Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.53 n.3-4 San José Sep. 2005
The Tadpoles of Hyla rufitela (Anura: Hylidae)
Heinz Hoffmann
INASAG, the Costa Rican Private Tadpole Research Center, Apartado 153, Barva-3011, Costa Rica; heinz.hoffmann@gmx.com
Received 18-II-2005. Corrected 22-VII-2005. Accepted 05-IX-2005.
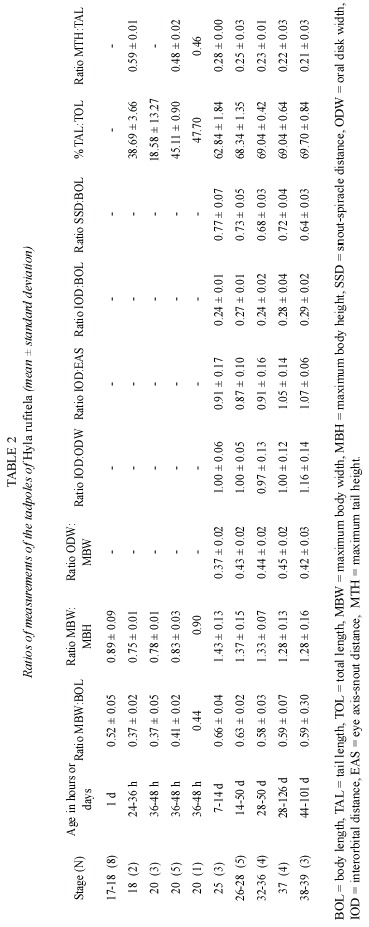
Abstract: The validity of the published description of Hyla rufitela is questioned because the origin of the described tadpoles does not correspond to the distribution of this species. The present description, based on tadpoles that were hatched from two egg masses from the Caribbean lowland of Costa Rica, includes tadpoles raised to adult frogs to verify the species. The newly described tadpoles differ from the previously described and illustrated ones not only in color but also by its more slender body shape (body height:body length, 0.43:0.47), the more slender tail (% tail length of total length, 69%:63%), the more conspicuous spiracle and its position, as well as by the larger oral disk width (oral disk width:body width, 0.44:~0.33). The young tadpoles continually ingest mud as is typical of bottom feeders. Nevertheless, older larvae feed on plant material like rotting mellow leaves of Piper auritum. The tadpoles metamorphosed between 44 and 114 days (x=80.3 days) under the described breeding conditions. Rev. Biol. Trop. 53(3-4): 561-568. Epub 2005 Oct 3.
Key words: Hyla rufitela, tadpole, larvae, description, morphology, allometry, development, ecology.
Duellman (1970) described tadpoles from the Colorado Islands, Panama, and from the Golfito region in southwestern Costa Rica as Hyla rufitela Fouquette 1961. He described the first larvae stages 20 to 25 (according to Gosner, 1960) from the Panama population, but the later stages he described using the relevant morphological data and the illustration from the Golfito sample of Costa Rica. The same author repeats the description in 2001, in the enlarged edition of the same title.
Duellman cited a few records of H. rufitela from northeast and also from southwest Costa Rica where H. rosenbergi is common. This means that both species would be sympatric in the Golfito region, as contrasted with Savages conclusion that both species are not sympatric. According to Savage, H. rufitela is relatively widespread, but only in the northern and eastern Atlantic slope of Costa Rica, while H. rosenbergi is restricted to the middle and south pacific slope. The Museum of the Universidad of Costa Rica collection includes 29 specimens of H. rufitela from 18 different sites, but all from the Atlantic slope. Although Lips and Savage (1996) give a tad-pole key that allows correctly identify H. rufitela larvae, Savage (2002) concluded that the larvae are still unknown, and that Duellmans account and illustration are based on tadpoles of Hyla rosenbergi.
The present study clarifies the H. rufitela – H. rosenbergi distinction through a new and more complete description of live tadpoles of H.rufitela. It also provides further ecological observations about H. rufitela.
For this study, some tadpoles were reared to froglets and adults to prove the identity of the species. Samples of the described tadpoles were deposited in the authors collection, in the Smithsonian Institution of the Natural Museum of Natural History, Washington, and also as sample # UCR 17393 in the collection of the Museum of Zoology of the Universidad de Costa Rica, together with an adult frog of the same population (# UCR 17394).
Materials and methods
The described tadpoles of H. rufitela originate from a population of the Caribbean Lowlands of Costa Rica at an elevation of 70 m (Ombú Experimental Farm, Cantón Pococí, Puerto Limón province; 10°16N, 83°43W). This farm is mainly covered with pasture and banana experimental plots, but a small swampy forest in the center of the farm has been preserved as biotope and was enlarged in the early nineties by an adjacent reforested area. In this forest, the males of H. rufitela call mostly from leaves and small branches over a small, slow moving, naturally draining watercourse. Calla plants on the border of this brook are the preferred perching places.
Most of the observed males perch about 70 cm above water, but on a few occasions I found them calling as high as 1.20 m above the water or just slightly to the side of the water source, over land. On December 4, 2002, I obtained the egg mass from a frog pair that I found the night before in amplectic position perching nearby the drain at 60 cm above the ground. That night, the pair adhered their egg clutch in the form of a surface film, on a semi-submerged cane 25 cm distance from the shore. The blackish eggs floated in one single layer, each one in the midst of a transparent jelly of extremely thin consistence. The clutch had a nearly rectangular form, covering 15 x 12 cm. At the oviposition site, the water was about 10 cm deep above the muddy bottom. On May 14, 2004, I found another egg mass in a dammed pond that is surrounded by Calla plants (Fig. 1).
The egg mass of the first clutch was transferred into a 33 x 50 cm container with water and mud from the ovipositioning site, with a water depth of 10 cm. The water was oxygenated with a slow air flow pump. Meanwhile the very fragile jelly was in the process of disintegration, the embryos dropped to the muddy bottom. Most of the eggs were transferred into a container with clear water without mud, but with plants and artificial aeration by air pump. About 2.5 days after oviposition, most larvae hatched at stages 19 to 20, according to Gosner (1960), but at the same time some larvae developed quicker and in stage 23 they were already swimming in the water container. These swimming tadpoles swam towards the waters surface, then sank downwards again. One day later, all the larvae died under these conditions. Nevertheless, the remaining larvae in the container with the original mud covered bottom survived and developed. These larvae were used for most of the tadpole description. They ate mud continually like typical bottom feeders. In this way, they presumably obtained detritus with the necessary organic material for nutrition within a larger quantity of indigestible material. The gut changed quickly to the color of the yellow mud, and the intestinal contents showed a similar structure as that of the mud. This observation suggests that the larvae feed unselectively during the early life stages. They ate mud continually like typical bottom feeders. In this way, they presumably obtained detritus with the necessary organic material for nutrition within a larger quantity of indigestible material. The gut changed quickly to the color of the yellow mud, and the intestinal contents showed a similar structure as that of the mud. This observation suggests that the larvae feed unselectively during the early life stages. Nevertheless, older larvae feed on plant material like rotting mellow leaves of Piper auritum. Forty-four days after oviposition, the first two froglets of total 44 tadpoles had already metamorphosed. The last one metamorphosed at 114 days (x = 80.3 days) under the previously described artificial rearing conditions.
To document ontogenetic changes during growth, 19 tadpoles between stages 17 and 20, in addition to 19 living tadpoles between stages 25 and 39, were taken for measurements and descriptions. The tadpoles were staged according to Gosner (1960). The conveniences and limitations of the type of allometric data collected here is discussed in publication (Hoffmann 2004).
Description
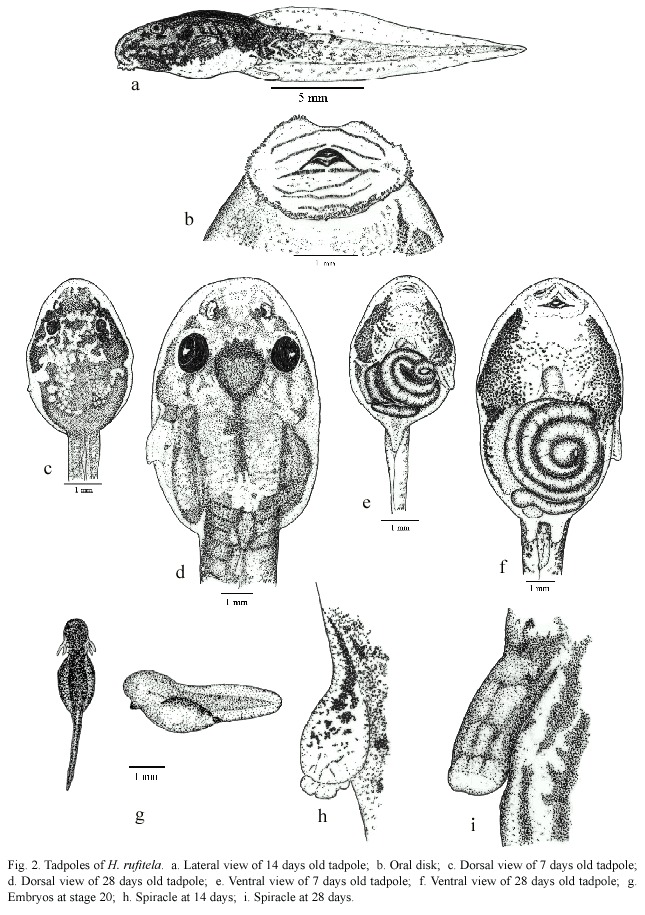
The eggs are blackish gray, and the developing embryos are still darker than the dark grayish cover of the yolk reserve. The hatchlings drop out of the eggs jelly with just the slightest disturbance, being in stage 20. The hatchlings are laterally compressed, they are higher than they are wide (Fig. 2g). From stage 25 on, the tadpoles start to change their compression axis, becoming more dorsoventral depressive (Fig. 2a, c-f). In these stages of development, the relation of body width to body length changes from ratios of about 0.40 to about 0.60 (Table 1 and 2). Therefore, at stage 25 or higher the body shape is characterized as depressed, following McDiarmid and Altig (1999).
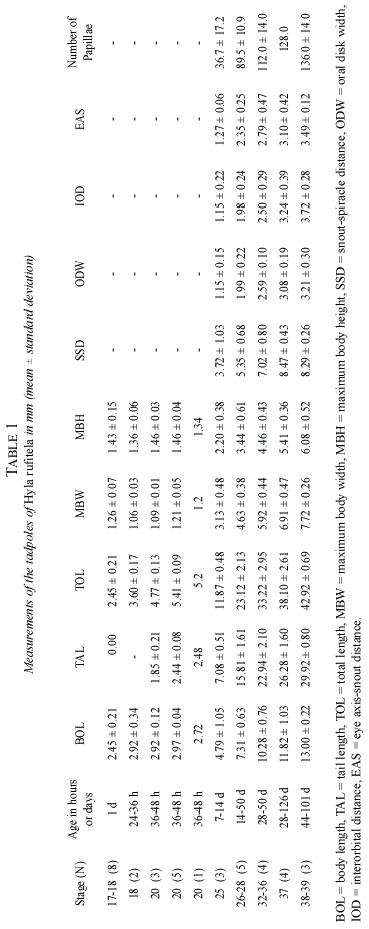
In dorsal view (Fig. 2c and d), the tadpoles at stage 25 and higher stages are of oval shape and the body is posteriorly truncated. The shape of the snout is between rounded and truncated, in both lateral and dorsal views. The nostrils are midway between the widely separated eyes and the tip of the snout. In small larvae, the conspicuous sinistral spiracle is pear-shaped with poor pigmentation; at later stages, the spiracle enlarges more quickly as the body grows and the spiracle becomes hose-shaped (Fig. 2h and i). The notably large sized spiracle of big tadpoles opens slightly above the mid-line level of the body at about two-thirds to three-quarters of the distance from the tip of the snout to the bodys distal end at the edge of the beginning of the anal tube.
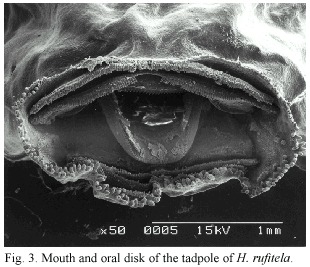
The anal tube is long and opens slightly dextrally. The mouth is ventral and the oral disks width is large, i.e., equal to about 44% of the greatest width of the body. The disk is provided with a broad gap on the upper lip; the rest of the mouth is bordered by a large number of papillae. The papillae are arranged in a single row on the dorsolateral part of the mouth; the ventrolateral and ventral lip is surrounded by a double row of papillae. The number of papillae increases with larvae growth from about 30 at stage 25, to about 150 at stage 38 (Fig. 2b and 3). The tooth row formula is 2/4. The A-rows are longer than the P-rows following terminology of McDiarmid and Altig (1999). A1 is continuous and double rowed, at least on the illustrated specimen on Fig. 3 that was taken with electronic microscopic photography; A2 has a small gap in the center; the length of the tooth rows P 1 to P3 are more or less equal; in some specimen P4 might be interrupted (Fig. 3). The upper and lower beaks are pigmented and serrated. Whereas the upper beak is W-shaped, the lower beak is V-shaped.
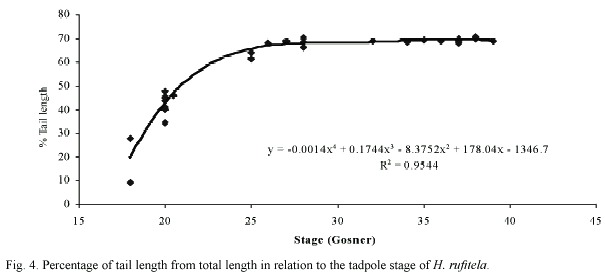
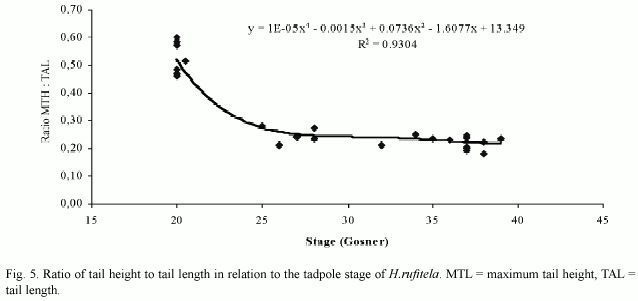
The tail begins to develop at stage 18 and acquires its definitive shape and proportion in relation to the total length at stage 26. The proportion of the tail length increases in relation to the total length from about 18% at stage 18, to 63% at stage 25, and remains at about 69% from stage 26 to stage 39 (Fig. 4). The ratio of maximal tail height to total tail length changes noticeably from stages 18 to 26, decreasing in these stages from 0.48 to 0.23 (Fig. 5). Therefore, the tails of middle-size to large tadpoles of H. rufitela have a roughly similar shape. The upper tail fin is inserted in the body end and is noticeably higher than the lower fin on the highest part of the tail (about 39% vs. 24% of the total tail height). The musculature takes up about 37% of the total tail height at its highest point.
The dorsum of the body is for the most part pigmented by punctate dermal melanophores, but some irregular veins and spots remain transparent without pigmentation. This produces the dark marbled, blackish-brown pattern on the body. There are dispersed basket-shaped melanophores on the ventrolateral part of the body. Fine silvery shiny iridophores cover most of the dorsum in loose groups, the eyeballs, and the ventrolateral parts of the body. The ventral thorax is strongly pigmented laterally, but the center contains only a few dispersed melanophores. The abdomen is nearly completely transparent (Fig. 2e and f). The gut spiral has mainly the color of the ingested material; the liver is orange-colored.
Acknowledgments
I thank my assistant Félix Domingo Bonilla Jiménez who helps me rear the tadpoles into froglets. The Ministerio del Ambiente y Energía (MINAE) supported my amphibian research program and I thank especially Javier Guevara for granting me collecting permits. I am grateful to Isabel Picado for her support through her revision of the English manuscript. Special thanks are due Richard Wassersug, who revised the final manuscript and gave valorous comments and corrections.
Resumen
La existencia de dudas sobre la validez de la descripción de las larvas de Hyla rufitela realizada por Duellman en 1970 motivaron una nueva descripción con base en renacuajos criados de dos masas de huevos de H. rufitela de la zona caribeña de Costa Rica. Algunos se criaron hasta la edad adulta para verificar la identidad de la especie. Los renacuajos difieren de la descripción e ilustración original por su color, la esbeltez (proporción entre altura y longitud del cuerpo 0.43 vs. 0.47), la cola más delgada (% longitud de la cola de la longitud total del renacuajo 69% vs. 63%), por la posición y forma conspicua del espiráculo, por el disco oral más ancho (ancho del disco: ancho del cuerpo 0.44 vs. ~ 0.33) y por algunos datos alométricos. Las larvas pequeñas permanecen en el fondo alimentándose con materia orgánica del lodo. Los renacuajos mayores se alimentan de material vegetal en descomposición. En cautiverio las renacuajos terminaron su desarrollo en 44-114 días (promedio 80.3 días).
Palabras claves: Hyla rufitela, renacuajos, larvas, descripción, morfología, alometría, ontogénesis, ecología.
References
Duellman, W.E. 1970. The Hylid Frogs of Middle America. Monogr. Mus. Nat. Hist. Univ. Kansas, no. 1, 427 p. [ Links ]
Duellman, W.E. 2001. The Hylid Frogs of Middle America. Vol.2, Soc. Study Amph. Rept., Univ.Kansas. No. 1, 694 p. [ Links ]
Gosner, K.L. 1960. A simplified table for staging anuran embryos and larvae with notes on idenfication. Herpetologica 16: 183-190. [ Links ]
Hoffmann, H. 2004. Description of the previously unknown tadpole of Hyalinobatrachium pulveratum (Anura: Centrolenidae). Rev. Biol. Trop. 52: 219-228. [ Links ]
Lips K.R. & J.M. Savage. 1996. Key to the Known Tadpoles (Amphibia: Anura) of Costa Rica. Stud. Neotrop. Fauna Environm. 31: 17-26. [ Links ]
McDiarmid R.W. & R. Altig. 1999. Tadpoles. The Biology of Anuran Larvae. University of Chicago, Chicago, Illinois, USA. [ Links ]
Savage, J.M. 2002. The Amphibians and Reptiles of Costa Rica. University of Chicago, Chicago, Illinois, USA. [ Links ]