Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.53 n.1-2 San José Jun. 2005
Nesting biology of four Tetrapedia species in trap-nests (Hymenoptera:Apidae:Tetrapediini)
Evandro Camillo
Departamento de Biologia, Faculdade de Filosofia, Ciências e Letras de Ribeirão Preto-USP,14040-901, Ribeirão Preto, SP, Brasil; ecamillo@ffclrp.usp.br
Received 28-VIII-2001. Corrected 24-II-2003. Accepted 09-XI-2004.
Abstract
The nests used in this study were obtained from trap-nests (tubes of cardboard and cut bamboo stems) placed on Santa Carlota Farm (Itaoca Section-IS, Santana Section-SS and Cerrado-Ce), Cajuru, SP, Brazil. The number of nests and corresponding species obtained were as follows: 516 nests of T. curvitarsis, 104 of T. rugulosa, 399 of T. diversipes and 98 of T. garofaloi. The most abundant species from SS and Ce was T.curvitarsis, and from IS it was T. diversipes. In general, most nests were collected during the hot and wet season (September to April). The nests were constructed with sand and an oily substance, and a single female established them. The cells were constructed in a linear series, sometimes followed by a vestibular cell. The number of brood cells ranged from 1 to 10 in T. curvitarsis (n=200), and in T. garofaloi (n=51), from 1 to 8 (n=30) in T. rugulosa, and from 1 to 6 (n=37) in T. diversipes. The pollen mass (pollen + oily substance) contained a hollow, sometimes divided by a transverse ridge, on the exposed face of the pollen mass. The egg was vertically positioned in the lower part of the hollow. At times, the closing of a cell was initiated before provisioning was completed, with a construction of a collar at the cell limit. In some nests the final cellular partition also acted as a closure plug. Females began activities at 6:18 a.m.and ended between 3:31 and 6:26 p.m. Some females (T. curvitarsis , T. rugulosa and T. garofaloi ) did not spend the nights at their nests, returning to them only the following morning with additional material. In general, the development period (for males and females) was greater in nests collected near the end of the hot and wet season than it was for nests collected in other months. Sex ratios for each species were as follows: T. curvitarsis, 1:1; T. rugulosa , 1.6:1 female; T. diversipes, 1.9:1; T. garofaloi, 2.8:1. Males and females of T. diversipes exhibited statistically similar sizes and in the other three species the females were larger than the males. The mortality rates were statistically similar: 33.2% for T. curvitarsis, 25.8% for T. rugulosa, 26.8% for T. diversipes and 38.2% for T. garofaloi. The parasitoids were: Coelioxoides exulans, Leucospis cayenensis, Anthrax sp., Coelioxys sp., Coelioxoides sp. and individuals of the family Meloidae. Rev. Biol. Trop.53(1-2):175-186. Epub 2005 Jun 24.
Key words: Apidae, Tetrapedia, trap-nests, nesting biology, nest structure, natural enemies, life tables.
Michener and Moure (1957) divided Tetrapedia into subgenera Tetrapedia and Lagobata. The subgenus Tetrapedia occurs from the Neotropical region of Mexico to southern Brazil, and from northern Argentina to Ecuador. The subgenus Lagobata is restricted to Brazil. These authors separated this genus from the Exomalopsini (Anthophoridae) and placed it in a new tribe because of the differences in adults. This separation was reinforced when Michener and Lange (1958) characterized the tribe essentially through the biological data obtained in T. maura Cresson, 1878. Roig-Alsina and Michener (1993) placed Tetrapediini in the subfamily Apinae, family Apidae, when the authors extinguished the family Anthophoridae and created the tribe Anthophorini.
The subfamily Apinae has some genera that collect floral oil, including Centris, Ptilotopus and Epicharis (Centridini), Paratetrapedia ,Chalepogenus and Tapinotaspis (Tapinotaspidini), Ctenoplectra (Ctenoplectrini) and Tetrapedia (Tetrapediini) (Vogel 1974, 1976b,1981,Simpson et al. 1977, Roberts and Vallespir 1978, Michener and Greenberg 1980, Neff and Simpson 1981, Buchmann 1987). Other species are considered as probable collectors of oily substance, including Monoeca (Tapinotaspidini) (Vogel 1974, Roberts and Vallespir 1978) and Lanthanomelissa (Tapinotaspidini), Anthophora (Anthophorini) and Exomalopsis (Exomalopsini) (Neff and Simpson 1981). Only two genera of the family Melittidae were observed collecting floral oil: Macropis (Vogel 1976a, 1976b, Michener 1981, Cane et al.1983, Vogel and Michener 1985, Buchmann 1987) and Rediviva (Whitehead et al. 1984, Whitehead and Steiner 1985, 1992, Buchmann 1987, Steiner and Whitehead 1988, 1990,1991).
Among the studies carried out on Tetrapedia are the works of Michener and Lange (1958), on natural nests of T. maura, in Costa Rica, and Roubik (1987), on Tetrapedia sp., which nested in trap-nests in Panama.
Although T. curvitarsis Friese, 1899, T. rugulosa Friese, 1899, T. diversipes Friese, 1810 and T. garofaloi Moure, 1999 are widely distributed and are common in our region, they remain poorly understood. The purpose of this paper is to present biological data on these species obtained from trap-nests placed in three habitats of São Paulo State in south-eastern Brazil.
Material and methods
Study site: The study was carried out on the Santa Carlota Farm, SP (21° 18–27 S, 47° 12–17 W), Brazil. The farm, drained by several streams, involves an area of 6 300 ha, with altitudes ranging from 540 to 944 m. Approximately 2 200 ha of the total area have native vegetation preserved in isolated patches (semideciduous forests, riparian forests, cerrados (s.l.) and cerradões ), surrounded by abandoned orchards,pastures and cultivated land. There are also some areas in several stages of secondary growth.Rhodic Ferralsol and Orthic Ferralsol soils are predominant on the farm.
Trap-nests were placed in three areas of the farm: (1) Santana Section (=SS), characterized by abandoned orchards surrounded by sugar cane plantations, pastures and cerrado (s.l .) with abandoned houses, stables and sheds. Semideciduous forests and riparian forests were located about 300 m from the orchard; (2) Itaoca Section (=IS), characterized by abandoned houses surrounded by plots planted with sugar cane, corn, rice and sorghum. About 300 m away from the houses there are areas with semideciduous forests and riparian forests; (3) Cerrado (s.l .) (=Ce), structurally a semideciduous xeromorphic low-arboreal woodland or low forest, or a closed to somewhat open scrub.
Two distinct seasons are recognized in this region: (1) Cold and dry season (May to August) with monthly averages temperatures ranging from 18.2 to 22° C and precipitation from 0 to 116 mm; (2) Hot and wet season (September to April) with monthly average temperatures ranging from 23.2 to 27.9°C and precipitation from 8 to 368 mm.
Trap-nests: Three types of trap-nests (TN) were used. Two of them consisted of tubes made with black cardboard, with one end closed with the same material. The small nests (=SN) measured 5.8 cm in length and 0.6 cm in internal diameter, and the big nests (=BN) measured 8.5 cm and 0.8 cm, respectively. These tubes were inserted into horizontal holes drilled into wooden plates (30 x 11.5 x 5 cm). The other type of trap-nests consisted of bamboo canes (=B) with a nodal septum closing one end; these ranged from 7 to 25 cm in length and from 0.65 to 2.5 cm in internal diameter.
The wooden plates, with tubes and bamboo canes in blundies of 6-8 units were placed along the shelves of a shelter built at the study locations. The types of TN and their number were placed in study locations as follows: SS: 297 SN, 193 BN and 450 B; IS: 300, 120 and 200; Ce: 250, 150 and 200, respectively. The TN were inspected at least once a month during the period January/87 to December/88 in SS, from April/89 to March/91 in IS and from November/89 to October/91 in Ce. At each inspection those that contained completed nests were collected and transported to the laboratory. Each nest was replaced with a new TN. In the laboratory all nests were placed in a transparent glass tube and kept at room temperature until the adults emerged. After emergences the nests were opened and their contents analyzed.
Female nesting behavior was observed in six nests of T.curvitarsis, two of T. rugulosa and six of T. garofaloi , from 5th to 9th / December/88.These observations were made from 06:00 to 20:00 hours. The head width of the emerged individuals (25 females and 25 males) was taken as a measure of body size. Life tables were constructed showing the various causes of mortality at different stages in the life cycle and the relative number of immature individuals dying from each known cause. All specimens were deposited in the Entomology Collection of the Department of Biology, FFCLRP-USP.
Results
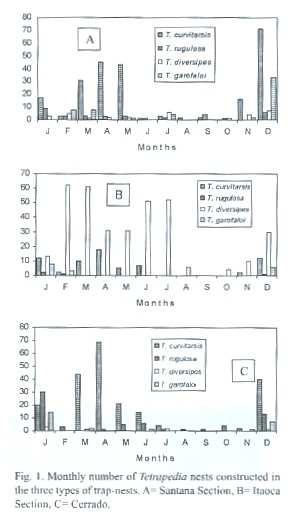
Nest numbers and seasonality: A total of 1117 Tetrapedia nests were obtained: 516 of T. curvitarsis, 104 of T. rugulosa, 399 of T. diversipes and 98 of T. garofaloi. Of these, 366 were from SS, 440 from IS and 311 from Ce. The most frequent species in SS and Ce was T. curvitarsis (63.9% and 68.8% of the collected nests, respectively) followed by T. garofaloi in SS (15.3%) and by T. rugulosa in Ce (20.9%). In IS the most frequent species was T. diversipes (79.8%) followed by T. curvitarsis (15.4%). The most nesting occurred in the hot and wet season (Fig.1). The number of nests of T. curvitarsis in SN, BN and B was 156, 340 and 20; of T. diversipes the numbers were 320, 78 and 1, and of T. garofaloi they were 17,72 and 9, respectively. T. rugulosa only nested in SN (n=71) and BN (n=33). In general, SN was the most utilized TN (50.5% of the collected nests), and the most abundant species was T. curvitarsis (46.2% of the collected nests).
Nest construction: After finding a suitable trap, the female leaves it returning with the oily substance to line the walls of the TN. During this time there appeared to be considerable confusion in recognition of tubes. After finishing the lining of the TN the female begins to collect sand or the oily substance (transported on her hind legs) for construction of the first cell. It was initiated with construction of the bottom wall of the cell. The thickness of these walls ranged from 0.04 to 0.7 cm (x=0.15 ± 0.1 cm,n=177) in T.curvitarsis, from 0.06 to 0.3 cm (x=0.12 ± 0.06 cm, n=26) in T. rugulosa, from 0.06 to 0.7 cm (x= 0.24 ± 0.17 cm, n=29) in T. diversipes and from 0.07 to 0.4 cm (x=0.17 ± 0.08 cm, n=43) in T. garofaloi. In 15 nests of T.curvitarsis and in 6 of T. diversipes, these walls were constructed recessed from the bottom.
For construction of one cell the number of flights to collect sand ranged from 12 to 17 (x=14.3 ±2.5,n=3) in T. curvitarsis and from 5 to 16 (x=13.3 ±4.2,n=7) in T.garofaloi; those to collect the oily substance ranged from 4 to 8 (x=6.0 ±2.0,n=3) and from 1 to 6 (x= 3.1 ±1.9,n=7), respectively. For provisioning, the number of flights to collect pollen ranged from 8 to 17 (x=12.5 ±4.6,n=4) in T. curvitarsis, from 13 to 19 (x=15.3 ±3.2, n=3) in T.rugulosa and from 5 to 9 (x=7.8 ± 1.8,n=5) in T. garofaloi; those flights to collect the oily substance ranged from 2 to 6 (x= 3.5 ±1.7,n=4) in T. curvitarsis and from 1 to 3 (x=1.6 ±1.1,n=3). In general,the duration of flights to collect sand ranged from 1.5 to 40 min (x=9.9 ±7.6 min,n=117) in T.curvitarsis, from 1.5 to 18 min (x=6.6 ±5.5 min,n=82) in T. rugulosa and from 2 to 62 min (x=13.2 ± 12.0 min, n=102) in T. garofaloi. The duration of flights to collect the oily substance ranged from 6 to 68 min (x=24.0 ±15.4 min, n=42) in T. curvitarsis, from 5 to 48 min (x=18.3 ± 9.9 min, n=20) in T. rugulosa and from 7 to 51 min (x=24.0 ±11.8 min, n=21) in T.garofaloi. The duration of flights to collect pollen ranged from 7 to 84 min (x=37.5 ±16.4 min,n=89) in T. curvitarsis, from 6 to 79 min (x=28.1 ±18.5 min, n=34) in T. rugulosa and from 13 to 67 min (x=37.6 ±11.2 min, n=54) in T. garofaloi. One case was observed in which the female lined the internal wall of the cell.
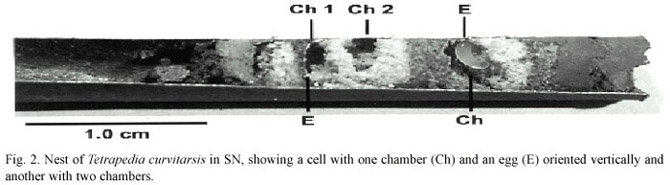
After finishing cell construction the female initiates cell provisioning. Before the female finishes these activities, she oviposits and begins the construction of cell partition, which had a form of a small ring that encircles the entire inner circumference of the nesting tube, thus constricting its diameter. Passing over the cell partition in construction, the female finished the provisioning. The pollen mass has a hollow area, sometimes divided by a transverse ridge, on the exposed face of the pollen mass. The egg was in a vertical position in the lower part of the hollow (Fig.2).
hen returning to her nest with material, the female enters head first but before depositing it, she turns inside the nest. The time spent by the female in the nest for depositing sand ranged from 1.5 to 10 min (x=3.0 ±1.8 min, n=51) in T. curvitarsis, from 2 to 12 min (x=5.6 ± 2.3 min, n=50) in T. rugulosa and from 1.5 to 6 min (x=2.7 ±1.1 min, n=55) in T. garofaloi. The time spent for manipulation of this material ranged from 9 to 24 min (x= 13.9 ±4.8 min, n=9), from 10 to 75 min (x= 29.9 ±19.2 min, n=10) and from 9 to 49 min (x=23.2 ±18.2 min,n=5), respectively. The time spent in the nest after returning with the oily substance ranged from 1 to 12 min (x=4.3 ±3.0 min,n=24) in T. curvitarsis, from 1 to 8 min (x=4.8 ±1.6 min,n=10) in T. rugulosa and from 1 to 9 min (x=4.3 ± 2.2 min, n=20) in T. garofaloi. The duration of time spent in the nest for manipulation of the oily substance (the female adds the oily substance to pollen or sand) ranged from 12 to 84 min (x=34.5 ±17.7 min, n=23), from 16 to 36 min (x=27.1 ±7.3 min, n=8) and from 9 to 22 min (x=14.3 ± 5.6 min, n=7), respectively.
The durations of time spent in the nest for pollen deposition ranged from 1.5 to 11 min (x=3.6 ±2.1 min,n=57) in T. curvitarsis, from 1.5 to 12 min (x=3.5 ± 2.3 min, n=53), in T. rugulosa and from 1 to 7 min (x=2.4 ± 1.3 min, n=39)in T. garofaloi. The time spent inside the nest for pollen manipulation ranged from 16 to 63 min (x=34.1 ± 18.9 min, n=7) in T. curvitarsis and from 18 to 112 min (x=47.1 ± 31.4 min, n=7) in T. rugulosa. Pollen manipulation was observed once in T. garofaloi, lasting 13 min.
After cell provisioning the female sometimes closed the small ring or completed all the closing of the cell. The thickness of cell partitions ranged from 0.08 to 0.45 cm (x=0.16 ± 0.07 cm, n=150) in T. curvitarsis, from 0.06 to 0.47 cm (x=0.13 ±0.05 cm, n=110) in T. rugulosa, from 0.04 to 0.24 cm (x=0.10 ±0.04 cm, n=100)in T.diversipes and from 0.06 to 0.34 cm (x=0.12 ± 0.03 cm, n=125) in T. garofaloi. In some nests the females were observed working at night when finishing the cell, partition or nest plug construction.
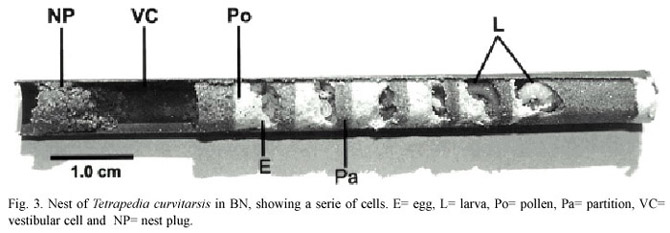
The cells were constructed in linear series, horizontally oriented. In some nests, between the last brood cell and the nest plug there was a space (vestibular cell), left empty or containing sand (Fig. 3). In 17 nests of T. curvitarsis, 3 of T. rugulosa and 9 of T. diversipes there was no vestibular cell. The lengths of these cells ranged from 0.4 to 9.0 cm (x=2.4 ±1.5 cm, n=115) in T. curvitarsis, from 0.2 to 4.0 cm (x= 1.9 ±1.2 cm,n=20) in T. rugulosa, from 0.5 to 1.2 cm (x=0.9 ±0.2 cm, n=7) in T. diversipes and from 0.2 to 5.3 cm (x=1.2 ±1.3 cm,n=20) in T. garofaloi .
In nests without a vestibular cell the last partition was also the nest plug. In these cases the thickness ranged from 0.14 to 0.6 cm (x= 0.3 ± 0.1 cm, n=17) in T. curvitarsis, from 0.4 to 0.6 cm (x=0.5 ± 0.1 cm,n=3) in T. rugulosa, from 0.2 to 0.4 cm (x=0.3 ±0.08 cm, n=7) in T. diversipes and from 0.12 to 0.9 cm (x=0.3 ± 0.2 cm,n=22) in T. garofaloi .
After closing the last brood cell, the female begins collecting material (the same as that for cell partitions) to construct a nest plug. After finishing, the females sometimes line it with an oily substance. To construct the nest plug the females of T. curvitarsis completed from 2 to 8 (x=5.6 ± 3.2, n=3) trips to collect sand and one trip to collect the oily substance. The time spent to construct the nest plug (observed once in each species) was 39 min in T. curvitarsis, 28 min in T. rugulosa and 19.5 min in T. garofaloi. The thickness of the nest plug ranged from 0.1 to 0.9 cm (x=0.35 ± 0.15 cm, n=115) in T. curvitarsis, from 0.15 to 0.5 cm (x=0.3 ± 0.1 cm, n=20) in T. rugulosa, from 0.14 to 0.3 (x=0.2 ±0.07 cm, n=7) in T. diversipes and from 0.1 to 0.7 cm (x=0.3 ± 0.2 cm, n=20) in T. garofaloi .
The number of brood cells constructed in each nest of T. curvitarsis ranged from 1 to 10 (n=200); in T. rugulosa from 1 to 8 (n=30); in T. diversipes (n=37) from 1 to 6 (n=37) and in T. garofaloi from 1 to 10 (n=51). The highest frequencies of cells were from 2 to 5, 3 and 4, 4 and 5 and 4 and 7, respectively.
The time spent for cell construction, provisioning, oviposition and construction of cell partition was 33:39 and 44:04 hs (n=2) in T. curvitarsis and from 29:05 to 49:48 hs (n=3) in T. garofaloi. In the only case observed in T. rugulosa the time spent was 66:06 hs.
The cell lengths of T. curvitarsis females ranged from 0.7 to 1.6 cm (x=1.1 ± 0.2 cm, n=119), of T. rugulosa from 0.7 to 1.0 cm (x= 0.9 ± 0.1 cm, n=33), of T. diversipes from 0.5 to 1.0 cm (x=0.7 ± 0.1 cm, n=28) and of T. garofaloi from 0.6 to 1.1 cm (x=0.8 ± 0.1 cm, n=35). Those of males ranged from 0.6 to 1.5 cm (x=1.1 ± 0.2 cm, n=119), from 0.5 to 1.1 cm (x=0.8 ± 0.2 cm, n=47), from 0.5 to 1.0 cm (x=0.8 ±0.1 cm, n=36) and from 0.6 to 1.0 cm (x=0.8 ± 0.1 cm, n=72), respectively. The cell diameters of T. curvitarsis females ranged from 0.5 to 0.9 cm (x=0.6 ± 0.07 cm, n=119), of T. rugulosa from 0.5 to 0.8 cm (x= 0.6 ± 0.05 cm, n=33) and of T. garofaloi from 0.5 to 0.7 cm (x=0.6 ± 0.04 cm, n=34). Those of males ranged from 0.5 to 0.8 cm (x=0.6 ± 0.06 cm, n=119), from 0.5 to 0.7 cm (x=0.6 ± 0.05 cm, n=42) and from 0.45 to 0.7 cm (x=0.6 ± 0.05 cm, n=72). The cells diameter of females (n=28) and males (n=36) of T. diversipes were the same as the TN utilized (0.6 cm).
Females activities: Female of T. curvitarsis began activities from 6:18 to 7:17 a.m. (n=16) and for T. garofaloi, activity occurred between 6:22 and 7:50 a.m.(n=14). Activity ended between 3:31 and 6:26 p.m.(n=17) and 3:50 and 6:18 p.m.(n=16), respectively. These observations were not made for T. rugulosa, because not all females spend the night at their nests. Species of Tetrapedia collected sand, the oily substance and pollen practically throughout the entire day. Obviously, this fact was related with stage of nest development. Some females did not spend nights at their nests, returning to them only the following morning with material for construction or provisioning according to the development of the nest. In these cases the last trip for T. curvitarsis occurred between 4:52 and 6:25 p.m.(n=4), for T. rugulosa between 5:42 and 6:25 p.m.(n=6) and for T. garofaloi between 6:10 and 6:19 p.m.(n=2). These females returned to the nests the next morning between 6:53 and 7:27 a.m. (n=6), 7:02 and 8:06 a.m.(n=7), and 7:50 and 8:23 a.m.(n=4), respectively.
In the 84.2% and 61.1% of the last flights, the females of T. curvitarsis and T. garofaloi , respectively, returned with the oily substance independently of the nest stage. In the other flights (15.8% and 38.9%) the female returned with sand that was utilized for construction of the cellular partition or nest plug. For T. rugulosa, in 50% of the flights (those before not spending the night at their nests) the females returned with the oily substance,37.5%with pollen and 12.5%with sand. Period of development, sex ratio and size of individuals: The developmental period was determined by the interval between the date of nest collection and the emergence of the adults. For T. curvitarsis, in nests collected near the end of the hot and wet season (March, April and part of May), the intervals for males (from 101 to 191 days) and females (from 103 to 211 days) were greater than those observed in nests collected during other months (from 33 to 55 and from 38 to 73 days, respectively). For T. rugulosa the same fact was observed for males (from 91 to 189 in hot and wet season and from 25 to 66 days in the cold and dry season) and females (from 95 to 99 and from 28 to 70 days, respectively). For T. diversipes this fact was evident only for males (from 92 to 170 and from 27 to 58 days); those for females ranged from 27 to 80 days. The intervals observed for males of T. garofaloi ranged from 102 to 290 days, except one case, which lasted 68 days; for females the intervals ranged from 104 to 290 days, independently of the season of the nest foundation.
The sex ratio observed in 894 cells of T. curvitarsis was 1.02 males to 1 female, which was significantly similar to a 1:1 (X2 = 0.13; p>0.05). In 116 cells of T. rugulosa it was 1.6 males to 1 female, in 108 cells of T.diversipes it was 1.9 males to 1 female and in 212 cells of T. garofaloi it was 2.8 males to 1 female. These sex ratios were significantly different from 1:1 (X2 =13.5; X2 = 92.9 and X2 = 50.1; respectively; p<0.05).
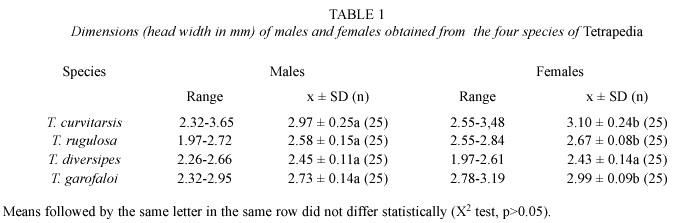
The size of males and females were significantly similar only in T. diversipes (Mann-Whitney, Z=0.1; p>0.05). In T. rugulosa, T. curvitarsis and T. garofaloi the females were significantly larger than the males (Mann-Whitney, Z=2.3; Z=2.2; Z=5.5; respectively; p<0.05)(Table 1). No significant differences existed with respect to the sizes of males and females of the four species (Kruskal-Wallis, H=59.2 and H=79.1, respectively; p<0.05).
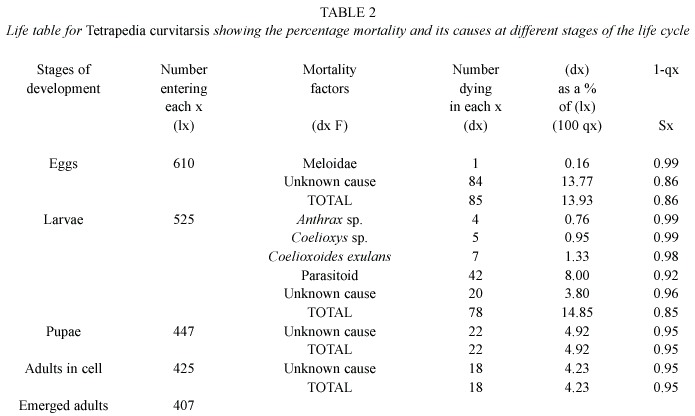
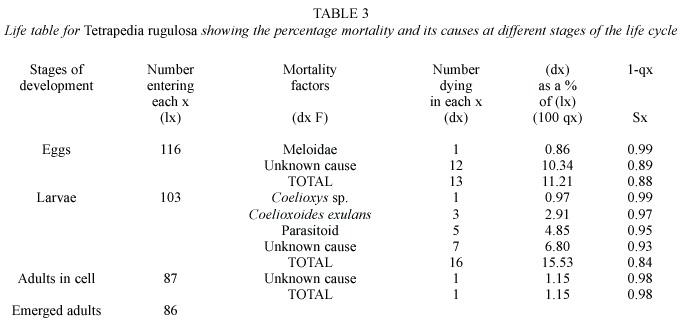
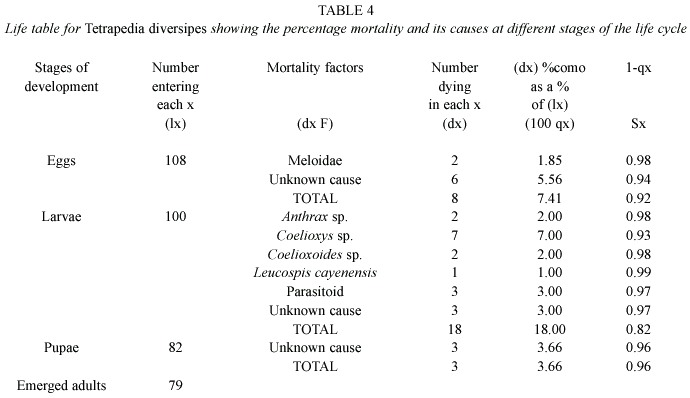
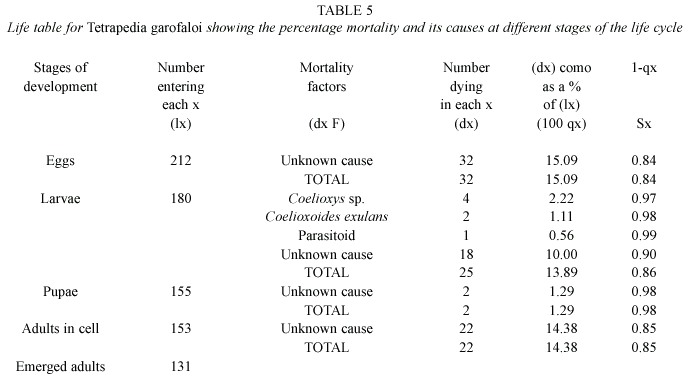
Life tables and mortality factors: Life tables (Tables 2, 3, 4 and 5) were constructed on the basis of 610 cells of T. curvitarsis, 116 of T. rugulosa, 108 of T. diversipes and 212 of T. garofaloi. In some cases, the parasitoids died during development, thus they were not identified. Unknown causes were responsible for 144 (70.9%) deaths of the 203 that occurred in T. curvitarsis, for 20 (66.6%) of the 30 in T. rugulosa, for 12 (41.4%) of the 29 in T. diversipes and for 74 (91.3%) of the 81 occurring in T. garofaloi. In the first and second species this factor was followed by unidentified parasitoids (20.7% and 16.6%, respectively), in the third by Coelioxys sp. (24.1%) and in the last by Coelioxoides exulans (Holmberg, 1887) (2.4%).
Analyzing each stage, it was observed that only in T. curvitarsis and T. diversipes, the greatest percentage of mortality in the larval stage was not due to unknown causes. In these cases the most common cause was unidentified parasitoid (42 or 53.8% in 78 deaths) and Coelioxys sp.(7 or 38.9% in 18 deaths), respectively. In general, the mortality rates occurring in T. curvitarsis (33.2%), T. rugulosa (25.8%), T. diversipes (26.8%) and T. garofaloi (38.2%) were significantly similar (X2 =3.24; p>0.05).
Discussion
The occurrence of the highest frequencies of nesting in the hot and wet season, seen in the four Tetrapedia species, was also observed in all species of solitary bees studied at same region in nesting in trap-nests (Camillo et al. 1994, 1995, Pereira 1997, Jesus and Garofalo 2000).
Lining the tube with the oily substance before beginning the nest construction was also observed by Jesus and Garofalo (2000) in Centris analis Fabricius, 1804. Pereira (1997) supposed that the substance utilized by Centris vittata Lepeletier, 1841 to line the trap-nests can be produced by the Dufours gland and this signal will be utilized for identification of the new nest and dissuade other females.
The studied species collected pollen and the oily substance in separate flights. The same fact was observed in C. analis in relation to pollen and nectar (Jesus and Garofalo 2000). According to Pereira (1997) females of C. vittata collected these two types of food in separate or in the same flight and floral oils were collected in distinct flights.
The beginning of the construction of cell partitions during the period of provisioning, observed in some nests of the studied species, is a behavior similar to that observed by Pereira (1997) and Jesus and Garofalo (2000) in C. vittata and C. analis, respectively. According to Pereira (1997) this behavior serves to protect cell contents from predators and parasitoids when the female is absent. The covering of the nest plug with oily material, observed in Centris species by Coville et al. (1983), Roubik (1989), Pereira et al .(1999), Jesus and Garofalo (2000), did not occur in nests of the studied species. The inner wall of the nest plug generally was not smooth and shining, but in some cases it was smoothed by the female.
The occurrence of prepupal diapause is a strategy used by some species of wasps and bees in response to adverse climatic conditions. Prepupal diapause observed in T. curvitarsis, T. rugulosa and T. diversipes occurred in nests collected in March, April and May (cold and dry pre-season).In T. garofaloi all immatures presented prepupal diapause.
The nest structure observed in studied Tetrapedia species was similar to that described for T.maura (Michener and Lange 1958). Contrary to our results, these authors observed that some wood particles occupied a small space at the bottom of the nest, and some had thin transverse layers of wood particles between cells.
Vogel (1974) suggested that Tetrapedia could be a floral oil collector because the females have a tarsal brush. According to Neff and Simpson (1981) these floral oil collecting structures in Tetrapedia are similar to those of other species such as Centris and Paratetrapedia. The supposition of Vogel (1974) was confirmed by Neff and Simpson (1981), Buchmann (1987) and Simpson et al. (1990).
In this study it was observed that T. curvitarsis, T. rugulosa and T. garofaloi collected the oily substance for provisioning and nest construction. Utilization of the oily substance in the larval food was also observed in other species of bees by Vogel (1974, 1981, 1984), Neff and Simpson (1981), Cane et al. (1983), Kevan and Baker (1983), Simpson and Neff (1983), Gottsberger (1985), Vinson et al. (1986), Buchmann (1987), Steiner and Whitehead (1988, 1991), Simpson et al. (1990, 1996, 1997), Whitehead and Steiner (1992). Collection of floral oil for nest construction was only observed by Simpson et al. (1990).
One of the more frequent parasitoids in nests of the studied species of Tetrapedia was Coelioxys sp., which was observed by Michener and Lange (1958) flying near nests of T. maura; Anthrax sp. was observed parasitizing nests of C. analis (Roubik 1989) and C. vittata (Pereira et al. 1999). Individuals of the family Meloidae were observed by Camillo et al. (1994) and Pereira et al. (1999) in C. vittata and Leucospis sp. by Roubik (1989) and Jesus and Garofalo (2000) in C. analis and Pereira et al. (1999) in C. vittata. The occurrence of Coelioxoides sp. in nests of the studied species was not observed in species of bees studied in the same region.
Some of the mortality rates of immatures in bees and wasps that constructed nests in trap- nests were near 50% (Krombein 1967, Danks 1971, Raw 1972, Jayasingh and Freeman 1980, Tepedino and Frohlich 1982, Tepedino and Parker 1983, Parker 1986). Some species presented mortality rates higher than 50% (Danks 1971, Jayasingh and Freeman 1980, Jesus and Garofalo 2000) and other species presented rates less than 50% (Camillo et al. 1993, 1994, Gathmann et al. 1994, Pereira et al. 1999) as observed in this study for all studied species.
Acknowledgments
To the owners of Santa Carlota Farm, Cajuru, SP, for allowing us free access to their land. Carlos Alberto Garofalo for his comments and suggestions and José Carlos Serrano for technical assistance.
Resumen
Los nidos usados en este estudio fueron obtenidos de nidos de trampa (tubos de cartón y tallos cortados de bambú) localizados en la finca Santa Carlota (Itaoca Sección-IS, Santana Sección-SS y Cerrado-Ce), Cajuru, SP, Brasil. El número de nidos y las especies correpondientes obtenidas fueron:516 nidos de T. curvitarsis, 104 de T. rugulosa, 399 de T. diversipes y 98 de T. garofaloi. La especie más abundante en SS y Ce fue T. curvitarsis, y la de IS fue T.diversipes. En general, la mayoría de los nidos fueron recolectados durante la estación caliente y húmeda (septiembre a abril). Los nidos fueron construidos con arena y una sustancia aceitosa, y una hembra solitaria se estableció en ellos. Las celdas fueron contruidas en una serie lineal, algunas veces contiguo a una celda vestibular. El número de celdas amplias se distribuyó desde 1 a 10 en T. curvitarsis (n=200), y en T. garofaloi (n=51), de 1 a 8 (n=30) en T. rugulosa, y de 1 a 6 (n=37) en T. diversipes. Las masas de polen (polen más sustancia aceitosa) contenían una depresión, algunas veces dividida por una carina transversal, sobre la cara expuesta de la masa de polen. Los huevos fueron puestos en la parte inferior de la depresión. A veces, el cierre de una celda fue iniciada antes de que el aprovicionamiento estuviera completo, con la construcción de un collar en el borde de la celda. En algunos nidos la partición final de las celdas también actuaba como un tapón. Las hembras empiezan sus actividades a las 6:18 a.m. y terminan entre 3:31 y 6:26 p.m. Algunas hembras (T. curvitarsis, T. rugulosa y T. garofaloi )no pasan las noches en sus nidos, regresando a ellos solamente la mañana siguiente con material adicional. En general, el periodo de desarrollo (para machos y hembras) fue mayor en nidos recolectados cerca del fin de la estación cálida y humeda que respecto a los nidos recolectados en otros meses. La proporción de sexos para cada especie fue: T. curvitarsis, 1:1; T. rugulosa, 1.6:1 hembras; T. diversipes, 1.9:1; T. garofaloi, 2.8:1. Machos y hembras de T. diversipes exhibieron tamaños estadísticamente similares y en las otras especies las hembras fueron más grandes que los machos. Las tasas de mortalidad fueron similares estadísticamente: 33.2% para T. curvitarsis, 25.8% para T. rugulosa, 26.8% para T. diversipes y 38.2% para T. garofaloi. Los parasitoides fueron Coelioxoides exulans, Leucospis cayenensis, Anthrax sp., Coelioxys sp., Coelioxoides sp.e individuos de la familia Meloidae.
Palabras clave: Apidae, Tetrapedia, nidos trampa, biología de la anidación, estructura de nido, enemigos naturales, tablas de vida.
References
Buchmann, S. L. 1987. The ecology of oil flowers and their bees. Ann. Rev. Ecol. Syst. 18:343-369. [ Links ]
Camillo, E., C. A. Garofalo & J. C. Serrano. 1993. Hábitos de nidificação de Melitoma segmentaria, Centris collaris, Centris fuscata e Paratetrapedia gigantea (Hymenoptera, Anthophoridae). Rev. Bras. Ent. 37: 145-156. [ Links ]
Camillo, E., C. A. Garofalo & J. C. Serrano. 1994. Aspectos bionômicos de Centris vittata (Hymenoptera, Anthophoridae) em ninhos armadilhas. Anais do I Encontro sobre abelhas, de Ribeirão Preto, SP. p. 298. [ Links ]
Camillo, E., C. A. Garofalo, J. C. Serrano & G. Muccillo. 1995. Diversidade e abundância sazonal de abelhas e vespas solitárias em ninhos armadilhas (Hymenoptera, Apocrita, Aculeata). Rev. Bras. Ent. 39:459-470. [ Links ]
Cane, J. H., G. C. Eickwort, F. E. Wesley & J. Spielholz. 1983. Foraging, grooming and mating behaviors of Macropis nuda (Melittidae) and use of Lysimachia ciliata oils (Primulaceae) in larval provisions and cell linings. Amer. Midl. Natur. 110:257-264. [ Links ]
Coville, R. E., G. W. Frankie & S. B. Vinson. 1983. Nests of Centris segregata (Hymenoptera, Anthophoridae) with a review of the nesting habits of the genus. J. Kansas Entomol. Soc. 56:109-122. [ Links ]
Danks, J. V. 1971. Biology of some stem-nesting aculeate Hymenoptera. Trans. Roy. Ent. Soc. London. 122: 323-399. [ Links ]
Gathmann, A., H. J. Greiler & T. Tscharnthe. 1994. Trap-nesting bees and wasps colonizing set-aside fields: succession and body size, management by cutting and sowing. Oecologia 98:8-14. [ Links ]
Gottsberger, G. 1985. Floral ecology reports on the years 1981 (79) to 1985. Prog. Bot. 47:384-417. [ Links ]
Jayasingh, D. B. & B. E. Freeman. 1980. The comparative populations dynamics of eight solitary bees and wasps (Aculeata: Apocrita: Hymenoptera) trap- nested in Jamaica. Biotropica 12:214-219. [ Links ]
Jesus, B. M. V. & C. A. Garofalo. 2000. Nesting behaviour of Centris (Heterocentris ) analis (Fabricius) in south-eastern Brazil (Hymenoptera, Apidae, Centridini). Apidologie 31:1-13. [ Links ]
Kevan, P. G. & H. G. Baker. 1983. Insects as flower visitors and pollinators. Ann. Rev. Entomol. 28:407-453. [ Links ]
Krombein, K. V. 1967. Trap-nesting wasps and bees. Life histories, nests and associates. Smithsonian. Washington, DC. 570 p. [ Links ]
Michener, C. D. 1981. Classification of the bee family Melittidae, with a review of species of Meganomiinae. Contr. Amer. Entomol. Inst. 18:1-135. [ Links ]
Michener, C. D. & J. S. Moure. 1957. A study of the classification of the more primitive nonparasitic anthophorine bees. Bull. Amer. Mus. Nat. Hist. 112:395-452. [ Links ]
Michener, C. D. &R. B. Lange. 1958. Observations on the ethology of neotropical Anthophorine bees (Hymenoptera: Apoidea). Univ. Kansas Sci. Bull. 39:69-96. [ Links ]
Michener, C. D. & L. Greenberg. 1980. Ctenoplectridae and the origin of long-tongued bees. Zool. J. Linn. Soc. 69:188-203. [ Links ]
Neff, J. L. & B. B. Simpson. 1981. Oil-collecting structures in the Anthophoridae (Hymenoptera): morphology, function and use in systematics. J. Kansas Entomol. Soc. 54:95-123. [ Links ]
Parker, F. D. 1986. Nesting, associates, and mortality of Osmia sanrafaelae Parker. J. Kansas Entomol. Soc. 59:367-377. [ Links ]
Pereira, M. 1997. Comportamento de nidificação e abundância sazonal de Centris (Hemisiella) vittata Lepeletier (Hymenoptera: Apidae: Apinae: Centridini). Dissertação de Mestrado. FFCLRP-USP. Ribeirão Preto, SP. 85 p. [ Links ]
Pereira, M., C. A. Garofalo, E. Camillo & J. C. Serrano. 1999. Nesting biology of Centris (Hemisiella ) vittata Lepeletier in sotheastern Brazil (Hymenoptera, Apidae, Centridini). Apidologie 30:1-12. [ Links ]
Raw, A. 1972. The biology of the solitary bee Osmia rufa (L.) (Megachilidae). Trans. Roy. Entomol. Soc. Lond. 124:213-229. [ Links ]
Robberts, R. B. & S. R. Vallespir. 1978. Specialization of hairs bearing pollen and oil on the legs of bees (Apoidea: Hymenoptera). Ent. Soc. Am. 71:619-627. [ Links ]
Roig-Alsina, A. & C. D. Michener. 1993. Studies of the phylogeny and classification of long-tongued bees (Hymenoptera: Apoidea). Univ. Kansas Sci. Bul. 55: 123-173. [ Links ]
Roubik, D. W. 1987. Notes on the biology of Anthophorid bee Tetrapedia and the mite Chaetodactylus panamensis Baker, Roubik and Delfinado-Baker (Acari: Chaetodactylidae). Int. J. Acarol. 13:75-76. [ Links ]
Roubik, D. W. 1989. Ecology and natural history of tropical bees. Cambridge University. 514 p. [ Links ]
Simpson, B. B. & J. L. Neff. 1983. Evolution and diversity of floral rewards, p. 360-372. In C. E. Jones & R. L. Little (eds.). Handbook of pollination biology. Van Nostrand-Rheinhold, New York. [ Links ]
Simpson, B. B.; J. L. Neff & D. Seigler. 1977. Krameria, free fatty acids and oil-collecting bees. Nature 267: 150-151. [ Links ]
Simpson, B. B.; J. L. Neff & G. Dieringer. 1990. The production of floral oils by Monttea (Scrophulariaceae) and the function of tarsal pads in Centris bees. Pl. Syst. Evol. 173:209-222. [ Links ]
Steiner, K. E. & V. B. Whitehead. 1988. The association between oil-producing flowers and oil-collecting bees in the Drakensberg on South Africa. Monogr. Syst. Bot. Missouri Bot. Gard. 25:259-277. [ Links ]
Steiner, K. E. & V. B. Whitehead. 1990. Pollinator adaptation to oil-secreting flowers -Rediviva and Diascia . Evolution 44:1701-1707. [ Links ]
Steiner, K. E. & V. B. Whitehead. 1991. Oil flowers and oil bees:further evidence for pollinator adaptation. Evolution 45:1493-1501. [ Links ]
Tepedino, V. J. & D. R. Frohlich. 1982. Mortality factors, pollen utilization, and sex ratio in Megachile pugnata Say (Hymenoptera: Megachilidae), a candidate for commercial sunflower pollination. J. New York Entomol.Soc.90:269-274. [ Links ]
Tepedino, V. J. & F. D. Parker. 1983. Nest size, mortality and Sex ratio in Osmia marginata Michener. Southwest. Entomol. 8:154-167. [ Links ]
Vinson, S. B., G. W. Frankie & R. E. Coville. 1986. Nesting habitats of Centris flavofasciata (Hymenoptera: Apoidea: Anthophoridae) in Costa Rica. J. Kansas Entomol. Soc.60:249-263. [ Links ]
Vogel, S. 1974. Ölblumen und ölsammelnde Bienen.Akad. Wiss. Abh. Math. Naturwiss. KI, 7:1-267. [ Links ]
Vogel, S. 1976a. Lysimachia: Ölblumen der Holarktis. Naturwiss. 63:44-45. [ Links ]
Vogel, S. 1976b. Oil collecting bees of the Old World and their flowers. Abstr. XV Int. Entomol. Congr. 19-27. [ Links ]
Vogel, S. 1981. Abdominal oil-mopping –a new type of foraging in bees. Naturwiss. 68:627-628. [ Links ]
Vogel, S. 1984. The Diascia flower and its bees –an oil- based symbiosis in Southern Africa. Acta Bot. Neerl. 33:509-518. [ Links ]
Vogel, S. & C. D. Michener. 1985. Long bee legs and oil-producing floral spurs and a new Rediviva (Hymenoptera, Melittidae, Scrophulariaceae). J. Kansas Entomol. Soc. 58:359-364. [ Links ]
Whitehead, V. B. & K. E. Steiner. 1985. Oil-collecting bees in South Africa. Afr. Wildlife 39:144-147. [ Links ]
Whitehead, V. B.& K. E. Steiner. 1992. Two new species of oil-collecting bees of the genus Rediviva from the summer rainfall region of South Africa (Hymenoptera, Apoidea, Melittidae). Ann. S. Afr. Mus. 102:143-164. [ Links ]
Whitehead, V. B., E. A. C. L. E. Schelpe & N. C. Anthony. 1984. The bee Rediviva longimanus Michener (Apoidea: Melittidae), collecting pollen and oil from Diascia longicornis (Thumb.) Druce (Scrophulariaceae). South Afr. J. Sci. 80:286. [ Links ]