Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.54 n.2 San José Jun. 2006
Differential leaf gas exchange responses to salinity and drought in the mangrove tree Avicecennia germinans (Avicenniaceae)
M.A. Sobrado
Laboratorio de Biología Ambiental de Plantas, Departamento de Biología de Organismos, Universidad Simón Bolívar, Apartado 89.000, Caracas, Venezuela. Fax: (58-212) 906 3064; msobrado@usb.ve
Received 20-IX-2001. Corrected 02-X-2005. Accepted 23-II-2006.
Abstract. Leaf gas exchange was assessed in Avicennia germinans L. grown under different NaCl concentrations (0-40), after salt-relief, and then during drought. Stomatal conductance (gs) and net photosynthetic rate (Pn) decreased with increasing NaCl concentration, and intrinsic water use efficiency (Pn / gs) increased. Under desalinization Pn / gs declined. Thus, gs did not change in plants grown at low NaCl concentration (10), but increased up to 30-32% at higher NaCl concentration (20 - 40). However, Pn was only slightly enhanced (10- 15%). Under drought, Pn decreased by as much as 46% in plants grown at low NaCl concentration (10) and by 22% at high NaCl concentration (40). Thus, Pn / gs decreased and water use efficiency was lower during drought compared to estimates prior to salt-relief. Rev. Biol. Trop. 54(2): 371-375. Epub 2006 Jun 01.
Key words: Avicennia germinans, mangrove, NaCl, stomatal conductance, water use efficiency.
Salinity-dependent reduction in carbon assimilation is a function of stomatal limitation of CO2 diffusion into leaves, of ion toxicity, and of the impairment of essential ion uptake (Flowers et al. 1977, Ball and Farquhar 1984a, b, Flowers and Yeo 1986, Munns 1993). One remarkable feature of the leaf gas exchange of mangrove species is their low transpiration and high water use efficiency (Ball and Farquhar 1984a, b). As NaCl concentration increases, mangrove water use characteristics become conservative (Ball 1988, Clough and Sim 1989, Medina and Francisco 1997). Avicennia germinas L. is a mangrove well known for its ability to thrive in areas with both high and fluctuating salinity. Drought enhances the salinity effect on mangrove species performance and distribution (Lugo and Snedaker 1974).Effect of drought on photosynthesis of A. germinans has been conducted under field conditions where interplay with NaCl concentration was unavoidable (Smith et al. 1986, Sobrado 1999a). Here, leaf gas exchange was assessed in A. germinans grown under a variety of salinity conditions, after salt was removed, and finally when watering was withheld. Thus, soil NaCl concentration increases with drought progression were avoided.
Materials and methods
Ten plants of A.germinans L. Stearn per treatment were grown in pots with sand at salinities of 0, 10, 20, 30 and 40 NaCl. Plants were kept in a glasshouse with natural sunlight and photoperiod for six months. Gas exchange measurement was taken prior to salt-relief in each plant group. Then plants were removed from salinity by washing the salt repeatedly, and pots were watered with full Hoagland solution (desalinization). After three days of salt relief, gas exchange measurements were taken again in each plant group. Afterwards, watering was withheld for seven days (drought), and leaf gas exchange measurements were taken again. Five of the ten control plants grown without salt addition were maintained under irrigation and five plants were subjected to drought. Gas exchange measurements were taken at midday in two fully expanded sunny leaves per plant per treatment, by using a portable gas analyzer model LCA-2 (Analytical Development Co., Herts, UK). Average irradiation for measurements was 1600 μmol m-2 s-1, leaf temperature was between 29-32 ºC (prior to salt relief) and 33-34 ºC (drought), relative humidity was 70- 75% and ambient CO2 concentration was 350 µmol mol-1. Leaf water potential (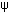
Results
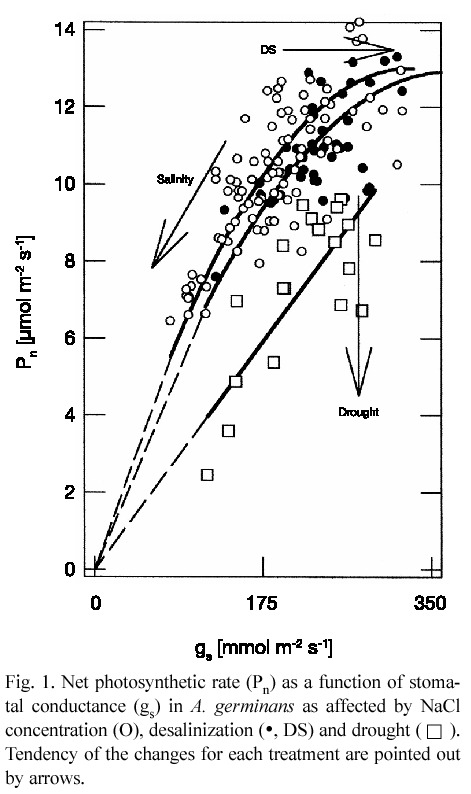
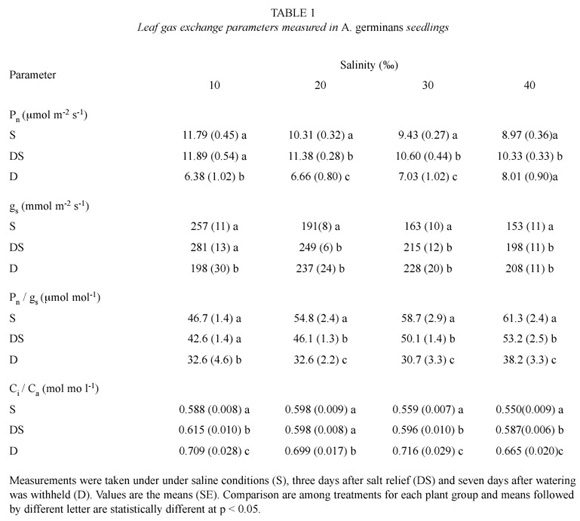
In control plants (0), the mean values of measurements in the three stages were net photosynthetic rate (Pn) of 14.9 ±0.5 µmol m-2 s-1, gs of 365.5 ±20.4 mmol m-2 s-1 and Ci/Ca of 0.597±0.022. Similarly, leaf water potential (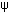
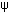
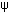
The 7-d drought caused a comparable 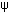
Discussion
In this study, leaf gas exchange parameters, under salinity or after salinity relief, pointed to increasing water use efficiency as NaCl concentration became higher, as has been previously found (Smith et al. 1989, Medina and Francisco 1997, Sobrado 1999b). However, non-stomatal limitations overrode effect of drought on Pn and water use efficiency was lowered. Leaf ion concentration of A. germinans can remain high up to 10 days after salt is eliminated from the soil (Suárez et al. 1998, Suárez and Sobrado 2000). Thus, plants grown under higher NaCl concentration are osmotically adjusted and have high leaf turgor and leaf water content for a given 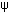
In conclusion, there were differences in the response of Pn and gs to NaCl concentration and drought. Stomatal functioning appears to be consistent with maximizing carbon gain by water loss under salinity. However, the reverse trend occurs during desalinization and drought. Declining water use efficiency as consequence of drought in the field overrides the opposite effect as result of salinity (Sobrado 1999a) Thus, the interaction of drought with salinity, as found in the field, may modulate the overall ecophysiological performance of A. germinans in drought-prone areas. In A. germinans, an accurate control of water loss to salt secretion capacity is required to maintain the balance of salt influx-efflux at leaf level (Sobrado 2001). Failure in water loss control may lead to salt build-up in leaves and to enhancement of leaf senescence.
Acknowledgments
Financial support was provided by DIDUSB.
Resumen
Se estudió el intercambio de gases en las hojas de Avicennia germinans L. en varias concentraciones de NaCl (0-40), después de la desalinización y durante la desecación. La conductancia de los estomas (gs) y la tasa de fotosíntesis (Pn) decrecieron con el incremento en la concentración de NaCl, y se incrementó la eficiencia en el uso intrínseco de agua (Pn / gs). Bajo desalinización Pn / gs declinó. Así, gs no cambia en el crecimiento de las plantas a bajas concentraciones de NaCl (10), pero se incrementó hasta 30-32% a las concentraciones de NaCl más altas (20 - 40). Sin embargo, Pn aumentó ligeramente (10-15%). En desecación Pn fue reducido hasta un 46% a bajas concentaciones (10) de NaCl, y a un 22% a altas concentraciones (40) de NaCl. Así, Pn / gs decrecieron y la eficiencia en el uso de agua fue menor durante desecación en comparación con los evalolres stimados previos a la desalinización.
Palabras clave: Avicennia germinans, manglar, NaCl, conductancia, estomas, eficiencia de uso de agua.
References
Ball, M.C. 1988. Salinity tolerance in the mangrove Aegiceras corniculatum and Avicennia marina. I. Water use in relation to growth, carbon partitioning, and salt balance. Aust. J. Plant Physiol. 15: 447-464. [ Links ]
Ball, M.C. & G.D. Farquhar. 1984a. Photosynthetic and stomatal responses of two mangrove species, Aegiceras corniculatum and Avicennia marina, to long-term salinity and humidity conditions. Plant Physiol. 74: 1-6. [ Links ]
Ball, M.C. & G.D. Farquhar. 1984b. Photosynthetic and stomatal responses of the grey mangrove, Avicennia marina, to transient salinity conditions. Plant Physiol. 74: 7-11. [ Links ]
Clough, B.F. & R.G. Sim. 1989. Changes in gas exchange characteristics and water use efficiency of mangroves in response to salinity and vapor pressure deficit. Oecologia 79: 38-44. [ Links ]
Flowers, T.J., P.F. Troke & A.R. Yeo. 1977. The mechanism of salt tolerance in halophytes. Annu. Rev. Plant Physiol. 28: 89-121. [ Links ]
Flowers, T.J. & A.R.Yeo. 1986. Ion relations of plants under drought and salinity. Aust. J. Plant Physiol. 13: 75-91. [ Links ]
Jarvis, P.G. & T.A. Mansfield. 1980. Reduced responses to light, carbon dioxide and abscisic acid in the presence of sodium ions. Plant Cell Environm. 3: 279-283. [ Links ]
Lugo, A.E. & S.C. Snedaker. 1974. The ecology of mangroves. Annu. Rev. Ecol. Syst. 5: 39-64. [ Links ]
Medina, E. & M. Francisco. 1997. Osmolality and 13C of leaf tissue of mangrove species from environments of contrasting rainfall and salinity. Estuarine Coast Shelf. Sci. 45: 337-344. [ Links ]
Munns, R. 1993. Physiological processes limiting plant growth in saline soils: some dogmas and hypothesis. Plant, Cell and Environ. 16: 15-24. [ Links ]
Naidoo, G. & D. J von Willert. 1994. Stomatal oscillations in the mangrove Avicennia germinans. Functional Ecol. 8: 651-657. [ Links ]
Smith, J.A.C., M. Popp, U. Lüttge, W.J. Cram, M. Diaz, H. Griffith, H.S.J. Lee, E. Medina, C. Schäfer, K.H. Stimmel & B. Thonke. 1989. Ecophysiology of xerophytic and halophytic vegetation of a coastal alluvial plain in northern Venezuela.VI. Water relations and gas exchange of mangroves. New Phytol. 11: 293-307. [ Links ]
Sobrado, M.A.1999b. Drought effect on photosynthesis of the mangrove Avicennia germinans under contrasting salinities. Trees 13: 125-130. [ Links ]
Sobrado, M.A. 1999b. Leaf photosynthesis of the mangrove Avicennia germinans as affected by NaCl. Photosynthetica 36: 547-55. [ Links ]
Sobrado, M.A. 2001. Effect of high external NaCl concentration on the osmolality of xylem sap, leaf tissue and leaf glands secretion of the mangrove Avicennia germinans (L.). Flora 196: 63-70. [ Links ]
Suárez, N., M.A. Sobrado & E. Medina. 1998. Salinity effects on the leaf water relations components and ion accumulation patterns in Avicennia germinans L. seedlings. Oecologia 114: 299-304. [ Links ]
Suárez, N. & M.A. Sobrado. 2000. Adjustments in leaf water relations of the mangrove, Avicennia germinans (L.) L., grown in a salinity gradient. Tree Physiol. 20: 277-282. [ Links ]
Tomlinson, P.B. 1986. The Botany of mangroves. Cambridge University, London. [ Links ]
Waisel, Y., A. Eshel & M. Agami. 1986. Salt balance of leaves of the mangrove Avicennia marina. Physiol. Plant. 67: 67-72. [ Links ]