Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.54 suppl.3 San José Dec. 2006
Fragmentation of the gastrodermis and detachment of zooxanthellae in symbiotic cnidarians: a role for hydrogen peroxide and Ca2+ in coral bleaching and algal density control
I. M. Sandeman
Department of Biology, Trent University, Peterborough, Ontario, K9J 7B8, Canada. Tel.: 705 652-8754, Fax: 705 748-1205; isandem@pipcom.com
Received 01-VI-2006. Corrected 02-X-2006. Accepted 13-X-2006.
Abstract: Coral bleaching involves the detachment of zooxanthellae and the simultaneous fragmentation of the gastrodermis. Results obtained with a cell permeant fluorescent probe for calcium ions (Ca2+) indicates that "thermal" bleaching is the result of a temperature related breakdown of the Ca2+ exclusion system. "Solar" bleaching, which takes place at lower temperatures and is driven by light, is the result of a build-up of photo-synthetically produced hydrogen peroxide in the tissues. Gastrodermal tissue with its symbionts, scraped from between septa of corals, was observed under controlled conditions of high light and temperature. Pieces of gastrodermis round off, zooxanthellae move to the surface, protrude from the surface and after a delay, detach, surrounded by a thin layer of host cytoplasm, inclusions and plasma membrane. The higher the temperature and light level the shorter the delay and higher the rate of algal detachment. Fragmentation by the ballooning-out and detachment of small spheres of cytoplasm (bleb formation) takes place simultaneously. This is likely to be due to oxidation, by hydrogen peroxide (H2O2), of -SH groups on the cytoskeleton and its attachment to the plasma membrane. Ground, polished and stained thin acrylic resin sections reveal similar processes taking place in artificially bleached corals. Isolated zooxanthellae and whole corals are shown to release H2O2 in the light. This process of algal detachment and fragmentation that takes place at normal sea temperatures may underlie the mechanism limiting algal populations in the gastrodermis and may be localized to areas with a concentration of algae near the membrane. At above-normal temperatures under the synergistic effect of light and temperature, the rate of production of H2O2 exceeds the rate at which can it be lost by diffusion or destroyed and H2O2 accumulates. This results in damage to the calcium exclusion system, detachment of zooxanthellae into the coelenteron and fragmentation of the gastrodermis. Rev. Biol. Trop. 54 (Suppl. 3): 79-96. Epub 2007 Jan. 15.
Keywords: Zooxanthellae, coral bleaching, Hydrogen peroxide, Ca2+, algal density, algal detachment, gastrodermal fragmentation.
Coral bleaching is the loss of symbiotic algae (zooxanthellae) or of the pigments of the photosymbionts from host organisms (Williams and Bunkley-Williams 1990). An early account of coral bleaching (Yonge and Nicholls 1931) reported mortality and recovery of corals on the Great Barrier Reef after an episode of 35.1°C temperatures (37.1-37.8°C in pools). Histological sections of affected corals showed few zooxanthellae in the gastrodermis and algae in the process of being ejected. Many healthy zooxanthellae and wandering cells containing algae were also reported to be present in the coelenteron. More recently, serious and widespread episodes of coral bleaching and mortality have increased, particularly over the last two decades. Bleaching episodes have been associated with higher-than-usual coastal sea temperatures (Ogden and Wicklund 1988, Glynn 1991, Jokiel and Coles 1990, Glynn and DCroz 1990), in the range 30-34°C(Hoegh-Guldberg 1999) and with the involvement of light in what has been referred to as solar bleaching (Fitt and Warner 1995, Brown et al. 2000a, b). Other causes include cold shock (Muscatine et al. 1991), increased UV levels (Jokiel 1980, Gleason and Wellington 1993, Drollet et al. 1995, Lesser 1996), reduced salinity (Goreau 1964), oxygen stress (Lesser 1996, 1997, Downs et al. 2000, 2002), with the possible involvement of reactive oxygen species (Dykens et al. 1992, Nii and Muscatine 1997, Downs et al. 2002), bacteria (Kushmaro et al. 1996, 1998, Banin et al. 2000), and damage to zooxanthellae (Szmant and Gassman 1990, Brown et al. 1995) or their photosynthetic mechanisms (Jones et al. 1998, Warner et al. 1999). In anemones the expulsion of algae increased with irradiation (McCloskey et al. 1996) and under hyperthermic conditions different algae were released at different rates from the same host species (Perez et al. 2001). Reactive oxygen species (ROS), including superoxide and hydroxyl radicals, are generated (Dykens et al. 1992, Nii and Muscatine 1997). Superoxide dismutase (SOD), catalase and peroxidase have been demonstrated in zooxanthellae (Tyler and in cnidarians (Dykens 1984, Dykens and Shick 1984, Lesser et al. 1990).
Mechanisms involved in the loss of symbiotic algae were reviewed by Gates et al. (1992). They included apoptosis and necrosis (Searle et al 1982, Glynn et al. 1985), exocytosis with the release of isolated algae (Steen and Muscatine 1987, Fang et al. 1998), zooxanthellae pinched off by the host cell (Glider 1983),and detachment of host cells with their complement of zooxanthellae (Gates et al. 1992) comparable with wandering cells (Yonge and Nicholls 1931). Apoptosis or programmed cell death (PCD) and necrosis were reported during hyperthermic stress-induced bleaching over a period of four days for the anemone Aiptasia (Dunn et al. 2002). Over seven-day treatments the zooxanthellae were also affected by PCD and necrosis. Under the Oxidative Theory of Coral Bleaching (Downs et al. 2002) apoptotic or necrotic processes are initiated by ROS, and mortality or recovery depends upon the extent of the oxidative damage produced by ROS. This last theory is built mainly on information from studies of sea anemones rather than from corals, and this is understandable given the difficulties that the coral skeleton places on observations.
The present study was undertaken to investigate mechanisms of bleaching specifically in corals. The initial approach followed a comment (Halliwell and Gutteridge 1999) on the study of irreversible cell injury: "one of the simplest methods, often not used, is simply to look under the microscope and see what the cells are doing! ".The aim was to observe and photograph living coral gastrodermal tissue using a microscope built into a chamber in which both light and temperature could be controlled to simulate bleaching conditions. A second aim was to see if similar events take place in the living whole coral exposed to simulated bleaching conditions. Sections of coral tissues are normally made after dissolving away the hard skeleton, a process that can distort tissues and permit loss of small loose fragments. Embedding in acrylic resin and grinding sections avoids these disadvantages. Several studies of cnidarian bleaching (Dykens 1984, Dykens and Shick 1984, Lesser et al. 1990, Dykens et al. 1992, Nii and Muscatine 1997) have focussed on the possible involvement of superoxide radicals produced in photosynthesis under photoinhibitory conditions. However, in the presence of superoxide dismutase (SOD), superoxide dismutates to form hydrogen peroxide, which, unlike the superoxide radical, is able to pass through biological membranes with relative ease (Halliwell and Gutteridge 1999, Downs et al. 2002). Hydrogen peroxide is known to cause membrane damage via lipid peroxidation (Halliwell and Gutteridge 1999) and to induce apoptosis (Slater et al. 1995). Calcium has an important role in maintaining the structural integrity of the cell. Cells normally have low levels of free Ca2+ and calcium exclusion is linked with the sodium pump system of the plasma membrane and an ATP-dependent pump of the endothelial reticulum (ER).Disruption of the calcium exclusion system leads to apoptotic or necrotic death of the cell (Halliwell and Gutteridge 1999). A common consequence of intracellular increase of Ca2+ is cell blebbing, or ballooning out of the membrane. This results from damage to the cytoskeleton and its connections to the plasma membrane by peroxide oxidation of essential –SH groups of the trans- membrane channels and cytoskeletal proteins (Halliwell and Gutteridge 1999).The ATP driven calcium exclusion system is temperature dependent. Coupling with ATP ceases at 42°C in human erythrocytes (Rega 1986) and sarcoplasmic reticulum (Meltzar and Berman 1984). Hydrogen peroxide is known to be secreted by the cyanobacterium Anacystis nidulans (Patterson and Myers 1973). Superoxide and hydrogen peroxide are secreted by the "red tide" dinoflagellate Cochlodinium polykrikoides and other free living dinoflagellates (Kim et al. 1999). The third aim in this study was to determine whether hydrogen peroxide is released by either zooxanthellae or whole corals, and if so, determine how its production is related to light and temperature, and thus to examine a possible role for hydrogen peroxide in coral bleaching
Materials and methods
Coral sampling and preparation: All sea water used for experiments was passed through a column of 4-8 mesh activated charcoal (Sigma) and millepore-filtered (0.45µ). Small pieces of live coral, Agaricia agaricites and Porites porites, were collected in the back reef at Discovery Bay, Jamaica and were held in the sea water table at the Discovery Bay Marine Laboratory of the University of the West Indies. A coring device was used to obtain 2 cm diameter x 1 cm thick samples from the massive corals Siderastrea siderea and Montastrea faveolata. The area of pieces of coral used for experiments was estimated with aluminium foil (Marsh 1970). Small Agaricia colonies proved to be a particularly useful source of gastrodermal tissue. Tissue between parallel setae was scraped out with a needle, under sea water in a solid watch glass. Zooxanthellate tissue was brown and could be distinguished from epidermal tissue. It was easily removed with a fine pipette. Some pieces of tissue were motile as a result of ciliary or flagellar activity. Scraping tissue off Agaricia plates produced relatively little mucus. This was not the case for the other coral species investigated in which the mucus had to be vigorously teased or cut to allow removal of the gastrodermal tissue. Slides were prepared in advance with a square of parafilm smeared thinly with silicone stopcock grease on both sides. A 1 cm hole in the parafilm allowed the introduction of sea water and gastrodermal tissue to be introduced. A coverglass pressed down gently made an effective seal that prevented evaporation and, as judged from cell motility, remained viable for 24 hr. The parafilm spacer was omitted with the x100 oil immersion lens and a thin layer of silicon grease was used to seal the preparation.
Observations: The base and stage of a Wild M20 Research Microscope was built into an insulated box constructed of 5 cm thick extruded polystyrene. The temperature within the box was set and maintained within 0.1ºC by a small electrically heated coil controlled by a temperature sensor, regulator and fan. Light was introduced to the microscope mirror through a plate of heat absorbing and UV blocking glass (Edmunds Scientific). A door on one side of the box and a plexiglass window on the top enabled focussing and other adjustments to be made. Phase contrast, dark field, and light field observations could be made and photographs taken in the normal way. Illumination was measured directly on the microscope stage with a LI-COR Quantum Radiometer (Model LI-250). The sensor was shielded to an area of 1 mm2. Coral bleaching at Discovery Bay takes place when sea water temperatures reach about 32ºC and the highest irradiances experienced in the surface waters are about 2000 µ mol.m-2.s-1. The majority of experiments were carried out at 30ºC, 32ºC or 34ºC. Three irradiance levels were used, 850, 1250 and 2000 µ mol.m-2.s-1. The area outside the central illuminated field of the microscope permitted dark comparisons to be made at any temperature.
Staining: The integrity of cell and fragment membranes was investigated by the vital dye exclusion method using trypan blue (McCarthy and Evan 1998). Sea water saturated with trypan blue was used in experiments in which gastrodermal tissue was exposed to bleaching conditions. Live cells with membrane integrity do not take up stain and can be distinguished from dead cells. Material released from gastrodermal tissue or by coral pieces during artificial bleaching were collected and concentrated by centrifugation. Bisbenzimide and acridine orange (20 µ g.ml-1 in sea water) were used to stain nuclei. Acridine orange and Hoechst 33342 (0.1 µ g.ml-1 sea water) stained living cells and Hoechst 33258 (0.1 µ g.ml-1 sea water) (Sigma-Aldrich) stained material that had been fixed (modified Karnovsky solution, see below). Fluorescence was observed and photographed with a Leica DMIRE2 inverted fluorescence photomicroscope.
Sections: Freshly collected pieces of Agaricia were maintained at 32ºC in a stirred constant temperature bath, under moderate illumination (600-800 µ mol.m-2.s-1) provided by a 60-watt halogen spotlight, with a heat filter and 80B optical filter. Pieces were taken out at intervals and fixed in modified Karnovsky solution (2.5% glutaraldehyde, 2% paraformaldehyde in 0.1 M Sorenson or cacodylate buffer). Small pieces from the edge of Agaricia colonies were dehydrated, embedded in LR White (hard) acrylic resin (Sigma) and cured at 60ºC under nitrogen. Slices (< 1 mm thick) were cut with a diamond saw. The slices, mounted on glass slides with thermoplastic cement, were ground with a series of abrasives to a thickness of less than 0.05 mm. The sections were then ground and polished on one side, heated, detached and turned over. After re-attachment they were ground and polished on the second side and stained with toluidine blue. After washing in distilled water and drying, the sections each received a drop of immersion oil and a coverglass. The thickness of the section was not critical but the surface had to be scratch free. With a short staining time of 30 s only a very thin layer at the surface was stained and the lower levels of the sections remained optically clear.
Hydrogen peroxide released by zooxanthellae: Production of H2O2 by zooxanthellae was measured with a technique similar to one used by Patterson and Myers (1973) based on the reduction of scopeletin fluorescence (Andreae 1955) in the presence of horseradish peroxidase (Sigma). A stock solution of 2 mg/L scopoletin (Sigma) was kept at 4ºC. In preliminary experiments it was found that a suspension of zooxanthellae strongly quenched the blue fluorescence and if stirred zooxanthellae clumped and adhered to surfaces. It was also apparent that the UV used to excite fluorescence generated significant quantities of H2O2, so the time for which the UV LED was switched on was kept to a minimum. Zooxanthellae were obtained with the water pik method (Johannes and Wiebe 1970). After homogenisation they were cleaned by low speed centrifugation for three minutes, at least four times, followed by resuspension with a vortex mixer between spins. The algae were then resuspended, strained through cheesecloth and counted using a haemocytometer. The density of the algal suspension was adjusted to 8.0 x 105 algae.ml-1 and total volume of 8 ml. To this was added an equal volume of sea water containing 1 ml of scopoletin stock (2 mg l-1) and 300 units of peroxidase. The final mixture contained 4.0 x 105 algae and gave close to a full scale deflection in the fluorometer (Fig. 1). One ml of the algal suspension was added to fifteen 1.5 ml plastic centrifuge tubes. Six tubes, three illuminated, three dark, were kept at room temperature at 27°C, and six tubes were placed in a temperature controlled chamber at 32°C. Illumination of 250 µ mol.m-2.s-1 was provided by a white LED (RL5-W18030, Super Bright LEDs Inc.) built into a cap that fitted over each centrifuge tube. After 15 min, during which the tubes adjusted to temperature, the initial fluorescence of the three remaining tubes was read in the fluorometer and the LEDs were switched on. After 1 hr the tubes were shaken and centrifuged for 5 min at high speed to deposit the algae and suspended matter. Then 0.6 ml of the supernatant was withdrawn from each with a pipette and transferred to a fluorometer sample vial. Three readings of fluorescence were taken for each vial and averaged. After 1 hr of illumination the fluorescence of the "light " and "dark" tubes from the two temperatures were read as above. The difference between the averages before and after illumination gave the average change in fluorescence during the period of illumination. Calibration was carried out with commercial 6% hydrogen peroxide solution diluted with distilled water (x 4000). 10 µ L containing 15 ng of H2O2 was introduced into each vial using a microsyringe, stirred gently and the fluorescence re-read. The commercial hydrogen peroxide was later assayed by iodometric titration (Vogel 1961) and the results adjusted.
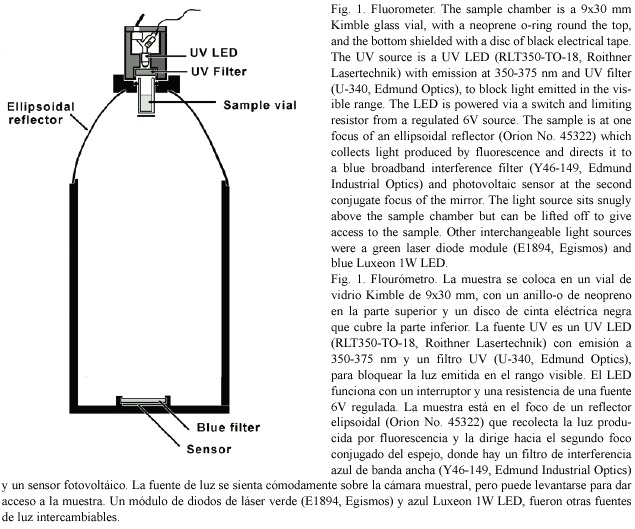
H2O2 released by whole corals: Small (2.5-5 cm2) pieces of thin plates of the coral Agaricia agaricites were placed on a layer of plastic mesh in a beaker with 40 ml of sea water. The layer of mesh rested on an insulated heater coil surrounding a central area with a magnetic stirrer bar. The beaker was continuously stirred and a thermistor sensor and controller maintained the temperature constant to within 0.1°C. Temperatures were monitored with a lab thermometer. Illumination was provided by a 60-watt narrow beam halogen spotlight with a heat filter and 80B optical filter. Peroxidase (500 units) and 2.5 ml of stock scopoletin solution (2.0 mg/l in sea water) were added to the sea water in the beaker. Fluorescence was monitored at intervals of 3-5 min by transfering 0.6 ml of the sea water medium from the incubation beaker to the chamber of the fluorometer. The fluorescence of the sample was read, compared to a sea water blank and the sample was then returned to the incubation beaker. The initial concentration of scopoletin was adjusted to give a full scale reading. If in the course of an experiment the concentration of scopoletin was reduced to below 20% of the initial reading more of the stock scopoletin was added and the same amount of medium removed to keep the total volume the same. Rates of change of fluorescence were obtained by regression analysis from four or five consecutive readings. Each experiment was calibrated by spiking the incubation medium with a known quantity of hydrogen peroxide and observing the change in fluorescence. Measurements were made of hydrogen peroxide release involving three sets of conditions. First, the temperature was changed while keeping illumination constant. Second, the illumination was changed and temperature was kept constant. Third, illumination and temperature (32 °C) were kept constant and hydrogen peroxide release was monitored over a period of about 7 hr.
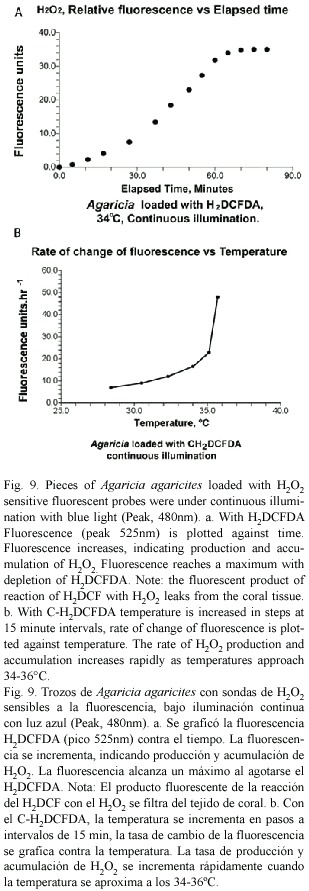
Two intracellular probes sensitive to H2O2 were used in this study. The first, 2, 7-ddichlorofluorescin diacetate (H2DCFDA) (Sigma), is permeable to living cells. Once loaded it deacetylates to H2DCF which reacts with peroxides to form the green fluorescing 2, 7-dichlorofluorescein. The second (C-H2DCFDA), 6- carboxy-2 ,7 -dichlorodihydrofluorescein diacetate di(acetoxy methyl ester) (Molecular Probes), works in a similar way to H2DCFDA but has two additional negative charges which reduce the tendency of the fluorescent product of its reaction with H2O2 to leak out of the cell. Stock solutions in DMSO (10 mmol.l-1) were prepared (Blackstone 2001) and small pieces of Agaricia were incubated for 1 hr in a 10 µ mol.l-1 solution in sea water in the dark. The pieces were rinsed in sea water and trimmed to fit into the fluorometer sample vials. The stimulating lamp was a blue Luxeon 1 watt LED (peak 480 nm) with a parabolic reflector, which produced a narrow beam of light, and broadband interference filter (410-480 nm). Fluorescence (peak 525 nm) was collected by the ellipsoidal mirror of the fluorometer (Fig.1) and passed through a broadband interference filter (510-590 nm) to the sensor. The fluorometer was operated in a temperature controlled chamber. To assess the effect of temperature the temperature was raised by steps at intervals of 15 min with the light source on.
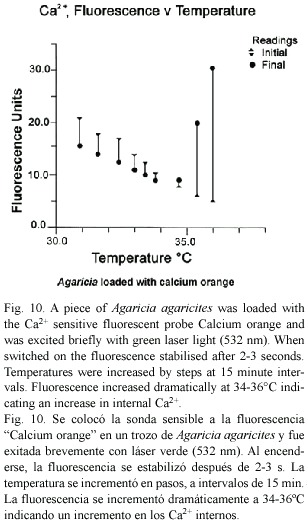
Intracellular Ca2+: The acetoxymethyl (AM) ester of the cell permeant calcium indicator Calcium Orange (Molecular Probes Inc.) is permeable to living cells. On entry it is hydrolysed to release the fluorescent calcium indicator in a charged form that is less permeant to cell membranes. 50 m g of Calcium Orange was dissolved in 0.1ml of a 20% solution of Pluronic® F-127 in dimethyl sulfoxide (DMSO). This was added to 20 ml of sea water to give a 4 µ M working solution. Small pieces from the edge of thin plates of Agaricia agaricites were incubated for 30 min, rinsed in sea water, trimmed and placed with 0.6 ml sea water in a fluorometer sample vial in the fluorometer (Fig. 1) in a temperature controlled chamber. Excitation at 532 nm was provided by a green laser diode module (E1894, Egismos Technology Corporation) and the fluorescence (peak at 575-580 nm) was collected and passed through a broadband interference filter (560- 640 nm) to the sensor. The temperature of the chamber was raised by steps at 15 min intervals. At each temperature the laser module was switched on briefly to take a reading of the fluorescence.
Results
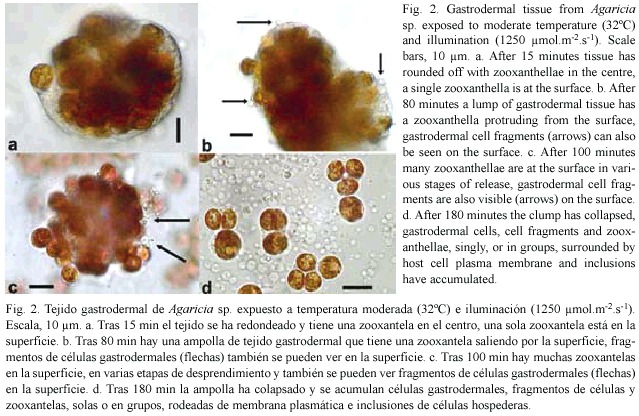
Direct observations: Clumps of gastrodermal cells, under moderate light (1250 µ mol.m-2.s-1) and temperature (32°C), formed spherical masses within 5-15 min with a clear coherent layer on the outside and zooxanthellae packed in the centre (Fig. 2a). After a delay period of up to 2 hr, individual zooxanthellae moved to the surface (Fig. 2a), protruded through it (Fig. 2b, c,), and eventually detached, singly, or in groups (Fig 2c). Most of the detached zooxanthellae were surrounded by a thin layer of host cytoplasm, plasma membrane, and cell inclusions (Fig. 2d). Host nuclei could be distinguished with regular and phase contrast optics and it was clear that host cytoplasm surrounding zooxanthellae did not always have a nucleus. At the same time small spherical blebs, not containing algae but sometimes with inclusions, appeared on the surface of the clump (Fig. 2b, c) and eventually detached (Fig. 2d). Most of the fragments and algae that were released sank to the surface of the slide nearby, but a few algae, surrounded by host material with a flagellum, swam away. After several hours, all that remained was an area strewn with algae and cell fragments, with a heap in the centre representing what was left of the original gastrodermal tissue.
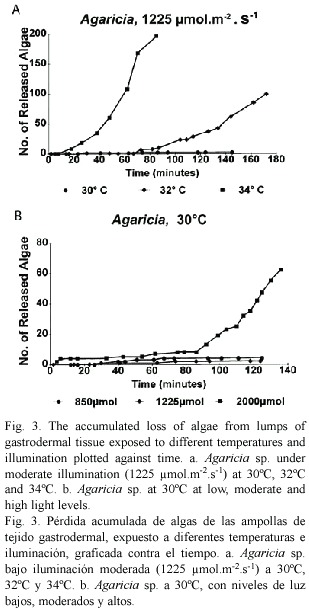
Observations on the delay periods and detachment rates were made at 30, 32 and 34°C, with three illumination levels: 850, 1250 and 2000 µ mol.m-2.s-1 (referred to as low, intermediate and high). Initially Agaricia agaricites was studied (37) but tissue from other species (Montastrea faveolata (13), M. annularis (7), Siderastrea siderea (12) and Porites porites (3) was used for comparison and confirmation that the same processes took place. Examples of the results can be seen in figures 4 and 5; they were chosen from experiments in which the rounded off balls of gastrodermal tissue were of a similar size, with about 200-300 zooxanthellae, and from the same coral colony. Generally, the higher the temperature and light level, the shorter was the delay, and the higher the rate of algal loss. More specifically, under a moderate light level (Fig. 3a) there was no loss of algae at 30°C but at 32°C after a delay of 75 min, algae detached at a rate of about 70/hr. At 34 °C, after a delay of 15 min algae detached at a rate of 200/hr. At 30°C (Fig. 3b) clumps of gastrodermal cells from Agaricia rounded off but no algae were shed at the lower and intermediate light levels. At the high light level, algal detachment commenced after 85 min. At 34°C algal loss took place at all light levels, almost immediately at the higher light level and after a delay of 7-8 min for the lower levels. In the dark areas of the slides at 30-32°C there was little or no algal loss from the tissue over the time period of the experiments but when left overnight at 32-34°C the rounded off tissue in the dark areas had also completely fragmented. At 30°C some algal detachment had taken place overnight but the cells appeared to be healthy and flagellar action was still evident. At 35-36°C in the dark, detachment of algae and fragmentation commenced after about 30 minutes and was complete within two hours (Fig. 2d). When temperatures were raised for tissue from Agaricia ciliary action ceased at about 36-37°C and epidermal cells started sliding apart at 37.5°C.
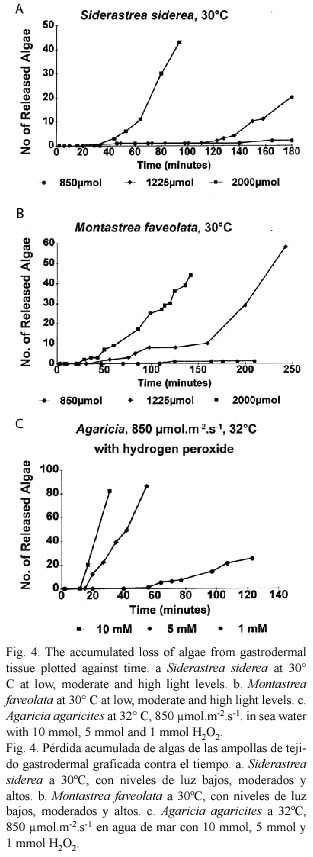
Gastrodermal cells from other species showed differences in their responses. Siderastrea siderea (Fig. 4a) was more sensitive than Agaricia at 30°C (Fig. 3b) in that algal loss took place after 120 min at the intermediate light level. At 34°C massive fragmentation started quickly and only later did algae move to the surface and detach. Montastrea faveolata (Fig. 4b) seemed to be slightly less sensitive than Agaricia in that the delay times were longer. At 30°C and intermediate light, algal detachment started at a very low rate (3/hr) and this changed after a 2.5 hr delay to a much higher rate (50/hr). Porites porites does not tend to bleach at Discovery Bay when other species are affected and gastrodermal tissue required a temperature of 34°C for algal detachment to occur and then only after 4.5 hr under high light conditions.
Staining: Observations were made of gastrodermal tissue in sea water saturated with trypan blue under high light/temperature conditions. Unlike the debris present, which took up the stain, neither the cell fragments nor the coral tissue surrounding released algae took up the stain. This indicated that the membranes retained their integrity as in apoptosis and were not necrotic. Further, the fragments retained their integrity at least overnight.
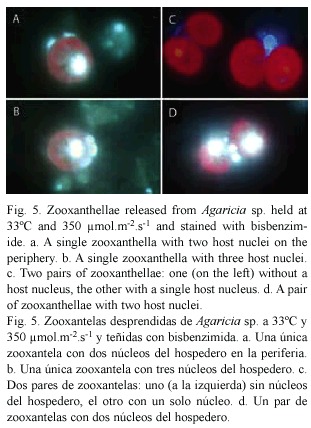
Live material released from corals under high light and temperature conditions were stained with bisbenzimide (Hoechst 33342). Zooxanthellae were readily visible as a result of the red fluorescence of the chloroplast. Nuclei, particularly of the coral host, were visible due to the blue fluorescence of DNA (Fig. 5). The nuclei did not show condensation of the chromatin that is typical of apoptosis in higher organisms. In the fixed material stained with Hoechst 33258 the host nuclei were smaller, the nuclei of the algae were brighter and condensed and generally the fluorescence was less brilliant. Pairs of zooxanthellae surrounded by host cytoplasm, similar to the one seen in Fig. 2d were present. Figure 5c shows two pairs of zooxanthellae held together by host cytoplasm, one with, the other without, a host nucleus. Both pairs are too far apart and too large to be the result of a recent cell division, the resulting individuals remaining together within a cyst wall. Also seen were pairs of zooxanthellae with two nuclei present (Fig. 5d). Some of the single zooxanthellae had two (Fig. 5a) or three (Fig. 5b) host nuclei. These observations suggest that it is unlikely that these zooxanthellae are in their original host cells.
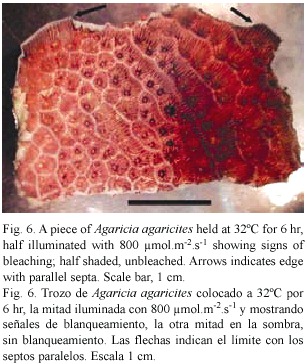
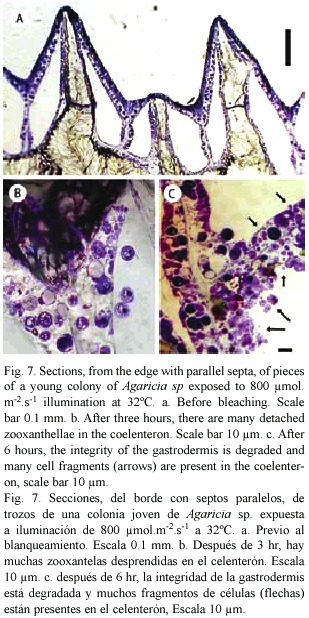
Sections: Small pieces of Agaricia sp. held in sea water at 32°C and exposed to light (300-800 µ mol.m-2.s-1.) generally showed signs of bleaching after 6-8 hr. Figure 6 shows an example in which one half of a piece of Agaricia was illuminated and the other half more or less shaded. The half exposed to light is much lighter in colour, especially on the ridges. In the central area where light and dark areas meet and the light intensity is lower, the patches of dark brown colouration round the mouths of the polyps indicate the accumulation of zooxanthellae prior to their loss. In sections of unbleached, freshly collected Agaricia (Fig. 7a) free zooxanthellae are not present in the coelenteron. Pieces of coral at 32°C showed no visible signs of bleaching after 2-3 hr in light but sections showed free zooxanthellae in the coelenteron (Fig. 7b). After 5-6 hr of exposure to light, pieces of coral began to show signs of bleaching, i.e. were lighter in colour on the ridges. The sections showed, in addition to detached algae, many small cell fragments present in the coelenteron. The cell layers, especially the gastrodermis, appear degraded, with fragmented contents (Fig. 7c). The thin layers lining the skeleton had also detached from the skeleton. At 30°C zooxanthellae appeared in the coelenteron but only after a much longer period of illumination, and there was some detachment of the two thin cell layers from the skeleton. The sections confirm that detachment of the zooxanthellae and fragmentation observed in gastrodermal tissue also takes place in intact corals under similar conditions of high light and temperature.
Hydrogen peroxide: Gastrodermal tissue from a colony of Agaricia placed in sea water containing hydrogen peroxide responded (Fig. 4c) in a similar way to algae exposed to high light/temperature (Fig. 3a, b, 4a, b), i.e., algal detachment took place after a delay period and the higher the concentration of H2O2 the shorter was the delay period and the higher the rate of detachment. Response to 10 and 5 mmol H2O2 was much stronger and faster but response to 1 mmol H2O2 was at a comparable level.
Isolated zooxanthellae were found to produce and release hydrogen peroxide. In the 12 coral samples from four host species the H2O2 released at 27°C varied considerably (51.4- 191.2, mean 81.3 ng.hr-1.106 algae-1). At 32°C production was slightly lower (4.8-174.8, mean 57.4 ng.hr-1. 106 algae-1). In the dark there was a small reduction in fluorescence representing H2O2 production of 10.9 ng.hr-1.106 algae-1 at 27°C and 5.2 ng.hr-1.10 6 algae-1 at 32 °C. The above rates are based on the difference between light and dark production and thus confirm the light related nature of the H2O2 production. The production of H2O2 by isolated zooxanthellae at 27°C and 32°C was not significantly different.
The rates at which hydrogen peroxide was released from Agaricia pieces from different colonies varied by as much as an order of magnitude. Individual pieces generally showed higher rates of release at above or below ambient temperatures. The mean rates of release of H2O2 (in µ g.cm-2.hr-1) recorded were as follows: 28ºC, 0.67 (n=2); 30ºC, 0.47 (n=7); 32 ºC, 0.46 (n=7); 34ºC, 1.03 (n=6); 36ºC, 0.66 (n=4). These results are similar to those obtained for isolated zooxanthellae. Production of H2O2 was light related. When illumination was cut off, the rate of H2O2 release decreased and when illumination was resumed, or increased, the rate of H2O2 release generally increased. Agaricia pieces showed higher production at 34°C.
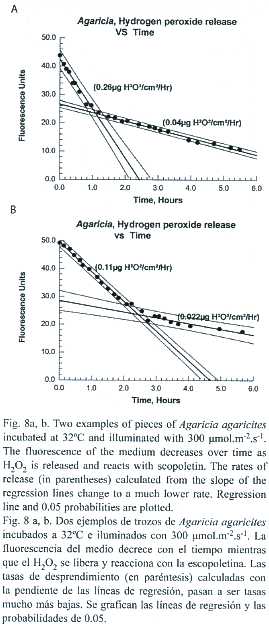
The release of H2O2 by pieces of Agaricia held at 32ºC with illumination of 300 µmol.cm-2.hr-1 was monitored over several hours. The pattern of the results obtained was similar (Fig. 8a, b) for eight pieces of Agaricia tested. Hydrogen peroxide was released at a constant rate between 0.11 and 0.40 µ g.cm-2.hr-1 (mean 0.219 µ g.cm-2.hr-1) for a period of 65-160 min (mean 122 min) at which point the rate dropped to a much lower but constant rate (0.025- 0.15 µ g.cm-2.hr -1, mean 0.06 µ g.cm-2.hr-1).Two of the coral pieces started showing signs of bleaching at the point of change with a lightening of the colour, particularly on the ridges. Regression analysis indicated that there is a possible (p=0.10) inverse relationship between the initial rate of hydrogen peroxide release and the time at which the rate of hydrogen peroxide release changed. The lower rate at which hydrogen peroxide was released from coral pieces probably represents the rate at which loss is by diffusion (note: in these experiments the chamber was stirred fairly vigorously and continuously). The initial higher rate may involve some active mechanism, such as active circulation of the contents of the coelenteron, assisting in the elimination of hydrogen peroxide.
Initial experiments in which coral pieces loaded with H2DCFDA were excited with blue light for brief periods showed little change in fluorescence level over the temperature range of 28-36°C. When the light was kept on permanently the fluorescence levels increased steadily, indicating accumulation of H2O2. In a typical experiment (Fig. 9a) fluorescence at 34°C increased but eventually leveled off as the H2DCF became depleted. In these experiments with H2DCFDA the fluorescent product of reaction with H2O2 leaked out of the tissues and the sea water in the vial fluoresced strongly. With the improved version, C-H2DCFDA, there was little leakage. The rate of change of fluorescence (Fig. 9b) increased slowly as the temperature was raised until about 34°C. After that it increased rapidly, indicating increases in the internal concentration of H2O2 and an increase in the rate at which it was being produced.
Intracellular Ca2+: When the green laser module was switched on to excite Agaricia loaded with calcium orange there was an immediate and rapid change in fluorescence. The fluorescence stabilized within 2-3 s (Fig. 10). As temperature was increased fluorescence levels decreased, but at about 34°C fluorescence levels began to increase dramatically, indicating rapidly increasing Ca2+ levels in the tissues of the coral as the temperature increased through 35-36°C.
Discussion
The direct observations of coral gastrodermal tissue suggest that changes to the membranes of the gastrodermal layer can be produced by the synergistic action of light and high temperature. Two key events were observed: first, the movement of zooxanthellae to the surface, their protrusion and detachment by what appeared to be a pinching-off process. The detached algae were surrounded by a thin layer of host membrane with host cell inclusions. The second key event, also taking place at the surface of the gastrodermal tissue, was the blebbing, or ballooning out and detachment of smaller spherical fragments of cytoplasm, also with host cytoplasmic inclusions. These are probably different aspects of the same phenomenon: if a bleb forms where a zooxanthella is in contact with or near the membrane, the alga becomes enclosed by the bleb with nearby host cytoplasm and inclusions. The detached zooxanthellae were surrounded by host cytoplasm but many lacked nuclei, and some possessed two or three nuclei. The interpretation by Gates et al. (1992) that they are detached host cells seems unlikely. The fixed and ground sections confirm that under conditions of high light and temperature the same events take place in intact corals. Zooxanthellae are released into the coelenteron and later fragmentation and extensive damage to the gastrodermis take place (Figs. 7b, c). The time period over which the events take place is rather longer in the intact coral than in excised gastrodermal tissue.
The results from the direct observations of algal detachment, the length of the delay period and rates of algal detachment, indicate a synergistic action of temperature and light. From the observations of what happens to similar sized portions of tissue from the same colony under different combinations of temperature and illumination (Figs. 3a, b, 4a, b), the conclusion can be drawn that higher temperature and stronger illumination shorten the delay period and increase the rate of algal detachment.
Both isolated zooxanthellae and whole corals have been shown in this study to release hydrogen peroxide under high light conditions. The experiments with H2DCFDA confirm the production of H2O2. The experiments with C-H2DCFDA indicate that H2O2 accumulates in the tissues and that the rate of accumulation increases with temperature. With H2O2 (1 mmol) added to the sea water medium containing gastrodermal tissue (Fig. 4c), rates of detachment and length of delay paralleled what had taken place as light intensity was increased but without added H2O2 (Figs. 3 and 4a, b). Together these observations provide a very strong pointer for the involvement of hydrogen peroxide. The observed delay before zooxanthellar detachment takes place probably corresponds to the period during which H2O2 builds to the level at which it damages the plasma membrane.
Calcium has a major role in maintaining the structural integrity of the cell. A known consequence of increase of Ca2+ in the cell is blebbing, or ballooning out of the membrane (Halliwell and Gutteridge 1999). The experiments with calcium orange in this study confirm that internal Ca2+ in coral tissues increases dramatically as temperatures approach 35-36°C. This is consistent with other reports (Rega 1986, Meltzar and Berman 1984) of the temperature related nature of the calcium exclusion system in higher organisms. Hydrogen peroxide and other peroxides can interfere with the calcium balance in the cell in several ways. Peroxides are known to damage the Ca2+ extrusion systems of the plasma membrane and ER; and also the structural integrity of membranes by oxidising essential SH groups of the transmembrane channels and cytoskeletal proteins (Halliwell and Gutteridge 1999).
It is thus possible to interpret "thermal" bleaching as a temperature related breakdown of the calcium exclusion system of the plasma membrane. Internal calcium increases and results in fragmentation and detachment of zooxanthellae by blebbing. "Solar" bleaching, which is light driven and takes place at lower temperatures, results from the accumulation of hydrogen peroxide and subsequent damage to the calcium exclusion system. The results are the same.
Experimental detachment of algae and fragmentation took place at 30°C, which is below the temperature at which bleaching takes place in the back reef at Discovery Bay. This suggests the possibility that the processes leading to detachment and fragmentation may provide the mechanism by which algal numbers are maintained at a relatively constant level in the gastrodermis. In regions where algae are denser hydrogen peroxide levels will also be greater and localized blebbing and algal detachment may result. This process may also account for the observed reduction of pigment associated with bleaching (Kleppel et al. 1989). If more heavily pigmented zooxanthellae release more H2O2, they would tend to detach first, leaving a population with less pigment.
Under conditions of high light and temperature, with still higher H2O2 production and accumulation, the rate of damage may outstrip the ability of the protective mechanisms to respond or to make repairs, and damage on a much wider scale will result. If the point is reached at which the integrity of the gastrodermis is compromised to the extent that sea water in the coelenteron is no longer circulated, H2O2 will accumulate even more quickly, leading to further breakup of the gastrodermal layer and bleaching. This is what may have occurred when the rates of H2O2 released by whole corals decreased (Fig. 9a, b) and symptoms of bleaching appeared. Water motion helped to explain the differential survival of corals during a bleaching event (Nakamura and van Woesik 1997) and is an important component of the loss of H2O2 by diffusion. The potential for bleaching may increase in calm weather with lack of currents and/or wave action to assist in the dispersal of H2O2.
Apoptosis (Searle et al. 1982) has been invoked as a mechanism involved in bleaching (Dunn et al. 2002, Downs et al. 2002). The membrane blebbing and fragmentation with retention of membrane integrity observed in this study are very similar to the blebbing and production of apoptotic bodies which are typical of apoptosis (Halliwell and Gutteridge 1999). The nuclei, however, did not show aggregation of the chromatin at the nuclear membrane that is also typical of apoptosis in higher organisms. The observed events may indicate a primitive form of apoptosis and should be investigated further. A possible advantage to the coral of this apoptotic-like process is that the fragments with integral membranes and algae surrounded by host cytoplasm could be recognized, reabsorbed and thus efficiently recycled elsewhere in the colony.
The mechanism, suggested here and demonstrated for scleratinian corals, fits in well with the Oxidative Theory of Coral Bleaching (Downs et al. 2002) and encompasses roles for light and changes of temperature. Any condition that increases the photoinhibitory production of hydrogen peroxide such as oxygen stress, metal ions or UV, may enhance bleaching. Simarly, conditions that interfere with the calcium pump of the plasma membranes may also lead to bleaching. Bleaching resulting from ionic imbalances caused by salinity changes (Goreau 1964) probably fits into the latter category. Many other cnidarians with photosynthetic symbionts are affected by bleaching conditions (Lasker et al. 1984) as are other reef invertebrates such as sponges (Vicente 1990). The mechanism described here is of a general nature and will probably apply to other organisms containing photosynthetic symbionts. It is interesting that H2O2 production rates of zooxanthellae in this study are much lower than those (0.1-2.0 nmol.min 1.104 cells1) found for Cochlodinium polykrikoides and other free-living red-tide dinoflagellates (Kim et al. 1999). Low H2O2 production may be a requirement for a successful intracellular symbiotic existence. The bleaching of corals involving bacteria (Kushmaro et al. 1996, 1998, Banin et al. 2000) may have a different mechanism involving toxins. It is possible that superoxide radicals and/or hydrogen peroxide could be produced by vibrios either through photosynthesis or, if luminescent, by the reduction of oxygen by flavoproteins. Another possibility is that the toxins produced by vibrios might affect the calcium pump directly. It also possible that some of the infective coral diseases that result in loss of zooxanthellae, and bleached areas of coral may involve the same mechanisms of algal detachment and fragmentation described in the present study.
Some studies have reported visible damage to zooxanthellae (Szmant and Gassman 1990, Brown et al. 1995) as a result of bleaching, others none (Yonge and Nicholls 1931, Glynn et al. 1985). In the present study very few (<5) abnormal zooxanthellae were seen and those only after periods at the highest temperature and irradiance level. As was the case for the sea anemone Aiptasia sp. (Dunn et al. 2002), damage to the zooxanthellae probably takes place only after long exposures to high temperature and within host tissue that has already been severely affected. The main physical symptom of bleaching, loss of colour resulting from the physical loss of algae may appear well after the processes described in the present study have taken place. Yonge and Nicholls (1931) reported that the maximum rate of loss of zooxanthellae took place 11 days after exposure to high temperature and continued for up to 27 days. The changes in photosynthetic efficiency of zooxanthellae in bleached corals that have been reported (Jones et al. 1998, Warner et al. 1999) may be due to the relocation of large numbers of algae in the coelenteron and consequent change in pH and availability of CO2, rather than to damage to their photosynthetic mechanisms. Confirmation of the latter comes from a report (Ralph et al. 2001) that released algae are photosynthetically competent.
The results presented here lead to an interpretation of bleaching in corals as the result of interference with the calcium exclusion system by high temperature or the accumulation of photosynthetically produced hydrogen peroxide. This interpretation generally confirms the Oxidative Theory of Coral Bleaching (Downs et al. 2002). That theory, however, proposes that coral bleaching is a last resort against oxidative stress. The present study suggests that the events that occur may be part of a delicate mechanism underlying the normal process limiting symbiotic algal populations in their host and maintaining a relatively constant, acceptable level of oxidative stress. Imbalances, with which the protective system cannot cope, caused by changes in environmental conditions such as increasing temperatures, may lead to severe bleaching and death of the coral.
Acknowledgments
I am grateful to Trent University and the staff of the Discovery Bay Marine Laboratory of the University of the West Indies for their support and encouragement. I also thank Craig Brunetti of Trent University for help and the use of his fluorescence microscope.
Resumen
El blanqueamiento de los corales implica el desprendimiento de zooxantelas y la simultánea fragmentación de la gastrodermis. Resultados obtenidos con una sonda florescente de iones Calcio (Ca2+) para células permeables, indican que el blanqueamiento "térmico" es el resultado de una interrupción del sistema de exclusión de Ca2+, provocada por la temperatura. El blanqueamiento "solar", que tiene lugar a temperaturas más bajas y está determinado por la luz, es el resultado de una acumulación de peróxido de hidrógeno producido fotosintéticamente en los tejidos. Para ver el proceso, se raspó tejido gastrodermal, junto con sus simbiontes, de entre los septos de los corales y se observó en condiciones controladas de luz y temperatura. Primero, los trozos de gastrodermis se dan vuelta, luego las zooxantelas se mueven hacia la superficie, sobresalen desde ella y tras un tiempo, se desprenden, rodeadas de una delgada capa de citoplasma del hospedero, inclusiones y membrana plasmática. Mientras mayor sea la temperatura y el nivel de luz, menor es el tiempo que tardan las zooxantelas en desprenderse y mayor es la tasa de desprendimiento algal. La fragmentación producida por el inflamación y el desprendimiento de pequeñas esferas de citoplasma (formación de ampollas), tiene lugar simultáneamente. Probablemente, esto es causado por oxidación en el citoesqueleto de grupos -SH por el peróxido de hidrógeno (H2O2), y por su acoplamiento a la membrana plasmática. Secciones de resina acrílica delgada molida, pulida y teñida revelan que en corales blanqueados artificialmente se llevan a cabo procesos similares. Se ha demostrado que tanto las zooxantelas aisladas como los corales enteros, liberan H2O2 en la luz. Debajo este proceso de desprendimiento algal y fragmentación que tiene lugar a temperaturas normales en el mar, puede encontrarse el mecanismo que limita las poblaciones algales en la gastrodermis, el cual podría estar localizado en áreas con concentraciones de alga cerca de la membrana. A temperaturas más altas de lo normal, bajo el efecto sinérgico de la luz y la temperatura, la tasa de producción de H2O2 excede la tasa a la cual éste puede destruirse o perderse por difusión y, por ende,se acumula. Esto resulta en daño al sistema de exclusión de calcio, desprendimiento de zooxantelas hacia el celenterón y fragmentación de la gastrodermis.
Palabras clave: zooxantela, blanquamiento de corales, peróxido de hidrógeno, Ca2+, densidad algal, desprendimiento algal, fragmentación gastrodermal.
References
Andreae, W. A. 1955. A sensitive method for the estimation of hydrogen peroxide in biological materials. Nature 175: 859-860. [ Links ]
Banin, E., T. Israely, A. Kushmaro, Y. Loya, E. Orr & E. Rosenberg. 2000. Penetration of the coral-bleaching bacterium Vibrio shiloi into Oculina patagonica. Appl. Env. Microbiol. 66: 3031-3036. [ Links ]
Blackstone, N. W. 2001. Redox state, Reactive oxygen species and adaptive growth in colonial hydroids. J. Exp. Biol. 204: 1845-1853. [ Links ]
Brown, B. E., M. D. A. Le Tissier & J. C. Bythell. 1995. Mechanisms of bleaching deduced from histological studies of reef corals sampled during a natural bleaching event. Mar. Biol. 122: 655-663. [ Links ]
Brown, B. E., R. P. Dunne, M. S. Goodson & A. E. Douglas. 2000a. Bleaching pattern in reef corals. Nature 404: 142-143. [ Links ]
Brown, B. E., R. P. Dunne, M. E. Warner, I. Ambarsari, W. K. Fitt, S. W. Gibb & D. G. Cummings. 2000b. Damage and recovery of Photosystem II during a manipulative field experiment on solar bleaching in the coral Goniastra aspera. Mar. Ecol. Prog. Ser. 195: 117-124. [ Links ]
Downs, C. A., E. Mueller, S. Phillips, J. E. Fauth & C. M. Woodley. 2000. Oxidative stress and seasonal coral bleaching. Mar. Biotechnol. 2: 533-544. [ Links ]
Downs, C. A., J. E. Fauth, J. C. Halas, P. Dustan, J. Bemiss & C. M. Woodley. 2002. Oxidative stress and seasonal coral bleaching. Free Radical Biol. Med. 33: 533-543. [ Links ]
Drollet, J. H., M. Faucon & P. M.V. Martin. 1995. Elevated sea water temperatures and solar Uv-B flux associated with two successive coral mass bleaching events in Tahiti. Mar. Freshwater Res. 46: 1153-1157. [ Links ]
Dunn, S. R., J. C. Bythell, M. D. A. Le Tissier, W. J. Burnett & J. C. Thomason. 2002. Programmed cell death and cell necrosis activity during hyperthermic stress- induced bleaching of the symbiotic sea anemone Aiptasia sp. J. Exp. Mar. Biol. Ecol. 272: 29-53. [ Links ]
Dykens, J. A. 1984. Enzymic defences against oxygen toxicity in marine cnidarians containing endosymbiotic algae. Marine Biology Letters 5: 291-301. [ Links ]
Dykens, J. A. & J. M. Shick. 1984. Photobiology of the symbiotic sea anemone, Anthopleura elegantissima: Defences against photodynamic effects, and seasonal photoacclimatization. Biol. Bull. 167: 683-697. [ Links ]
Dykens, J. A., J. M. Shick, C. Benoit, G. R. Buettner & G. W. Winston. 1992. Oxygen radical production in the sea anemone Anthopleura elegantissima and its endosymbiotic algae. J. Exp. Biol. 168: 219-241. [ Links ]
Fang, L-S., J-T. Wang & K-L. Lin. 1998. The subcellular mechanism of the release of zooxanthellae during coral bleaching. Proc. Natl. Sci. Counc. Rep. China, Pt B, Life Sci 22: 150-158. [ Links ]
Fitt, W. K. & M. E. Warner. 1995. Bleaching patterns of four species of Caribbean reef corals. Biol. Bull. 189: 298-307. [ Links ]
Gates, R. D., G. Baghdasarian & L. Muscatine. 1992. Temperature stress causes host cell detachment in symbiotic cnidarians: Implications for coral bleaching. Biol. Bull. 182: 324-332. [ Links ]
Gleason, D. F. & G. M. Wellington. 1993. Ultraviolet radiation and coral bleaching. Nature 365: 836-838. [ Links ]
Glider, W. V 1983. The biology of the association of Symbiodinium microadriaticum with Aiptasia pallida: an anemone-alga symbiosis. Ph.D. Diss., Univ. Nebraska, Lincoln, Nebraska. 102 p. [ Links ]
Glynn, P. W , 1991. Coral reef bleaching in the 1980s and possible connections with global warming. Trends Ecol. Evol. 6: 175-179. [ Links ]
Glynn, P. W & L. DCroz. 1990. Experimental evidence for high temperature stress as the cause of El Nino-coincidental coral mortality. Coral Reefs 8: 181-191. [ Links ]
Glynn, P. W , E. C. Peters & L. Muscatine. 1985. Coral tissue microstructure and necrosis: relation to catastrophic coral mortality in Panama. Dis. Aquat. Org. 1: 29-337.
Goreau, T. F 1964. Mass expulsion of zooxanthellae from Jamaican reef communities after Hurricane Flora. Science 145: 383-386. [ Links ]
Halliwell, B. & J. M. C. Gutteridge. 1999. Free Radicals in Biology and Medicine. (Third Edition) Oxford University Press, Oxford, England. 936 p. [ Links ]
Hoegh-Guldberg, O. 1999. Climate change and worlds coral reefs: Implications for the Great Barrier Reef. Mar. Freshw. Res. 50: 839-866. [ Links ]
Johannes, R. E. & W. J. Wiebe. 1970. A method for determination of coral tissue biomass and composition. Limnol. Oceanogr. 5: 822-824. [ Links ]
Jokiel, P. L. 1980. Solar ultraviolet radiation and coral reef epifauna. Science 207: 1069-1071. [ Links ]
Jokiel, P. L. & S. L. Coles. 1990. Responses of Hawaiian and other Indo-Pacific reef corals to elevated temperatures. Coral Reefs 8: 155-162. [ Links ]
Jones, R. J., O. Hoegh-Guldberg, A. W. D. Larkum & U. Schreiber. 1998. Temperature-induced bleaching of corals begins with impairment of the CO2 fixation mechanism in zooxanthellae. Plant, Cell Environ. 21: 1219-1230. [ Links ]
Kim, C. S., S. G. Lee, C. K. Lee, H. G. Kim & J. Jung. 1999. Reactive oxygen species as causative agents in the ichthyotoxicity of the red tide dinoflagellate Cochlodinium polykrikoides. J. Plankt. Res. 21: 2105-2115. [ Links ]
Kleppel, G. S., R. E. Dodge & C. J. Reese. 1989. Changes in pigmentation associated with the bleaching of stony corals. Limnol. Oceangr. 34: 1331-1335. [ Links ]
Kushmaro, A., Y. Loya, M. Fine & E. Rosenberg. 1996. Bacterial infection and coral bleaching. Nature 380: 396. [ Links ]
Kushmaro, A., E. Rosenberg, M. Fine, Y. B. Haim & Y. Loya. 1998. Effect of temperature on bleaching of the coral Oculina patagonica by Vibrio AK-1. Mar. Ecol. Prog. Ser. 171: 131-137. [ Links ]
Lasker, H. R., E. C. Peters & M. A. Coffroth. 1984. Bleaching of reef coelenterates in the San Blas Islands, Panama. Coral Reefs 3: 183-190. [ Links ]
Lesser, M. P. 1996. Elevated temperatures and ultraviolet radiation cause oxidative stress and inhibit photosynthesis in symbiotic dinoflagellates. Limnol. Oceanogr. 41: 271-283. [ Links ]
Lesser, M. P. 1997. Oxidative stress causes coral bleaching during exposure to elevated temperatures. Coral Reefs 16: 187-192. [ Links ]
Lesser, M. P., W. R. Stochaj, D. W. Tapley & J. M. Shick. 1990. Bleaching in coral reef anthozoans: effects of irradiance, ultraviolet radiation, and temperature on the activities of protective enzymes against active oxygen. Coral Reefs 8: 225-232. [ Links ]
Marsh, J. A. 1970. Primary productivity of reef-building calcareous red algae. Ecology 51: 255-263. [ Links ]
McCarthy, N. J & G. I. Evans. 1998. Methods for detecting and quantifying apoptosis. Curr. Top. Develop. Biol. 36:259-278. [ Links ]
McCloskey, L. R., T. G. Cove & E. A. Verde. 1996. Symbiont expulsion from the anemone Anthopleura elegantissima (Brandt) Cnidaria; Anthozoa).. J. Exp. Mar. Biol. Ecol. 195: 173-186. [ Links ]
Meltzar, S & M. C. Berman. 1984. Effects of pH, temperature, and calcium concentration on the stoichiometry of the calcium pump of sarcoplasmic reticulum. J. Biol. Chem. 259: 4244-4253. [ Links ]
Muscatine, L., D. Grossman & J. Doino. 1991. Release of symbiotic algae by tropical sea anemones and corals after cold shock. Mar. Ecol. Prog. Ser. 77: 233-243. [ Links ]
Nakamura, T. & R. Van Woesik. 1997. Water flow rates and passive diffusion partially explain differential survival of corals during the 1998 bleaching event. Mar. Ecol. Prog. Ser. 149: 163-171. [ Links ]
Nii, C. M. & L. Muscatine. 1997. Oxidative stress in the symbiotic sea anemone Aiptasia pulchella (Carlgren, 1943): Contribution of the animal to superoxide ion production at elevated temperature. Biol. Bull. 192: 444-456. [ Links ]
Ogden, J. & R. Wicklund. 1988. Introduction, p.1-9. In J. Ogden & R. Wicklund (eds.).Mass Bleaching of Corals in the Caribbean: A Research Strategy. Natl. Undersea Res. Prog. Rep. 88-2, NOAA, Washington DC. [ Links ]
Patterson, C. O. P. & J. Myers. 1973. Photosynthetic production of hydrogen peroxide by Anacystis nidulans. Plant Physiol. 51: 104-109. [ Links ]
Perez, S. F., C. B. Cook & W. R. Brooks. 2001. The role of symbiotic dinoflagellates in the temperature-induced bleaching response of the subtropical sea anemone Aiptasia pallida. J. Exp. Mar. Biol. Ecol. 256: 1-14. [ Links ]
Ralph, P. J., R. Gademann & A. W. D. Larkum. 2001. Zooxanthellae expelled from bleached corals at 33ºC are photosynthetically competent. Mar. Ecol. Prog. Ser. 220: 163-168. [ Links ]
Rega, A. F. 1986. Other properties and coupling of Ca2+ transport and ATP hydrolysis: p. 91-104. In: A. F. Rega & P. J. Garrahan, The Ca2+ pump of plasma membranes. CRC Press, Boca Raton, Florida, USA. [ Links ]
Searle, J., J. F. R. Kerr & C. J. Bishop. 1982. Necrosis and apoptosis: distinct modes of cell death with fundamentally different significance. Pathol. Annu. 17: 229-259. [ Links ]
Slater, A. F. G., C. Stefan, I. Nobel, D. J. van den Dobbelsteen & S. Orrenius. 1995. Signalling mechanisms and oxidative stress in apoptosis. Toxicol. Lett. 82/83: 149-153. [ Links ]
Steen, R. G. & L. Muscatine. 1987. Low temperature evokes rapid exocytosis of symbiotic algae by a sea anemone. Biol. Bull. 172: 246-263. [ Links ]
Szmant, A. & N. J. Gassman. 1990. The effects of prolonged bleaching on the tissue biomass and reproduction of the reef coral Montastrea annularis. Coral Reefs 8: 217-224. [ Links ]
Tytler, E. M. & R. K. Trench. 1986. Activities of enzymes in 
Vicente, V. P 1990. Response of sponges with autotrophic endosymbionts during the coral-bleaching episode in Puerto Rico. Coral Reefs 8: 199-204. [ Links ]
Vogel, A. I. 1961. A text-book of quantitative inorganic analysis including elementary instrumental analysis. Longmans, Green, London. 363 p. [ Links ]
Warner, M. E., W. K. Fitt & G. W. Schmidt. 1999. Damage to photosystem II in symbiotic dinoflagellates: A determinant of coral bleaching. Proc. Natl. Acad. Sci. USA 96: 8007-8012. [ Links ]
Williams, E. H., Jr & L. Bunkley-Williams. 1990. The world wide coral bleaching cycle and related sources of coral mortality. Atoll Res. Bull. 335: 1-71. [ Links ]
Yonge, C. M. & A. G. Nicholls. 1931. Studies on the physiology of corals IV. The structure, distribution and physiology of the zooxanthellae. Great Barrier Reef Expedition 1928-229, Scient. Rep. 1: 135-176.