Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.56 n.2 San José Jun. 2008
Diversity, natural history and conservation of amphibians and reptiles from the San Vito Region, southwestern Costa Rica
Georgina Santos-Barrera1, Jesus Pacheco2, Fernando Mendoza-Quijano3, Federico Bolaños4, Gerardo Cháves4 , Gretchen C. Daily5, Paul R. Ehrlich5 & Gerardo Ceballos2
1. Universidad Nacional Autónoma de México, Facultad de Ciencias, Museo de Zoología, A.P. 70-399, C.P. 04510, México D. F; gsantos@miranda.ecologia.unam.mx
2. Instituto de Ecología, Universidad Nacional Autónoma de México, A.P. 70-275, C.P. 04510, México D. F., Mèxico.
3. Instituto Tecnológico de Huejutla, Hidalgo, México, Km 5.5 Carr. Huejutla-Chalahuiyapa, C.P. 43000, Huejutla de Reyes, Hidalgo, México.
4. Escuela de Biología, Universidad de Costa Rica, San José, Costa Rica.
5. Center for Conservation Biology, Stanford University, California, USA.
Abstract: We present an inventory of the amphibians and reptiles of the San Vito de Coto Brus region, including the Las Cruces Biological Station, in southern Costa Rica, which is the result of a survey of the herpetofauna occurring in mountain forest fragments, pastures, coffee plantations, and other disturbed areas. We found 67 species, included 26 species of amphibians and of 41of reptiles. We describe the distribution patterns of the community on the basis of the life zones, elevation, fragmentation, and degree of anthropogenic impact. We also provide some nouvelle data on the systematics of some select taxa, their geographical ranges, microhabitats, activity, and other relevant ecological and natural history features. Finally, we comment on the present conservation status of the herpetofauna in the region. Previous literature and collection records indicate a higher number of species occurring in this area, which suggests that some declines have occurred, especially of amphibians, in last decades. Rev. Biol. Trop. 56 (2): 755-778. Epub 2008 June 30.
Keywords: Amphibians, reptiles, Costa Rica, San Vito, checklist, habitat fragmentation, conservation.
Wildlife inventories are a fundamental tool for conservation because they are the basis for selecting priority sites and for evaluating species conservation status and population extinctions, aid in conservation planning and priority setting and help to identify priority species for conservation action and recovery planning; and support educational programmes (Caughley and Gunn 1996, Daily et al. 2003, Baillie et al. 2004, Lips et al. 2004, Pineda and Halftter 2004, Santos-Barrera & Pacheco 2004, Ramírez-Bautista et al. 2006, Urbina-Cardona et al. 2006). The relevance of having solid biodiversity inventories has become apparent in recent decades because of the alarming increase in the rate of loss and decline of populations and species. Biodiversity loss is, perhaps, the only truly irreversible environmental problem resulting from human activities (Ceballos and Ehrlich 2002, Stuart et al. 2004). Of major concern is the conservation future of most amphibian and reptile species.
Declines of amphibian populations in the tropics have been extensively documented in Latin America, especially in relation to the anthropogenic activities or their environmental impacts such as habitat destruction, emerging infection diseases, and global warming, to the point to consider that amphibians are the group experiencing the most important declines or extinctions from all vertebrates (Young, et al. 2001, Collins & Storfer 2003, Stuart et al. 2004, Pounds et al. 1999, 2006).
Amphibians and reptiles are major components of Neotropical vertebrate faunas (Heatwole and Sexton 1966, Scott 1976, Fauth et al. 1989). Indeed, the Neotropics probably maintain the highest diversity of both amphibian and reptiles on Earth (Young et al. 2001). Within the Neotropics, Costa Rica has a high diverse herpetofauna which has been relatively well inventoried (e.g. Taylor 1954, 1955, Scott et al. 1983, Savage and Villa 1986, Savage 2002). Most information about natural history and ecology in Costa Ricas natural areas come however, from the Guanacaste tropical dry forests, the cloud forest of Monteverde and the La Selva Biological Station (Janzen 1983, Savage 2002, Bell and Donnelly 2006). There are however regions like the southwest and the Cordillera de Talamanca that remain little known, and even new species have been recently described (e. g. Lips 1996, ONeill and Mendelson 2004).
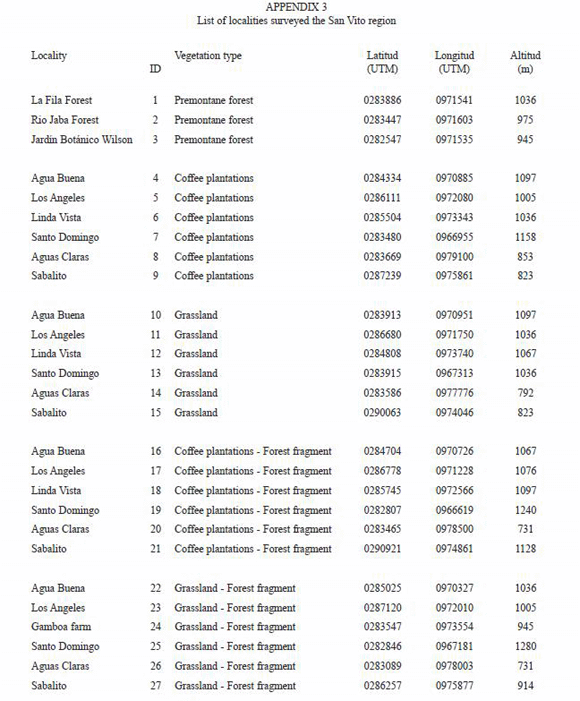
In this study we evaluate the species composition and structure of the herpetological communities of the San Vito region including Las Cruces forest reserve in southern Costa Rica, as part of a major project that focous on the effects of forest fragmentation on the biodiversity of mammals, birds, insects, and plants in human-dominated landscapes (Ricketts et al. 2001, Daily et al. 2003, Ricketts 2004, Mayfield and Daily 2005, Pacheco et al. 2006). The landscape in the San Vito region originally was dominated by montane and premontane cloud forests, but over the past 40 years the forest has been converted into pastures, crops, and coffee plantations. The main forest patch is the 266 ha LCBS of Premontane tropical forest. The herpetofauna has been more or less well studied for at least four decades, recording to date 77 species of amphibians and 102 of reptiles in the whole (Scott 1983, OTS 1994, Schlaepfer 2000, Schlaepfer and Gavin 2001). The main objectives of this study are to: (1) Provide a recent inventory of the reptiles and amphibians of the San Vito region; (2) Describe the patterns of species distributions across the matrix of habitats in a human-dominated landscape; (3) Contribute to the knowledge of the species conservation status and; (4) Present species accounts for all species recorded.
Materials and methods
Study site: Las Cruces Biological Station (LCBS) of the Organization of Tropical Studies (OTS) and the Wilson Botanical Garden (WBG) are located at south western Costa Rica in Coto Brus, Puntarenas Province, Costa Rica about 30 km from the Panama border at extreme coordinates: 08º4704, 82º5709º, ranging from 1 100 m to 1 500 m of elevation. The study area comprises a circumference of about 20 km ratio along LCBS (Fig. 1). Landscapes are mostly fragments of premontane forest immersed in a matrix of coffee plantations and pasture lands which are intensively managed since 20 to 35 years ago (Daily & Ehrlich, 1995). The main forest reserve area of LCBS is possibly the only continuous less disturbed forest still present in the area (Schlaepfer and Gavin, 2001). LCBS and WBG are located 5 km south of the town of San Vito de Coto Brus. The area was formerly covered with premontane humid forests and belongs to the 23 Costa Rican Biotic Unit which means that predominant climate conditions are of a 10 months of temperate/tropical to very wet season with 2 months of "winter" or dry season (Herrera and Gomes, 1993).
Methods: We conducted six surveys during 2002 to 2004 covering wet and dry seasons. Diurnal and nocturnal Visual Encounter Surveys were used (VES hereafter, Crump and Scott 1994) in combination with a trapping arrays system using drift-fences and pitfall and funnel traps. We sampled 27 sites that included the following types of vegetation with differential grades of disturbance: pristine and secondary forests patches: Las Cruces Forest (LCF), grasslands or pasture lands (PL), coffee plantations (CP), and combinations of forestgrassland (FP), and forest-coffee plantations (FCP). Transects were standardized for two observers walking along a defined transect of 2 hours/2 km duration in each site resulting in walking surveying of 4 kilometers per site. Along this transect each amphibian and reptile individual observed was identified and recorded directly in the field.
The trapping arrays consisted of two parallel lines departed 10 m. Each line contained three pitfall traps setting at 5 m along the line. Finally, two funnel traps were located on each side of the fence. Total trapping effort consisted of 27 study sites with 162 pitfall and 108 funnel traps. VES effort consisted of 4 hours per site/ person, for 156 hours in each sample season. VES effort including diurnal and nocturnal transects was of 648 hours.
All individuals collected and/or observed through the traps and transects were identified directly in the field and then released. No mark-recapture techniques were applied as an attempt to avoid negative impacts on the wild populations and because they were not relevant for the purposes of this study. Some specimens of difficult identification in the field were collected and transported to the laboratory for precise revision and later released at the same locality of capture the day after. Additional records as well as specialized taxonomic literature for identification for the area were compiled (Savage and Lips 1993, Harris 1994, Campbell and Savage 2000, Duellman 2001, Savage 2002, Solórzano 2004). Records at Universidad de Costa Rica (UCR), Museo de Zooogía, Facultad de Ciencias, Universidad Nacional Autónoma de México (MZFC) and other incidental records were also included.
The species were classified according to their relative abundance, following an arbitrary classification based on their proportion in respect of the total observations in the surveys with the following criteria, relevant for this study only: rare, 1 to 10 records; common, 11 to 100 records; abundant, 101 to 200 records, and very abundant, more than 200 records. Relative abundance provides a qualitative basis for ecological comparisons between taxa and habitats, although no specific criteria has been developed to define species abundance since this feature highly depends on several intrinsic and extrinsic factors. A Chi-Square (X2) was used to test about differences in species richness among habitats.
Results
Species composition, relative abundance and habitat distribution: A total of 1,678 individuals were recorded comprising 67 species of amphibians and reptiles. There were 26 species of amphibians, representing 16 genera, 10 families, and three orders. Reptiles included 41 species, 34 genera, 1 order, and 11 families. Amphibians represented 59% of all records in this study (986 records), while reptiles represented 41% of the records (692 records, Appendixes 1 and 2).
The relative abundance of species was very variable, with species recorded from one to 447 records: indeed, 21 species were recorded just once. Most amphibians such as the toad Ollotis conifera were rare (16, 59.3%), 8 species (30%) such as the tree frog Smilisca phaeota were common, 2 species were abundant (i.e. the leaf litter frog Craugastor crassidigitus and Pristimantis ridens), and one was very abundant (Craugastor stejnegerianus; 415 records). Similarly, most reptiles such as the false coral snake, Erythrolampus mimus were rare (36 species, 86%); followed by 4 common species such as the lizard, Ptychoglosus plicatus (9.5%), only 2 species were abundant an very abundant, the whip-tail lizard Ameiva quadrilineata (67 records) and the anole Norops polylepis (447 records), respectively.
Abundance varied across the different habitats. Most amphibians were recorded at coffee plantations (15 species, 53.57%), followed by the montane forest fragments (14 species, 50%), and LCF and pasture lands (10 species, 35.71%). The highest number of exclusive amphibian species was recorded in coffee plantations with 5 species (17.85%) followed by the LCF and forests patches with 4 species (14.28%), and one species (3.57%) in the pasture lands. The most abundant amphibian species at the forests patches was the leaf litter frog Craugastor stejnegerianus (353 records), and one of the least recorded was the toad Ollotis conifera (one record). In the coffee plantations the most abundant species was Craugastor stejnegerianus (41 records) and the least recorded the frog Leptodactylus poecilochilus (one record). Finally, at pasture lands the most abundant species was Smilisca phaeota (23 records) and the least recorded was the bufonid toad Rhaebo haematiticus (one record).
In contrast, most reptile species were recorded in pasture lands (16 species, 37.21%) followed by the LCF (15 species, 34.88%), coffee plantations (14 species, 32.55%) and forest fragment (13 species, 30.23%) (Fig. 2, Appendix 1). Several species were exclusively found in one habitat. There were 9 species exclusive of the LCF (20.93%), 8 in the PL (18.6%), 7 in the CP (16.28%) and 4 in the FP (9.30%).
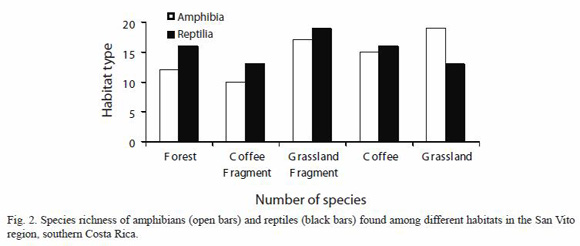
Reptiles also varied in abundance among habitat types. We found that the most common species at forest patches was the anole Norops polylepis (336 records), and one of the rarest the pit-viver snake Atropoides nummifer (1 record). In the coffee plantations the most abundant species was again N. polylepis (67 records) and the rarest the coluber snake Stenorrhina degenhardti (1 record). Finally, at the pasture lands, the most abundant species was N. polylepis (44 records) and the rarest the pit-viper snake Porthidium nasutum (1 record). There were no significant differences in the distribution of amphibians and reptiles among the habitats (x2=2.13, gl=4, p=0.713) however most habitats have more reptiles while the grasslands have more amphibian species. The number of amphibian (x2=3.64, gl=4, p=0.303) and reptile (x2=1.64, gl=4, p=0.802) species were not significant relating the habitats.
Discussion
Inventory and long-term changes in herpetological diversity: As has been pointed out by Duellman (1999), the distribution of amphibian species in Costa Rica is concentrated in the southern lowlands of the country, the highlands of the Central America isthmus and the Caribbean region. It is clear that the geographic distribution of amphibians is determined by the types of vegetation, being
the richest one the wet forest. Our study focused on the wet forests of the southern part of the country where we found interesting differences with previous studies, for example several taxa have apparently disappeared from the area (Scott et al. 1983). We found 6 new records of amphibians and 2 of reptiles for the region. This can be interpreted as a result of the intensity of our field work, but also it may reflect differences in the habitat and microhabitats sampled. Unfortunately the lack of systematic inventories precludes the solid evaluation of the possible trends we documented. For example, the harlequin frog, (Atelopus varius) was formerly a very abundant species, there were no records in our study, and we attribute the lack of records to a local extinction event. From a series of interviews with the local people we concluded that the species started to decline in the early 90s (also F. Bolaños pers. obs). It has been speculated that this species has experienced several local extinctions across its geographic range and thus it has been recently classified as critically endangered (IUCN, 2006). The main cause for declines in this severely impacted genus is thought to be the presence of the emerging infectious disease Chytriomycosis driven by global warming (La Marca et al. 2005, Pounds, et al. 2006). Other species of amphibians that may be declining in the region are Ollotis conifera and Lithobates warszchewitschii, both species were previously observed as abundant, but only rarely recorded in this study. One species that potentially occurs in the region but not observed in our study is Ollotis aucoinae (ONeill and Mendelson 2004). The most diverse group are the snakes (45 species) and lizards (26), we were unable to find turtles, caiman and the eye lash snake Bothriechis schlegelii, however we know that the red-cheeked mud turtle Kinosternon scorpioides dwells well in the Rio Jaba, caimans and eye lash snakes have been barely seen in the area since at least 10 years as well as the fer-de-lance Bothrops asper that used to be very common but recorded just once in this study at a private coffee farm.
Conservation status and population trends: Classification of conservation status for all the species recorded in this study can be seen in the Appendix 1 (IUCN 2006). The data indicate that a very large number of species are at risk of extinction. Their persistence in this region is an indicator of their relatively large number. The 58 endangeres species of amphibians include 32 species classified as least concern (LC), 8 as critically endangered (CRA2 ac), 5 vulnerable (VUB1 ab [iii]), 6 endangered (EnB1 ab [iii]; ENA2 ace), 4 near threatened (NT), and 2 as data deficient (DD). The commercial trade of species (CITES 2007), does not list species of amphibians occurring in the San Vito region. However, 7 species of snakes are included in the Appendix 1. Five of these are pit vipers like the colorful species Bothriechis schlegeli. The other one is a species commonly used as pet, Boa constrictor. The Red Data Book of Costa Rica (2005) includes one species of amphibian, Silverstoneia nubicola and the snakes Boa constrictor and Clelia clelia as well as the Water tegu, Neusticurus apodemus as threatened species.
With the available information to date, we can conclude that the only one species that has probably gone locally extinct is the harlequin frog, Atelopus varius which maintains few populations in Costa Rica (Pushendorf pers. comm.). The lack of recorded but expected species can not be attributed to induced extinctions since natural fluctuations in amphibian populations are common (Pechmann and Wilbur 1994). Our observation indicate that even when extensive deforestation and agriculture has occurred in the area, a high diversity of historically known herpetofauna is often still present (Scott 1983). These species might be maintained as a result of the persistence of some large to medium forest patches that provide suitable microhabitats for certain species (Daily et al. 2003, Bell and Donnelly 2006).
In relation to the emerging infection disease caused by the Chytrid fungus Batrachochytrium dendrobatidis (Bd), it is important to note that the presence of this pathogen has not been confirmed in the San Vito amphibian fauna. The nearest known records are from Las Tablas (Lips et al. 2003) and San Isidro del General (Pushendorf 2003). Lips and colleagues (2003) examined four specimens of Atelopus varius from the Wilson Botanical Garden collected in the early 90s, all of them resulted negative for the Bd test. In terms of the infection dispersal, continuous research is needed in order to assess the present status of wild amphibian populations from southern Costa Rica since important mass mortalities events of amphibians have been reported in nearby region in both Costa Rica and Panama (Lips 1998, 1999).
Finally, this research and similar studies evaluating local faunas are fundamental to detect long-term changes in vertebrate communities in the tropics, which are essential to understand long-term dynamics and the impact of anthropogenic activities.
Acknowledgments
We are grateful to the following colleagues and friends that provided invaluable help during the development of this research. Luis Diego Gomes provided support during our stay in Las Cruces Field Station. For field assistance we would like to thank Sofía Rodríguez, Randy Figueroa, Alberto Mendoza, Jesús Ilama, and Eduer Sandi. Thanks also to the personnel at Las Cruces Forest Station, especially to Rodolfo Quiros for providing literature and advice in several ways. César Ríos and Heliot Zarza kindly assisted with figures preparation, and Ana Davidson revised a preliminary version of this manuscript.
Resumen
En este artículo se presenta un inventario de los anfibios y reptiles de la región de San Vito de Coto Brus incluyendo la Estación de Biología Las Cruces, en el sur de Costa Rica. Se llevó a cabo una evaluación de las poblaciones de anfibios y reptiles presentes en los parches de bosque, potreros, cafetales y otras áreas perturbadas de la región. Como resultado de esta evaluación se registraron 26 especies de anfibios y 41 de reptiles lo que suma un total de 67 especies. Asimismo se describen los patrones generales de distribución de las especies basándose en los tipos de vegetación así como en la altitud, fragmentación y grado de perturbación antrópica en el área. Se proporcionan algunos datos sobre la sistemática de las especies, su distribución geográfica, el microhábitat que ocupan, su actividad y otros datos ecológicos y biológicos relevantes. Finalmente, se presenta una breve discusión sobre la conservación de la herpetofauna en la región. Algunos registros de la literatura y de colecciones científicas muestran que el número de especies de la herpetofauna conocidos en esta región era probablemente mayor, lo que podría indicar que han ocurrido algunas declinaciones importantes de especies, especialmente de anfibios en las últimas décadas.
Palabras clave: Anfibios, reptiles, Costa Rica, San Vito, inventarios de especies, fragmentación del hábitat, conservación.
Received 28-III-2007. Corrected 22-X-2007. Accepted 04-II-2008.
References
Bell, K.E. & M.A. Donnelly. 2006. Influence of forest fragmentation on community structure of frogs and lizards in Northeastern Costa Rica. Conserv. Biol. 20: 1750-1760. [ Links ]
Campbell, J.A. & J.M. Savage, 2000. Taxonomic reconsideration of Middle American frogs of the Eleutherodactylus rugulosus Group (Anura: Leptodactylidae): a reconnaissance of subtle nuances among frogs. Herp. Monographs 14: 186-292. [ Links ]
Caughley, G. & A. Gunn. 1996. Conservation Biology in theory and practice. Blackwell Science, Cambridge, Massachusetts, USA. [ Links ]
Ceballos, G. & P.R. Ehrlich. 2002. Mammal population losses and the extinction crisis. Science 296: 904-907. [ Links ]
Collins, J.P. & and A. Storfer. 2003. Global Amphibian Declines: Sorting the hypothesis. Divers. Distrib. 9:89-98. [ Links ]
Crump, M.L. & N.J. Scott Jr. 1994. Visual Encounter Surveys, p. 84-92. In W.R. Heyer, M.A. Donnelly, R.W. McDiarmid, L.C. Hayek & M.S. Foster (eds.). Measuring and Monitoring Biological Diversity, Standard Methods for Amphibians. Smithsonian Institution, Washington D. C, USA. [ Links ]
Daily, G.C. & P.R. Ehlich. 1995. Preservation of biodiversity in small rainforest patches: rapid evaluations using butterfly trapping. Biod. Conserv. 4: 35-55. [ Links ]
Daily, G.C., G. Ceballos, J. Pacheco, G. Suzán, & A. Sánchez-Azofeifa. 2003. Countryside Biogeography of Neotropical Mammals: Conservation Opportunities in Agricultural Landscape of Costa Rica. Conserv. Biol. 17: 1-11. [ Links ]
Duellman, W.E. 1999. Distribution patterns of amphibians in Middle America, p. 111-210. In W.E. Duellman (ed.). Patterns of distribution of amphibians. A global perspective. The Johns Hopkins University, Baltimore, Maryland, USA. [ Links ]
Duellman, W. E. 2001. The hylid frogs of Middle America. Two volumes. Society for the Study of Amphibians and Reptiles. Lawrence, Kansas, USA. [ Links ]
Fauth, J.E., B.I. Crother & J.B. Slowinski. 1989. Elevational patterns of species richness, evenness, and abundance of the Costa Rican leaf-litter herpetofauna. Biotropica 21:178-185. [ Links ]
Harris, D.M. 1994. Review of the teiid lizard genus Ptychoglossus. Herpetol. Monographs 8:226-275. [ Links ]
Heatwole, H. & O.J. Sexton. 1966. Herpetofaunal comparisons between two climatic zones in Panama. Amer. Midl. Natur. 75:45-60. [ Links ]
Herrera, W. & L.D. Gomes. 1993. Mapa de unidades bióticas de Costa Rica (1: 685 000). Instituto de la Caza Fotográfica y Ciencias de la Naturaleza (INCAFO), Madrid, Spain. [ Links ]
Janzen, D. H. 1983. Costa Rican natural history. Chicago: University of Chicago, Chicago, Illinois, USA. [ Links ]
La Marca, E., K.R. Lips, S. Löters, R. Puschendorf, J.V. Rueda-Almonacid, R. Schulte, C. Marty, F. Castro, J. Manzanilla-Puppo, J.E. García-Pérez, F. Bolaños, G. Chaves, E. Toral, A. Pounds & B. Young. 2005. Catastrophic population declines and extinctions in Neotropical harlequin frogs (Bufonidae: Atelopus). Biotropica 37:190-201. [ Links ]
Lips, K.R. 1996. New tree-frog from the Cordillera de Talamanca of Central America with a discussion of systematic relationships in the Hyla lancasteri group. Copeia 3: 615-626 [ Links ]
Lips, K.R. 1998. Decline of a tropical montane amphibian fauna. Conserv. Biol. 12: 106-117. [ Links ]
Lips, K.R., 1999. Mass mortality and population declines of anurans at an upland site in western Panama. Conserv. Biol. 13: 117-125. [ Links ]
Lips, K.R., D.E. Green, & R. Papendick. 2003. Chytridiomicosis in wild frogs from southern Costa Rica. J. Herpetol. 37: 215-218. [ Links ]
Lips, K.R., D.E. Green, F. Brem, R. Brenes, J.D. Reeve, R.A. Alford, J. Voyles, C. Carey, L. Livo, A. P. Pessier, & P. Collins. 2006. Emerging infectious disease and the loss of biodiversity in a Neotropical amphibian community. Proc. Nat. Acad. Sci. USA, 103: 3165-3170. [ Links ]
Mayfield, M.M. & G.C. Daily. 2005. Countryside biogeography of Neotropical herbaceous and schrubby plants. Ecol. Appls. 15: 423–439. [ Links ]
Mendoza-Quijano, F., Santos-Barrera, G., & J. Pacheco. 2002. Eleutherodactylus fitzingeri (Common Rain Frog). Clutch size and parental care. Herp. Review. 33: 125. [ Links ]
OTS. 1994. Las Cruces Biodiversity Data Base, Herpetology, Birds, Mammals. Las Cruces Biological Field Station. Organization for Tropical Studies, San Jose, Costa Rica. [ Links ]
ONeill, E.M. & J.R. Mendelson III. 2004. Taxonomy of Costa Rican Toads Referred to Bufo melanochlorus Cope, with the Description of a New Species. J. Herpetol. 38: 487-494. [ Links ]
Pacheco J., G. Ceballos, G. Daily, P. Ehrlich, G. Suzan, B. Rodríguez-H, & E. Marcé. 2006. Diversidad, historia natural y conservación de los mamíferos de la región de San Vito de Coto Brus, Costa Rica. Rev. Biol. Trop. 54: 1-20 [ Links ]
Pechmann, J.H., & H.M. Wilbur. 1994. Putting declining amphibian populations in perspective: natural fluctuations and human impacts. Herp. Rev. 50: 65-84 [ Links ]
Pineda, E. & G. Halffter. 2004. Species diversity and habitat fragmentation: frogs in a tropical montane landscape in Mexico. Biol. Conser. 117: 499-508 [ Links ]
Pounds, J.A., M.P. Fogden, & J.H. Cambell. 1999. Biological response to climate change on a tropical mountain. Nature 398:611-615. [ Links ]
Pounds, J.A., M.R. Bustamante, J.A., L.A. Coloma , J.A. Consuegra, M.P. Fogden, P.N Foster, E. La Maza, J.L. Masters, A. Merino-Viteri, R. Puschendorf, S.A. Ron, G. Ramirez-Bautista A., L. Canseco-Marquez, & F. Mendoza-Quijano (eds). 2006. Inventarios Herpetofaunísticos de México: Avances en el conocimiento de su biodiversidad. Pubs. Soc. Herpetol. Mex. No. 3. 346 pp [ Links ]
Sánchez-Azofeifa C., J. Still & B.E. Young. 2006. Widespread amphibian extinctions from epidemic disease drive by global warming. Nature 439:161-167. [ Links ]
Pushendorf, R. 2003. Atelopus varius (Harlequin frog) fungal infection. Herp. Rev. 34:355. [ Links ]
Ricketts, T.H. 2004. Tropical forest fragments enhance pollinator activity in nearby coffee crops. Cons. Biol., 18:1262-1271 [ Links ]
Ricketts, T.H., G.C. Daily, P.R. Ehrlich & J.P. Fay. 2001. Countryside biogeography of moths in a fragmented landscape: biodiversity in nature and agricultural habitat. Cons. Biol. 15:378-388. [ Links ]
Santos-Barrera, G. & J. Pacheco. 2004. Status of three species of toads in North-western Mexico. Froglog No. 63: 1-2. [ Links ]
Savage, J.M. 2002. The amphibians and reptiles of Costa Rica. A herpetofauna between two continents, between two seas. The University of Chicago Press. Chicago and London, USA-England. [ Links ]
Savage, J.M. & J.D. Villa. 1986. Introduction to the herpetofauna of Costa Rica. Contributions to Herpelogy 5. Oxford, Ohio: Society for the Study of Amphibians and Reptiles, Lawrence, Kansas, USA. [ Links ]
Savage, J.M. & K.R. Lips. 1993. A review of the status and biogeography of the Lizard genera Celestus and Diploglossus (Squamata: Anguidae), with description of two new species from Costa Rica. Rev. Biol. Trop. 41: 817-842. [ Links ]
Schlaepfer, M.A. 2000. Frogs and lizards on the edge. Amigos Newsletter 54: 12-14. [ Links ]
Schlaepfer, M.A. & T.A. Gavin. 2001. Edge Effects on lizards and frogs in tropical forest fragments. Cons. Biol. 15:1079-1090. [ Links ]
Scott, N.J. Jr., 1976. The abundance and diversity of the herpetofaunas of tropical forest litter. Biotropica 8: 41-58. [ Links ]
Scott, N.J. Jr., J.M. Savage, & D.C. Robinson. 1983. Reptiles and Amphibians. Introduction. p. 367-374
In Janzen D.H. (ed.). Costa Rican Natural History. University of Chicago Press, Chicago, USA.
Solórzano, A. 2004. Snakes of Costa Rica. Instituto Nacional de Biodiversidad, Heredia, Costa Rica. [ Links ]
Stuart, S. N., J. S. Chanson, N. A. Cox, B. E. Young, A. S. L. Rodrigues, D. L. Fischman, & R. W. Walker. 2004. Status and trends of amphibian declines and extinction worldwide. Science 306: 1783-1786. [ Links ]
Taylor, E.H. 1954. Additions to the known herpetological fauna of Costa Rica withcomments on other species. No. I, Univ. Kansas Sci. Bull. 36: 597-639. [ Links ]
Taylor, E.H. 1955. Additions to the known herpetological fauna of Costa Rica with comments on other species. No. II, Univ. Kansas Sci. Bull. 37:499-575. [ Links ]
Urbina-Cardona, N., M. Olivares-Pérez, & V.H. Reynoso. 2006. Herpetofauna diversity and microenvironment correlates across a pasture-edge-interior ecotone in tropical rainforest fragments in the Los Tuxtlas Biosphere Reserve of Veracruz, Mexico. Biol. Conserv. 132: 61-75. [ Links ]
Young, B.E., K.R. Lips, J.K. Reaser, R. Ibáñez, A.W. Salas, J.R. Cedeño, L.A. Coloma, R. Ron, E. La Marca, J.R. Meyer, A. Muñoz, F. Bolaños, G. Chaves & D. Romo. 2001. Population Declines and Priorities for Amphibian Conservation in Latin America. Conser. Biol. 15: 2013-2023. [ Links ]
Internet references
CITES, 2007. UNEP-WCMC, 12 February, 2007.UNEPWCMC Species Database: CITES-Listed Species. http://earthwatch.unep.net/data/wcmc/index.php (downloaded 1 March 2008). [ Links ]
IUCN, 2006. 2006 Red List of Threatened Species. www.iucnredlist.org. (downloaded 1 March 2008). [ Links ]
Accounts of species of amphibians and reptiles studied at the San Vito region. Localities are referred in the Appendix 3.
Number of collected specimens are indicated in parenthesis
Class AMPHIBIA
Order Anura
Family Brachycephalidae
Craugastor crassidigitus Taylor, 1952.
A moderate sized frog that have the posterior tight surface uniform orange or red. Highly webbing reaching proximal end between toes II-III-IV. The color of the dorsal surfaces is usually gray to dark brown with light orange or dark brown spots. This species was one of the most common in the area, most of the times resting
at low elevations on small leafs of short bushes. Recorded specimens: 137. Localities: 1, 3, 8, 11, 12, 13, 14, 16, 17, 18, 19, 22, 23, 24, 25.
Craugastor fitzingeri (O. Schmidt, 1857).
A moderate to large sized frog that can be easily distinguished from other species of the genus by its light-spotted posterior tight surfaces. The dorsal surfaces is commonly with small granules. A midgular light stripe is present. The dorsal surfaces is grayish or brownish. It has a mid-dorsal yellow to orange stripe present in adult males. The ventral surfaces white to yellowish. This species was very common at the San Vito region; we have records from forest patches and even in pasture lands adjacent to streams and pools. On March 29, 2001 Mendoza-Quijano et al. (2002) found one female individual caring a clutch sized 85
eggs, each egg measuring 2 mm of diameter. Recorded specimens: 39. Localities: 1, 5, 6, 9, 11, 14, 15, 18, 20, 21, 23, 24, 26, 27.
Craugastor podiciferus (Cope, 1875).
A moderate sized frog that can be distinguished from other Eleutherodactylus and Craugastor species by having one to three heel tubercles, and because of its dorsal coloration which consisted of a light brown or tan to yellowish ground sometimes with dark blotches in the flanks. The venter light is yellow and clearly smooth. This species lacks toe webbing and expanded digital disks (Savage, 2002). All the individuals were found into the leaf litter of premontane forest patches. Recorded specimens: 15. Localities: 19, 21, 23, 25, 26.
Craugastor stejnegerianus (Cope, 1853).
A small to tiny frog traditionally confused with E. bransfordi, a very similar species from north central Costa Rica, both species are sympatric only at the Laguna del Arenal area. C. stejnegerianus can be distinguished by having prominent tubercles on hands and feet, the thenar tubercle is considerably smaller than the palmar tubercle; adult males lack nuptial pads. Dorsal coloration is dark brown to dark gray frequently without marks. Upper surfaces of tights reddish colored. This was definitive the most common species in this study, always found on the ground or even beneath leaf litter. Active throughout the year. Recorded specimens: 388. Localities: 1, 2, 3, 4, 5, 6, 7, 12, 13, 16, 17, 18, 19, 20, 21, 22, 23, 25, 25, 26, 27.
Pristimantis cruentus (Peters, 1873).
A medium sized frog that can be distinguished because of its distinctive large heel tubercle and numerous supraocular tubercles, with one enlarged. The most common color pattern is of bright yellow to light brown in the dorsum with some dark small marks. The groin and anterior surfaces of the tights are marked with several darker yellow to orange spots with golden borders (Savage, 2002). We commonly found it calling under the leaves in small shrubs, up to 1.5 m within premontane forest patches. Recorded specimens: 35. Localities:
1, 2, 3, 5, 10, 12, 15, 16, 18, 19, 20, 23, 24, 25, 27.
Pristimantis ridens (Cope, 1866).
A tiny frog with enlarged digital disks. Anterior and posterior surfaces of the thighs, feet and disks red colored. It has various supraocular warts present. Snout clearly enlarged. The dorsum is light brown to yellow tan with a dark brown supra tympanic mark. The venter is yellow to yellowish with scattered points and marks. According to our observations this species is more frequent in low vegetation, over leaves and tree steams, sometimes jumping as high as 3 m. Recorded specimens: 159. Localities: 1, 2, 3, 4, 10, 11, 12, 13, 14, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27.
Pristimantis vocator Taylor, 1955.
This is a tiny and sometimes inconspicuous frog with a narrow head. Without toe webbing, small digital disks; dorsal coloration gray with a narrow interorbital dark line and an elongate indefinite darker median area and
a white spot on heel. Entire ventral surface and concealed parts of the limbs pigmented, the pigment forming a reticulation leaving very numerous small rounded light areas; a dark line follows the supratympanic fold. Almost always found into pasture-lands and open areas calling hidden on the grass, in areas adjacent to forest patches. Recorded specimens: 25. Localities: 1, 14, 17, 20, 23, 24, 25.
Family Bufonidae
Rhinella marina (Linné, 1758).
This is the largest toad in Costa Rica. The parotoid glands are clearly great swallowed and sub triangular, the rest of the cranial crests well developed; a tarsal fold is present (Savage, 2002). The dorsum in adults is dark brown cinnamon with irregular chocolate blotches; venter cream with a dusky pigment; this species in the San Vito region occurs on all types of vegetation, although according to Savage (2002) is more evident in disturbed areas and around human settlements. At present, this species is considered as a weed species; however, not frequently recorded at high elevation forests. Recorded specimens: 19. Localities: 1, 8, 10,
11, 14, 15, 23, 24, 26.
Ollotis conifera (Cope, 1862).
A moderate sized toad with conspicuous dorsal black tipped warts and a lateral line of warts on each side of the body. The parotoid glands are small and sub triangular. Color on dorsal surfaces is light greenish ground. Venter cream. This toad is a common inhabitant of moist and rain montane or premontane forests where uses to climb the trees. The specimen recorded was found close to a Las Cruces Field Station building. Recorded specimens: 2. Localities: 3, 15.
Rhaebo haematiticus (Cope, 1862).
A moderate to large sized toad having large parotoids glands and a distinct inner tarsal fold. Lacking any cranial crests (Savage, 2002). The skin is conspicuously smooth. The ground color is of olive gray to olive dull brown with a broad dark brown lateral band that runs from tip of the mouth to the groin, sometimes with dark brown to black dots on the dorsum. This species dwells in the ground covered by leaf litter in premontane wet forests. We found three specimens, two into the reserve forest and one at a forest patch highly visited by tourists. Recorded specimens: 3. Localities: 3, 15.
Family Centrolenidae
Centrolene prosoblepon (Boettger, 1892).
A small sized glass frog lacking protuberant nostrils and with moderate toe webbing. The web not reaching distal subarticular tubercle on toe IV or base of disks on toes III and IV. The adult males of this species have an obvious projecting blunt humeral hook (Savage, 2002). The color of the dorsum is light green, usually
with many small black dots, dark green bones in life, and a white parietal sheath. Venter transparent, with the digestive tract visible. In the San Vito it was found in large pasture lands in large pasture plants into grasslands adjacent to a premontane moist forest and under the foliage of a tree into a small forest patch. Recorded specimens: 4. Localities: 11, 14, 22.
Dendrobatidae
Silvertoneia nubicola (Dunn, 1924).
A very small dendrobatid frog having and oblique lateral line extending from the groin to the upper eyelid. The venter is yellow and males with black throat. Commonly confused with C. talamancae, however this last species lacks the oblique lateral light strips. The three individuals that we recorded were found active
during the day in a humid premontane forest patch adjacent to the main reserve forest. Recorded specimens: 3. Localities: 24.
Family Hylidae
Agalychnis callidryas (Cope, 1862).
This is a relatively large tree frog with large webbed feet and dilated toe tips. The dorsum is bright green, sometimes with round white spots, the throat and belly are creamy white, the hands and feet are orange with the anterior and posterior surfaces of the thigh dark blue. The sides of the body are dark blue to purple with prominent ivory horizontal borders and vertical bars, and the iris of the eye is blood red (Duellman, 1970, Savage, 2002). We documented the presence of larvae along a small stream adjacent to coffee and banana plantation as well as advertisement calls during the night at nearby small streams in a rainy night. Recorded specimens: 2. Localities: 9, 26.
Dendropsophus ebraccatus (Cope, 1874).
This is a moderate to small tree frog. The most conspicuous character is the distinctive dorsal coloration. This species has a well developed axillary membrane, extensive finger webbing, and usually a dark brown lateral
band extending from the tympanum at least to the mid body (Savage, 2002). The characteristic color pattern of dorsum in this species is of one or more bold dark brown blotches, usually broadly bordered by white or yellow; bright yellow to orange thighs, the usual figure marked is a roughly hourglass. We recorded it at temporary pools over grasslands and lowland premontane forest. This species exhibits a great variation on dorsal color pattern. It is active exclusively at night. We recorded congregations after heavy rains when temporally ponds were formed. This species was found in the same pools with other anuran species like
Rana sp. Recorded specimens: 7. Localities: 3, 9, 10, 14.
Dendropsophus microcephalus (Cope 1855, 1856).
A small slender yellowish tree-frog having minimal finger webbing, venter granular, tympanum indistinct and iris bronze (Savage, 2002). The dorsum is yellowish with a welldeveloped dorsolateral white stripe bordered
below by a narrow dark brown stripe running from the eye above the tympanum to mid body. The tights are uniformly yellow. We found three individuals in grasslands near to lowland premontane wet forests where temporary pools were formed with abundance of grasses and shrubs. It is active only at night. Recorded
specimens: 3. Localities: 10, 27.
Hypsiboas rosenbergi (Boulenger, 1898).
A moderate sized tree-frog, distinguished from other hylid species by the tan, orange, yellowish dorsal color pattern. Bright eyes and a protuberant prepolex. Venter is cream immaculate. The single recorded individual was a juvenile characterized by its dark dots on the dorsum and was found was perching on a leaf in a coffee-plantation. Recorded specimens:1. Localities: 8.
Ptychohyla legleri (Taylor, 1958)
A medium sized bright green tree frog with a very large tympanum and snout moderately long (Savage, 2002). Color in the dorsum is bright green with a white upper labial stripe running below the eye from the snout to the groin. Ventral surfaces light to bright yellow. We recorded nine individuals of this species after a truly heavy rainfall and exclusively at a relatively disturbed premontane forest patch where a small temporary stream was formed. Recorded specimens: 9. Localities: 25
Smilisca phaeota (Cope, 1862).
A moderate to large sized frog, relatively long legged. A white labial stripe is present and a dark brown mask running from the nostril and reaching the insertion of arms. The flanks are marked with a fine dark brown to
black venation. This character distinguished this species from S. baudinii. Although some variation of the color pattern of this species can be seen, the most common is of uniform dark olive to bright green ground dorsum with dark brown blotches. This is a common inhabitant of the premontane wet forest in the area even
in pasture lands and coffee plantations. It is frequently confused with S. baudinii because of the similar color pattern of the dorsum; both species are sympatric at some forest patches. Recorded specimens: 41. Localities: 3, 9, 11, 12, 13, 14, 17, 18, 21, 23, 24, 26, 27
Smilisca sordida (Peters, 1863).
A moderate to large tree-frog with a variable color pattern of the dorsum. This species differs from another Smilisca species by having large webbed fingers and extensively webbed toes (most toes have only the distal phalanx free of web), snout obtuse to rounded in profile, dorsal surfaces and tympanum smooth, a prominent series of warts along postero-ventral margin of lower arm (Savage, 2002). The dorsal color pattern varies from gray, to tan, greenish brown, or reddish brown marked with darker blotches. The flanks and posterior surfaces marked with bluish white or cream flecks or bluish white spots in a darker ground. This species
was recorded almost always on broad leafs at relatively high plants. Recorded specimens:
15. Localities: 11, 12, 13, 15, 23, 25, 26.
Leiuperidae
Engystomops pustulosus (Cope, 1864).
A small frog that resembles a toad. This species is clearly differentiated by possessing parotoid glands, dorsum with numerous warts and a conspicuous outer tarsal tubercle. Dorsum is brown or dark gray with darker gray
blotches. Venter is light brown or light gray with black reticulation or suffused black marks. Aggregations of this species were common in disturbed areas where temporary pools were formed by heavy rains. Sometimes we found them under logs and rocks in grasslands at forest borders where males called loud. Recorded
specimens: 12. Localities: 8, 15, 21, 23.
Leptodactylidae
Leptodactylus melanonotus (Hallowell, 1860, 1861).
A moderate sized frog with well developed toe and fingers webbing. Plant tubercles are black, lips marked with dark bars. Dorsal surfaces are dark brown with some spots and an interorbital dark bar or triangle. Venter light
brown with some pigmentation and reticulation. A series of ventrolateral glands are present in both sexes. This species was recorded once into a small pond in an open grasslands hidden at the basis of a plant. Recorded specimens: 1. Localities: 13
Leptodactylus poecilochilus (Cope, 1862).
A medium to large sized frog that can be distinguished by having one t three pairs of dorsolateral folds, smooth soles and tarsi, and a longitudinal white stripe across the posterior thigh. The dorsal surfaces are light brown to grayish with marks. I has a vertebral light line is often present, the venter is white. We found only two female individuals hidden into the ground at a flooded open pasture land. Recorded specimens: 2. Localities: 15, 21.
Family Ranidae
Lithobates taylori (Smith, 1959).
A large sized frog with relatively short legs. Dorsolateral fold distinct but discontinuous, this feature distinguish this species from its relative R. forreri. The dorsum is brown or gray-brown with large darker brown spots, the venter is white cream with no marks. This species was always associated to medium sized pools or temporary ponds formed after heavy rains in pasture lands and other open areas. It was active also in daylight. Recorded specimens: 6. Localities: 4, 10, 11.
Lithobates warschewitschii (O. Schmidt, 1857).
A moderate sized frog very distinct by having red coloration on the lower ventral surfaces and underside legs and between toes. Lateral and tights surfaces with several yellow dots and bands. Dorsum is tan brown with green to blue dots. The venter is cream or light yellow with no marks. This species is an inhabitant of forest litter sometimes found along roads sides at rainy nights. Clearly absent in our last survey in the study area (2004). Observations of individuals of this species in the area are clearly scarce, in previous years we had the chance to see abundant populations at the forest and even along the border of the roads at rainy nights.
Recorded specimens: 1. Localities: 3.
Order CAUDATA
Family Plethodontidae
Bolitoglossa lignicolor (W. Peters, 1873).
A salamander with 13 conspicuous coastal grooves and 12 folds; 3.5 folds between adpressed limbs; foot considerably thicker than hands. Dorsum is frequently brown with a broad darker band. The ventral surfaces are brown with numerous light spots. The coastal surfaces, tail and limbs dark gray or black. It is a species
active at night, arboreal, and frequently remains under logs and root crevices during the day. We captured three individuals in funnel traps at a grassland hill adjacent to a riparian forest patch. Another individual was collected at night while feeding at a coffee plant. Recorded specimens: 4. Localities: 8, 14.
Order GYMNOPHIONA
Family Caecilidae
Dermophis parviceps (Dunn, 1924).
This species is recognized by having eyes under the skin and lacks the inner mandible teeth. Dorsal color pattern is of lavenderplumb gray with some small white spots or speckles that became lighter in the borders,
ventral surfaces are light lavender. It is known from tropical lowlands in the Atlantic versant. It is terrestrial commonly found under logs and soil. It is apparently uncommon into open and disturbed areas (Leenders, 2001; Savage, 2002). However, in this study we found all the specimens in highly disturbed areas such as
pasture grasslands and coffee plantations that sometimes were adjacent to forests patches. Recorded specimens: 8. Localities: 4, 5 13, 22, 23.
Class REPTILIA
Order SQUAMATA
Suborder SAURIA
Family Anguidae
Diploglossus bilobatus (OShaughnessy, 1874)
A small to medium-sized lizard. Body covered with small cicloid scales that may have weak medium keel. Claws hidden within a scaly sheath so that only the tips are exposed. Dorsal surfaces dark brown to black in juveniles. Flanks with green to tan marks. The species occurs in lowlands, rainforest and premontane wet forest. We found one female during the day basking on a river rock, with 6 eggs that hatched in captivity. Localities: 3.
Corytophanidae
Basiliscus basiliscus (Linné, 1758).
A large basilisk that can be distinguished from B. plumifrons (Atlantic) by having well developed dorsal and caudal crests and rounded or pointed head crest. A pair of white stripes run on each side of the body, more conspicuous in juveniles and almost absent in females. In addition occipital scales are smooth and larger in B. basiliscus. Color of the dorsum is brown to olive in adults and yellowish green with brown cross bands in juveniles. The species was found into premontane forest patches hidden under big branches of litter. Recorded specimens: 1. Localities: 1, 8.
Family Gymnophtalmidae
Neusticurus apodemus Uzzell, 1966
This is a small and very distinguishable species of lizard. The head is pointed and dorsal scalation consists of a mixture of small flat scales and other enlarged keeled and tuberculated forming keels. Venter covered with flat
and quadrangular scales. Dorsal surfaces are dark brown to gray, arms covered with yellow spots. Venter is dark brown. Males have the chin colored with orange dots. This species occurs in forest floor and premontane forest. According to Savage (2002) is associated to streams but we found all four specimens on the forest ground into the forest. Recorded specimens: 4. Localities: 2, 20, 24.
Ptychoglossus plicatus (Taylor, 1949)
Medium-sized lizard that can be distinguished by the presence of elongate and mucronate dorsal scales in contrast with the quadrangular smooth ventral scales. Head mahogany brown, dorsum is black slightly reddish brown, venter cream immaculate. This species occurs in premontane wet forests and lowland wet forest.
It is a common species found in leaf litter into the forest, under rocks and debris. On March 2001 we found a pregnant female with three eggs distinguished through the ventral skin. Recorded specimens: 22. Localities: 1, 2, 3, 7, 16, 17, 18, 20, 22, 24, 25.
Corytophanes cristatus (Merrem, 1822).
A moderate sized Corytophanid species with a clearly distinctive helmet-shaped casque with raised lateral ridges extended to the back as a serrated skin flap. Color of dorsum is brown olive to reddish brown with darker marks, few black lines radiating from eyes to upper lip. The venter is lighter brown to light brown-green with some dark dots. Specimens were active during day and night perched at the tree trunks, and were recorded only at forest patches. Recorded specimens: 4. Localities: 17, 21, 23.
Family Polychrotidae
Norops aquaticus (Taylor, 1956)
Amoderate sized anole lizard distinguished from other anoles mainly by its coloration. Dorsal surfaces are olive green with a coastal darker line and darker cross bands on a yellow ground. Head with a bright yellow line running from the eye to the end of body. We found this lizard during the day, always active along the
river borders and under rocks into the forest. Recorded specimens: 6. Localities: 2
Norops biporcatus (Wiegmann, 1834)
A large anole that can be distinguished by its large size and coloration, the dorsum is bright green, in addition it has strongly keeled ventral, and tuberculate dorsal and lateral scales. The male dewlap is tricolor white on the base with blue and orange above. We found it only in forested areas perching in tree steams or trunks
or even on the ground. Recorded specimens: 5. Localities: 7, 10, 14, 16.
Norops capito (W. Peters, 1863)
A large anole with very short head. Dorsal and lateral scales are smooth, flattened and juxtaposed. Males dewlap is greenish yellow. Dorsal color is greenish or yellow olive to brownish frequently with dots and a v-shaped interorbital band. Venter light yellow with several marks or dots. This species inhabit in
tropical montane and premontane humid forest. Recorded specimens: 12. Localities: 1, 2, 24.
Norops limifrons (Cope, 1862)
This is a small anole with long legs. It can be distinguished by its white upper lip, the smooth or slightly keeled ventral scales, small granular dorsal and lateral scales and rugose or keeled scales in the prefrontal region. Dorsal surfaces are gray brown to olive, some females with a narrow area or yellow dorsal stripes with a series of diamond-shaped middorsal dark blotches. Venter is white to pale-yellow. Males dewlap is white with a distinct basal spots. This was a very common and wide ranging species in our study site from forests to coffee and banana plantations and even in disturbed areas. Recorded specimens: 23. Localities: 1, 24, 27.
Norops polylepis (Peters, 1873, 1874)
This is a small anole with relatively long legs. The most common and abundant species in the area with smooth ventral scales. Dorsal surfaces are brown to reddish brown with a middorsal line of blotches, as in N. limifroms. Some females exhibit a median line of diamondshaped blotches or a broad middorsal stripe, other are bordered with dark venter and numerous dots. Males dewlap is large and orange. This species occurs in premontane humid forest and rainforest in other areas. We recorded a large number of individuals in forest fragments and coffee plantations but less seen in forest patches. Recorded specimens: 311. Localities: 1, 2, 3, 4, 5, 7, 8, 9, 10, 11, 12, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27.
Anolis woodi (Dunn, 1940)
This is a large-sized anole with long legs, blue eyes, slender and attenuate tail and keeled ventral scales. Dorsal surfaces are dark olive to dark brown, sometimes with a red suffusion ventral light olive brownish. Males dewlap dark olive green to black. This species inhabits only at montane and premontane forest. It
was a rare species. Recorded specimens: 9. Localities: 1, 25.
Family Scincidae
Mabuya unimarginata (Cope, 1862)
It is a large lizard with long legs when comparing with other skinks. Dorsal is brown or upper brown with lateral dark brown bands bordered below by light yellow or cream stripes. A pair of enlarged nuchals and a transparent disc in the lower eyelid. Ventral whitish to bluish-white. The species was recorded in a
great variety of habitats from dry to wet forest, coffee and banana plantations and in secondary or disturbed areas. Recorded specimens: 7. Localities: 7, 11, 15, 23, 25.
Sphenomorphus cherriei (Cope, 1893)
This is a small lizard, characterized by having the upper lip spotted with dark pigment, lacking enlarged nuchals (occipitals) and the transparent disk in the lower eyelid. Color of the dorsum is bronze with numerous
tiny spots and a black stripe running from the nostril through the eye, behind the neck and farther and bordered below by a narrow cream stripe. Ventral surfaces are cream to yellow with no marks. This species was found it in the forest floor at premontane forest, disturbed and secondary forest. Recorded specimens: 3. Localities: 5, 9, 24.
Family Teiidae
Ameiva festiva (Lichteinstein and Von Mertens, 1856)
This species is a medium-sized lizard. The most conspicuous feature observed is a vertebral stripe changing from light-blue in juvenile to yellow in adults and lately disappearing. Coastal surfaces are marked with numerous longitudinal yellow dots arranged in three parallel rows along the coastal surfaces of the body. This species occurred in a large variety of microhabitat from cultivated areas and relatively disturbed premontane to tropical forest. Recorded specimens: 8. Localities: 4, 8, 9, 11, 16, 17, 20, 21, 22, 23, 25.
Ameiva quadrilineata (Hallowell, 1860, 1861)
A medium-sized lizard that can be recognized by its 4 longitudinal light yellow stripes at the side, and a clear ventral brown band. A clearly sexual dimorphism, can be observed in this species. This was one of the most abundant species in our study area, it was found in cultivated areas, pasture and secondary forest.
Recorded specimens: 47. Localities: 6, 7, 8, 9, 11, 13, 14, 15, 17, 20, 21, 22, 26, 27.
Family Xantusidae
Lepidophyma reticulatum (Taylor, 1955)
A medium-sized lizard with a conspicuous smooth and enlarged head contrasting in texture with the tuberculated body. Color on the dorsal is dark brown to dark black ground with numerous large rounded yellow dots. Gular surface is covered with a reticulum instead of uniformly light ground dark spots of L. flavimaculatum. This species inhabits premontane humid forest and humid lowlands. In our study
it was recorded in pastures beneath rocks and logs. Recorded specimens: 3. Localities: 3, 10.
Order SERPENTES
Family Boidae
Boa constrictor Linné, 1758
A large and heavy-bodied snake characterized by having dorsal smooth and small scales. Color on dorsum is light brown to gray-brown, head frequently with median elongate dark brown bands. Dorsum with 22 to 35 darkbrown or black dorsal blotches. This species inhabits in several habitats but it was extremely rare in our study site. We just saw one individual in a small area of grassland. Recorded specimens: 1. Localities: 2.
Family Colubridae
Dendrophidium percarinatum (Cope, 1893)
A medium-sized coluber snake with brown uniform dorsum or with indistinct dark longitudinal strips, usually cross banded with dark- edges narrow light bands anteriorly. Venter is white or yellowish immaculate becoming yellow posteriorly. We found it in premontane forest. Recorded specimens: 2. Localities: 12, 21.
Erythrolamprus mimus (Cope, 1868, 1869)
A medium-sized coluber snake, with a coloration resembling coral snakes. Most of the rings on the body split dorsally by white areas, having the single black rings separated from the red ones by broad light rings. This is an uncommon species mostly diurnal that inhabit in premontane and tropical lowlands forest.
Recorded specimens: 1. Localities: 1.
Geophis hoffmani (Peters, 1859)
A small snake characterized by a uniform dorsal dark brown to black color. Venter yellow to cream color. This was a common species in low and uplands, found here only in coffee plantations. Recorded specimens: 3. Localities: 9.
Imantodes cenchoa (Linné, 1758)
A medium-sized coluber snake, head thin and compressed, with bulging eyes. Distinguished by other Imantodes species by the presence of enlarged middorsal row scales and a pattern of dorsal reddish rounded blotches. This is a common inhabitant of secondary forest, cultivated lands and marginal premontane and humid tropical forest, found only at night in this study. Recorded specimens: 6. Localities: 4, 10, 21, 24.
Leptodeira septentrionalis (Kennicot in Baird, 1859)
A medium-sized coluber snake. The dorsal color pattern is of light brown to reddish ground with numerous (38 to 70) dark brown rounded spots. Vertical pupil. Venter cream, sometimes with orange marks. This species
occurs in lowland moist and wet forest also in premontane forest. In this study we recorded one individual at relatively disturbed forest. Recorded specimens:1. Localities: 2.
Liophis epinephalus (Cope, 1862)
This is a medium-sized coluber snake with smooth dorsal scales. Dorsal surface of body reddish marked with black bands. Red venter marked with black spots or black bands. Commonly found riparian habitats and open areas. In this study we found one individual in a disturbed area. Recorded specimen: 1. Localities: 10.
Mastigodryas melanolomus (Cope, 1868)
A medium-sized snake with a pattern of alternating dark dorsal and lateral blotches and boldly marked labials, chin and throat in juveniles. Dorsum brown. Venter immaculate. This species occurs in a great variety of habitats from lowlands and premontane humid forest to highly disturbed areas such as pasture and cultivate
areas. Recorded specimens: 5. Localities: 3, 9, 12, 20.
Ninia maculata (Peters, 1861)
A small coluber snake with dark brown ground dorsal color covered with black spots and marks. The venter is yellow or cream with numerous quadrangular spots. A conspicuous black nuchal collar presents. Scales strongly keeled. The species occurs in premontane and lowland forest. It is very common in disturbed
and urbanized areas, commonly a leaf-litter inhabitant. Recorded specimens: 5. Localities: 4, 11, 23, 24.
Oxybelis aeneus (Wagler, 1824)
The bejuquilla is a medium-sized snake clearly distinguished by its attenuate body and elongate head. Dorsum is brown to gray-brown with a conspicuous black border on the mouth. A sleigh depression runs from the orbit to the tip of the snout. Venter white to yellowish. This species is relative common in dry and humid
lowlands. In this study one individual was recorded in a premontane humid forest patch. Recorded specimens: 1. Localities: 8.
Oxyrhopus petolarius (Linné, 1758)
This is medium-sized coluber snake, clearly distinguished by its orange to pink-orange bands. The venter is cream with no marks. It is terrestrial and wide ranging in America. We recorded one individual in a premontane forest and one in an urbanized areas. Recorded specimens: 2. Localities: 26.
Rhadinaea decorata (Günther, 1858)
A small-sized coluber snake. Dorsal surface are brown colored with a pair of longitudinal light stripes. The head have light postocular spots. Venter is cream immaculate, some individuals are colored with orange at
the rear venter. This is a typical lowland species mostly found it in tropical humid forest, but also in relatively disturbed areas. Recorded specimens: 1. Locality: 23, 24.
Scaphiodonthophis annulatus (Duméril, Bibron and Duméril, 1854)
A medium-sized snake frequently with a tricolor banded pattern of monads (each read band separated by a yellow-black.yellow series of bands). The venter is white or yellow immaculate or marked with brown dots. In
adults a light interocular bands is present. We found one individual in a premontane forest patch. Recorded specimens: 1. Localities: 2.
Spillotes pullatus (Linné, 1758)
A very large species clearly distinguished by its black ground color on the dorsum marked with alternating oblique or transversal yellow lines. Dorsal scales keeled arranged in 14 to 18 rows at mid-body. We found it in a pasture land. Recorded specimens: 1. Locality: 24.
Stenorrhina degenhardtii (Berthold, 1845)
A medium-sized coluber snake with an enlarged rostral and shovel-shaped snout. Dorsum is brown to gray-brown with or without darker brown blotches. Ventral surfaces heavily marked with dots and spots or with a medium dark strip. Color pattern in this species is highly variable. This snake inhabits lowlands
and premontane humid forest. We recorded it in coffee plantations. Recorded specimens: 2. Localities: 7, 25.
Tantilla alticola (Boulenger, 1903)
A small-sized coluber snake with 15 rows of dorsal scales. Brown dorsum and red venter. A nuchal collar is present and a post ocular light spot. This is a fossorial secretive species found in lowland and premontane forest and even in the rain forest. The specimens were found under leaf-litter. Recorded specimens: 2.
Localities: 24, 26.
Tantilla schistosa (Bocourt, 1883)
A very small coluber snake similar in dorsal coloration to T. alticola but lacking pre and postocular spots. A conspicuous nuchal collar can be seen. The dorsum is brown, and venter red immaculate. We found it at the margins of a coffee plantation and premontane tropical forest. Recorded specimens: 1. Localities: 16.
Trimetopon pliolepis (Cope, 1894)
A small species of coluber snake. With just one prefrontal shield. Dorsal color pattern is of brown ground with a dark middorsal stripe and four lateral stripes on each side of the body. A light nuchal collar interrupted at the midline. The venter is white to yellow. The species occurs in lowland and premontane humid forest.
We found one individual in the edge of the primary forest under the ground. Recorded specimens: 1. Localities: 10
Family Elapidae
Micrurus alleni K. Schmidt, 1936
A medium-sized coral snake with the head cap usually forming an oblong or lanceolate figure along the interparietal suture. Commonly bicolor but sometimes with a tricolor dorsal pattern. Venter tricolor (black, red, yellow). This species occurs in lowland an intermediate elevation in wet forest and premontane wet forest. At this part of the country is sympatric whit M. nigrocinctus. Recorded specimens: 1. Localities: 19.
Micrurus nigrocinctus (Girard, 1854, 1855)
A medium-sized coral snake with a tricolor ringed pattern. The black head cap extending posteriorly to the orbits and sometimes extending to anterior parietals. A nuchal black ring is frequently touching the posterior tips of the parietal. The only specimen recorded was a juvenile found at a coffee plantation. Recorded
specimens: 1. Localities: 7.
Family Viperidae
Atropoides mexicanus (Rüppell, 1845)
A medium-sized pitviper with dark spots on the supralabials. A dorsal color pattern of dark diamonds with lateral points directly downward. Smaller lateral blotches aligned with the dorsal diamonds. Venter immaculate
but sometimes mottled with brown. This species occurs in premontane wet forest and rain forest. The only one individual recorded was found under a rock into a forest patch. Recorded specimens: 1. Localities: 18.
Bothrops asper (Garman, 1883, 1884)
A large and dangerous pitviper clearly distinguished by having light out lined dark triangular lateral blotches that often fuse across the back to form butterfly shaped figures. Dorsal color is olive green to greenish gray or brown. Venter light yellow immaculate but sometimes at the middle venter can have little spots or
marks. The single individual was found in a coffee plantation: 1. Localities: 24.
Porthidium nasutum (Bocourt, 1868)
A small to medium-sized pit viper with a sharply snout usually elongated into a proboscis. Dorsal ground color is brown to gray with 15 to 23 marking arrow dark. This species occurs in lowland moist forest and marginally premontane wet forest, frequently associated to disturbed or anthropogenic altered places. We recorded a
juvenile at a pasture land adjacent to a stream. Recorded specimens: 1. Localities: 14.