Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.54 n.1 San José Mar. 2006
Microbial degradation of palm (Elaeis guineensis )biodiesel
Giselle Lutz1, Max Chavarría1, María Laura Arias2 & Julio F. Mata-Segreda1
1 Laboratory of Bio-organic Chemistry, School of Chemistry, University of Costa Rica, 2060, Costa Rica; glutz@equi.ucr.ac.cr, mcharria@equi.ucr.ac.cr, jmata@cariari.ucr.ac.cr
2 Faculty of Microbiology, University of Costa Rica, 2060, Costa Rica; mlarias@cariari.ucr.ac.cr
Received 18-III-2005. Corrected 26-VII-2005. Accepted 01-VIII-2005.
Abstract: The kinetics of biodegradation of palm-derived fatty methyl and ethyl esters (Elaeis guineensis biodiesel) by a wild-type aerobic bacterial population was measured at 20 °C, as the rate of oxygen uptake by a manometric technique. The methyl and ethyl biodiesels were obtained by potassium-hydroxide catalysed transesterification of palm oil, respectively. The bacterial flora included the genera Bacillus, Proteus, Pseudomonas, Citrobacter and Enterobacter. The rate of oxygen uptake for palm biodiesel is similar to the quantity observed in the biodegradation of 1.0 mM solutions of simple substrates such as carbohydrates or amino acids.Palm methyl or ethyl biodiesel is subjected to facile aerobic biodegradation by wild-type bacteria commonly present in natural open environments. This result should lessen any environmental concern for its use as alternative fuel, solvent or lubricant. Rev. Biol. Trop. 54(1): 59-63.Epub 2006 Mar 31.
Keywords: Palm biodiesel, fatty esters, Elaeis guineensis, wild-type bacterial degradation, kinetics.
The limited resources and increasing price of petroleum, as well as global environmental concerns have been the main reasons for exploring the use of natural triacylglyceras alternative sources of fuels. Biodiesel has an important role amongst biomass products, mainly due to the environmental problems associated to Diesel fuel. The term biodiesel commonly indicates the mixtures of methyl or ethyl fatty esters obtained upon transesterification of TAG with either methanol or ethanol. The source of TAG can be either animal or vegetal (Mittelbach and Remschmidt 2004, Tashtoush et al. 2004), but also from recycled fatty materials (Mittelbach and Remschmidt 2004, Çetinkaya and Karaosmanolu 2004).
Technological emergencies with petroleum or petrochemical materials are one of the most serious environmental problems. When accidental or negligent spills occur, the hydrocarbon components may reach underground water reservoirs before becoming immobilised in the soil phase. After diffusion dynamics come to equilibrium, hydrocarbons become partitioned into ground water, soil pore space and to the surfaces of soil particles. Persistency of these pollutants in the environment depends on the intrinsic biodegradability of the constituting hydrocarbons. Thus, microorganisms capable of degrading hydrocarbons play an important role in the soil and aquatic environment.
Fatty acids and their derivatives such as esters are natural cellular fuels. Therefore, it is important to consider the environmental impact of fatty esters, in relation to petrochemical products. The rate of biodegradation of soybean methyl biodiesel has been found as fast and complete as that of glucose by the action of soil microbial populations (Zhang et al .1995).
Triacylglycerols from palm (Elaeis guineensis) is one of the possible sources of TAG for the synthesis of biodiesel in Costa Rica, although its chemical composition is different from the common highly unsaturated examples in the literature such as cotton, rapeseed, soybean, sunflower, etc. We report in this article a kinetic study of the biodegradation of palm fatty esters, by an aerobic wild-type bacterial population model.
Materials and methods
Materials: All reagents were common chemicals taken from the shelf. The biodiesel samples were synthesised from the transesterification of palm oil with either methanol or ethanol, and potassium hydroxide as catalyst. The purity of the materials, as fatty monoesters, was determined to be at least 98% by proton NMR. (Lutz, unpublished results).Diesel fuel was obtained from a neighbourhood service station.
Bacterial population: The wild-type bacterial cocktail marketed as Superbugs ® was used. Routine isolation techniques (Vanderzant and Splittstoesser 1992) were employed in determining the material to contain spores or vegetative forms of the following genera: Bacillus, Proteus, Pseudomonas, Citrobacter and Enterobacter .
Kinetic measurements and data treatment: Experiments were carried out by dissolving the soluble substrates in distilled water (1.0 mM). For the cases of sucrose, starch and egg albumin, the monomer-unit concentration was also 1.0 mM (virtual molecular weight of glucosyl and fructosyl moieties taken as 162 and 110 for aminoacyl moieties). To 250 cm3 of each substrate aqueous solution, 0.1 g of the aerobic bacterial preparation was added.No mineral nutrients were added to avoid supporting microbial growth.
The rate of bacterial aerobic degradation of the different materials was followed by determining oxygen consumption by means of Oxytop ® micromanometers, at 20 ºC. The overall kinetic law of this kind of processes is adequately described by the equation (Mihelcic 2001):
-d [O2]/dt =k m [O2] [Organic material]
where m is bacterial density and k the corresponding rate constant. All experiments were carried out in duplicate, with initial substrate concentration of 1.0 mM and bacterial preparation at 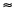
After 1-3 day induction periods, the amount of oxygen uptake varied linearly with time during the follow-up period of 10-20 days, thus allowing the calculation of the initial rate of oxygen consumption (ri) under steady-state conditions. For substrates readily metabolised (short length of induction period), ri was calculated as:
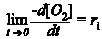
Thus, relative k values can be analysed for the different soluble substrates, from the observed ri/m values. The rationale is based on the fact that millimolar substrate concentrations are high enough to saturate the bacterial biochemical systems.
For the cases of the insoluble materials, the experiments were done by thorough mixing of 1.00 cm3 of the samples with 1.0 dm3 of distilled water. To an aliquot portion of 250 cm3 of the suspension, 0.1 g of Superbugs ® was added.
The values of initial rates were obtained by least-squares fitting of the [O2]-time data pairs in the linear range observed in each experiment. All comparisons between the ri/m magnitudes were done by Student t-test null hypothesis.
Results
All substrates, except petroleum-derived Diesel fuel, showed apparent zeroth-order kinetics.
The rates of bacterial degradation of the different materials are given in table 1, and expressed as ri/m. The mean standard deviation in all experiments was ±10%.
No significant difference was found between the rates of degradation of the soluble substrates and those observed for palm methyl or ethyl biodiesels (0.09 ± 0.01 mM h-1 vs. 0.12 ± 0.01 mM h-1/g).
Discussion
The microbial population used in this experiment proved capable of degrading both Diesel fuel and biodiesel. Indeed, biodegradation of petroleum derivatives has been observed for pure cultures of all the bacterial genera contained in the microbial material used in this study (Maachi et al .2001, Marchal et al. 2003, Obire and Nwaubeta 2002, Riis et al. 2003, Williams et al .1998).
The zeroth-order kinetic result observed for all substrates, except for Diesel fuel, suggests that substrate transport across bacterial cell walls and the metabolic machinery were operating under saturation conditions. For carrier-mediated transport, typical values for half-saturation concentrations are from one up to five orders of magnitude lower than the 1.0 mM substrate concentrations used in this work. (Lengeler et al .1999).
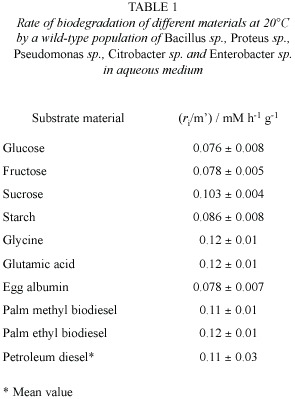
Fig. 1 shows palm ethyl biodiesel as a readily biodegradable material, compared to the case in Fig. 2 for Diesel fuel. The last proved more refractory for biodegradation, in the sense that a larger extent of oxygen uptake was observed by the fatty esters. The petroleum-material system approaches asymptotically to a maximum oxygen uptake of 4.1 mM by day 12, whereas the fatty esters went on to up to 6-7 mM by day 27.
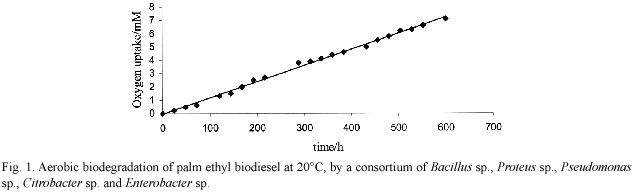
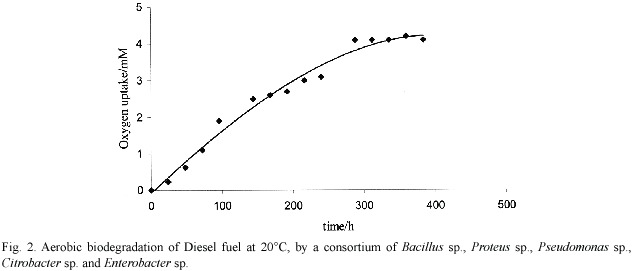
This result had already been observed, and can be rationalised in terms of the recalcitrant resistance to biodegradation of the major isoalkane and cycloalkane fraction -circa 50%- present in Diesel fuel (Marchal et al .2003).
The mean value theorem allows the estimation of the rate of Diesel fuel biodegradation, in the time range from 0-384 h (Cogan et al. 1960). The observed mean rate of biodegradation for Diesel fuel of 0.11 ± 0.03 mM h-1/g is similar to those observed for the other organic substrates, as expected from the catabolic pathways for carbohydrate, lipid and protein:
In conclusion, palm methyl or ethyl biodiesel is subjected to facile aerobic biodegradation by wild-type bacteria commonly present in natural open environments. This result should lessen any environmental concern for its use as alternative fuel, solvent or lubricant.
Resumen
La cinética de la biodegradación de los ésteres metílicos y etílicos derivados de palma (biodiesel) por una población silvestre de bacterias aeróbicas fue medida a 20 °C, como medición manométrica del consumo de oxígeno. Los ésteres metílicos y etílicos se obtuvieron por transesterificación del aceite de palma con metanol y etanol,respectivamente. La flora bacteriana incluyó a los géneros Bacillus, Proteus, Pseudomonas, Citrobacter y Enterobacter. Las velocidades de consumo de oxígeno para las muestras de biodiesel fueron similares a lo observado en la biodegradación de disoluciones 1.0 mM de sustratos sencillos solubles en agua, tales como carbohidratos, aminoácidos y albúmina de huevo.
References
Çetinkaya, M. & F. Karaosmanolu. 2004. Optimization of base-catalyzed transesterification reaction of used cooking oil. Energy Fuels 18:1888-1895. [ Links ]
Cogan, E. J., R. Z. Norman & G. L. Thompson. 1960. Calculus of functions of one argument. Prentice-Hall, Englewood Cliffs, New Jersey, USA, p. 143-144. [ Links ]
Lengeler, J. W., G. Drews & H. G. Schlegel. 1999. The biology off the prokaryotes. Thieme, Stuttgart, Germany, p.205-206. [ Links ]
Maachi, R., M. Abousseoud & T. Chaabane. 2001. Kinetics of biodegradation of petroleum by Pseudomonas sp. Desalination 139:367. [ Links ]
Marchal, R., S. Penet, F. Solano-Serena & J. P. Vandecasteele. 2003. Gasoline and diesel oil biodegradation. Oil & Gas Science and Technol.–Rev. IFP 58:441-448. [ Links ]
Mihelcic, J. R. 2001. Fundamentos de ingeniería ambiental. Limusa Wiley, México, D. F., p. 299-303. [ Links ]
Mittelbach, M. & C. Remschmidt. 2004. Biodiesel. The comprehensive handbook. Boersedruck Ges. M. B. H., Vienna, pp. 27-35. [ Links ]
Obire, O. & O. Nwaubeta. 2002. Effects of refined petroleum hydrocarbons on soil physicochemical and bacteriological characteristics. J. Appl. Sci. Environ. Manag. 6: 39-44. [ Links ]
Plohl, K., H. Leskovek & M. Bricelj. 2002. Biological degradation of motor oil in water. Acta Chim. Slov. 49:279-289. [ Links ]
Riis, V., S. Kleinsteuber & W. Babel. 2003. Influence of high salinities on the degradation of diesel fuel bacterial consortia. Can. J. Microbiol. 49: 713-721. [ Links ]
Tashtoush, G. M., M. I. Al-Widyan & M. M. Al-Jarrah. 2004. Experimental study on evaluation and optimization of conversion of waste animal fat into biodiesel. Energy Conversion Manag. 45: 2697-2711. [ Links ]
Vanderzant, C & D. F. Splittstoesser. 1992. Compendium of methods for the microbiological examination of food. APWA, Washington, D. C. [ Links ]
Williams, D. L., K. D. Kriel, G. A. Stewart, R. C. Hulse, J. E. Holsomback, Jr. & J. R. Stewart. 1998. Bioremediation of oil-contaminated soils by stimulating indigenous microbes. Environ. Geosci. 5: 1-8. [ Links ]
Internet reference
Zhang, X., C. L. Peterson, D. Reese, G. Möller & R. Haws. 1995. Biodegradability of biodiesel in the aquatic environment. Department of Agricultural Engineering, University of Idaho, Moscow, Idaho, USA. (Downloaded: December 5, 2004, http://biodiesel.org/resources/reportsdatabase/reports/mar/19950601_mar-009.pdf). [ Links ]














