Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.54 suppl.3 San José Dec. 2006
A recent increase in the abundance of the echinoid Diadema antillarum in Dominica (Lesser Antilles): 2001-2005
S. C. C. Steiner1 & S. M. Williams1, 2
1 Institute for Tropical Marine Ecology Inc. (ITME), P. O. Box 944, Roseau, Commonwealth of Dominica; admin@itme.org
2 University of Puerto Rico, Mayagüez, Department of Marine Science, P. O. Box 9013, Mayagüez, PR 00681, USA.
Received 01-VI-2006. Corrected 02-X-2006. Accepted 13-X-2006.
Abstract: Diadema antillarum populations at many Caribbean locations have failed to recover from the pathogen-induced mortality events of the 1980s. It has become clear that the massive decline of this herbivorous urchin and the wide-spread absence of a population recovery lead to numerous long-term ecological consequences and reef degradation. While few quantitative studies on pre-mortality exist, great effort has been put forth to monitor remaining populations and their recovery. However, the patchy distribution of D. antillarum coupled with paucity of long-term studies based on the same methods applied at the same locations undermines the value of local as well as regional comparisons. In Dominica, ongoing quantitative assessments of D. antillarum began in 2001. Surveys of D. antillarum abundance are being carried out in 4-month intervals at six 100 m2 sites; spread over 38 km along the west coast. The density of D. antillarum has differed significantly between sites, ranging from 0.81(SD= 0.04) to 3.13 m-2 (SD= 2.10), and increased by 61.11% during the first five years of this study. Seasonal fluctuations, possibly related to spawning aggregations, are also evident. The current abundance of D. antillarum on Dominican reefs contrasts that of Caribbean locations with recorded incidents of mass mortality events, and in some cases resembles pre-mortality densities from the early 1970s. Prior to this study, no systematic quantitative assessments of D. antillarum were carried out in Dominica. It is thus unclear in what way Dominicas D. antillarum were affected by the mass mortality events observed elsewhere in the 1980s. The increase in D. antillarum density so far observed may thus be the recovery from a pathogen-induced disturbance or from Hurricane Lenny in November 1999. Locally, D. antillarum is important grazer on Dominicans reefs, where over-fishing has drastically reduced the number of herbivorous fishes. On a regional scale, the islands D. antillarum may represent a source of larvae for downstream islands, given the duration of the echinoplutei stage and a range of larval dispersal of hundreds of kilometers. Rev. Biol. Trop. 54 (Suppl. 3): 97-103. Epub 2007 Jan. 15.
Keywords: Diadema antillarum, population density, Dominica, Lesser Antilles, Caribbean.
Twenty years after Caribbean-wide mass mortalities of the herbivorous echinoid Diadema antillarum Philippi, its populations have failed to recover (Lessios 2005). Although the causative pathogen has not been identified, post-mortality surveys throughout the Caribbean clearly demonstrate that such mass mortality events lead to long-lasting cascading ecological consequences and phase shifts (Liddell and Ohlhorst 1986, Hughes et al. 1985, Hughes et al. 1987, Levitan 1988, Carpenter 1990, Hughes 1994). However, pre-mortality density data, necessary to gauge population recovery, is only available from a few locations (Lessios 1988a) with very distinct types of D. antillarum habitats and this complicates a direct comparison of data from different regions. Furthermore, there is a paucity of studies that encompass multi-year surveys carried out with the "same methods at exactly the same locations" (Lessios 2005). Given D. antillarums patchy distribution within and between reefs, as well as site-specific disturbance regimes, it is essential that new and ongoing studies be based on long-term surveys and consistent methodology applied at sites broadly spread along a particular coastal region or island.
Long-term quantitative assessments of D. antillarum in Dominica began in 2001 and have since then been carried out in 4-month intervals, at six locations along the islands west coast. The purpose of this ongoing project is to establish a quantitative data base on D. antillarum for Dominica in order to (a) weigh the severity of possible future abundance fluctuations, (b) provide complementary data to gauge local "reef health", and (c) elucidate the status of D. antillarum in the Eastern Caribbean within a broader regional context of recorded mortality events. This report presents emerging results based on data recorded up to July 2005.
Materials and methods
At each of six sites, spread over a distance of 38 km along the west coast (Fig. 1), the end points of two 25 m long transects were permanently marked by concrete nails installed at depths ranging from 1-5 m. The selection of west coast sites was based upon the preliminary finding that D. antillarum were virtually absent along northeastern and eastern side of Dominica (windward). Each site was monitored in 4-months intervals, starting in July 2001, by mounting transect lines between the permanent makers and measuring the density of D. antillarum, counting all specimens whose test (not just spines) were at least partially within 1 m of either side of the transect. Thus, 100 m2 were monitored at each site. Surveys were carried out during the day, between 6-11 am. Due to poor visibility and/or turbulent seas, surveys could not be carried out at Tarou Point and Scotts Head in November 2001, Taby Bay and at one of two transects at Tarou Point in July 2002, and Tarou Point in July 2003.
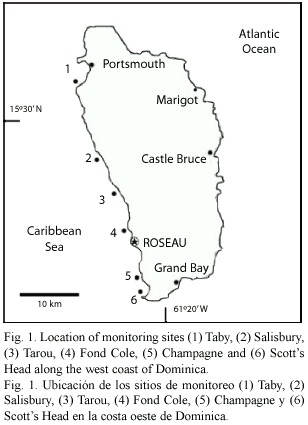
Density data were analyzed by using analysis of variance (ANOVA) with a randomized complete block design (RCB). Two factors which are repeated measures were included: seasons (March, July and November) and years (2001-2005). Each transect from each site represents one block (12 blocks in total).
Results
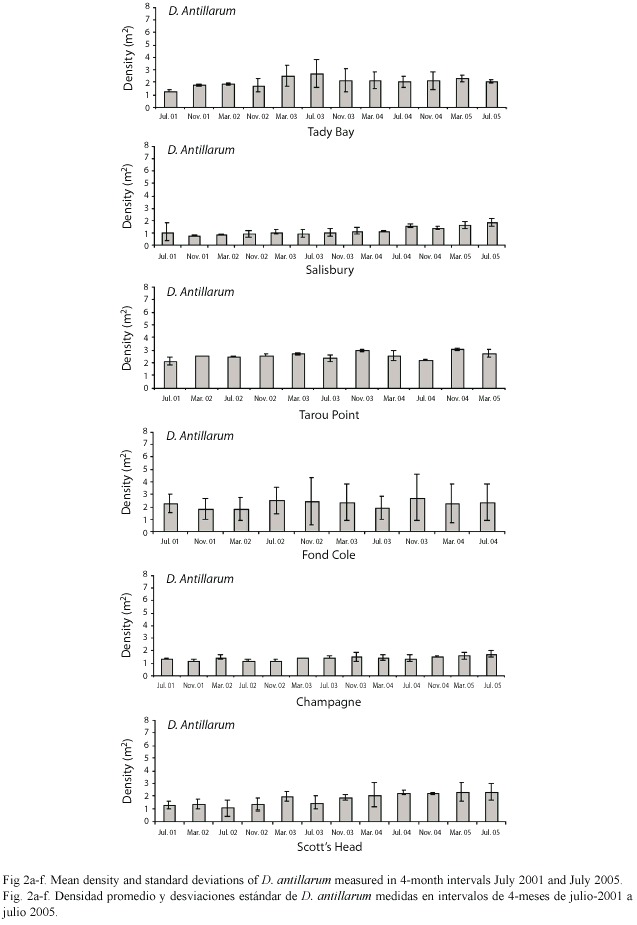
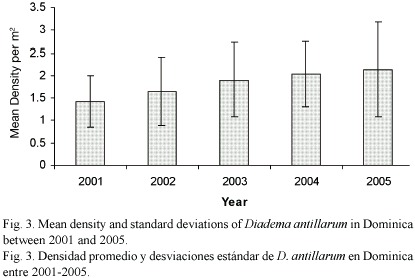
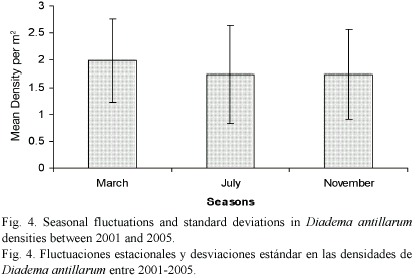
The mean density of D. antillarum per site (100 m2) differed significantly (ANOVA, p<0.0001) and ranged from 0.81 m-2 (SD= 0.04) at Salisbury in November 2001 to 3.13 m-2 (SD= 2.10) at Fond Cole in July 2005 (Fig. 2a-f). Between 2001 and 2005, mean densities increased at all sites by 29.06% at Champagne to 83.68% at Salisbury. The overall mean densities for each of the five years also differed significantly (ANOVA, p<0.0001) and increased by 61.11% with annual increases ranging from 6.8% (2003-2004) to 16.6% (2001-2002) (Fig. 3). On a seasonal scale, densities peaked in March and a significant difference (ANOVA, p= 0.05) was detected between the March, July and November surveys (Fig. 4).
Discussion
Five years into this ongoing study, the number of D. antillarum has increased at all sites by 29.06% to 83.68%, leading to an overall density increase of 61.11% between July 2001 and March 2005. Because the study sites are spread over 38 km, this recent increase is representative for Dominicas west coast shallow (11-5 m) D. antillarum habitats, and not a localized incident. The density values recorded in Dominica since 2001, contrast earlier findings from other Caribbean locations which were surveyed after the mass mortality of D. antillarum in the early 1980s. In St. John 0.27 m-2 Diadema were observed by Karlson and Levitan (1990), 0.51 – 0.60 m-2 in the Florida Keys by Forcucci (1994), 1.15 m-2 in Jamaica by Moses and Bonem (2001) and 0.33-0.80 m-2 in the Dry Tortugas by Chiappone et al.(2001). The differences may be related to actual differences in the population densities due to habitat availability and quality, or differences in sample size and frequency. Interestingly, Dominican density values are closer to pre-mortality density data from Florida in the 1970s (Bauer 1980) and Panama in the early 1980s (Lessios 1995).
Quantitative data on the abundance of D. antillarum in Dominica from times preceding the large scale die-offs reported in other parts of the Caribbean do not exist. It is therefore difficult to assess whether the current population is the result of a successful recovery in progress, such as the ones reported for St. Croix (Miller et al. 2003) and Puerto Rico (Weil et al. 2006), or whether the die-offs of the early 1980s did not affect Dominica, or only marginally. If the same causative pathogen which affected D. antillarum of the western Caribbean was introduced to Dominica and other Windward Islands (e.g. Barbados, see Hunte et al. 1986), this may have occurred via ballast waters (Steiner and Williams 2006) as reported for Curaçao by Bak et al. (1984). Surface currents are less plausible mechanisms for the spread of this pathogen from Central America to Dominica (see Lessios 1988a, Phinney et al. 2001). Alternatively, storms may have caused drastic decreases in the abundance of D. antillarum in Dominica in the past. The islands narrow shelf and reef habitats are characterized by the lack of energy-dissipating structures (Steiner 2003), such as wide fringing reefs, barrier reefs or mangrove forests. Hence, D. antillarum has little structural protection from storm-related disturbance, both from the sea (surge) and from land (sediment run off). The density increase noted since 2001 in this study may thus be part of a recovery from Hurricane Lenny 1999 (Steiner and Williams 2006) because no major storms have affected Dominica since then. Locally, the current population represents an important grazing component, controlling algal growth in reef communities (Steiner and Williams 2006).Low densities of herbivorous reef fishes (Komoroske 2002) and fish predators of D. antillarum (Pettersen 2002) related to over-fishing (pers comm., Guiste, Fisheries Division) may account for this situation. Thus, a drastic reduction in the D. antillarum population of Dominica could lead to a pronounced increase in algal cover on many reefs.
The density peaks observed during March surveys may be linked to aggregative behavior (see Levitan 1988) among spawning D. antillarum. In Dominica spawning D. antillarum have been observed aggregating on elevated reef structures as early as February, and this was was not a strictly nocturnal activity. Given the inherent spatial competition in this reproductive behavior set in a structurally complex reef environment, the time frame of temporary relocations (cryptic towards more exposed areas) may exceed that of diurnal movements related to grazing, thus leading to the seasonal density peaks recoded here. These observations are in congruence with Lewis (1966) findings of peaks in gonad volume in D. antillarum during March-April, and a completion of spawning by June-July in Barbados. Consequently, the lower densities of D. antillarum in July and November, recorded in this study, may be the result of "dismantling" spawning aggregations. Considering a duration of echinopluteii in D. antillarum between 4-7.5 weeks (Hunte and Younglao 1988, Carpenter 1990, Eckert 1998) and post-settlement growth rates of 1 mm per week (Bak 1985), the presence of a greater number of small (< 1cm) D. antillarum during July surveys (Steiner and Williams 2006) also indicates that spawning commenced several months earlier, as witnessed in Dominica and/ or upstream islands (e.g. Martinique).
In a regional context, Dominicas D. antillarum population is a potential source of larvae for downstream islands such as The Saints which are only 18 km to its North. The range of larval dispersal is known to be hundreds of kilometers (Lessios 1988a) and downstream islands may thus be stepping stones towards the Leeward Islands and the Greater Antilles. It remains unclear, however, why test diameters in Dominica (Steiner and Williams 2006) are similar to those recorded prior to the mass mortality in St. Croix (Ogden et al. 1973, Carpenter 1990), but larger than those recorded in Boca Raton and Cozumel (Bauer 1980), and Panama (Lessios 1995), where test sizes have remained "small", 20 years after mass mortality events (Lessios et al. 1984, Lessios 1988b, Carpenter 1990, Miller et al. 2003). If "small test sizes" is a telling symptom or long –lasting effect in D. antillarum populations which suffered from a mass mortality within the last 20 years, the abundance of comparatively "large" D. antillarum is not. Hunte et al. (1986) report that pathogen-induced mortality events affected adult D. antillarum (> 4 cm) more severely than juveniles. It is thus possible that Dominicas D. antillarum were affected less severely and recovered at a quicker rate as the common presence of adult echinoids (Highsmith 1982, Pearce and Scheibling 1990), specifically D. antillarum (Hunte and Younglao 1988, Forcucci 1994), are among the main cues for larval settlement.
A closer look at the biotic and abiotic factors shaping D. antillarum populations will be necessary to seprate site-specific phenomena from regional events (see also Cowen et al. 2000, Edmunds and Carpenter 2001, Lessios 2005). For example, D. antillarum habitats from Dominica (narrow shelf, marginal reef systems and little carbonate substrate) different greatly from D. antillarum habitats in San Blas, Panama (wide shelf, extensive reef systems and carbonate frameworks) resulting in different combinations of habitat parameters (e. g. reef rugosity, predator abundance, oceanic and terrestrial influences) which can be expected to lead to different carrying capacities of D. antillarum. In Dominica, "habitat preference" (within a spectrum of turbulence regimes) of D. antillarum is highlighted by its virtual absence on the windward side of the island, while the leeward side harbors a well developed population. Nevertheless, longterm surveys based on consistent methodology applied at the same locations are still fundamental prerequisites for investigations of the status of D. antillarum and its role on Caribbean reefs, and should be supported.
Resumen
Las poblaciones de Diadema antillarum de muchas localidades del Caribe, no se han recuperado después de los eventos de mortandad, inducidos por un patógeno, en la década de 1980. Se ha podido demostrar que la muerte masiva de este erizo herbívoro y la ausencia de recuperación, han tenido consecuencias ecológicas que han producido la degradación de los arrecifes. Existen pocos estudios pre-mortalidad, pero se han realizado grandes esfuerzos para monitorear las poblaciones restantes y su recuperación. Sin embargo, la distribución en parches de D. antillarum combinado con pocos estudios a largo plazo usando la misma metodología en los mismos sitios, mina la posibilidad de comparaciones locales y regionales. En Dominica, el monitoreo cuantitativo de D. antillarum empezó en 2001. Cada cuatro meses se realizan determinaciones de abundancia de D. antillarum en seis sitios de 100 m2; distribuidos a lo largo de 38 km de la costa oeste. La densidad de D. antillarum, fue significativamente diferente entre sitios, con ámbitos de 0.81 (SD= 0.04) a 3.13 m-2 (SD= 2.10), y aumentó 61.11% durante los primeros cinco años de estudio. Fluctuaciones estacionales, posiblemente relacionadas con agregaciones reproductivas, son evidentes. La abundancia actual de D. antillarum en los arrecifes de Dominica contrasta con otros sitios en el Caribe que sufrieron mortandades masivas y en algunos casos se parece a densidades pre-mortalidad de inicios de la década de 1970. Antes de este trabajo, no se había realizado ningún estudio sistemático de Diadema en Dominica, por lo que no es claro como fueron afectadas las poblaciones en este sitio. El aumento en la densidad de D. antillarum observado hasta ahora puede deberse a recuperación tras una perturbación por un patógeno o por el Huracán Lenny de noviembre 1999. Localmente, D. antillarum es un herbívoro importante en los arrecifes de Dominica donde la sobrepesca ha disminuido el número de peces herbívoros. En escala regional, las poblaciones de D. antillarum de la isla de Dominica pueden representar una fuente de larvas corriente abajo, dada la duración de estadio equiinopluteo y el ámbito de dispersión de las larvas, que es de cientos de kilómetros.
Palabras clave: Diadema antillarum, densidad poblacional, Dominica, Antillas menores, Caribe.
Acknowledgments
Logistic and financial support for this study was provided by the Institute for Tropical Marine Ecology Inc. (ITME RP04-001).
References
Bak, R. P. M. 1985. Recruitment patterns and mass mortalities in the sea urchin Diadema antillarum. Proc. 5th Int. Coral Reef Symp., Tahiti 5: 267-272. [ Links ]
Bak, R. P. M., M. J. E Carpay & E. D. Ruyter van Steveninck. 1984. Densities of the sea urchin Diadema antillarum before and after mass mortalities on the coral reefs of Curacao. Mar. Ecol. Prog. Ser. 17: 105-108. [ Links ]
Bauer, J. C. 1980. Observations on geographical variations in population density of the echinoid Diadema antillarum within the western North Atlantic. Bull. Mar. Sci. 30: 509-515. [ Links ]
Carpenter, R. C. 1990. Mass mortality of Diadema antillarum. I. Long-term effects of sea urchin population-dynamics and coral reef algal communities. Mar. Biol. 104: 67-77. [ Links ]
Chiappone, M., S. L Miller, D. W. Swanson, J. S Ault & S. G. Smith. 2001. Comparatively high densities of the long-spined sea urchin in the Dry Tortugas, Florida. Coral Reefs 20: 137-138. [ Links ]
Cowen, R. K., K. M. M. Lwiza, S. Spongaugle, C. B. Paris & D. B. Olsen. 2000. Connectivity of marine populations: open or closed? Science 287: 857-859. [ Links ]
Eckert, G. L. 1998. Larval development, growth and morphology of the sea urchin Diadema antillarum. Bull. Mar. Sci. 63: 443-451. [ Links ]
Edmunds, P. J. & R. C. Carpenter. 2001. Recovery of Diadema antillarum reduces macroalgal cover and increases abundance of juvenile corals on a Caribbean reef. P. N. A. S. 98: 5067-5071. [ Links ]
Forcucci, D. 1994. Population density, recruitment and 1991 mortality event of Diadema antillarum in the Florida Keys. Bull. Mar. Sci. 54: 917-928. [ Links ]
Highsmith, R. C. 1982. Induced settlement and metamorphosis of the sand dollar (Dendraster excentricus) larvae in predator-free sites: adult sand dolar beds. Ecology 63: 329-337. [ Links ]
Hughes, T. P 1994. Catastrophes, phase shifts and large scale degradation of a Caribbean coral reef. Science 256: 1547-1551. [ Links ]
Hughes, T. P , B. D. Keller, J. B. C. Jackson & M. J. Boyle. 1985. Mass mortality of the echinoid Diadema antillarum Philippi in Jamaica. Bull. Mar. Sci. 36: 377-384. [ Links ]
Hughes, T. P , D. C. Reed & M. J. Boyle. 1987. Herbivory on coral reefs: community structure following mass mortalities of sea urchins. J. Exp. Mar. Biol. Ecol. 113: 39-59. [ Links ]
Hunte, W. & D. Younglao. 1988. Recruitment and population recovery of Diadema antillarum (Echinodermata; Echinoidea) in Barbados. Mar. Ecol. Prog. Ser. 45: 109-119. [ Links ]
Hunte, W., I. Cote & T. Tomascik. 1986. On the dynamics of the mass mortality of Diadema antillarum in Barbados. Coral Reefs 4: 135-139. [ Links ]
Karlson, R. H. & D. R. Levitan. 1990. Recruitment-limitation in open populations of Diadema antillarum: an evaluation. Oecologia 82: 40-44. [ Links ]
Komoroske, L. 2002. Predatory and grazing fish associated with Diadema antillarum in western Dominican coral reefs. ITME Research Reports 12: 15-20. [ Links ]
Lessios, H. A. 1988a. Mass mortality of Diadema antillarum in the Caribbean: what have we learned? Ann. Rev. Ecol. Syst. 19: 371-393. [ Links ]
Lessios, H. A. 1988b. Population dynamics of Diadema antillarum (Echinodermata: Echinoidea) following mass mortality in Panama. Mar. Biol. 99: 515-526. [ Links ]
Lessios, H. A. 1995. Diadema antillarum 10 years after mass mortality: still rare, despite help from a competitor. Proc. R. Soc. Lond. B 259: 331-337. [ Links ]
Lessios, H. A. 2005. Diadema antillarum populations in Panama twenty years following mass mortality. Coral Reefs 24: 125-127. [ Links ]
Lessios, H. A., D. R. Robertson, & J. D. Cubit. 1984. Spread of Diadema mass mortality throughout the Caribbean. Science 226: 335-337. [ Links ]
Levitan, D. R. 1988. Asynchronous spawning and aggregative behavior in the sea urchin Diadema antillarum (Philippi), p 181-186. In R. D. Bruke, P. V Mladenov, P Lambert & P. L. Parsley (eds.). Echinoderm Biology. Balkema, Rotterdam, Holland. [ Links ]
Lewis, J. B. 1966. Growth and breeding in the tropical echinoid Diadema antillarum Phillipi. Bull. Mar. Sci. 16: 151-158. [ Links ]
Liddell, W. D. & S. L. Ohlhorst. 1986. Changes in benthic community composition following the mass mortality of Diadema at Jamaica. J. Exp. Mar. Biol. Ecol. 95: 271-278. [ Links ]
Miller, R. J., A. J. Adams, N. B. Ogden, J. C. Ogden & J. P. Ebersole. 2003. Diadema antillarum 17 years after mass mortality: is recovery beginning on St. Croix? Coral Reefs 22: 181-187. [ Links ]
Moses, C. S. & R. M. Bonem. 2001. Recent population dynamics of Diadema antillarum and Tripneustes ventricosus along the north coast of Jamaica, WI. Bull. Mar. Sci. 68: 327-336. [ Links ]
Ogden, J. C., R. A. Brown & N. Salesky. 1973. Grazing by the echinoid Diadema antillarum. Formation of halos around West-Indian patch reefs. Science 182: 715-717. [ Links ]
Pearce, C. M. & R. L. Scheibling. 1990. Induction of settlement and metamorphosis in the sand dollar Echinarachnius parma: evidence for an adult-associated factor. Mar. Biol. 107: 363-369. [ Links ]
Pettersen, L. 2002. Fish and invertebrate predators of Diadema antillarum in Dominica. ITME Research Reports 12: 21-25. [ Links ]
Phinney, J. T., F. Muller-Karger, P. Dustan, & J. Sobel. 2001. Using remote sensing to reassess the mass mortality of Diadema antillarum 1983-1984. Conserv. Biol. 15: 885-881. [ Links ]
Steiner, S. C. C. 2003. Stony corals and reefs of Dominica (Lesser Antilles). Atoll Res. Bull. 498: 1-15. [ Links ]
Steiner, S. C. C. & S. M. Williams. 2006. The density and size distribution of Diadema antillarum in Dominica (Lesser Antilles):: 2001-2004. Mar. Biol. in press. [ Links ]
Weil, E., J. L. Torres & M. Ashton. 2006. Population characteristics of the sea urchin Diadema antillarum in La Parguera, Puerto Rico, 17 years after the mass mortality event. Rev. Biol. Trop. 53: 219-231. [ Links ]














