Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.56 n.4 San José Dec. 2008
Linear Alkylbenzene Sulfonate tolerance in bacteria isolated from sediment of tropical water bodies polluted with detergents
Kehinde I.T. Eniola & Albert B. Olayemi
1. Environmental and Public Health Research (EPHR) Laboratory,
2. Department of Microbiology, University of Ilorin, P.M.B. 1515, Ilorin, Kwara State, Nigeria; kennyeniola@yahoo.com, ephr_lab@yahoo.co.uk
Abstract: The discharge of untreated detergent-bearing waste introduces linear alkylbenzene sulfonates (LAS) to the aquatic environment. The surfactant persists in some streams and rivers in Nigeria, some is adsorbed to suspended materials and end in the sediment of the receiving water bodies. In this study, bacteria isolated from sediments of some tropical detergent-effluent-polluted streams were tested for tolerance to LAS using the media dilution technique. LAS-tolerance was indicated by growth of the bacteria in the presence of the surfactant. The pH, concentrations of surfactant, population of heterotrophic bacteria and population of LAS-tolerant bacteria in the sediments were determined. A direct relationship (r= 0.9124) was found between the alkaline conditions (pH= 8.2-12.0) and high surfactant concentrations (45-132 mg/g) in the sediment. The sediments harboured a high population and a wide variety of bacteria; the populations of viable heterotrophic bacteria (vHB: 2.9×105 to 1.2×107 cfu/g) and LAS tolerant bacteria (LTB: 1.5×104 to 1.2×106 cfu/g) had a direct relationship (r= 0.9500). An inverse relationship resulted between each of them and the concentration of surfactant in the sediment, rvHB/ LAS = -0.9303 and rLTB/ LAS = -0.9143, respectively. Twelve bacteria species were isolated from the sediment: Alcaligenes odorans, Bacillus subtilis, Burkholderia cepacia, Citrobacter freundii, Citrobacter diversus, Escherichia coli, Micrococcus luteus, Micrococcus albus, Pseudomonas putida, Pseudomonas stutzeri, Staphylococcus aureus and Streptococcus faecalis. Most of them were adapted to the surfactant with their maximum acceptable concentrations ranging between 0.03 and >1.0% (w/v). The sediments could serve as source of adapted organisms which can be used in bio-treatment of LAS-bearing waste. Rev. Biol. Trop. 56 (4):7-15. Epub 2008 December 12.
Key word: LAS, surfactant, tolerance, detergent, effluent.
Linear alkylbenzene sulfonate (LAS) is an amphiphilic surfactant with a high solid-adsortion coefficient and readily adsorbs onto colloidal surfaces, suspended particles and solid substrates in water (Painter and Zabel 1989). It is widely used in the detergent Industry and gets into the environment via a variety of sources (Larson et al. 1995, CLER 1996, Takada et al. 1997). LAS is removed from the environment by a variety of processes; however, biodegradation and sorption to suspended materials are reported to be the main mechanisms (WHO large 1996, Takada et al. 1997, Schulze and Tiehm 2004). It has been estimated that between 20 to 50% of LAS is removed from water by adsorption unto solids. The adsorption of the surfactant is influenced by the physicochemical properties of the sediment, by those of the surfactant and by those of the solution with which it is in contact (Dentel et al. 1993).
Cavalli et al. (2000) reported that the suspended solids in the water column are the major constituents that form the sediment along the water course. LAS that adsorbed to suspended particles would likely end up in the sediment if the supporting particles are or have a higher density than water. However, adsorbed LAS may occur in suspension if the particles are sufficiently small. LAS can also be associated with the cell membrane in living organisms including bacteria (Folker and Landner 2000). The settling of suspended solids to which LAS has adsorbed and the slow pace of degradation/ persistence of LAS in sediment (Dentel et al. 1993, Jacques Whitford Limited 2006) would result in persistence of LAS in sediments.
The sediment of water bodies have been shown to contain a higher concentration of LAS than the overlaying water (de Ferrer et al. 1996, Olayemi et al. 2003). Bacteria resident in the sediments will therefore be exposed to higher concentrations of the surfactant for a longer time than bacteria that are in the overlying water column. This kind of exposure could be crucial as indicated by Takada et al. (1997) in explaining the accelerated degradation of LAS when sediments from sites previously exposed to LAS were added to their preparation. Brock et al. (1994) had pointed out that surfaces were important because they allow attachment of organisms, accumulate nutrients and may even serve as nutrient themselves.
A study by Eniola and Olayemi (2003) showed that there was a difference in the composition of bacterial flora of water and sediment of some detergent polluted water bodies in Ilorin, Nigeria. This study examines the LAS-tolerance of bacteria isolated from sediments from some detergent-effluent-polluted streams in Ilorin Nigerian. The tolerances will be assessed in terms of ability or failure to grow in the presence of 0.1% (w/v) of the surfactant, using the media dilution technique.
Materials and methods
Study Area/ Sampling points
The study was undertaken using the major river Asa River and two of its tributary streams: Okun and Osere streams in Ilorin, North Central Nigeria (8o 28N, 4o 38E to 8o 31N, 4o 40E). The river, through the tributaries, regularly receives untreated detergent-bearing-effluent from a soap and detergent industry as well as residential buildings. Samples of sediment were collected from two points along the course of each of the water body Samples were collected by digging about 1cm into the sediment using a sterile spatula. The samples were collected into sterile sampling bottles. Samples were collected in triplicate and taken to the laboratory for analysis in an improvised ice chest.
Physicochemical Analysis of Samples
A suspension of the sediment was prepared by weighing 2g of the sediment into 20ml of sterile distilled water and agitated gently to avoid forming. The pH of the suspension was determined using a pH meter. The concentrations of surfactant were determined using Hyamine titration method (GSD 2002 unpublished). Suspension of the sediment was filtered using Whatmann filter paper (12.5cm) and 10ml of the filterate was measured into a conical flask and 5ml of dimedium bromide indicator, 15ml of chloroform, 20ml of distilled water and 2 drops of phenolphtalein indicator were added to it. The mixture was titrated against 0.004N Hyamine solution. The end point was indicated by colour changes from an initial pink to purple then blue. Concentration of surfactant in the sediment was calculated from the titre.
Bacteriological Analysis
The populations of heterotrophic bacteria in the sediments were enumerated using pour plate method (Eniola and Olayemi 1999). Tenfold serial dilutions of the suspension of the sediment were prepared by pipeting 1ml of the sediment suspension into 9ml of sterile diluent. This was repeated to get the 7th dilution and 1ml each of the sixth and seventh dilutions were measured into separate sterile Petri dish. Cool molten nutrient agar was then poured into the plates, swirled to mix the content properly and then allowed to set. The plates were incubated at 37ºC for 48 hours. The populations of bacteria that could tolerate 0.1% (w/v) of LAS were also enumerated by plating another 1ml of the seventh dilution on nutrient agar into which LAS was incorporated. The isolates were purified, characterized and identified according to Bergeys Manual of Determinative Bacteriology (Buchanan and Gibbons 1974).
The bacteria isolated were tested for tolerance to LAS using media dilution method. Nutrient broth containing varying concentrations (0.03, 0.05, 0.07, 0.09, 0.1 %) of LAS were prepared and inoculated with standardized pure cultures of the isolates. The tubes were incubated at 37ºC for 48 hours. Un-inoculated tubes of the broth were incubated to serve as control. Data obtained were subjected to statistical analysis using the Analysis of variance and Pearson product-moment correlation test.
Results
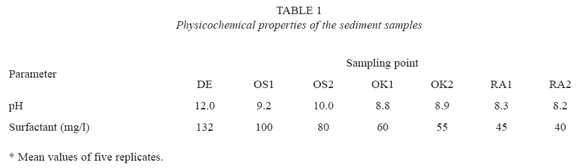
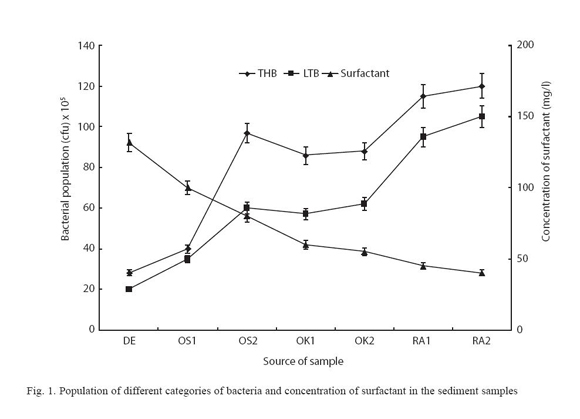
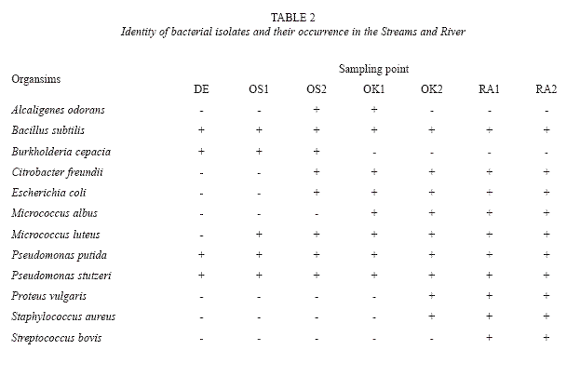
The sediment samples were found to generally be alkaline in nature; with pH ranging from pH 8.2 to pH 12. The surfactant concentration were generally high ranging from 45mg/g to 132mg/g. The pH and concentrations of surfactants in the samples are shown Table 1. The populations of viable heterotrophic bacteria in the samples were generally high. It ranged between 2.9× 105 and 1.2× 107 cfu/g. The population of bacteria that could tolerate 0.1% of LAS was also generally high. Their population ranged between 1.5× 104 and 1.2× 106 cfu/g. The populations of the viable heterotrophic bacteria and bacteria that could tolerate 0.1% of LAS are shown on Figure 1. Twelve bacterial isolates were identified; their identity of the organisms and distribution of the bacteria isolated are shown on Table 2. The bacteria showed varying degree of tolerance to LAS; the highest concentration of LAS that each of them could tolerate are shown on Table 3. Statistical analysis showed that a direct relation existed between the pH and concentration of surfactant (r= 0.9124) and between the populations of heterotrophic bacteria and LTB (r= 0.9500). On the other hand, an inverse relationship existed between populations of heterotrophic bacteria and surfactant concentration (r= -0.9303) and between LTB and surfactant concentration (r = -0.9143)
Discussion
The sediments were consistently alkaline in nature which is attributable to the presence of the surfactant. The surfactant, a sodium salt is alkaline in nature (WHO 1996). Statistical analysis also showed that a direct relationship exited between the concentrations of surfactant in the sediments and their corresponding pH (r= 0.9144). The high concentrations of surfactant are likely to be consequences of discharge of untreated or poorly treated effluents. The concentrations were higher than those reported for rivers in United States (Rapid Creek: 190 mg/kg, Mississippi River 0.01 to 0.95 mg/ kg) and Italy (Tiber River: 1.8 mg/kg). This is buttressed by the submission of OECD SID (2005) that LAS concentrations in sediment below poorly functioning treatment plants or treatment plants having only primary treatment may be higher.
The high concentration of surfactant in the sediment samples could have also resulted from gradual accumulation, at the bottom, of suspended materials to which the surfactant had adsorbed.This is consistent with the assertion of Cavalli et al. (2000) that suspended solids in the water column form the sediment along the water course. Dentel et al. (1993) estimated that 20 to 50% of LAS in effluent is adsorbed to solids due to the high adsorption coefficient of the compound. The concentration of LAS in the sediments is however not uniformly spread, Leon et al. (1999) reported that sediment concentration of LAS decreases with depth; but the decrease was not significant in the first few centimeters due to a thin oxic layer. This is attributable to the fact that degradation of LAS proceeds faster in oxic environments and proceeds at very slow rate, if at all, in anoxic conditions. The slow rate of LAS degradation in sediment allows the accumulation of surfactant in the sediment.
The generally high populations of viable heterotrophic bacteria encountered are typical of water bodies that receive organic pollutants. Similar high populations of heterotrophic bacteria have being reported by Olayemi (1994). The populations of bacteria in the sediments were higher than populations reported for the water bodies by Eniola and Olayemi (1999). A study by Olayemi et al. (2003) showed that the sediment of the water bodies haboured a higher population of heterotrophic bacteria than the water column. The high population of bacteria showing ability to grow in the presence of up to 0.1% of LAS (LAS-tolerant bacteria- LTB) is attributable to the ability of bacteria to be reoriented in response to environmental change and to available nutrient. This is consistent with the submission of some previous workers (Hardman 1989, Takada et al. 1997).
The observed variation in degree of LAS-tolerance showed by the bacteria is consistent with the report of Folker and Lander (2000). The tolerance pattern is also consistent with reports which showed that Pseudomonas putida showed different Ecotoxicity concentration (EC50) depending on the exposure (contact) time and concentration. variations were also reported in the toxicity effect on organisms exposed to LAS in sediment (OECD SID, 2005). Members of Enterobacteriaceae have been observed to show widely varying degree of tolerance to surfactant; but high surfactant tolerance was not uniform to them (Krammer et al. 1980).
Micrococcus luteus and a species of Pseudomonas isolated from the water column were identified as capable utilizing of detergent surfactant (Eniola and Olayemi 2002). A. odorans, Micrococcus luteus and Pseudomonas putida have being found to be able to initiate primary degradation of LAS (Eniola, 2005). The ability of these organisms to tolerate high concentrations of LAS makes them important in removal of LAS from water containing high concentration of the surfactant like untreated effluents. The ability of the organisms to withstand high concentration of the surfactant is likely to have resulted from their reorientation/ adaptation due to exposure to the surfactant in effluents as suggested by Takada et al. (1997).
This study clearly demonstrates that the sediment bacteria varied in their response to LAS. This difference in response could be used to monitor levels of surfactant pollution in water. Some of the organisms could also be used in biotreatment of LAS-containing waste water.
Resumen
La descarga de desechos que contienen detergentes liberan sulfonatos de alquibenceno lineal (LAS) al ambiente acuático. El tensoactivador persiste en algunos arroyos y ríos de Nigeria, en parte es absorbido por materiales en suspensión y termina entre los sedimentos de los cuerpos de agua receptores. En este estudio, bacterias aisladas de los sedimentos de algunos arroyos tropicales que reciben efluentes contaminados con detergentes, fueron analizadas para determinar su tolerancia a los LAS, utilizando la técnica de dilusión del medio. Las bacterias se consideraron tolerantes a los LAS cuando continuaron creciendo aún en presencia del tensoactivador. En los sedimentos también se determinó acidez (pH), concentración de tensoactivador, poblaciones de bacterias heterotróficas y de bacterias tolerantes a los LAS. Se encontró una relación directa (r= 0.9124) entre condiciones alcalinas (pH= 8.2-12.0) y concentraciones altas de tensoactivador (45-132 mg/g) en los sedimentos. Además, los sedimentos mostraron albergar a una población grande y variada de bacterias; las poblaciones de bacterias heterotróficas (VHB: 2.9×105-1.2×107 cfu/g) y bacterias tolerantes a los LAS (LTB: 1.5×104 -1.2×106 cfu/g), mostraron una relación directa (r= 0.9500). Por otra parte, una relación inversa se encontró entre cada una de ellas y la concentración de tensoactivador en los sedimentos, rvHB/ LAS = -0.9303 y rLTB/ LAS = -0.9143 respectivamente. Doce especies de bacterias fueron aisladas de los sedimentos: Alcaligenes odorans, Bacillus subtilis, Burkholderia cepacia, Citrobacter freundii, Citrobacter diversus, Escherichia coli, Micrococcus luteus, Micrococcus albus, Pseudomonas putida, Pseudomonas stutzeri, Staphylococcus aureus y Streptococcus faecalis. La mayoría de esas especies muestra adaptaciones al tensoactividor, siempre que éste se encuentre en concentraciones entre 0.03 y 1.0% (w/v). Los sedimentos pueden servir como una fuente de organismos que pueden ser utilizados en el bio-tratamiento de desechos que contengan LAS.
Palabras clave: LAS, tensoactivador, tolerancia, detergentes, efluentes.
Received 24-VIII-2007. Corrected 30-VI-2008. Accepted 31-VII-2008
References
Brock, T., D. Madigan, M. T. Martinko, J. M. & J. Parker 1994. Biology of microorganism (6th editon). Prentice Hall, Englewood Cliffs, Nueva Jersey, USA. [ Links ]
Buchanan, K. E. & N. E. Gibbons. 1974. Bergeys manual of determinative bacteriology (8th edition). The Williams and Wilkins co. Baltimore, Maryland, USA [ Links ]
Cavalli, L., Cassani, G. Pravettoni, S., Nucci, G. LAzzarin,M. Zatta, A. and L. vigano (2000). Surfactant in Sediments. CLER Review 6: 100- 111. [ Links ]
De Ferrer, J., A. Moreno, M.T. vaquero, & L. Comellas. 1996. The fate of LAS in untreated sewage and high sludge soils. CLER Review. 2: 28-37. [ Links ]
Dentel, S. K., H. E. Allen, C. Srinivasarao, & J. Divincenzo. 1993. Effects of surfactants on sludge dewatering and pollutant fate. Third year completion report project number 06. Water Resources Center. University of Delaware. Delaware, USA. [ Links ]
Eniola, K.I.T. 2005. Biodegradability studies on detergent surfactant in effluent using resident bacteria of detergent-polluted freshwater bodies in Ilorin, Nigeria. Ph.D Thesis submitted to Department of Microbioogy, University of Ilorin, Kwara, Nigeria. [ Links ]
Eniola, K.I.T. & A.B. Olayemi. 1999. Impact of effluent from a detergent producing plant on some water bodies in Ilorin. Internat. J. Environ. Health Res. 9: 335-340. [ Links ]
Eniola, K.I.T. & A.B. Olayemi. 2002. Some aspects of bacterial-detergent interaction in freshwater environments. Biosc. Res. Comm. 14: 645- 649 [ Links ]
Folker, J. & L. Landner. 2000. Risk assessment of LAS in sewage sludge and soil. Report to Kemisk- Tekniska Loverantorbundent (KTF), Stockholm, AF- Miljoforskar Gruppen Stockholm- Sweden. [ Links ]
Hardman, D.J. 1989. Microbial control of environmental pollution: The use of genetic techniques to engineer organisms with novel catalytic capabilities. In Environmental Biotechnology. Forster, C.F. and D.A. John Wase (eds). Ellis Horwood, London, England. [ Links ]
Jacques Whitford Limited. 2006. vancouver Island Transmission Reinforcement Project Technical Data Report: Potential Effects of Alkylbenzene Release to the Marine Environment. Project No. BCv50466.27, Whitford, British Columbia Transmission Corporation, British Columbia, Canada. [ Links ]
Krammer, V. C., D. M. Calabrese & K.W. Nickerson. 1980. Growth of Enterobacter cloacae in the presence of 25% sodium dodecyl sulfate. App. Environ. Microbio. 40.5. 973- 976 [ Links ]
Larson, R. J., T. M. Rothgeb, R. J. Shimp, T.E. Ward, & R.M. ventullo. 1995. Kinetics and practical significance of biodegradation linear alkylbenzene sulfonate (LAS) in the environment. The CLER Review 1: 4-19. [ Links ]
Leon, V., R. Ponce, E. Gonzalez-Mazo, J.M. Forja, & Gomez-Parra. 1999. Factor analysis of Linear alkyl-benzene sulphonate (LAS) vertical distribution in coastal sediments of Cadiz Bay (southwest Spain). Bol. Inst. Esp Oceanogr.15:509-516. [ Links ]
OECD SIDS. 2005. SIDS Initial Asssesment Report for 20th SIAM. UNEP. Kenya. [ Links ]
Olayemi, A. B., K.I.T. Eniola, S. Awe & T.M. Kayoe-isola. 2003. Distribution of Bacteria in three detergent-effluent-polluted water bodies in Ilorin, Nigeria. NISEB Journal. 3.3. 79- 86. [ Links ]
Painter, H.A. & T.F. Zabel. 1989. The behaviour of LAS in sewage treatment. Tenside surfactant detergents26. 108- 115. [ Links ]
Schulze, S. & A. Tiehm. 2004. Assessment of microbial natural attenuation in groundwater polluted with gasworks residues. Water Sciences and Technology 50. 5. 347- 353. [ Links ]
Leon V., R. Ponce, E. Gonzalez-Mazo, J.M. Forja & A. Gomez-Parra. 1999. Factor analysis of Linear alkylbenzene sulphonate (LAS) vertical distribution in coastal sediments of Cadiz Bay (southwest Spain). Bol. Inst. Esp Oceanogr.15:509-516. [ Links ]
Olayemi, A. B. 1994. Bacteriological water assessment of an urban river in Nigeria. Intern. J. Environ. Health Res. 4:156-164. [ Links ]
Takada, H., K. Mutoh, N. Tomita, T. Miyadzu & N. Ogura. 1997. Rapid removal of LAS by attached biofilm in an urban stream. The CLER Rev. 3:54-65. [ Links ]
WHO. 1996. International Programme on Chemical Safety. Environmental health criteria 169. Linear alkylbenzene sulfonates and related compounds. World Health Organization. Geneva, Swess. [ Links ]
Internet reference
Council for LAB/LAS Environmental Research (CLER).1996. The Council for LAB/LAS Environmental Research (CLER); www.cler.com (downloaded July 28, 2008). [ Links ]