Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.53 n.3-4 San José Sep. 2005
Effect of 2,4-D, hydric stress and light on indica rice (Oryza sativa) somatic embryogenesis
Allan Meneses1, Dora Flores2,3, Miguel Muñoz1, Griselda Arrieta1 & Ana M. Espinoza1,4
1 Centro de Investigación en Biología Celular y Molecular, Universidad de Costa Rica, San José, Costa Rica, menesesa@catie.ac.cr
2 Centro de Investigación en Biotecnología, Instituto Tecnológico de Costa Rica, Cartago; dflores@itcr.ac.cr
3 Escuela de Biología, Instituto Tecnológico de Costa Rica, Cartago.
4 Escuela de Agronomía, Facultad de Ciencias Agroalimentarias, Universidad de Costa Rica, San Pedro de Montes de Oca; amespino@racsa.co.cr
Received 04-II-2005. Corrected 03-VIII-2005. Accepted 26-VIII-2005.
Abstract: With the purpose of increasing the embryogenesis regeneration process in vitroplants obtained from somatic embryos of the indica rice variety CR-5272 (Oryza sativa L.), two independent experiments were performed. The first experiment consisted in the effect of combination of three concentrations of the gelling agent PhytagelTM (1.8, 2.4, and 3 gL-1) and four 2,4-D concentrations (2.26, 4.52, 6.78, and 9.05 m M) on the induction and subsequent regeneration of embryogenic calli. On the second experiment, the pre-regeneration phase was modified; calli were subjected to darkness or diffuse light conditions for one, two, and three weeks. In embryogenesis induction, 35% calligenesis was obtained using the MS culture medium supplemented with 6.78 m M of 2,4-D and 2.4 gL-1 PhytagelTM , whereas on the control treatment (MS medium supplemented with 9.05 m M of 2,4-D and 3 gL-1 PhytagelTM ) 24% calligenesis was obtained. In addition, regeneration percentages were improved (22% and 16% for calli induced with the above treatments, respectively). Furthermore, in light exposure experiments, the best result was obtained by exposing the embryogenic calli to darkness for one week in pre-regeneration, followed by direct light exposure during the regeneration phase. Rev. Biol. Trop. 53(3-4): 361-368. Epub 2005 Oct 3.
Key words: Rice, somatic embryogenesis, light, plant regeneration.
Somatic embryogenesis induction and embryo regeneration are determined by the interaction of growth regulators and the composition of the culture media along with the genotype and growth conditions (Figure 1).
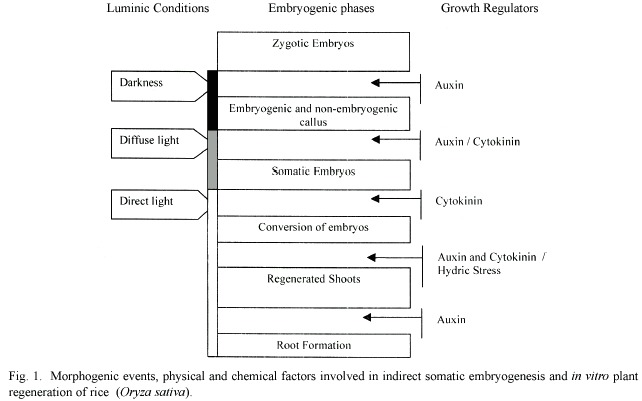
In monocotyledons, using auxins as promoters of cell indiferentiation it is possible to induce indirect somatic embryogenesis. Among these, 2,4-dichlorephenoxyacetic acid (2,4-D) is one of the growth regulators most commonly used to obtain embryogenic calli in cereals at concentrations between 10 -5 M and 10 -7 M (Salisbury and Ross 1992, Jimenez 2000). Endress (1994) suggests that 2,4-D generates DNA hypermethylation, which maintains the cells in a highly active mitotic stage and, therefore, in a pro-embryonic phase. In the specific case of rice, indirect somatic embryo-genesis from mature embryos is induced by the auxin 2,4-D at concentrations between 4.52 and 9.05 m M under darkness conditions. In contrast, plantlet regeneration from somatic embryos requires the use of cytokinins, either 6-bencyl-aminopurin or kinetin (Chen et al.1998, Valdez et al. 1996 ). However, a different response has been observed as a function of the genotype, since indica varieties are characterized by having low regeneration rates of in vitro somatic embryos, in contrast to the japonica varieties (Jain 1997, Kyungsoon et al. 2002, Valdez et al. 1996). In this sense, the concentration used and the subculture period control the embryogenic response and the reconversion of embryos to plants (Tsukuhara and Hirosawa 1992, Mitsuoka et al. 1994, Jimenez 2000). Vitroplant regeneration from somatic embryos of the variety CR-5272 is approximately two to three plantlets per callus, even though adequate cytokinins concentrations and sufficient light are administered to induce organogenesis. Rueb et al. (1994) inform that the use of high 2,4-D concentrations are necessary to induce somatic embryogenesis in rice, but at the same time they exert an inhibitory effect in vitroplant regeneration because regulator residues remain within the embryo cells.
In addition, it is known that light also plays an important role in embryogenesis induction and vitroplant regeneration. In Arabidopsis thaliana and Cydonia oblonga Mill (quince), it was determined that red light conditions (650/750 nm) promote a high conversion level of somatic pro-embryos to more advanced stages, whereas white light promotes vitroplants (Kaldenhoff et al. 1994, DOnofrio et al. 1998).
The purpose of this research was to determine the effect of different concentrations of 2,4-D and Phytagel as a gelling agent in the induction of embryogenic rice calli and their later regeneration. In addition, the evaluation of embryogenic calli regeneration was performed by applying three different light conditions for several periods along the pre-regeneration phase.
Materials and methods
Two independent experiments were performed with the purpose of modifying different stages of the somatic embryogenesis protocol of the rice variety CR-5272 and dilucidating the effect of these changes on plant-let regeneration. On the first experiment, the effect of the combination of different 2,4-D and Phytagel concentrations was evaluated on somatic embryogenesis and embryo regeneration. On the second experiment, the effect of light type and duration of the pre-regeneration phase was evaluated on the regeneration of somatic embryos.
Experiment 1: Mature rice embryos (Oryza satva L. cv CR-5272) were used for callus induction. For in vitro establishment, dehulled seeds were subjected to a superficial disinfection process using a 3.5% a.i. of sodium hypochlorite (NaOCl) with Tween 20 (one drop per 100 ml volume) for 20 minutes under constant stirring, followed by three washes with distilled sterile water in a sterile flow chamber.
This step was repeated twice, and seeds were finally washed with sterile distilled water and were innoculated on the culture media.
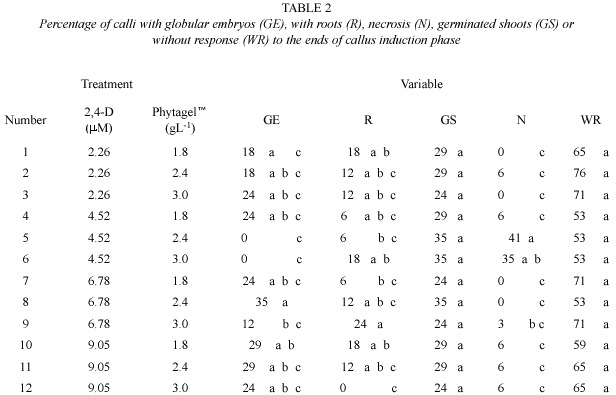
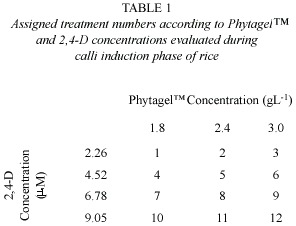
The MS medium was used (Murashige and Skoog 1962), supplemented with three concentrations of the gelling agent Phytagel (SIGMA) (1.8, 2.4, and 3.0 gL-1) and four concentrations of 2,4-D (2.26, 4.52, 6.78, and 9.05 m M) in the induction of embryogenic calli. For this, the 2,4-D concentration treatments were combined with each one of the Phytagel treatments, for a total of 12 treatments. Each consisted of five repetitions with 17 seeds (Table 1).
Four weeks later, the number of calli with globular-stage embryos, necrosed calli, calli with roots, germinated explants and not responding explants was evaluated. Furthermore, the number of vitroplants obtained from the calli of the three best treatments of the calligenesis phase was recorded. For pre-regeneration, the embryogenic calli were cultivated for a period of two weeks under diffuse light in a MSKA culture medium: MS (1962) supplemented with 9.3 µM kinetin (Kin), 2.69 µM Naphthalene acetic acid (NAA), 3% maltose, and 6gL-1 Phytagel. In the regeneration phase, a regeneration medium (MSR) was used: MS culture medium + 9.3 µM Kin + 2.69 µM NAA, as well as a control treatment with free-hormone MS medium. All culture media were supplemented with the MS vitamins, 3% maltose, 3 gL-1 Phytagel, and pH 5.8. Four repetitions per treatment were used, with 10 calli per repetition. Seven weeks later, the number of calli with plantlets, necrosed calli, and non-regenerating calli was evaluated.
Experiment 2: The calligenesis induction medium described by Valdez et al. (1996) was used: MS + 9.05 m M 2,4-D + 3% maltose + 3 gL-1 Phytagel, pH 5.8. Each treatment consisted of eight repetitions, with 15 disinfected seeds per repetition. The embryogenic calli kept for four weeks in this culture medium were subcultured to the pre-regeneration medium: MS + 9.3 µM Kin + 2.69 µM NAA + 3% maltose + 6 gL-1 Phytagel, pH 5.8 (MSKA). In this phase, the calli were exposed to three different light conditions: total darkness (0 lux), diffuse light (592 +/- 2.76 lux), and direct light (2067 +/- 17.49 lux), and were kept on these light conditions for one, two and three weeks. After each period, all the calli were subcultured to the regeneration medium: MS + 9.3 µM Kin + 2.69 µM NAA + 3% maltose + 3gL-1 Phytagel, pH 5.8 (MSR) under direct light. The variables evaluated were: number of calli with plantlets, number of green calli, number of calli with roots, and number of necrosed calli. The data were analyzed using the Statistix 4.0 statistical software.
Results
Experiment 1: The calli formed on the culture media solidified with 1.8 gL-1 Phytagel were characterized by having an intense yellow color, small size, and compact and globular form. In these treatments, the percentages obtained of globular callus (GE) formation were 18% (T1), 24% (T4 and T7) and 29% (T10). An increase in this percentage was observed in relation to the increase of 2,4-D concentration In these treatments, the highest percentage of globular calli resulted from using 9.05 µM of 2,4-D.
In the calli obtained with culture media gelled with 2.4 gL-1 Phytagel, both friable and globular forms were observed on a same callus. The GE percentage was variable among treatments. Thus, using low 2,4-D concentrations (2.26 and 4.52 µM), the embryogenic response was 18% (T2) and 0% (T5), whereas increasing the 2,4-D concentration to 6.78 and 9.05 µM, this percentage increased to 35% (T8) and 29% (T11), respectively. Concerning embryo formation, the treatment that used 6.78 µM of 2, 4-D and 2.4 gL-1 Phytagel generated the best results Significant differences according to comparison mean test by Tukey (
The calli induced in media with 3.0 gL-1 Phytagel (included in the control treatment) were light yellow in color, composed of larger globular areas than those obtained in the treatments in which a lower Phytagel concentration was used. No significant differences were observed regarding the calligenesis percentage in the treatments with the lowest (T3) and highest (T12) 2,4-D concentrations, since in both cases it was 24%. In a similar way, T5 and T6 (both with 4.52 µM of 2,4-D) did not present callus formation. Also, in both of these treatments, the highest percentage of necrosed calli was obtained, 41% and 35% respectively. In the remaining treatments, this percentage was low and oscillated between 0% and 6%.
The presence of roots in the calli was detected in all treatments, but was greater in treatments T1 and T10, in which 1,8 gL-1 Phytagel was used. The rhyzogenesis was reduced by increasing the Phytagel concentration. Thus, the control treatment T12 (9.05 µM 2,4-D / 3 gL-1 Phytagel) was the only one that did not exhibit root formation. In all the evaluated treatments, the percentage of explants that did not respond was between 53% and 76%, and was higher than the percentage of embryo formation (Figure 2).
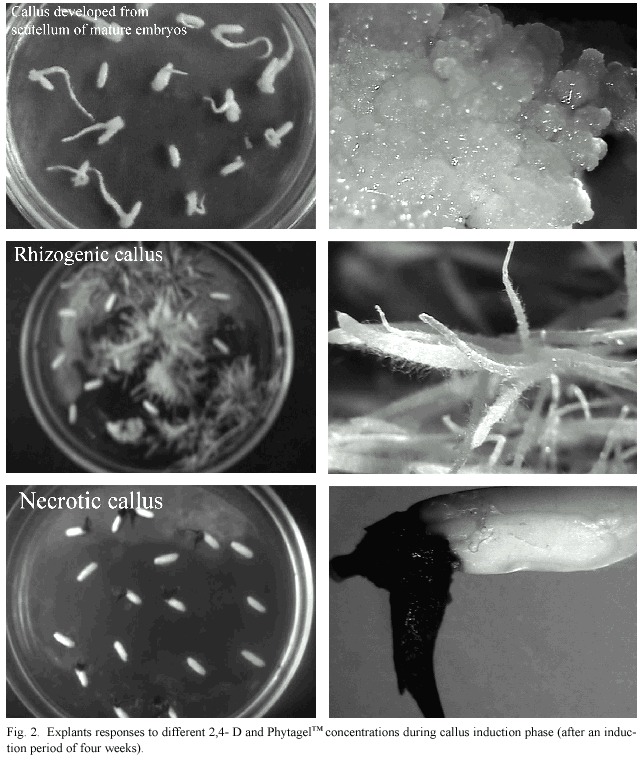
Considering all the variables evaluated, it was observed that the combination of 6.78 µM 2,4-D and 2.4 gL-1 Phytagel (T8) allowed to obtain the highest percentage of embryogenic calli with the least amount of necrosed material.
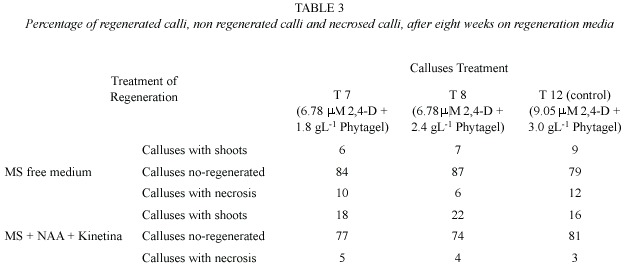
In the regeneration phase, the percentage of regenerated calli, non-regenerated calli, and necrosed calli induced in the calligenesis treatments T7, T8, and T12 evaluated in two regeneration media (with and without growth regulators) was recorded. In the calli from the above treatments, 18%, 22%, and 16%, regeneration was obtained, respectively, while in the media lacking cytokinins, plantlet regeneration was 6%, 7%, and 9% (Table 3).
The addition of growth regulators to the medium decreased the percentage of necrosed calli, but even with the growth regulators, the percentage of non-regenerating calli was high (between 74% and 81%) and similar to that of the treatments without growth regulators (between 79% and 84%), in which no regeneration would be expected. In these calli, green areas were observed; however, these did not develop into plantlets or just presented a morphogenic response (Figure 3).
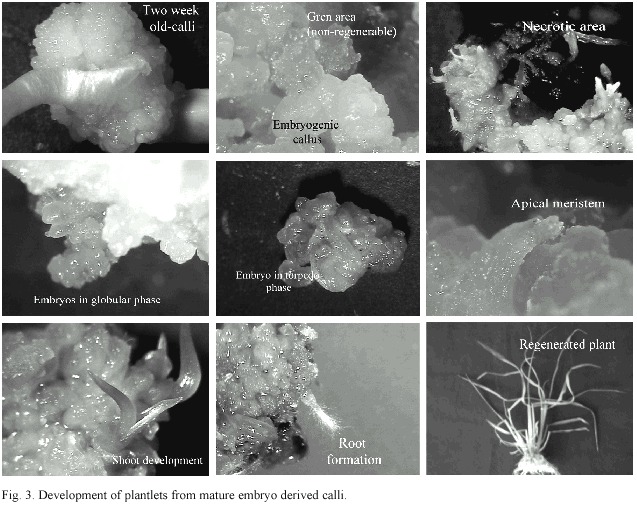
Experiment 2: On table 4, the average percentages of callus with plantlets, green areas, roots, and necrosis, obtained during the pre-regeneration study, are shown.
It was observed that the regeneration percentage decreased as the culture time in the pre-regeneration phase increased, for both light conditions evaluated. Thus, 13% and 11% regeneration was obtained when the material was kept for one week in pre-regeneration in darkness and diffuse light, respectively. Besides, no difference was observed between keeping the explants under diffuse light or in total darkness for one week, since for both treatments a high percentage of calli with green areas was observed (85% in darkness and 74% in diffuse light).Discussion
By increasing 2,4-D concentration and decreasing the concentration of Phytagel in the culture media, a better response of embryogenic calli was observed, since the mobility and absorption of the growth regulator and culture media nutrients was favored with this condition. With a high Phytagel concentration (3 gL-1), the embryogenic response decreases even though the auxin concentration is increased. The indirect somatic embryogenesis protocol in rice (Oryza sativa L) described by Valdez et al. (1996) uses a Phytagel concentration of 3.0 gL-1. However, concentrations similar to those recommended (1.8 and 2.4 gL-1) and lower than those suggested by Valdez et al. (1996) with 9.05 and 2.26 µM 2,4-D, root formation and seed germination were observed without obtaining embryogenic calli. Studies performed by Al-Khayri et al. (1996) determined that inoculated seeds in MS media (1962) with 4% sucrose, 10gL-1 agar-agar and 2.26 µM to 4.52 µM of 2,4-D produced roots and shoots due to low auxin concentration, whereas in those cultivated with 9.05 µM 2,4-D, the germination process was inhibited and calli were obtained from embryonic regions such as the scutellum. Under the conditions of this experiment, the best treatment for embryogenic callus induction was the combination of 6.78 µM of 2,4-D + 2.4 gL-1 Phytagel. Parallel to that, the regeneration rate of these calli was also the most successful. In addition, it was observed that the calli do not produce vitroplants in a synchronized way. In this sense, Mitsuoka et al. (1994) affirm that callus regeneration is produced slowly, since this process initiates as the amount of intercellular 2,4-D in the calli decreases. The auxin is necessary for the formation of rice embryogenic calli, but it negatively affects their redifferentiation process.
Concerning the effect of duration and type of light used in the pre-regeneration phase, no difference was observed by keeping the explants under diffuse light or in total darkness for one week, since for both treatments a similar percentage of regenerated plants was obtained (13% and 11%, respectively). In addition, a high percentage of calli with green areas was obtained for calli subjected to darkness (85%) and diffuse light (74%). The calli were cultured in media with a double concentration of PhytagelTM with the purpose of promoting hydrid stress, which at the same time promotes abscisic acid (ABA) biosynthesis and, therefore, the development of somatic embryos.
However, a detrimental effect was observed by prolonging the pre-regeneration phase for two or three weeks, independently of the type of light used. In addition, the high percentages of calli with green areas indicate that the photosynthetic processes begin when the calli are exposed to a light stimulus. However, more than this stimulus is required to activate the morphogenesis processes in the embryos.
Although the type of cells with embryogenic potential was good, two situations could have occurred during the regeneration process: an inhibitory factor may hinder bud production, or else a greater or different stimulus is required to continue the process. Therefore, although the number of regenerated plants was increased in comparison to the study performed by Vega (1996), it is necessary to search for other factors that may affect the in vitro culture of the variety CR-5272, such as the use of osmoregulators to dehydrate embryogenic calli through the application of high mannitol concentrations and gelled agents (Al-Khayri et al. 1996, Jain et al. 1996). One alternative would be to cultivate the calli in growth regulator-free media for one week before initiating the regeneration phase
to eliminate all possible "toxic residues" of the calligenesis promoting auxin (2,4-D) (since there is information about the inhibitory effect it causes on rice calli regeneration). Another one would be to use novel regeneration techniques as temporary immersion systems.
Acknowledgments
Authors thank Anabelle Muñoz and Erwin Gamboa for their comments during the development of this research. Tania Quesada and Marco Antonio Flores for their help in the English translation of the manuscript and finally to CIGRAS for supplying rice seed.
References
Al-Khayri, J., C. Shamblin, R. McNew & E. Anderson. 1996. Callus induction and plant regeneration of U.S. rice genotypes as affected by medium constituents. In vitro cell. Dev. Biol-Plant. 32: 227-232. [ Links ]
Chen, L., S. Zhang, R.N. Beachy & C.M. Fauquet. 1998. A protocol for consistent, large-sacale production of fertile transgenic rice plants. Plant Cell Rep. 18: 25-31. [ Links ]
D Onofrio, C., S. Morini & G. Bellocchi. 1998. Effect of light quality on somatic embriogenesis of quince leaves. Plant Cell Tiss. Org. Cult. 53: 91-98. [ Links ]
Endress, R. 1994. Plant Cell Biotechnology. Springer- Verlag. Berlin, Alemania. 353 p. [ Links ]
Jain, R.K., S. Jain & R. Wu. 1996. Stimulatory effect of water stress on plant regeneration in aromatic Indica rice varieties. Plant Cell Rep. 15 (6): 449-454. [ Links ]
Jain, R.K. 1997. Effect of some factors on plant regeneration from indica rice cells and protoplast - A Review. Indian J. Exp. Biol. 35: 323-331. [ Links ]
Jiménez, V. 2000. Endogenous Hormone Levels in Wheat, Maize, Barley, Carrot, Grapevine and Citrus Tissues and relationship to their in vitro somatic embryogenesis. Verlag Grauder, Stuttgart, Germany. 173 p. [ Links ]
Kaldenhoff, R., U. Henningsen & G. Ricther. 1994. Gene activation in suspension-cultured cells of Arabidopsis thaliana during blue light-depended plantlet regeneration. Planta 195: 182-187. [ Links ]
Kyungsoon, L., J. Hyesung & M. Kim. 2002. Optimization of a mature embryo-based in vitro culture system for high-frecuency somatic embryogenic callus induction and plant regeneration from japonica rice cultivars. Plant Cell Tiss. Org. Cult. 71: 237-244. [ Links ]
Mitsuoka, K., H. Honda, H. Xing & H. Unno. 1994. Effect of intracellular 2,4-D concentration on plantlet regeneration of rice (Oryza sativa L.) callus. Appl. Microbiol Biotechnol. 42: 364-366. [ Links ]
Murashige, T. & F. Skoog. 1962. A revised medium for rapid growth and bio-assays with tobacco tissue cultures. Physiol. Plant. 15: 473-497. [ Links ]
Rueb, S., M. Leneman, R.A. Schilperoot & L. Hensgens. 1994. Efficient plant regeneration through somatic embryogenesis from callus induced on mature rice embryos (Oryza sativa L.). Plant Cell Tiss. Org. Cult. 36: 259-264. [ Links ]
Salisbury, F. & C. Ross. 1992. Fisiología Vegetal. 4 ed. Traducido por Biol. Virgilio González Velásquez. Editorial Iberoamérica. p.135-136. [ Links ]
Tsukahara, M. & T. Hirosawa. 1992. Simple dehydration treatment promotes plantlet regeneration of rice (Oryza sativa L.) callus. Plant Cell Rep. 11: 550-553. [ Links ]
Valdez, M., M. Muñoz, J. Vega & A.M. Espinoza. 1996. Plant regeneration of indica rice (Oryza sativa L.) cultivars from mature embryo-derived calli. Rev. Biol. Trop. 44 (3) / 45 (1): 1996-1997. [ Links ]
Vega, J. 1996. Embriogénesis somática en arroz (Oryza sativa L. cultivar CR-5272): Anatomía e Histología de los procesos embriogénicos. Tesis de maestría, Escuela de Biología, Universidad de Costa Rica, Costa Rica. 98 p. [ Links ]