Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.50 n.2 San José Jun. 2002
by GA3, light and salinity
K. Kathiresan and N. Rajendran
Centre of Advanced Study in Marine Biology, Annamalai University, Parangipettai 608 502, Tamil Nadu, India. Fax: +91- 4144-43555 (off.), +91- 4144-22265. e-mail: cdl_mangrove@sancharnet.in ; cdl_aucasmb@sancharnet.in; casenvis@yahoo.co.in
Received 02-III-2001. Corrected 16-IX-2001. Accepted 22-III-2002.
Abstract
The growth performance of Rhizophora apiculata Blume (mangrove) seedlings in the presence and absence of exogenous gibberellic acid (GA3 ) under different combinations of salinity and light was analyzed. Root and shoot growth responses of 75-day old seedlings in liquid-culture, were measured. It was concluded that light exhibited a significant inhibitory effect on all the growth parameters-number of primary roots, primary root length, shoot elongation, number of leaves, total leaf area; and, the GA3 treatment singly or in combinations with light, showed a significant influence on the total leaf area and primary root length.
Key words: Rhizophora, growth, GA3, light, salinity.
Rhizophora apiculata, a mangrove tree species of tropical coastal environments, propagates through viviparous propagules. Due to nutritional, hormonal and environmental factors, these propagules exhibit low sprouting, resulting in inefficient establishment of seedlings in their natural habitats (Kathiresan and Thangam 1989, 1990, Kathiresan et al. 1990, 1995, 1996, Smith et. al. 1995, e.g. Kathiresan 2000). There are only discrete studies available on the growth of mangrove seedlings as influenced by salinity, light and growth regulators. Of the growth regulators, the Gibberellic acid (GA3 ) is capable of reversing the salinity-induced stress on hypocotyl elongation (Banyal and Rai 1983, Smith et al. 1995). However, there is no clear study so far available for mangroves, on the combined effects of salinity and light in the presence and absence of exogenous growth regulators, although a similar aspect has already been studied, but with endogenous levels of GA3 (Smith et al. 1995). Hence, this study has been undertaken to analyze the influence of salinity, GA 3 and light on seedlings of a mangrove species.
Materials and methods
Propagules of R. apiculata Blume with a length of 28 ± 2 cm were collected during September 1999, from Pichavaram mangrove forest (Lat. 11° 27 N; Long. 79° 47 E), south-east coast of India. The propagules were placed separately in 500 ml beakers containing 300 ml of filtered seawater adjusted with tap water to three levels of salinity viz., 0 (tap water alone), 15 and 30 g 1-1, and with GA3 at three concentrations of 0,5 and 50 mg 1-1, at different combinations of GA3 and salinity. The seawater-cultured propagules were placed under two light conditions – one with a maximum of 1 700 µmol m-2 s-1 and another with a maximum of 25 µmol m-2 s-1, (the latter is mentioned as "shade" in the text). The former was kept in open sunlight (outside the room and with a maximum temperature of 30° C) and the latter in a room with diffused sunlight (with a maximum temperature of 26° C). The filtered seawater was replenished every 3 days. Five propagules and three replicates for each treatment were maintained for analysis. After 75 days, the data on number and length of primary roots, shoot elongation, number of leaves, and total leaf area, were collected.
Results
Effects on number of primary roots per seedling: This was significantly influenced only by light, and not by all other treatments singly or in combinations (Table 1). The light inhibited the average number of primary roots, ranging from 12.3 to 29.4% as compared to shade condition, in different saline conditions and GA3 treatments.
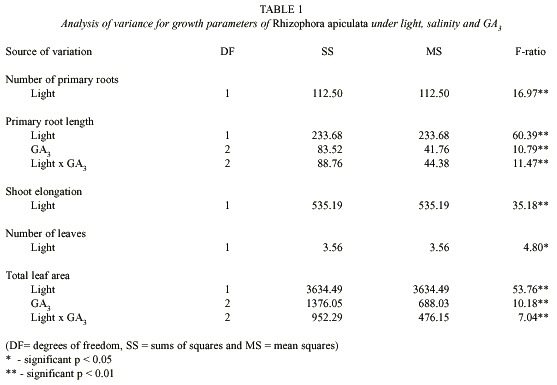
Effects on primary root length: Light and GA3 significantly influenced this, either singly or in combinations (Table 1), but not by salinity. The light inhibited the root elongation ranging from 6.3 to 61.8% in different saline conditions and GA3 treatments, under high light as compared to shade condition (Fig. 1). The GA3 had only a marginal effect on root length under light condition. But the growth regulator induced, in general, better root growth under shade.
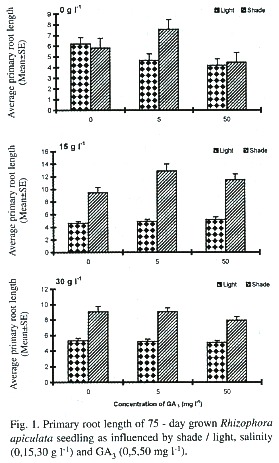
Effects on shoot elongation: This was significantly influenced only by light (Table 1), and not by all other treatments singly or in combinations. The light inhibited the shoot elongation, ranging from 29.7 to 91.05% as compared to shade condition, in different saline conditions and GA3 treatments (Fig. 2). The salinity at 15 g 1-1 enhanced the shoot elongation especially in the GA3 –treated seedlings grown under shade by 164.9 and 191.2% in 5 and 50 mg 1-1 growth regulator treatments, respectively.
Effects on number of leaves per seedling: This was significantly influenced only by light (Table 1) and not by all other treatments singly or in combinations. The light inhibited the number of leaves, ranging from 25 to 60% as compared to shade condition.
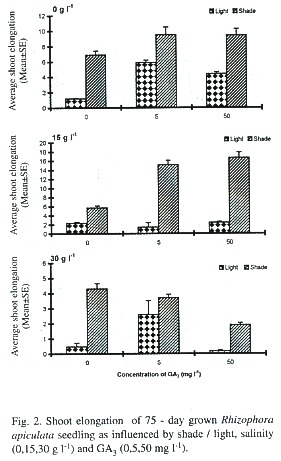
Effects on total leaf area per seedling: Light and GA3 significantly influenced this, either singly or in combinations (Table 1), but not by salinity. The light inhibited the leaf area expansion, ranging from 31.9 to 79.5%, as compared to shade condition (Fig. 3). The GA3 at 5 and 50 mg 1-1 increased the leaf area by 119.3% and 124.6% respectively, as compared to GA3 -untreated control, under non-saline, light conditions. The GA3 enhanced the leaf area by 57.05% and 15.33% at 5 and 50 mg 1-1 respectively, as compared to GA3 –untreated control, under non-saline, shade conditions (Fig. 3).
Discussion
Of the three factors studied, the light had striking inhibitory effects on all the growth parameters studied (Table 1; Figs. 1-3). The mangroves exposed to higher solar radiation were earlier reported to exhibit a low photosynthetic rate and growth (Bjorkman et al. 1988). Light was found to induce dormancy and non-rooting behaviour of floating propagules of Kandelia candel (Fan and Chen 1993). This was further observed by Kathiresan and Moorthy (1993) that in R. apiculata the shade-grown plants showed better growth performance than under full sunlight, a response commonly observed in many non-mangrove plants that is likely mediated by phytochrome system (Smith et al. 1995). This result is in accordance with our field observations that mangrove seedlings, which are existing in fully exposed areas, often fail to develop in spite of the fact that all other environmental factors appear favourable. The strategy adopted by the red mangrove R. mangle to light conditions was recently suggested Smith and Lee (1999). They observed growth especially in the internodal region of the seedlings when exposed to high photosynthetic photon flux density (PPFD) and to low ratio of Red and Far red spectral quality.
The exogenous application of GA3 had a significant effect on the primary root length and leaf area (Table 1; Figs. 1, 3) but, not on the number of roots and leaves. Thus this GA3 effect was more on the elongation of cells rather on the initiation of cells. Similar beneficial effects of GA3 were earlier recorded in mangroves - R. mangle (Smith et al. 1995), and in Avicennia marina (Kathiresan and Moorthy 1994).
The GA3 induced growth was found lower under light than in shade (Figs. 1, 3). This can be attributed to the high intensity of light which was already known to either destroy or to inhibit the biosynthesis of certain endogenous gibberellins that are required for growth of R. mangle seedlings (Smith et al. 1995).
The negative effects of sunlight may be related to the high doses of UV-B radiation that the mangrove receives. However, there have been few studies of UV-B and mangroves (Lovelock et al. 1992, Moorthy 1995, Smith and Snedakar 1995). Mangroves posses mechanisms to deal with the high sunlight. Hence, the mangroves occur in tropical habitats where they are exposed to high light intensities. The intense light can damage the mangroves and drop the photosynthesis rate (Cheeseman et al. 1991, Cheeseman 1994). This could explain why Rhizophora seedlings establish and sprout most readily under the shady canopy of larger trees (Kathiresan and Ramesh 1991). Also, Kathiresan and Moorthy (1993) demonstrated that the seedlings grow faster in the shade and show more efficient photosynthesis. Shading can also have nega-tive effects. For example, the mangrove seedlings under a closed canopy showed lower growth in south Florida (Koch 1997) and Belizean mangals (Ellison and Farnsworth 1993). Species like A. marina show good resistance to high sunlight, hot and dry conditions and are well adapted to arid zones (ElAmry 1998). Shade tolerance differs among mangrove species. Clarke and Allaway (1993) found that shading had no effect on growth or survival of A. marina. McKee (1995) noted that shading with brief periods of light exposure increased biomass and growth in A. germinans and Laguncularia racemosa and the same experiment had little effect on R. mangle. In dense mangrove forests, shaded saplings have lower shoot biomass than those exposed to the sun. The saplings may compensate for this by increasing development of the pneumatophores (Turner et al. 1995). Seedlings of R. mangle are apparently adapted for the shaded understory environment while mature trees do better in the sunlight canopy (Farnsworth and Ellison 1996).
In the present study, there was no significant combined effect of salinity and GA3, recorded. However, Banyal and Rai (1983) found that GA3 reversed the inhibition of hypocotyl elongation of Brassica campestris L., under osmotic stress which indicates that the decrease in endogenous concentrations may be a major consequence of salt stress. The exogenous application of gibberellins stimulated stem elongation and leaf area expansion in R. mangle propagules floating in 35 g 1-1 sea-water (Smith et al. 1995). But the present result does not support the salinity-induced effect of GA3 on the mangrove seedlings.
Acknowledgments
The authors are grateful to the Director and the authorities of Annamalai University for providing facilities.
Resumen
Se analizó el crecimiento de las semillas de mangle Rhizophora apiculata Blume en presencia y ausencia de ácido giberelico (GA3 ) exógeno bajo diferentes combinaciones de salinidad y luz. Se midió crecimiento de la raíz y el brote en plántulas de 75 días de germinación en medio de cultivo líquido. Se concluye que la luz presenta un efecto inhibitorio significativo respecto a todos los parámetros de crecimiento – número de raíces primarias, longitud de la raíz primaria, elongación de la plántula, número de hojas, área total de las hojas; y el tratamiento de GA3 solo o en combinaciones con luz, mostró una influencia significativa sobre el área total de las hojas y longitud de la raíz primaria.
References
Banyal, S. & V.K. Rai. 1983. Reversal of osmotic stress effects by gibberellic acid in Brassica campestris. Recovery of hypocotyl growth, protein and RNA levels in the presence of GA. Physiol. Plant. 59: 111-114. [ Links ]
Bjorkman, O., B. Demmig & J.T. Andrews. 1988. Mangrove photosynthesis: Response to high irradiance stress. Australian J. Plant Physiol. 15: 43-61. [ Links ]
Cheeseman, J.M. 1994. Depressions of photosynthesis in mangrove canopies. pp. 377-389. In N.R. Baker, (ed.), Photoinhibition of Photosynthesis: From Molecular Mechanisms to the Field. [ Links ]
Cheeseman, J.M., B.F. Clough, D.R. Carter, C.E. Lovelock, O.J. Eong, & R.G. Sim. 1991. The analysis of photosynthetic performance in leaves under field conditions: A case study using Bruguiera mangroves. Photosynthesis Res. 29(1): 11-22. [ Links ]
Clarke, P.J. & W.G. Allaway. 1993. The regeneration niche of the grey mangrove (Avicennia marina): effects of salinity, light and sediment factors on establishment, growth and survival in the field. Oecologia 93: 548- 556. [ Links ]
ElAmry, M. 1998. Population structure, demography and life tables of Avicennia marina (Forssk.) Vierh. at sites on the eastern and western coasts of the United Arab Emirates. Mar. Freshwat. Res. 49(4): 303-308. [ Links ]
Ellison, A.M. & E.J. Farnsworth. 1993. Seedling survivor-ship, growth and response to disturbance in Belizean mangal. Am. J. Bot. 80(10): 1137-1145. [ Links ] Fan, H. & J. Chen. 1993. Impacts of light on rooting of mangrove Kandelia candel propagules. J. Guang Acad. Sci. 9(2): 73-76. [ Links ]
Farnsworth, E.J. & A.M. Ellison. 1996. Sun-shade adaptability of the Red Mangrove, Rhizophora mangle (Rhizophoraceae): Changes through ontogeny at several levels of biological organization. Am. J. Bot. 83(9): 1131-1143. [ Links ]
Kathiresan, K. 2000. Areview of studies on Pichavaram mangrove, southeast India. Hydrobiologia 430: 185-205. [ Links ]
Kathiresan, K. & T.S. Thangam. 1989. Effect of leachates of mangrove leaf litter on rooting of Rhizophora seedlings. Geobios 16: 27-29. [ Links ]
Kathiresan, K. & T.S. Thangam. 1990. Effect of phenolics on growth of viviparous seedlings of Rhizophora apiculata Blume. Geobios 17: 142-143. [ Links ]
Kathiresan, K. & M.X. Ramesh. 1991. Establishment of seedlings of a mangrove. Indian For. 17(3): 93-95. [ Links ]
Kathiresan, K. & P. Moorthy. 1993. Influence of different irradiance on growth and photosynthetic characteristics in seedlings of Rhizophora species. Photosynthetica 29(1): 143-146. [ Links ]
Kathiresan, K. & P. Moorthy. 1994. Hormone- induced physiological responses of tropical mangrove species. Bot. Mar. 37: 139-141. [ Links ]
Kathiresan, K., G.A. Ravishankar & L.V. Venkataraman. 1990. Auxin-phenol induced rooting in a mangrove Rhizophora apiculata Blume. Curr. Sci. 59: 430-432. [ Links ]
Kathiresan, K., P. Moorthy & S. Ravikumar. 1995. Studies on root growth in seedlings of a tropical mangrove tree species. Int. Tree Crops J. 8(2/3): 183-187. [ Links ]
Kathiresan, K., N. Rajendran & G. Thangadurai. 1996. Growth of mangrove seedlings in intertidal area of Vellar estuary southeast coast of India. Indian J. Mar. Sci. 25: 240-243. [ Links ]
Koch, M.S. 1997. Rhizophora mangle L. seedling development into the sapling stage across resource and stress gradients in subtropical Florida. Biotropica 29(4): 427-439. [ Links ]
Lovelock, C.E., B.F. Clough, & I.E. Woodrow. 1992. Distribution and accumulation of ultraviolet-radiation- absorbing compounds in leaves of tropical mangroves. Planta 188(2): 143-154. [ Links ]
McKee, K.L. 1995. Interspecific variation in growth, biomass partitioning and defensive characteristics of neotropical mangrove seedlings: response to light and nutrient availability. Am. J. Bot. 82: 299-307. [ Links ]
Moorthy, P. 1995. Effects of UV-B radiation on mangrove environment: Physiological responses of Rhizophora apiculata Blume. Ph.D. Thesis, Annamalai University, India. 130 p. [ Links ]
Smith, S.M. & D.W. Lee. 1999. Effects of light quantity and quality on early seedling development in the red mangrove, Rhizophora mangle L. Bull. Mar. Sci. 65(3): 795-806. [ Links ]
Smith, S.M. & S.C. Snedaker. 1995. Developmental responses of established red mangrove, Rhizophora mangle L., seedlings to relative levels of photosynthetically active and ultraviolet radiation. Florida Sci. 58(1): 55-60. [ Links ]
Smith, M.S., Y. Yell Yang & Yuji Kamiya, 1995. Environmental controls on the early growth and development of the red mangrove, Rhizophora mangle L.: relationship to endogenous gibberellins. pp. 150-160. In M. Ajmal Khan & I.A. Ungar (eds.), Biology of Salt Tolerant Plants. University of Karachi, Karachi. [ Links ]
Turner, I.M., W.K. Gong, J.E. Ong, J.S. Bujang & T. Kohiyama. 1995. The architecture and allometry of mangrove saplings. Funct. Ecol. 9(2): 205-212. [ Links ]