci, plant extracts, wild "tacaco", Sechium pittieri, phagodeterrence.
Despite being a cosmopolitan and probably the main agricultural pest worldwide, Bemisia tabaci (Gennadius) is a particularly serious problem in the tropics, due to its ability to transmit a whole gamut of viruses such as carlaviruses, closteroviruses, criniviruses, luteoviruses, nepoviruses, potyviruses, and begomoviruses, among which the latter predominates (Brown and Bird, 1992; Brown, 1994).
In the case of tomatoes, the main begomovirus (family Geminiviridae), formerly geminiviruses, so far reported for Costa Rica is the Tomato Yellow Mottle virus (ToYMov) (Polston and Anderson, 1997). Its impact on yields depends on plant age at time of infection, so that the critical period of susceptibility for several begomoviruses encompasses about the first 50-60 days after emergence (Franke et al. 1983, Schuster et al. 1996). In Costa Rica, since the action threshold for the vector is very low (0.3 adults/plant) (Hilje 2001), a preventive approach would be desirable, focusing on this critical early period to minimize contact between the vector and the host plant. Therefore, a possible approach could be the application of environmental-friendly repellent or deterrent substances (Hilje 1993, 2001), among which botanical products merit further investigation, as it has been shown that B. tabaci adults can be affected by substances present in some plant extracts (Cubillo et al. 1999; Hilje and Stansly, 2001, Aguiar et al. 2003). One of such plants is wild "tacaco" (Sechium pittieri (Cogn.) C. Jeffrey, Cucurbitaceae), so that the objective of this research was to confirm preliminary results (Hilje and Stansly 2001), as well as to test some of its fractions to gain insight into more specific groups of substances causing phagodeterrence.
Materials and methods
Location: Greenhouse experiments were carried out at CATIE, in Turrialba, within the Caribbean watershed of Costa Rica, at 9° 52 N, 83° 38 W and 640 m.a.s.l., with annual averages of 22 ºC, 2479 mm (rainfall) and 87% RH.
Extract preparation: Plant material was collected from a single location and at the same time, in Orosi (Cartago), Costa Rica, in order to avoid undesirable variability due to geographic or seasonal differences.
Hydroalcoholic extracts were prepared at the Natural Products Research Center (CIPRONA), Universidad de Costa Rica, as follows: fruits, dried in an oven at 40 ºC, were ground and extracted with 70% methanol in a suitable flask for 24 h; the solvent was drained and the residue was treated again with methanol for 24 h. The pooled extracts were filtered through a Whatman No. 4 filter paper, and concentrated at 40 ºC using a rotary evaporator. The final residue was freeze-dried to eliminate any water remaining in the crude extract.
The crude extract was batch-fractionated on the synthetic resin Diaion HP-20 (Mitsubishi Chemical Industry). The resin was washed with portions of water, water: methanol (1:1), methanol, and diethyl ether and then the solvents were reversed. Three consecutive samples of 15.0 g (45.0 g in total) were placed on the column and eluted with 1 L of each of the solvents, starting with water and finishing with diethyl ether. The solvent sequence was reversed after each run, in order to have the column in the initial conditions. The corresponding fractions were pooled and the solvents were evaporated and the residue freeze-dried, if necessary, to give 8.40 g (18.67% of the crude extract) of the aqueous fraction; 5.38 g (11.96% of the crude extract) of the water: methanol fraction; 8.71 g (19.36% of the crude extract) of the methanol fraction, and 0.33 g (0.73% of the crude extract) of the diethyl ether fraction.
The dry weight of each fraction was used to determine its equivalent dose with respect to the weight of the crude extract. A description of the procedure used is shown in the next section.
Both the freeze-dried crude extract and the respective fractions were kept hermetic, refrigerated and in darkness, to avoid their chemical decomposition or contamination by fungi.
Preparation of fraction solutions: Initially the crude extract was evaluated for biological activity at 0.1, 0.5, 1.0, and 1.5% w/v (weight/volume); so, the fractions were evaluated at the corresponding concentrations, equivalent to those of the crude extract, according to the relative concentration of the fraction in the crude extract. In the text, we refer to the concentrations of the fractions as 0.1, 0.5, 1.0, and 1.5% w/v but these are equivalent concentrations. The real fraction concentration is different, as described below. To prepare the solutions, the amount required for each fraction, in accordance to the yield of the fractioning process, was weighed and dissolved into 100 ml of the respective solvent.
Thus, 32 treatments were obtained, corresponding to four concentrations (0.1, 0.5, 1.0 and 1.5% w/v) of each of the four fractions. Therefore, such treatments (w/v) were: 0.01867%, 0.09335%, 0.1867% and 0.2800% (water fraction), 0.01195%, 0.05975%, 0.1195% and 0.17925% (methanol: water fraction), 0.01935%, 0.09675%, 0.1935% and 0.29025% (methanol fraction), and 0.00073%, 0.00365%, 0.0073% and 0.01095% (diethyl ether fraction).
A stock solution was prepared at the highest concentration; other solutions were prepared by dilution. In the case of the fractions with organic solvents (methanol and diethyl ether) it was necessary to change the solvents, because of phytotoxicity. It was found that, when methanol or diethyl ether was used, necrosis of the leaves occurred. Thus, the methanol fraction was dissolved in 20% methanol in water, and the diethyl ether fraction was dissolved in 20% acetone in water. These mixtures were found to be the least toxic, most compatible solvents for each fraction. The solvent for the water: methanol fraction was maintained as 50% methanol in water.
All dissolutions were prepared just before the experiment was set up, with distilled water as a carrier.
Experiments: Both the crude extract and four fractions (water, water: methanol, methanol, and diethyl ether) were assessed for their feeding deterrence and oviposition response on B. tabaci adults (belonging to biotype A). Insects were taken from a colony kept in a greenhouse at CATIE, and reared on a mixture of eggplant and tomato plants.
The crude extract and four fractions were tested by means of restricted-choice experiments (in a closed space), whereas only the most promising doses of each fraction were compared through an unrestricted-choice experiment (in an open space).
Restricted-choice experiments: In this case, adult whiteflies were exposed to a treated and an untreated potted tomato plant within a sleeve cage, so that they had to choose between two alternatives or remain flying and resting on the cage´s walls.
A first experiment was run with a crude freeze-dried extract, aiming at confirming previous results by Hilje and Stansly (2001), as well as determining the minimum dose at which deterrence could be detected, to use it as reference for further experiments. Then an experiment was run for each of the four fractions, tested at the equivalent of such a dose along with the respective solvents, as controls, to rule out any effects of them on B. tabaci adults.
For these restricted-choice experiments, two pots, each one with a tomato plant, were placed in a sleeve cage. One of the plants was sprayed with either the crude extract, each one of its fractions or the control solvents, whereas the other was treated with distilled water. Experiments were run using a completely randomized design (as environmental conditions were quite homogenous inside the greenhouse where cages were located), with four replicates, the experimental unit being represented by each potted plant receiving a given treatment.
The crude extract, as well as each one of its fractions were tested in individual experiments at the doses of 0.1, 0.5, 1.0 and 1.5% w/v, or their equivalent (see above). They were compared with an insecticide (endosulfan), a control treatment (distilled water), and Aceite Agrícola 81 SC, which is an agricultural oil that strongly deters whitefly adults (Hilje, unpubl.), and the emulsifier Citowett. Endosulfan (Thiodan 35% CE; Hoechst, Germany) (350 g a.i./L) was used at its recommended commercial dose (2.5 mL/L water), as was Aceite Agrícola 81 SC (1.5% v/v) (Mobil Oil Corp., Memphis, Tennessee). Citowett (BASF, Germany) was added to all treatments at the same dosage (0.25 mL/L, i.e., 0.025%, which is half of what is commercially recommended) to improve adherence to foliage.
In all cases, treatments were applied to tomato plants (var. Hayslip) with three true-leaves. This was done with a Devilbiss 15 hand-sprayer, with an adjustable tip (The Devilbiss, Somerset, Pennsylvania), which was connected to an air pump, under a constant pressure (10 kg/cm2). Plants from each treatment were sprayed separately with each solution in an isolated room, for which they were placed on a table and thoroughly sprayed to run-off.
Treated plants were introduced into sleeve cages (30 cm x 30 cm x 45 cm, with walls made of wood, a fine net, and glass) 30 min after being sprayed. Fifty B. tabaci adults were collected from the colony with a hand aspirator and released into each cage. Release took place between 8:30-10:30 h; 2 min later, the aspirator flask was checked, in order to count and release additional adults to replace those which had died due to handling.
Unrestricted choice experiments: In this case, tomato plants were exposed to flying whiteflies inside a greenhouse where their colony is maintained, so that they had no restrictions in choosing where to settle.
These experiments were performed for the lowest doses causing deterrence in the previous experiments: diethyl ether (0.1%), water (0.5%) and water: methanol (0.5%). They were compared with the same controls, except for endosulfan, in order not to disturb colony development.
Potted tomato plants (treated as in the restricted-choice experiments) were placed on a bench and arranged in a randomized complete block design, with four replicates. The experimental unit was represented by each potted plant receiving a given treatment.
Analysis: For the restricted-choice experiments, counts were made on the foliage of the whole plant. The criterion to appraise feeding deterrence was the number of adults on each plant at 48 h, in combination with the number of those surviving within that interval. Oviposition response was appraised by counting the number of eggs laid over 48 h on one leaf per plant; eggs were counted under a stereo-microscope.
Mortality was determined by counting the total number of living adults in each cage (on both plants) at 48 h, and subtracting the sum from 50; in this case, the experimental unit was represented by each cage (both two potted plants), as adult whiteflies are very mobile and would be expected to make contact with both plants and get killed if substances present in a given treatments are toxic.
For the unrestricted-choice experiments counts were also made on the foliage of the whole plant 1, 2, 8 and 15 days after plant treatment. Nymph numbers (instars I and II) were counted at the end of the experiment by counting a 1 cm2 square on one of the leaves, under a stereo-microscope.
To determine feeding deterrence and oviposition response for both types of experiments, the number of either adults or eggs present on each plant within each cage were subjected to analysis of variance (ANOvA), and mean values were compared using a Duncan´s test (SAS Institute, 1985), at a significance level of α = 0.05. For determining mortality, the total numbers of living adults in each cage were also subjected to ANOvA and mean values were compared using a Duncan´s test.
Since the coefficients of variation were high (> 20%) for data resulting from the unrestricted-choice experiments, they were transformed by the logarithmic method, in order to stabilize variance and meet the assumptions underlying ANOvA. In all cases, such assumptions were verified.
Results
Restricted-choice experiments: Because of the nature of the experiments, comparisons about feeding deterrence and oviposition response were valid only between both plants within each cage, and not between cages. However, for mortality, comparisons were valid between cages.
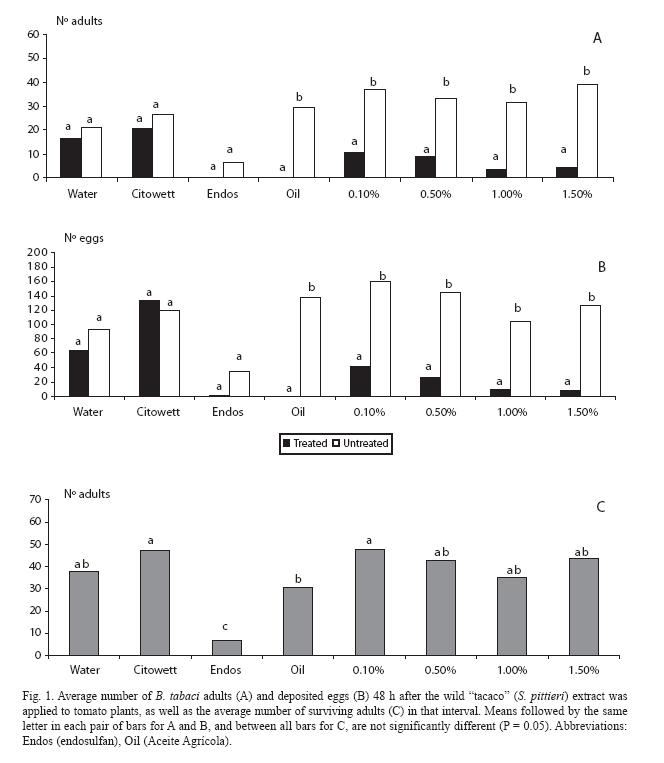
For the freeze-dried crude extract, there were very low numbers of adults on the treated plant for all doses (p < 0.05), as it also occurred with the mineral oil (Fig. 1A). In the extreme treatments (water control and endosulfan), as well as in the Citowett treatment, there was no difference between both plants (p > 0.05), but in the endosulfan treatment the numbers strongly decreased in both plants. Similar trends were observed for the oviposition response (Fig. 1B).
Regarding mortality, plants treated with endosulfan had the lowest number of surviving adults, in contrast with all the crude extract treatments (Fig. 1C), which did not differed among them (p > 0.05) nor with the water control.
In the following experiment, the lowest dose of the crude extract causing deterrence (0.5%) was selected as a baseline reference.
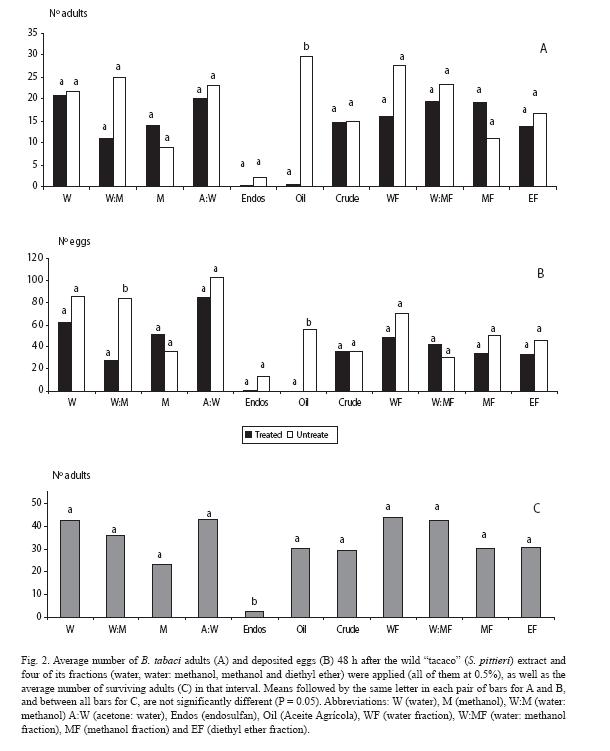
The number of adults did not differ (p > 0.05) for any of the doses, with differences detected only for the mineral oil (Fig. 2A). Trends held for the oviposition response, except for one of the solvents (methanol: water) (Fig. 2B). As for mortality, there was a sharp contrast between endosulfan and the rest of treatments (Fig. 2C).
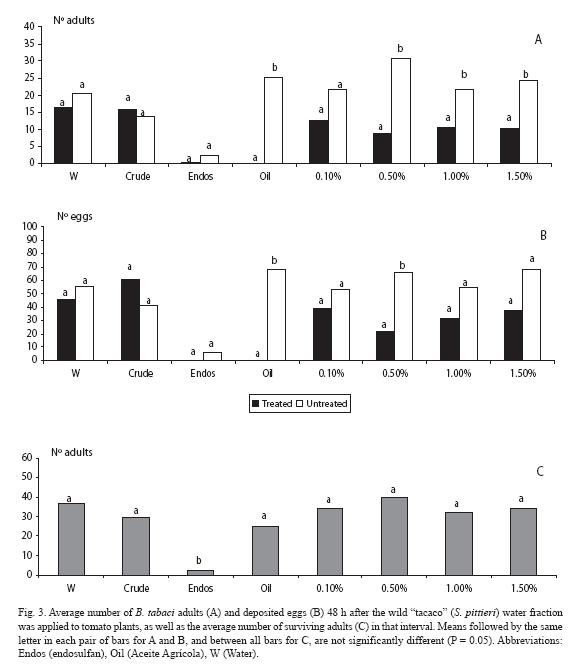
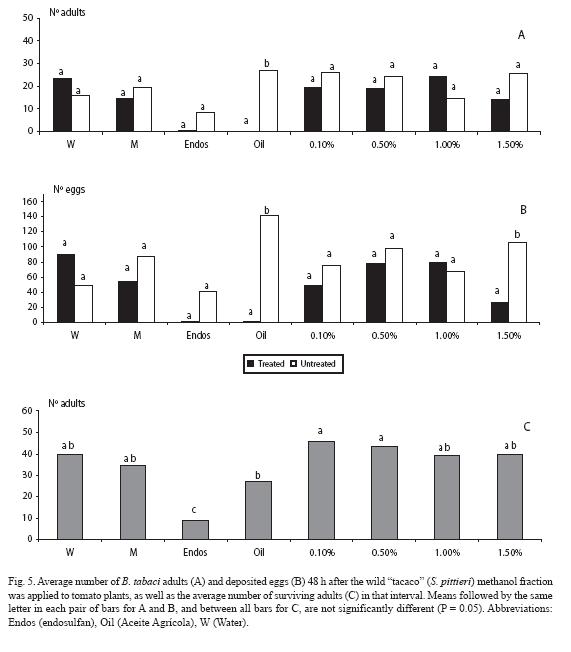
When analyzing the water fraction, fewer adults landed (p < 0.05) at the three highest wild "tacaco" doses (0.5, 1.0 and 1.5%), as well as in the mineral oil (Fig. 3A). Trends held for the oviposition response, but at the two highest doses there were no differences (p > 0.05) (Fig. 3B), although there were fewer eggs in those plants treated with these fractions. Concerning mortality, endosulfan clearly contrasted with the rest of treatments (Fig. 3C).
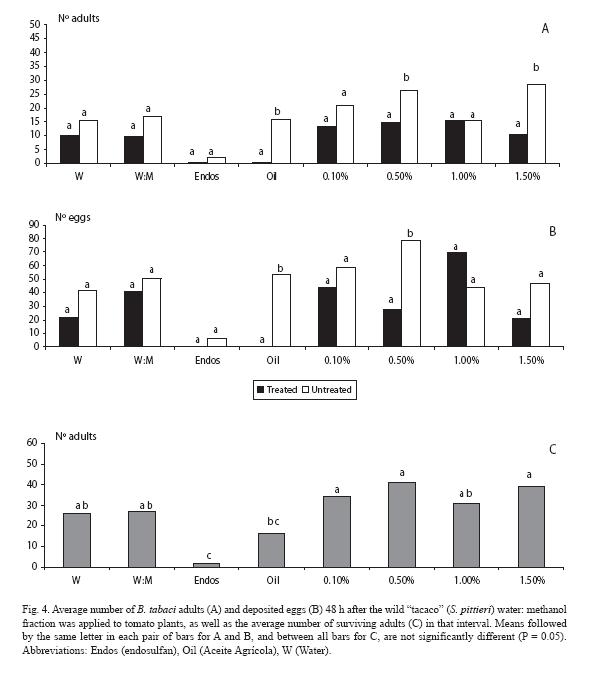
For the water: methanol fraction, there were fewer adults landed (p < 0.05) at the two wild "tacaco" doses (0.5 and 1.5%), as well as with the mineral oil (Fig. 4A). Trends held for the oviposition response, but at the highest dose there was no difference (p > 0.05), although there were fewer eggs in the plant treated with that fraction (Fig. 4B). As for mortality, endosulfan clearly contrasted with the rest of treatments, except for the mineral oil (Fig. 4C).
For the methanol fraction, numbers of adults did not differ (p > 0.05) for any of the doses, with differences detected only for the mineral oil (Fig. 5A). Trends held for the oviposition response, except for the highest dose, at which there were differences (p < 0.05) (Fig. 5B). Regarding mortality, endosulfan boldly contrasted with the rest of treatments (Fig. 5C).
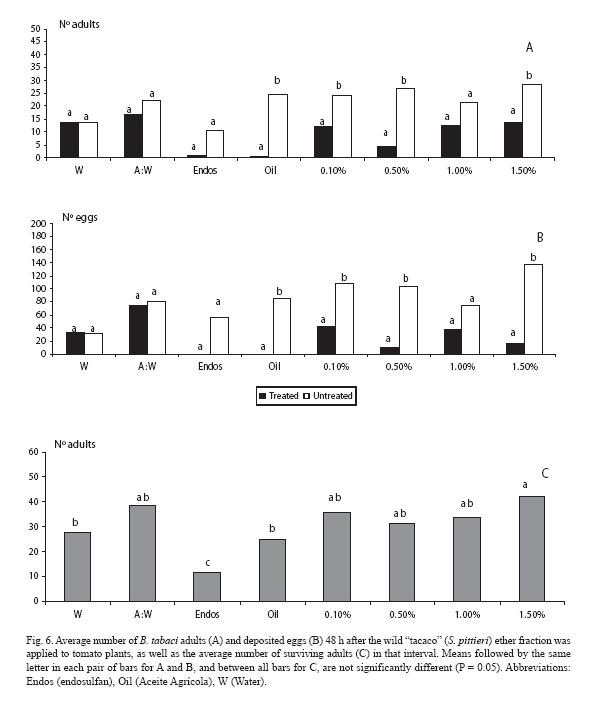
For the diethyl ether fraction, fewer adults landed (p < 0.05) only at all of the doses except at 1.0%, as it also occurred with the mineral oil (Fig. 6A). Similar trends were observed for the oviposition response (Fig. 6B). As for mortality, endosulfan neatly contrasted with the rest of treatments (Fig. 6C).
Unrestricted-choice experiments: In this case, comparisons about feeding deterrence and nymphal development were valid between treatments, as all of them were equally exposed to the same group of adult whiteflies.
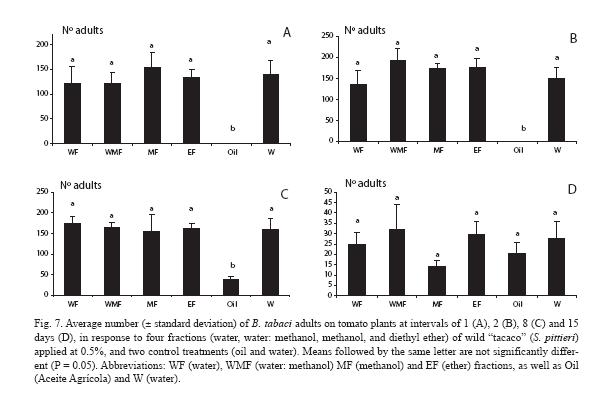
For the first count, 24 h after the fractions were sprayed, there was a very strong contrast between all treatments and the mineral oil, which yielded no adults (Fig. 7A). Such trends exactly held a day later, with adult numbers increasing for all treatments, whereas only one adult was counted in the mineral oil treatment (Fig. 7B). A week later (Fig. 7C) the same pattern was observed, but numbers increased or either increased or decreased depending on the treatments. Finally, two weeks later there were no differences among treatments (p > 0.05) (Fig. 7D).
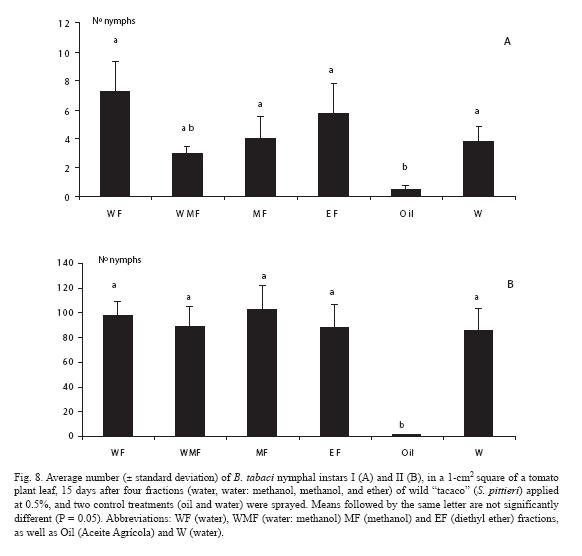
Regarding nymph numbers, they were lowest for the plants treated with the mineral oil, both for instars I (Fig. 8A) and II (Fig. 8B).
Discussion
Theoretical aspects regarding B. tabaci adults ability to react to plant substances (Walker and Gordh 1989) as well as the types of allomones to which they could respond (Warthen and Morgan 1990) and how their effects could be recognized (Walker and Perring 1994) have been discussed elsewhere (Flores et al. 2008).
Our clear-cut findings with the freeze-dried crude extract confirm preliminary results from Hilje and Stansly (2001) regarding the presence of phagodeterrent principles for B. tabaci in wild "tacaco" extracts. The lower number of landed adults on tomato plants after treating them with the extract was an expression of feeding deterrence. It would be expected that once adults land on a treated plant make contact with the deterrent substances, so that over time they tended to gather on the untreated plant.
However, in the experiment at which the lowest doses of the crude extract causing deterrence (0.5%) was compared to the fractions at that same dose, neither the crude extract nor any of the fractions performed well as phagodeterrents. This suggests that something went wrong with the extracts and the fractions, since the opposite occurred when each one of the fractions was tested individually.
Testing of fractions to gain insight into more specific groups of substances responsible for causing phagodeterrence, showed that they induced that effect, which occurred at doses as low as 0.1% (diethyl ether fraction) and 0.5% (water and water: methanol fractions), with no activity from the methanol fraction. In very few cases there were inconsistencies regarding adult responses to some of the control treatments, as well as to lack of the expected dose-dependent responses to the diethyl ether and the water: methanol fractions, as at 1.0% there were no differences although they occurred at 0.5 and 1.5%. They could be explained in terms of a great deal of variability between replicates of each treatment, especially due to adult age and vigor, different sex ratios in each replicate, etc., which were factors that were not controlled.
As for oviposition response, in general fewer eggs were recorded on treated plants, which can be explained by the lower number of females present on them, rather than in terms of oviposition deterrence, although the latter can not be ruled out altogether. Likewise, in the unrestricted-choice experiment, nymph numbers were probably a direct expression of female abundance and egg-laying activity in each treatment.
Concerning mortality, adults in plants treated with either the crude extract or the fractions showed low levels, always sharply contrasting with endosulfan. Adult mortality observed in treated plants, which was always dose-independent, may be attributed either to an indirect effect of strong deterrence, causing heat stress, energy depletion or dehydration (veierov 1996), or to direct toxicity of the crude extract or the fractions to very susceptible or weakened adults.
Chemicals causing the observed effects remain unknown, although probably they are a series of glycosides known as tacacosides, which are very bitter and irritating. Six of these bayogenin saponins have been isolated from fruits and aerial parts of S. pittieri and S. talamancense (Castro et al. 1997). Cucurbitacins, which have several kinds of activities, including toxicity and feeding deterrence (Mabry and Gill 1979), are not found in this plant.
The lack of biological activity of the methanol fraction was surprising, as it has been shown to be the most powerful fraction containing phagodeterrent substances to B. tabaci in concurrent experiments conducted with mother-of-cocoa (Gliricidia sepium, Fabaceae) (Flores et al. 2008a) and bitterwood (Quassia amara, Simaroubaceae) crude extracts and fractions (Flores et al. 2008b). This could be explained considering that the more polar components of this extract are glycosides of bayogenin (saponins) (Castro et al. 1997) and the diethyl ether fraction could contain some of the aglycone bayogenin as a product of partial decomposition (hydrolysis) of the saponins. So, eventually, a further experiment has to be performed to obtain some of the bayogenin itself and test the pure compound on whiteflies.
Despite promising findings in restricted-choice experiments, none of the fractions performed well as in the unrestricted-choice experiment, the same occurring with nymph numbers. None of them did better than the mineral oil, a trend that lasted for a week. This suggests that the deterrent principles decompose under the experimental conditions.
Most of what is known about wild "tacaco" responds to ethnobotanical knowledge but, being very bitter, this is normally a plant which people regard as a weed, in contrast to its congeneric species "tacaco", S. tacaco (= Frantzia tacaco) and "chayote" (S. edule) whose fruits are edible (León 1987). It is a perennial vine that occurs at a very wide altitudinal range (100-2500 m) from Nicaragua to Panama, where it grows on both wild and disturbed habitats, usually near rivers or creeks, and even in flooding areas (Lira 1995). Its fruits, 4-6 cm long and 3-4 cm wide, are green, kind of ovoid or fusiform, very bitter, and can appear all year round.
Wild "tacaco" is one of a myriad of neotropical plants with an untapped potential as sources of active principles against insect pests. In addition to whiteflies their extracts have insecticidal activity on mahogany shootborer, Hypsipyla grandella (Lepidoptera: Pyralidae) larvae (Mancebo et al. 2001), which is a pest of very valuable neotropical wood species, such as mahoganies and cedars. Moreover, bayogenin saponins isolated from fruits and aerial parts of S. pittieri as well as from S. talamancense have bioactive properties with antiproliferative activity (Castro et al. 1997).
Therefore, it would be attractive for the agrichemical industry to get involved into developing commercial deterrents or insecticides derived from tropical plants. By doing so, industry could also contribute to sustainable development through the utilization of a pristine resource from neotropical biodiversity, while benefiting rural dwellers by involving them in different stages of the domestication, planting and harvesting process for such valuable plant species.
Acknowledgments
This paper is a partial result of a CATIEs M.Sc. thesis by the Unidad de Manejo de la Cuenca del Río Reventazón (UMCRE), from the Instituto Costarricense de Electricidad (ICE).
Thanks are due to Arturo Ramírez and Gustavo López (CATIE) for their logistical support, to Juan Carlos Brenes (CIPRONA) for preparing the extracts for the experiments, and to Norman Farnsworth (Chief Editor of NAPRALERT), for granting access to the database.
Resumen
Bemisia tabaci es una plaga clave de hortalizas y otros cultivos, mundialmente, y representa un problema particularmente serio en los trópicos, debido a su habilidad para transmitir varios tipos de virus, especialmente begomovirus (Geminiviridae). Por tanto, un enfoque preventivo para enfrentar las epidemias virales podría consistir en la utilización de sustancias repelentes o disuasivas en las eta-pas tempranas del desarrollo de la planta (período crítico). Así, el extracto crudo y cuatro fracciones (agua, agua: metanol, metanol, y éter dietílico) de tacaco cimarrón (Sechium pittieri, Cucurbitaceae), fueron evaluadas por su posible actividad fagodisuasiva sobre B. tabaci en un invernadero, utilizando plantas de tomate, en Turrialba, Costa Rica. Tanto los experimentos de escogencia restringida como los de escogencia irrestricta mostraron dicha actividad en el extracto crudo y en algunas de sus fracciones. En el primer tipo de experimento (en jaulas de manga), las fracciones causaron fagodisuasión a dosis tan bajas como 0.1% (éter dietílico) y 0.5% (agua y agua: metanol), mientras que la fracción metanólica no la causó. Sin embargo, en los otros experimentos (plantas expuestas a B. tabaci dentro de un invernadero) ninguna de las fracciones funcionó bien, lo cual sugiere que los principios disuasivos se disiparon bajo esas condiciones experimentales.
Palabras clave: Bemisia tabaci, extractos vegetales, wild "tacaco", Sechium pittieri, fagodisuasión.
Recibido 16-II-2007. Corregido 08-V-2008. Aceptado 12-V-2008.
References
Aguiar, A., D.C. Kass, G.A. Mora & L. Hilje. 2003. Fagodisuasión de tres extractos vegetales sobre los adultos de Bemisia tabaci. Man. Integr. Plagas y Agroecol. (Costa Rica) 68: 62-70. [ Links ]
Brown, J.K. 1994. Current status of Bemisia tabaci as a plant pest and virus vector in agroecosystems worldwide, FAO Plant Prot. Bull. 42: 3-32. [ Links ]
Brown, J.K. & J. Bird. 1992. Whitefly-transmitted geminiviruses in the Americas and the Caribbean Basin: Past and present. Plant Dis. 76: 220-225. [ Links ]
Castro, V., E. Ramírez, G. Mora, Y. Iwase, T. Nagao, H. Okabe, H. Matsunaga, M. Katano & M. Mori. 1997. Structures and antiproliferative activity of saponins from Sechium pittieri and S. talamancense. Chem. Pharm. Bull. 45: 349-358. [ Links ]
Flores, G., L. Hilje, G.A. Mora & M. Carballo. 2008a. Antifeedant activity of botanical crude extracts and their fractions on Bemisia tabaci (Homoptera: Aleyrodidae) adults: I. Gliricidia sepium (Fabaceae). Rev. Biol. Trop. 56 (4): 2099-2113. [ Links ]
Flores, G., L. Hilje, G.A. Mora & M. Carballo. 2008b. Antifeedant activity of botanical crude extracts and their fractions on Bemisia tabaci (Homoptera: Aleyrodidae) adults: III. Quassia amara (Simaroubaceae). Rev. Biol. Trop. 56 (4): 2131-2146. [ Links ]
Franke, G., L. van Balen & E. Debrot. 1983. Efecto de la época de infección por el mosaico amarillo sobre el rendimiento del tomate. Rev. Fac. Agron. Univ. Zulia (venezuela) 6: 741-743. [ Links ]
Greathead A.H. 1986. Host plants. p. 17-26. In M.J.W. Cock (ed.). Bemisia tabaci-A Literature Survey. CAB Int. Inst. of Biol. Control, Silwood Park, UK. [ Links ]
Hilje, L. 1993. Un esquema conceptual para el manejo integrado de la mosca blanca (Bemisia tabaci) en el cultivo del tomate. Man. Integr. Plagas (Costa Rica) 29: 51-57. [ Links ]
Hilje, L. 2001. Avances hacia el manejo sostenible del complejo Bemisia tabaci-geminivirus en tomate, en Costa Rica. Man. Integr. Plagas (Costa Rica) 61: 70-81. [ Links ]
Hilje, L. & P.A. Stansly. 2001. Development of crop associations for managing geminiviruses vectored by whiteflies in tomatoes. Final Report. U.S. Dept. Agr. (USDA). CATIE. Turrialba, Costa Rica. 134 p. [ Links ]
León, J. 1987. Botánica de los cultivos tropicales. IICA, Costa Rica. 445 p. [ Links ]
Lira, R. 1995. Estudios taxonómicos y ecogeográficos de las Cucurbitaceae latinoamericanas de importancia económica. pp. 163-165. In Systematic and Ecogeographic Studies on Crop Genepools, No. 9. International Plant Genetic Resources Institute (IPGRI), Rome. [ Links ]
Mabry, T.M. & J.E. Gill. 1979. Sesquiterpenene lactones and other terpenoids. pp. 501-537. In G.A. Rosenthal and D.H. Janzen (eds.), Herbivores: Their interaction with secondary plant metabolites. Academic, New York. [ Links ]
Mancebo, F., L. Hilje, G.A. Mora & R. Salazar. 2001. Biological activity of Ruta chalepensis (Rutaceae) and Sechium pittieri (Cucurbitaceae) extracts on Hypsipyla grandella (Lepidoptera: Pyralidae) larvae. Rev. Biol. Trop. 49: 501-508. [ Links ]
Perring, T.M. 2001. The Bemisia tabaci species concept. Crop Prot. 20: 725-737. [ Links ]
Polston, J.E. & P.K. Anderson. 1997. The emergence of whitefly-transmitted geminiviruses in tomato in the Western Hemisphere. Plant Dis. 81: 1358-1369. [ Links ]
SAS Institute, 1985. SAS User Guide: Statistics, version 6 Edition, SAS Institute Inc, Cary, North Carolina. 956 p. [ Links ]
Schuster, D.J., P.A. Stansly & J.E. Polston. 1996. Expressions of plant damage of Bemisia. p. 153165. In D. Gerling & R.T. Mayer (eds.). Bemisia 1995: Taxonomy, Biology, Damage, Control and Management. Intercept, Andover, UK. [ Links ]
Veierov, D., 1996. Physically and behaviorally active formulations for control of Bemisia. p. 557-576. In D. Gerling & R.T. Mayer (eds.). Bemisia 1995: Taxonomy, Biology, Damage, Control and Management. Intercept, Andover, UK.
Warthen, J.D. & E.D. Morgan. 1990. Insect feeding deterrents. p. 23-134. In E.D. Morgan & N.B. Mandava (eds.). CRC Handbook of Natural Pesticides, vol. 6: Insect Attractants and Repellents. CRC Press, Boca Raton, Florida. [ Links ]