Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.54 suppl.3 San José Dec. 2006
Evaluating the potential of natural reproduction and artificial techniques to increase Acropora cervicornis populations at Discovery Bay, Jamaica
Norman J. Quinn1 & Barbara L. Kojis2
1 Discovery Bay Marine Lab, University of the West Indies, Discovery Bay, St. Ann, Jamaica. Fax 876-973-3092; norman.quinn@gmail.com
2 Division of Fish and Wildlife, St. Thomas, US Virgin Islands 00802
Received 09-VI-2006. Corrected 02-X-2006. Accepted 13-X-2006.
Abstract: Shallow water Acropora species have become uncommon on north coast Jamaican coral reefs owing to a number of factors. On many reefs, algae have taken their place. The result is loss of habitat for many species of fish and invertebrates and less attractive reefs with fewer fish. These reefs appear to be prime candidates for coral restoration. However, the potential for coral to naturally recover should be examined before efforts to restore reefs are undertaken. Reef restoration is unnecessary if the population has the capacity to recovery through natural means. We observed that the rate of settlement of Acropora spat in the Caribbean is much lower than the spat settlement rate of several other Caribbean coral families and much lower than Acropora spat settlement rates in the South Pacific. A very low percentage of apparently healthy colonies of A. cervicornis possessed developing gametes in July 2005, a month before spawning. It appears that the long-term survival of remnant A. cervicornis populations is threatened unless successful sexual reproduction is restored. Several techniques were used to test transplant methods for restoring A. cervicornis populations. The mean survivorship and growth rate of one technique was >75% and nearly 250% per annum, respectively. Working with hotel operators, environmental groups, and local fishers, we are attempting to reintroduce A. cervicornis to sites where it previously existed and increase coral biomass and complexity at these sites. Reefs with greater A. cervicornis biomass have larger edible fish populations. With localized protection of these restored reefs we anticipate an increase of larger edible fish not only on reefs within the restored protected areas but also on adjacent reefs. Furthermore, increased coral and fish biodiversity improves the attractiveness of the reef community for divers and snorkelers. Rev. Biol. Trop. 54 (Suppl. 3): 105-116. Epub 2007 Jan. 15.
Key words: coral reefs, coral restoration, reproduction, Jamaica, Caribbean.
Just three decades ago, Acropora cervicornis (Lamarck, 1816) was one of the most important reef-building corals on Jamaican reefs (Goreau and Wells 1967). However, this species has declined throughout the region, becoming locally extinct on many reefs (Knowlton et al. 1990). Hurricane Allen in 1980, reduced the A. cervicornis population at the West Fore Reef at Discovery Bay from 53 ± 10% to a negligible presence (Woodley et al. 1981, Crawford 1995). Eight years later when Hurricane Gilbert struck Jamaica, the few recovering stands of Acropora that had survived Hurricane Allen were smashed again (Woodley 1991). Incidents of predation and disease further reduced the A. cervicornis population on the north Jamaica coast (Bruckner et al. 1997) with no increase 25 years later (Wapnick et al. 2004, Quinn and Kojis 2005). The past dominance of Caribbean acroporids was attributed to their rapid growth rates and asexual reproduction by fragmentation (Tunnicliffe 1981).
The Caribbean wide decline of Acropora corals in recent decades has serious consequences for coral reef biodiversity, coastal geology, and the fisheries and tourism economies of the region. The decline is so serious that on May 4, 2006, NOAA announced its final determination to list both A. cervicornis and A. palmata as threatened under the Endangered Species Act. This is a first for reef-building scleractinian corals globally.
Even on reefs where measures to address the root causes of coral decline have been implemented, acroporan populations do not appear to be recovering, as larval recruitment is very sparse (Quinn and Kojis 2005). A likely hypothesis for the lack of larval-based recovery is that there are too few surviving colonies of A. cervicornis and A. palmata to produce the numbers of planulae required to initiate their recovery (Kojis and Quinn 1993). The long-term survival of Caribbean acroporans is threatened unless successful sexual reproduction occurs or successful methods of restoration are implemented.
Acropora cervicornis, in particular, is not recurring where it was formerly common (Wapnick et al. 2004). Given the historically low levels of sexual recruitment of A. cervicornis throughout the Caribbean (Bak and Engle 1979, Rylaarsdam 1983, Sammarco 1985), we tested the hypothesis that acroporan larval recruitment on the north coast of Jamaica is insufficient to promote the short or medium term recovery of this species.
Experimental Restoration Efforts on Jamaican Reefs: Efforts to rehabilitate select coral populations have varied from very expensive deployment of artificial structures (Edwards and Clark 1992) to simple coral culturing activities (Franklin et al. 1998, Yap et al. 1998, Lindahl 2003).The less expensive efforts involve reattachment of coral fragments in the expectation that the fragments will simulate a natural community as the fragments grow. Transplantation of coral fragments functions as a catalyst for recovery by increasing the live coral cover and topographic complexity on reefs where scleractinian coral cover is low and thereby supporting a larger, more diverse reef fish community (Quinn and Kojis 2007).
Given the lack of recovery and in some cases local extirpation of Acropora cervicornis on reefs of Discovery Bay we examined the feasibility of restoring populations by growing new colonies from small fragments of healthy populations of this species. Together with local stakeholders, including the Friends of the Sea, Montego Bay Marine Park, Northern Jamaica Conservation Association and several local hotels, we worked to restore the biomass of A. cervicornis on select reefs sites on the Jamaican north coast. The strategy involved propagation of A. cervicornis branches taken from healthy populations for experimental propagation in areas where large populations once existed.
Materials and methods
Coral Recruitment and Reproduction: Coral recruitment arrays were constructed by attaching four 208 cm2 unglazed terracotta tiles to a PVC array. The tiles were smooth on one side and had 12 ridges on the other side. Two tiles were oriented horizontally and two vertically on the array. The tiles were ~ 0.8 – 1 m above the substrate. The arrays were initially installed at the CARICOMP coral reef site on the West Fore Reef, Discovery Bay, Jamaica (WFR) (18º28.17N; 77º24.49 W) in late March / early April 2001 and were replaced in October 2001 ("summer 2001" sampling period), April 2002 ("winter 2001 / 02"), October 2002 ("summer 2002"), in April 2003 ("winter 2002 / 03"), and October 2003 ("summer 2003"). Paired arrays were positioned initially at 3, 9, 14, 19, 26 and 33 m depth. In April 2003, a third array was deployed at 3m, and paired arrays deployed at 5 m WFR (18º28.397 N; 77º27.83 W), and three other sites near Discovery Bay – 35 m Long Term Site (LTS) (18º28.44 N; 77º27.47 W), 9 m Dairy Bull (18º28.04N; 77º23.10 W), and 3 m and 10 m at Columbus Park Reef (18º27.49N; 77º24.50 W) and grouped in the "summer 2003" deployment period.
After removal from the site, the tiles were fixed in formalin and bleached. Scleractinian and milleporan corals were counted (standardized to number recruits m-2) and identified to family where possible using a binocular microscope.
Acropora cervicornis broadcast spawns once annually in summer, releasing gametes into the sea for external fertilization. In order to determine whether colonies of this species had ripe gametes, colonies were sampled in July 2004, before the predicted time of spawning. Twenty-two branches of A. cervicornis colonies were sampled at least 10 cm from the apical polyp from healthy colonies >50 cm in diameter at Diary Bull, Jamaica and Coki Beach, St. Thomas, U.S. Virgin Islands (USVI) (18º28.12 N; 64º51.35 W). The following year branches from 20 different colonies at Coki Beach were sampled on 26 July 2005, 5 days after the full moon. A further 25 branches were sampled from different colonies on 8 and 14 August 2005, 18 and 24 days after the full moon, respectively. In Jamaica, 50 branches were sampled on 20 July before the July full moon and 25 branches on 23 August, four days after the August full moon.
Only healthy colonies were sampled. Colonies exhibiting White Band Disease were not sampled. Most branches were broken near the dead branch base. Acroporan polyps near the apical polyp are often not reproductive. Branch diameters were measured at the widest point and branches were examined for visible gonads by breaking each branch perpendicular to the long axis of the branch with a hammer and chisel at a minimum of three locations starting with the basal portion of the branch. Both ends of each broken branch section were examined for ovaries and testes using a dissecting microscope. At least ten polyps in each sample were examined. The percentage of reproductive colonies was calculated.
Coral Transplantation
1 Effects of Sampling on Source Population: Several large populations of Acropora cervicornis were located near Discovery Bay at Dairy Bull (Idjadi et al. 2006) and East Rio Bueno. These populations were used for coral restoration experiments similar to those done in Puerto Rico (Bowden-Kerby 2001).
An important objective of a reef rehabilitation program is to ensure that the impact on the donor population is minimal and the growth rate remains similar to that prior to the breakage event. In June 2004, 160 fragments of Acropora cervicornis (5 - 14 cm in length) were collected from colonies at Dairy Bull at a depth of ~9 m. One fragment was collected per colony. Buckets filled with sea water were used to temporarily house the fragments while they were transported by boat to the laboratory. To test the impact of removing branches on source colonies, we attached colored cable ties to 40 source colonies ~2 cm below where fragments were broken off in September 2004. An additional 40 source population colonies, which had no branches removed, were used as a control and were tagged with different color cable ties at approximately the same position as on donor colonies but on broken branches. Three months later the colonies were reexamined to determine whole colony mortality rates.
2 A-frame Technique: Five Acropora cervicornis fragments were attached to each side of 14 wire mesh "A-frames" using plastic cable ties in June 2004. The A-frames were made of plastic coated wire mesh (parallelogram mesh 3 x 6 cm and rectangular 20 x 20 cm) cut to a size of 0.8 x 1.2 m, and bent in the middle at 90º (Fig. 1). The frames were tagged and two metal weights were attached to the end of each frame for increased stability. The A-frames were then deployed at: Columbus Park Reef (CPR) (5 frames, N = 50 fragments) - depth 6 m; East Back Reef (EBR) (5 frames, N = 50 fragments) (18º28.09N; 77º24.15W) - depth 4 m; the Blue Hole (BH) (3 frames, N = 30 fragments) (18º28.10 N; 77º24.51W) - depth 2 m, Canoe Channel (CC) (3 frames, N = 30 fragments) (18º28.15N; 77º24.54W) - depth 2 m; and outside the bay at West Fore Reef (WFR) (4 frames, N = 40 fragments) (18º28.19N; 77º24.53W) - depth 6 m. At Columbus Park Reef, the water was turbid with a visibility frequently <5 m and the bottom was composed of red / brown silt. A dense population of A. cervicornis had previously existed at this site for at least 6 000 year b. p. (Wapnick et al. 2004). The EBR site was subject to breaking waves and the water was clear (typically ~10m visibility). The substrate was sand and rubble with some Thalassia testudinum (Banks ex Köing, 1805). The A-frames at BH were located in a back reef, lagoon environment with slightly turbid water (typically < 9 m visibility) and mixed sand and rubble substrate. The CC site was just behind the reef crest at the WFR in an inter reef sandy area. This site received clear ocean water as waves broke over the crest and consequently had the clearest water of all the experimental sites. The sites substrate was sand with T. testudinum surrounded by predominantly algal dominated remnant coral structures. Black plastic shade cloth was suspended over the A-frames at the shallow Canoe Channel site to reduce light intensity and approximate light levels at the 9 m depth of the source population.

Transplanted branch fragments were monitored at irregular intervals for survivorship and growth. The percentage survivorship was calculated by dividing the number of live fragment by the total number of original fragments. We also recorded the fraction of each branch that survived. Missing fragments were counted as non surviving. Growth was measured from the fragment base to the apical polyp of the main branch and, then for each subbranch on a fragment, from the branching point to the apical polyp. The lengths of the main branch and each subbranch were added together and linear length and percentage growth calculated.
3 Cement Base Technique: A 50% - 50% cement - sand mixture was flattened, pressed and dried in the shape of a ~ 5 cm circular disk to construct 211 cement disks. Two holes were punctured through the disk before it hardened. After hardening, a nylon monofilament line was looped through the holes and one small (< 7 cm) fragment of Acropora cervicornis (N = 190) or A. prolifera (N = 21) were secured to each of the disks. Acropora prolifera is very similar to A. cervicornis genetically and morphologically, but is much less common than A. cervicornis. It was used to compare its robustness with A. cervicornis. Disks were secured to a wire mesh sheet with the nylon monofilament line. The fragments were monitored for survivorship and growth as described in the A-frame experiment.
4 Line Experiment: Sixteen Acropora cervicornis fragments were collected from colonies at Dairy Bull in June 2004. After ~ 4 hrs in shaded buckets, the fragments were attached to monofilament nylon lines at a depth of ~1.5 m and suspended ~ 0.5 m above the sandy substrate by reinforcing bar embedded in the sand at Canoe Channel. The line was shaded by shade cloth to approximate the reduced light levels at the source population.
The experiment was repeated in September 2004 with 40 fragments also collected at Dairy Bull. This time the fragments were deployed on lines in the Canoe Channel within 30 min of being collected. The cultures were monitored for both growth and survivorship. Growth rates and survivorship was determined as described in the A-frame experiment.
Results
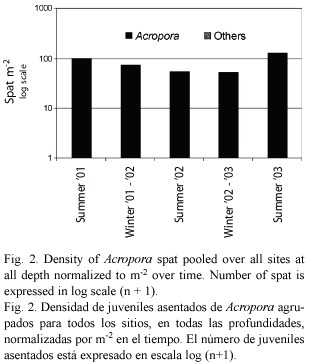
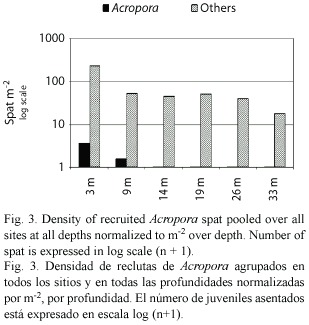
Coral Recruitment and Reproduction: Over a 42 month period only seven acroporid spat recruited to terracotta tiles deployed on the entire depth range of arrays on the WFR – three spat settled in "summer 2001" (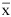
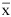
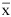
In 2004, 8% of the Acropora cervicornis sampled at Dairy Bull, Jamaica (N = 50) and 16% at Coki Beach, St. Thomas, USVI (N = 50) were reproductive. All of the reproductive samples contained both eggs and testes. Spawning was not observed, but it was presumed to have occurred just after the full moon in July 2004 as samples taken just before the August 2004 full moon did not have any gametes.
In 2005, the proportion of Acropora cervicornis colonies at Coki Beach with gametes was more than twice the proportion at Dairy Bull. Only 35% (N = 50) of colonies sampled in July and 0% (N = 50) of those sampled in August at Dairy Bull were reproductive compared with 70% in July (N = 50) and 80% in August (N = 50) at Coki Beach. Individual branch diameter from Dairy Bull samples were smaller (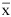
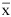
Coral Restoration
1. Effects of Sampling on Source Population: Of the 40 tagged colonies which had branches removed at Dairy Bull, 29 were relocated after three months. Of these 29 colonies, eight were dead (28% mortality) and 21 were alive (72% survival). In contrast, 32 of the 40 originally tagged control colonies were relocated after three months. Eight colonies were dead (20% mortality) and 24 colonies were alive (80% survival). A survey of the Acropora cervicornis population at Dairy Bull Reef found that 16% of the colonies (N = 200) had dead tips on colonies otherwise alive. This mortality is likely caused by bearded fire worm (Hermodice carunculata) predation. White Band Disease was also present at the base of 9% of the branches. Damage on colonies caused by damselfish (Stegastes fuscus) bites occurred on 5% of the colonies.
2. A-frame Experiment: Survivorship of transplants varied among sites. After 62 weeks Columbus Park Reef had the highest percent survivorship of Acropora transplants - 68%, with 29% at Blue Hole, and 4% at Canoe Channel (Fig. 4).The low survival rate at the Canoe Channel was a result of wave action from Hurricane Ivan, which passed south of Jamaica in September 2004.
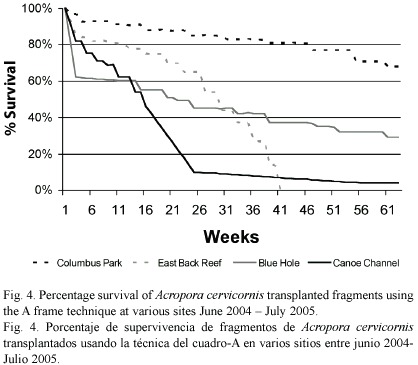
Transplants with the fastest growth rates were the ones with the greatest number of new branches. There was a high variability among fragments in the number of apical polyps produced. Each apical polyp has the potential to yield a new branch. About 83% of transplants developed at least two new apical polyps usually forming new branches, 8% of the transplants developed only one new apical polyp and 9% of the transplants produced no new apical polyps. The average linear growth rate of a single branch was 54% lower than the growth rate of transplants with many new branches. After 39 weeks, the overall mean net growth rate of the remaining live transplants on the A-frames was 7.3 cm (s. e. ± 7.0 cm; N = 63), ranging from a mean of 1.1 cm (s. e. ± 0.6 cm; N = 16) at Canoe Channel to 15.3 cm (s. e. ± 4.6 cm; N = 40) at Columbus Park (Fig. 5). This represents an overall mean growth rate of 0.19 cm week-1 or an annualized rate of 9.7 cm year-1 (s. e. ± 6.3 cm year-1).
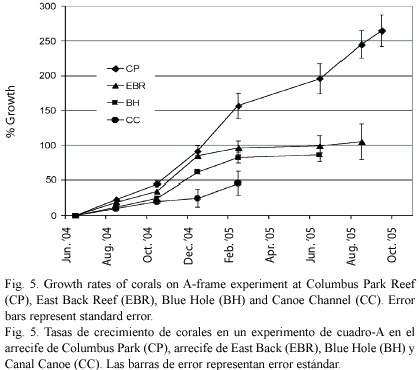
After this set of measurements a series of extreme events including hurricanes, higher than normal sea water temperatures and later coral bleaching occurred. Hurricanes Dennis and Emily in July 2005 demolished the A- frame experiment at the WFR and EBR. A massive coral bleaching event commenced in September 2005 and continued through June 2006 (Quinn and Kojis in press). By December 2005, nearly all the corals in the experiment had died and only small sections on a few colonies were alive. The results from the surviving colonies were not considered typical and will be reported in a separate paper.
Transplants on A-frames on sand did considerably better than when frames were placed on rock or rubble. Transplants on A-frames placed on rock or rubble were preyed upon by Hermodice fire worms, as well as Stegastes damselfish. However, if the frames were far from the reef, algal overgrowth became a problem due to a lack of grazing by herbivores. The method of weighing the frames down with cement blocks at some sites resulted in the inadvertent creation of ideal Stegastes habitat, with subsequent negative impacts to the corals. The size of the wire mesh also affected the outcome, with small mesh becoming ideal shelter for the damselfish and allowing more surface for the algal farming activities of the fish. Solutions to these problems were implemented in the second set of experiments in January and March 2005 when wire mesh with apertures of 20 x 20 cm were located on bare sand 1-3 m from rocky areas. These modifications inhibited colonization of the A-frames by damselfish by reducing the surface area for algal farming activities, and maintained a sufficient distance from the reef to reduce predation by fire worms and still allow herbivorous fish to visit the frames regularly for cleaning.
The source population at Dairy Bull experienced 100% bleaching from late September to mid December 2005. In January 2006, over 90% of the A. cervicornis population had died and was covered with filamentous algae. By June 2006 <2% of the population was alive. Many of the remaining colonies were being preyed upon by Coralliophila abbreviata or showed signs of bearded fire worm (Hermodice carunculata) predation.
3 Cement Disk Experiment: The survival and growth of the colonies on the cement disks was high during the first couple of months. Colonies quickly grew and established multiple growing branches. However, survivorship after 37 weeks was only 19% (only 40 of the original 211 fragments were still alive). All of the Acropora prolifera died within the first 12 weeks. Mortality was primarily owing to damage to colonies by Hurricane Ivan, which passed south of Jamaica in September 2004. Forty weeks after transplantation, the mean annualized growth rate of a branch, calculated by summing the growth of all surviving branch lengths on a colony, was 23 cm year-1 (s. e. ± 9.2 cm year-1). After 14 months (August 2005), many colonies exhibited partial mortality of branch tips and <10% of the surviving colonies exhibited significant growth. Several of the colonies developed multiple apical polyps but had little growth.
4 Line Experiment: Within two days of the start of the June 2004 experiment most of the transplants had bleached. After five days, we observed 100% bleaching and the start of algal growth over the white tissue. None of the colonies recovered. Five days after the September 2004 transplantation, none of the coral fragments had exhibited bleaching in spite of water temperature being at its annual maximum (Quinn and Kojis 2003). By the 12th week, 86% of the fragments were alive and an apical polyp had formed over the broken end of over 66% of the surviving fragments and tissue had overgrown the broken end of the remaining surviving fragments. All of the surviving fragments had also developed at least one new branch. After 26 weeks, the survivorship rate of the second attempt was 32%. Considering that Hurricane Ivan passed by Jamaica two weeks into the experiment, this is a rather low mortality. In August 2005, the remaining 16 colonies (40% survival rate) were doing extremely well with a mean annualized growth exceeding 25% (s. e. ± 42%) and mean annualized linear growth of 21.0 cm (s. e. ± 7.5 cm). Colonies were vigorously growing from both ends and had numerous new branches with no sign of Hermodice carunculata predation or Stegastes fuscus bites.
Discussion
Recovery of Acropora cervicornis in Jamaica has not been observed in the nearly 30 years since the onset of the decline of the coral reefs on the north coast of Jamaica., except at Dairy Bull (Idjadi et al. 2006) which lasted only a couple of years. The reasons for this lack of recovery are myriad, but foremost among them is the lack of new recruits most likely because of the severe reduction in gamete producing adults (Quinn and Kojis 2005), but also because of the life history of this species (Rylaarsdam 1983). The reef at Dairy Bull had recently been touted as an example of a reef that had recovered (Idaji et al. 2006). However, this recovery appears to be ephemeral, as surveys conducted in May/June 2006 revealed that <2% A. cervicornis population at Dairy Bull was still alive. The population of A. cervicornis at East Rio Bueno had much less mortality and still had 39% coral cover (Jan 2006), down from 47% coral cover (April 2004) (Quinn and Kojis unpubl. data).. The surviving colonies from both Dairy Bull and East Rio Bueno may be more tolerant of elevated sea water temperatures and therefore better candidates for future restoration experiments once they have completely recovered both their health and abundance. The higher survival rate of A. cervicornis at Rio Bueno may be a function of the more turbid water at this site, which may have protected the colonies from UV radiation which possibly works synergistically with temperature to cause bleaching.
Even when isolated populations recover, as in Dairy Bull (Idaji et al. 2006), their existence is threatened by bleaching and predation by Coralliophilia, Hermodice and Stegastes. As well, other factors such as a decline in water quality, recent and widespread outbreaks of coral diseases, are also inhibiting recovery and these have been recently reviewed by Precht and Aronson (2006).
One of the factors linked to the decline that would directly affect coral spat recruitment is the decline of grazing Diadema sea urchins (McClanahan 1994). Studies of the role of Diadema in coral recruitment have provided conflicting results. While recent work suggests that Diadema is important in clearing space for the settlement of coral spat (Edmunds and Carpenter 2001), work by Sammarco (1980), performed prior to the mass die off of Diadema found that the highest coral recruitment and diversity occurred at the lowest Diadema densities. It was at the highest Diadema densities and, concomitantly lowest algal cover, that Sammarco (1980) found that coral recruitment was greatly depressed owing to the biological disturbance created by the echinoids abrasive grazing mechanism. The presence of some Diadema increased coral recruitment by removing algae and making space available for settlement, while high densities of Diadema, particularly in the period just after settlement, was likely to remove spat and reduce settlement success. High algal cover does not seem to inhibit growth of A. cervicornis once it is established on a reef. At East Rio Bueno, Jamaica, a flourishing Acropora cervicornis population (~47% coral cover) exists with colonies commonly attached to the substrate even thought the reef is algal dominated (Quinn and Kojis unpubl. obs.).
Acropora spat settlement rates in the Caribbean are commonly lower than other many coral families (Rylaarsdam 1983) in the region and much lower than Acropora settlement rates on reefs in the South Pacific (Kojis and Quinn 2001, Quinn and Kojis 2006). It appears that the long-term survival of remnant A. cervicornis populations is threatened unless successful sexual reproduction is restored. At an Acropora workshop (Bruckner 2002), experimental restoration techniques were recommended as a mean to restore Acropora populations.
Acropora cervicornis is suitable for transplantation because of its fast growth rate and branching habit. Of the surviving A. cervicornis colonies in an experiment in Tobago, the growth rate ranged from 6.5 - 11.7 cm year-1 (Laydoo 1996) which is comparable to our annualized growth rates of 7.3 cm year-1 – 15.3 cm year-1, 
The effects of hurricanes, the bleaching event and A-frame location disrupted the experiments and rendered the results inconclusive. Owing to these events, colony mortality on the line and cement experiments were high and, therefore, these colonies were never transplanted from the experimental site to designated recovery reefs where survival and growth could be assessed. Source colonies, where the collection of fragments occurred, had only a slightly higher mortality rate than control colonies. A net benefit analysis of the transplantation efforts is not possible owing to the disruption to the experiments by natural events. The continued occurrence of many of the forces that contributed to the decline of the species years ago should be considered as a persistent threat to coral populations and their future impact assessed before any extensive coral restoration endeavor is attempted.
The survival of the transplanted colonies varies with site. In Tobago, West Indies, A. cervicornis transplantation has been done with survival of 30-35% of the colonies eight months after transplantation. Comparing these results with this experiment over the same period of time, one site (CC) had a lower survival rate (88%), two sites had a slightly higher survival rate - 37% (EBR) and 42% (BH), and one site (CPR) had a much higher survival rate (83%). The high survival at CPR was unexpected as the site was the most turbid and most affected by land runoff. However, the depth at the site more closely approximated the source population site and an extensive population of A. cervicornis had historically existed there (Wapnick et al. 2004).
The results from this study suggest that the success of restoration is very site specific. Local variation of abiotic conditions like wave exposure, salinity, depth, turbidity can influence survival. As well biotic influences such as abundance of fire worms and damselfish can influence growth and survival.
Considerations for Reef Management: The impact of humans touching corals during the restoration process has been raised. While it is believed by some people in the water sports industry that coral is damaged by touching and that humans can spread diseases by touching live coral, there is actually little evidence to support this. In a study in the Florida Keys, the average diver had ten contacts by hand or fins within a 45 min dive (Talge 1992). In later examination of the gross morphology and subsequent histological studies of corals that had been experimentally touched, Talge (1992) did not see any evidence of damage to the polyps or interference with their reproductive cycles. In our over 30 years of handling corals we have never noticed any subsequent disease or deleterious effects on colonies that we have lightly touched while carrying out research. It is not likely that carefully conducted manipulative experiments are likely to increase diseases in coral populations, particularly if done when water temperatures are cooler.
This project supported existing management strategies and heightened coral reef conservation awareness during workshops, providing information on the status of corals and highlighting the importance of protecting reef corals. The research sites were located within existing coral reef conservation areas, taking advantage of the protection afforded in these conservation areas, while helping contribute to the recovery of biodiversity within the management areas.
On Indian Ocean reefs, transplanted arborescent acroporid corals have been shown to enhance fish abundance and diversity (Lindahl 2003). Acropora cervicornis is one of only two open branching arborescent species in the Caribbean. It is a keystone species in the Caribbean and is of critical importance to biodiversity, fisheries, and tourism interests. It is particularly vital as fisheries habitat (Quinn and Kojis 2007) as it is the only large open-branched coral species of reef slope, back reef, and lagoonal environments in the Caribbean. Consequently, its severe decline represents a loss of essential fish habitat on many Caribbean reefs.
Important partnerships were formed with members of the tourism industry as their water sports staff participated in coral restoration workshops at DBML and some hotels even hosted their own workshops on their properties. The development of co management relationships is fundamental to the wider success of restoration efforts. The work has shown acceptance with resort water sports staff. They learned how to measure the growth and record the survival of the experiments as well as increasing their knowledge of marine conservation issues. We also expect that these activities increased awareness of the changes that have occurred on Caribbean reefs and encouraged the hospitality industrys appreciation of the natural beauty of coral reefs through their participation in reef conservation.
Due to the high cost and potential for failure associated with coral restoration, innovative techniques are needed. The restoration of complex coral reef habitat using cultivated fragments of fast growing corals, e. g. Acropora cervicornis, may assist in the restoration of coral reef ecosystem function, but is no substitute for reducing human threats to coral reefs.
Acknowledgments
This publication was made possible through support provided by the National Institutes of Health through the University of Mississippi under the terms of agreement No. R21 TW006645 funded by the Fogarty International Center and the National Institute for Research Resources for the International Cooperative Biodiversity Groups. The opinions expressed herein are those of the authors and do not necessarily reflect the views of the National Institutes of Health or the University of Mississippi. We are grateful for the primary financial assistance provided by USAID through the CWIP2 program and for the use of the facilities of the Discovery Bay Marine Laboratory, University of the West Indies and the Division of Fish and Wildlife, U.S. Virgin Islands. Additional financial support was provided by Tropical Discoveries Fund and Counterpart International. A Bowden-Kerby and M Stennett deserve particular recognition for their assistance in the initial phase of the coral restoration experiments. This is Discovery Bay Marine Laboratory publication #731.
Resumen
Las especies someras de Acropora se han vuelto poco comunes en los arrecifes de la costa norte de Jamaica debido a numerosos factores. En muchos arrecifes, las algas han tomado su lugar y el resultado es la pérdida de hábitat para muchas especies de peces e invertebrados y la pérdida de atractivo de los arrecifes, que tienen mucho menos peces. Estos arrecifes parecen ser excelentes candidatos para restauración de corales. Sin embargo, antes de emprender un esfuerzo de restauración, debe examinarse el potencial de los corales para regenerarse naturalmente, ya que si la población tiene la capacidad de recobrarse por sus propios medios, la restauración de arrecifes se vuelve innecesaria. Nosotros observamos que la tasa de colonización por adherencia a un sustrato duro de Acropora en el Caribe, es más baja que la de muchas otras familias de corales del Caribe y mucho menor que la de Acropora en el Pacífico sur. Un muy bajo porcentaje de colonias de A. cervicornis poseía gametos en desarrollo y estaba saludable en julio de 2005, un mes antes del desove. Al parecer, la supervivencia a largo plazo de las poblaciones remanentes de A. cervicornis está amenazada a menos que se restaure una reproducción sexual exitosa. Se utilizaron varias técnicas para probar los métodos de transplante para restaurar las poblaciones de A. cervicornis. La supervivencia promedio y la tasa de crecimiento resultantes de una de las técnicas fue de >75% y de casi 250% por año, respectivamente. Al trabajar con agentes de hoteles, grupos ambientalistas y pescadores locales, estamos tratando de reintroducir A. cervicornis en lugares donde anteriormente existía e incrementar la biomasa del coral y la complejidad de estos sitios. Los arrecifes con mayores biomasas de A. cervicornis tienen también mayores poblaciones de peces comestibles. Si se protegen estos arrecifes en restauración, prevemos que habrá un incremento de peces comestibles, no sólo en los arrecifes restaurados, sino también en los adyacentes. Más aún, el aumento en la biodiversidad de corales y peces mejora el atractivo del arrecife para los buzos.
Palabras clave: arrecifes de coral, restauración de corales, reproducción, Jamaica, Caribe.
References
Bak, R. P. M. & M. S. Engle. 1979. Distribution, abundance, and survival of juvenile hermatypic corals (Scleractinia)and the importance of life history strategies in the parent coral community. Mar. Biol. 54: 341-352. [ Links ]
Bowden-Kerby, W. A. 2001. Coral transplantation modeled after natural fragmentation processes: low-tech tools for coral reef restoration and management. Ph.D. Thesis, Univ. Puerto Rico, Mayagüez, Puerto Rico. 195 p. [ Links ]
Bruckner, A. W., R. J. Bruckner & E. H. Williams Jr. 1997. Spread of black-band disease epizootic through the coral reef system in St. Anns Bay, Jamaica. Bull. Mar. Sci. 61: 919-928. [ Links ]
Bruckner, A. W. 2002. Proceedings of the Caribbean Acropora workshop: Potential application of the U.S. Endangered Species Act as a conservation strategy. NOAA Technical Memorandum NMFS-OPR-24, Silver Spring, Maryland, USA. 199 p. [ Links ]
Crawford, J. A. 1995. The effects of Hurricane Allen at Discovery Bay, Jamaica, and a post-hurricane survey of the living hermatypic corals. Carib. J. Sci. 31: 237-242. [ Links ]
Edmunds, P. J. & R. C. Carpenter. 2001. Recovery of Diadema reduces macro algal cover and increases abundance of juvenile corals on a Caribbean reef. Proc. Natl. Acad. Sci. USA 98: 5067-5071. [ Links ]
Edwards, A. J. & S. Clark. 1992. Rehabilitation of coral reef flats using pre-cast concrete. Concrete 26: 16-19. [ Links ]
Franklin, H., C. A. Muhando & U. Lindahl. 1998. Coral culturing and temporal recruitment patterns in Zanzibar, Tanzania. Ambio 27: 651-655. [ Links ]
Goreau, T. F & J. W. Wells. 1967. The shallow–water Scleractinia of Jamaica: revised list of species and their vertical distribution range. Bull. Mar. Sci. 17: 442-453. [ Links ]
Idjadi, J. A., S. C. Lee, J. F. Bruno, W. F Precht, L. Allen-Requa & P. J. Edmunds. 2006. Rapid phase shift reversal on a Jamaican coral reef. Coral Reefs 25: 65-75. [ Links ]
Knowlton, N., J. C. Lang & B. D. Keller. 1990. Case study of natural population collapse: post hurricane predation of Jamaican staghorn corals. Smithsonian Contrib. Mar. Sci. 31: 1-25. [ Links ]
Kojis, B. L. & N. J. Quinn. 1993. Biological limits to Caribbean reef recovery. A comparison with western South Pacific reefs, p. 35-41. In R.N. Ginsburg (Compiler). Global Aspects of Coral Reefs, Health, Hazards, and History, Miami, Florida, USA. [ Links ]
Kojis, B. L. & N. J. Quinn. 2001. The importance of regional differences in hard coral recruitment rates for determining the need for coral restoration. Bull. Mar. Sci .69: 967-974. [ Links ]
Laydoo, R. S. 1996. Coral transplantation in reef management at Bucco Reef, south-west Tobago. Carib. Mar. Stud. 5: 67- 77. [ Links ]
Lindahl, U. 2003. Coral reef rehabilitation through transplantation of staghorn corals: effects of artificial stabilization and mechanical damages. Coral Reefs 22: 217-223. [ Links ]
McClanahan, T. R. 1994. Kenyan coral reef lagoon fish: effects of fishing, substrate complexity and sea urchins. Coral Reefs 13: 231-241. [ Links ]
Precht, W. F & R. B. Aronson. 2006. Death and resurrection of Caribbean coral reefs: A paleoecological perspective, p 40-77. In I. Côté & J. Reynolds (eds.). Coral Reef Conservation. Cambridge Univ., Cambridge, England. [ Links ]
Rylaarsdam, K. W. 1983. Life histories and abundance patterns of colonial corals on Jamaican reefs. Mar. Ecol. Prog. Ser. 16: 249 - 263. [ Links ]
Quinn, N. J. & B. L. Kojis. 2003. Variation in subsurface seawater temperature off Discovery Bay, Jamaica and the U.S. Virgin Islands. Rev. Biol. Trop. 51 (Suppl. 4): 181-187. [ Links ]
Quinn, N. J. & B. L. Kojis. 2005. Patterns of sexual recruitment of acroporid coral populations on the West Fore Reef at Discovery Bay, Jamaica. Rev. Biol. Trop. 53 (Suppl. 1): 83-90. [ Links ]
Quinn, N. J. & B. L. Kojis. 2006. Natural variability in the recovery of reef ecosystems, p. 83-997. In W.F Precht (ed.). Reef Restoration Handbook -The Rehabilitation of an Ecosystem Under Siege. CRC Press, Boca Raton, Florida, USA.
Quinn, N. J. & B. L. Kojis. 2007. Evaluating artificial means to increase Acropora coral populations and increase associated fish communities in Jamaica. Proc. 58th Meet. Gulf Carib. Fish. Inst., San Andres Island, Columbia, 7-11 November 58; in press. [ Links ]
Quinn, N. J. & B. L. Kojis. (in press) Assessing the potential for natural recovery and restoration of Scleractina coral after the 2005 coral bleaching event on Jamaicas reefs. World Resources Review. [ Links ]
Sammarco, P. W. 1980. Diadema and its relationship to coral spat mortality: grazing, competition, and biological disturbance. J. Exp. Mar. Biol. Ecol. 45: 245-272. [ Links ]
Sammarco, P. W 1985. The Great Barrier Reef vs. the Caribbean: comparisons of grazers, coral recruitment patterns and reef recovery. Proc. 5th Int. Coral Reef Congress, Tahiti 4: 391-397. [ Links ]
Talge, H. 1992. Impact of recreational divers on scleractinian corals at Looe Key, Florida. Proc. 7th Int. Coral Reef Symp., Guam 2: 1077-1182 [ Links ]
Tunnicliffe, V. 1981. Breakage and propagation of the stony Acropora cervicornis. Proc. Natl. Acad. Sci. USA 78: 2427-2431. [ Links ]
Wapnick, C., W. F Precht & R. B. Aronson. 2004. Millennial-scale dynamics of staghorn coral in Discovery Bay, Jamaica. Ecol. Lett. 7: 354-361. [ Links ]
Woodley, J. D., E. A. Chornesky, P. A. Clifford, J. B. C. Pearson, J. W. Porter, M. C. Rooney, K. W. Rylaarsdam, V. J. Tunnicliffe, C. M. Wahle, J. L. Wulff, A. S. C. Curtis, M. D. Dallmeyer, B. P. Jupp, M. A. R. Koehle, J. Niegel & E. M. Sides. 1981. Hurricane Allens impact on Jamaican coral reefs. Science 241:749-755. [ Links ]
Woodley, J. D. 1991. The effects of Hurricane Gilbert on coral reefs At Discovery Bay. Appendix 9, In UNEP Regional Seas Reports and Studies, No. 110. Pp. 135. [ Links ]
Yap, H. T., AR. M. Alvarez, H. M. Custodio & R. M. Dizon. 1998. Physiological and ecological aspects of coral transplantation. J. Exp. Mar. Biol. Ecol. 229: 69-84. [ Links ]