Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.50 n.2 San José Jun. 2002
coercion vs. female cooperation in sepsid flies (Diptera: Sepsidae)
William G. Eberhard
Smithsonian Tropical Research Institute, and Escuela de Biología, Universidad de Costa Rica, Ciudad Universitaria, Costa Rica. Fax: (506) 228-0001; e-mail: archisepsis@biologia.ucr.ac.cr
Received 16-XII-2001. Corrected 17-V-2002. Accepted 20-V-2002.
Abstract
Female resistance behavior that occurs prior to intromission does not by itself imply forced copulation. Such behavior may function instead as a test of the male in order to favor some males over others, or to induce the male to desist. Thus, male persistence and forcefulness may sometimes be better described as persuasion rather than coercion. Under the persuasion hypothesis, the male only gains intromission due to an active response of the female. Under the coercion hypothesis, male and female are opposed in a physical battle which the female loses if copulation occurs. In species in which males are morphologically incapable of forcing intromission without active female cooperation (Iargue here that this is probably a very common situation), data on the behavioral and ecological context in which resistance occurs can distinguish between the two possibilities. Partially congruent functions of resistance, seen from the female point of view, are female resistance to screen (male persuasion), and female resistance to avoid males non-selectively (male coercion). Sepsid flies illustrate these ideas. Females often struggle energetically in apparent attempts to dislodge mounted males and to prevent intromission, and males grasp females with powerful species-specific structures on their front legs and genitalia. This suggests the possibility of coerced intromission. But behavioral and morphological evidence demonstrate that active female cooperation occurs at the moment of intromission, and that males are probably dependent on this cooperation because they are not morphologically equipped to force their genitalia into those of an uncooperative female. Despite the impression from previous publications, male insects in general may seldom be able to achieve intromission by genitalic force. The species-specific forms of the grasping genitalia of male sepsids are probably not the result of an evolutionary arms race between coercive males and unselectively resistant females.
Key words: Sexual selection, cryptic female choice, male-female conflict, Sepsidae.
A female flees from an approaching male, and the male pursues and overtakes her, then hangs on. She struggles in his grasp, but finally they copulate. Did the male forcefully compel the female to mate, and is this copulation appropriately analyzed in terms of the conflict between selection on males to inseminate as many females as possible, and selection on females to avoid the costs of sexual interactions such as lost time, increased exposure to predation and disease (Daly 1978)? Or was the females resistance actually selective cooperation, functioning not under natural selection to avoid costly interactions, but under sexual selection to bias paternity in favor of males with particular traits such as a better ability to chase, or a better ability to induce her to stop resisting? These questions are seldom asked. Female behavior such as that just described is usually termed "resistance" rather than "screening", and it is often thought to stem from male-female conflict (e.g. Alexander et al. 1997, Jormalainen et al. 2000). I will argue here that the difference between resistance as unselective avoidance and resistance as screening is important, and that knowledge of the morphology and behavior of male and female genitalia can help provide answers. The example discussed will be sepsid flies, whose dramatic pre-copulatory female resistance behavior has led previous authors to assume male-female conflicts (Parker 1972a, b; Ward et al. 1992, Allen and Simmons 1996, Blankenhorn et al. 1997, 2000). The ideas will then be extended to other insects.
The basic contrast is between male coercion and male persuasion or, from the female point of view, between unselective avoidance vs. selective cooperation. It can be divided into two different but related questions: Why did the female resist? and Why did the female copulate? Interpretation of both the act of intromission and of the female behavior preceding it are affected. With respect to intromission, one possibility suggested by the male coercion hypothesis is that the female copulated because the male was able to physically force his genitalia into her body by brute force, by wearing out her resistance and then forcing his way in, or by inserting them quickly, before she was able to begin to resist penetration (I will call all of these "direct coercion" or "intromission by genitalic force") (Table 1). A forced intromission is one in which the male uses his genitalia to forcefully gain access to and enter into the females genital tract. For instance, the male might use a rigid structure to pry open or thrust through a closed external (or internal) entrance to the females reproductive tract. In some animals such as most birds, forced intromissions of this sort are not feasible (Gowaty and Buschaus 1998), because males lack intromittent genitalic structures and thus cannot inseminate (introduce sperm into) the female unless she opens her cloaca.
Another possible type of more indirect coercion can occur even though the male is unable to force his genitalia into the female. He may nevertheless force the female to cooperate by threatening or actually inflicting physical damage, or by imposing fitness costs such as preventing her from performing other selectively important activities such as feeding, oviposition, or avoidance of predators. He may thus make it less costly for her to cooperate and allow intromission than to continue resisting (I will call this "indirect coercion" or "intromission by imposition") (see also convenience polyandry of Thornhill and Alcock 1983).
The persuasion hypothesis suggests a third possibility, that the males behavior and morphology induced the female to selectively cooperate and to allow or even facilitate intromission after her initial resistance, not in order to avoid costs, but to obtain the benefit of having particular genes from this male in her offspring rather than those of other males. This would be a case of persuasion, or what I will call "intromission by selective female cooperation."
Female resistance behavior also has different possible interpretations. Perhaps the female resisted in order to avoid copulation with the male irrespective of any of his behavioral or morphological traits; following Westneat et al. (1990), I will call this "indiscriminate female resistance as avoidance" or "resistance to avoid") (for a related, but more narrow concept, see "female reluctance" of Blankenhorn et al. 2000). Unselective female resistence as avoidance is expected when copulation per se is disadvantageous to the female. Or perhaps the female resisted to test the male ("resistance as screening") ("active female choice" of Blankenhorn et al. 2000). Female resistence as screening could result in several benefits. It could be designed to filter out those individuals that were unable to physically overcome such resistance (e.g. Berry and Shine 1980, Crean and Gilburn 1998), that were unable to induce the female to cooperate, that are not conspecifics (Connolly and Cook 1973), or that were otherwise inappropriate mates. The distinction between resistance as avoidance and resistance as screening can be difficult to make in practice.
These categories of behavior in the two sexes (male coercion vs. persuasion, female resistence to avoid vs. resistance to screen) are likely to coevolve. In a species in which indiscriminate female resistence as avoidance is prevalent, male coercion will be favored over male persuasion. In contrast, if female resistance as screening is common, male persuasion is more likely to be favored. Similar cause-effect associations can be made starting from male rather than female behavior. For instance, if males are frequently coercing copulations, indiscriminate female resistance to avoid is more likely to be favored. But other combinations are also possible. A male performing behavior that evolved to persuade selective females could be rejected by non-selective female resistance to avoid; or a female performing resistance behavior that evolved to screen males could be physically coerced to copulate. The focus of the data of this paper is on female behavior, so I will generally emphasize female rather than male roles, but obviously the roles of the two sexes are tightly entwined.
Seen from the females point of view, the problem is to distinguish between (a) copulations that occur despite uncompromising, generalized female resistance to avoid that is sometimes finally overwhelmed by superior male force, as contrasted with (b) selective female resistance to screen that functions to favor some males over others. This distinction is important, as it contrasts two different benefits to the female resulting from avoidance of male copulation attempts: benefits due to natural selection (inter-sexual conflict of interest that results in indiscriminate resistance) (Holland and Rice 1998) and benefits due to sexual selection (mate assessment) that results in selective resistance (Arnqvist 1992, Blankenhorn et al. 2000). The danger of confounding resistance as avoidance with resistance as screening has been recognized by other authors. McKinney and Evarts (1997; p. 165) noted that "...we need to be cautious in using the word forced [as in forced copulation] because it is possible that females resist forced copulation attempts as a tactic to test male quality . " Thornhill and Alcock (1983; p. 404) described this problem in terms of the male: "there is a problem ... of distinguishing between aggressive courtship and forced copulation" (for similar points, see also Parker 1974, Weigensberg and Fairbairn 1994, Rutowski 1997, Brown et al. 1997, Eberhard 1998a, Blankenhorn et al. 2000).
The distinction between male coercion and persuasion is also crucial to understanding genitalic evolution. The "conflict of interest" hypothesis (Lloyd 1979, Alexander et al. 1997) explains the rapid divergent evolution that is typical of male genitalia as the result of an arms race between males and females to control events associated with copulation and sperm transfer and use. "Coercive" male-female interactions are distinguished from "luring" interactions, and rapid divergent genitalic evolution is predicted to be associated with male coercion rather than male persuasion. The cryptic female choice hypothesis (Eberhard 1985), in contrast, proposes that female resistance as screening has been the cause of rapid elaboration and diversification of male genitalic structures.
I will argue here that some progress with these questions can be achieved by a process of elimination that combines morphological and behavioral data. If male and female genitalic morphology have features that preclude intromission by force (I will argue that this is very common), then male coercion using intromission by genitalic force (Table 1) can be eliminated. This elimination focuses attention on the behavioral and ecological context in which the interaction occurs as the crucial source of information to discriminate between the remaining indirect male coercion and male persuasion. In this paper I will present behavioral and morphological data that argue strongly against the coercion hypothesis in several species of sepsid flies.
Copulation in sepsid flies
A tentative overview of the reproductive behavior of sepsid flies can be pieced together from descriptions of several species, mostly in the genera Sepsis, Archisepsis, and Microsepsis. Female sepsids generally mate near oviposition substrates such as dung and carrion (Parker 1972a, b, Pont 1979, Eberhard and Pereira 1996, Schulz 1999). Mounted males tightly clamp the bases of the females wings with species-specific clasping structures on their modified front legs ( ulc 1928, Hennig 1949, Parker 1972a, Pont 1979, Blackenhorn et al. 1997, Schulz 1999, Eberhard 2001a). Most mounting attempts fail, as do most copulation attempts once the male has succeeded in mounting (Parker 1972a, Ward et al. 1992, Eberhard and Pereira 1996). Female resistance behavior commonly associated with failures includes kicking and pushing at the mounted male with her legs, shaking violently from side to side, running or flying and crashing into surrounding objects and knocking the mounted male off, and bending her abdomen ventrally and thus preventing genitalic contact (Parker 1972a, Ward et al. 1992, Allen and Simmons 1996, Eberhard and Pereira 1996, Schulz 1999, Blankenhorn et al. 2000, M.L. Baena in prep.). However, rejected males are seldom physically displaced by these female maneuvers; instead they usually dismount in pauses between bursts of female resistance (M.L. Baena in prep., W. Eberhard in press a).
The genitalia of male sepsids have well-developed clasping structures (the surstyli) which are often species-specific in form (Pont 1979, Ozerov 1992, 1993, Silva 1993). During copulation the surstyli forcefully pinch the external surface of the females abdomen near her genital opening (Eberhard and Pereira 1996, Figs. 1, 2). A study of functional morphology concluded that the most likely function of surstyli is a combination of physical restraint and female stimulation (Eberhard and Pereira 1996), but later observations showed that they give the female long stylized series of rhythmic squeezes that differ between species, supporting only the stimulation hypothesis (Eberhard 2001b, in prep.). Insertion of the males intromittent genitalia is relatively rapid, taking as little as 1 - 2 min for maximum penetration (Eberhard and Huber 1998). Copulation lasts 15 - 20 min, and ends when the male dismounts and pulls his intromittent genitalia from the females vagina, a process involving forceful tugging that requires up to 20 - 30 s in some pairs (Parker 1972a, Ward et al. 1992, Eberhard and Pereira 1996, Eberhard and Huber 1998, Schulz 1999). Sperm transfer occurs during a short period, and begins about 10 min or more after the start of copulation (Eberhard and Huber 1998).
The only published study directly discussing questions regarding female resistance to avoid and female resistance to screen in sepsids is that of Allen and Simmons (1996). They mention both resistance to forced copulation and female screening as possible functions for female resistance in S. cynipsea, but then ignore the possibility of screening in their discussion. They assumed that the association they found between the symmetry of the male clamping organs and male mating success was due to the more effective force generated by symmetrical structures in holding onto the female, without considering the alternative that females resisted some males more energetically than others on the basis of the morphology of these organs, or that stimulation of the female with these structures rather than their ability to "forcefully overcome female resistance" (p. 740) could be important in inducing female cooperation.
There are no descriptions of how male and female genitalia are brought into play in sepsids at the moment of intromission. Although the males surstyli are clearly designed to exert force on the female, it is not certain whether or not males are morphologically capable of forcing intromission. The observations reported below address the question of intromission by genitalic force in sepsids. Combined with behavioral observations related to male coercion, they help throw light on the probable function of resistance behavior of females.
Materials and methods
Some morphological data were obtained from virgin, two to three day old males and females of A. discolor (Bigot), A. pleuralis (Coquillett), and M. armillata (Melander and Spuler) that had been reared from eggs laid in cow dung by females that were collected near San Antonio de Escazú, San José Province, Costa Rica. Males were allowed to mount females in small petri dishes. As soon as a male was securely mounted, the petri dish was held over a Dewar flask containing liquid N2 , and jarred sharply so that the flies fell (or flew) into the N2 . A total of 12 A. discolor, 20 A.pleuralis, and 22 M. armillata pairs were frozen. Judging by the positions of frozen pairs, this treatment induced the male to hold on tight to the female. The flask was then placed in a freezer at –20º C, where the N2 was allowed to evaporate. While still in the freezer, the frozen flies were then fixed in absolute ethanol at –20º C, and were held there for a week before being brought to room temperature. The flies thus did not thaw out before being fixed, and had no chance to change positions. Pairs of S. neocynipsea and Sepsidimorpha sp. near Lakeside, AZ and of Themira minor near Ithaca NY were frozen in the field using ethyl chloride spray, and immediately immersed in 80% ethyl alcohol. This technique conserves surstylus positions in other sepsids (Eberhard and Pereira 1996). Specimens examined in the SEM (S-2360N) were dehydrated from glutaraldehyde and Karnovsky, dried by sublimation, and coated with 20 µm of gold. The morphological terms for intromittent male genitalia follow those of Eberhard and Huber (1998).
Behavioral observations in captivity of A. discolor, A. pleuralis, A. diversiformis Ozerov, Palaeosepsis pusio, M. eberhardi Ozerov, M. furcata, and M. mitis were made under a dissecting microscope, and involved 2 - 5 day old virgin flies (all raised from females captured in the Valle Central of Costa Rica, except those indicated with "Pan", which were raised from females on Barro Colorado Island, Panama). Field observations were made on A. diversiformis, A. pleuralis, A. ecalcarata, and M. armillata using 2X magnifying lenses mounted on a headband, and involved flies of unknown reproductive histories observed on and near fresh cow dung in pastures near San Antonio de Escazú, San José Province, Costa Rica or, in the case of A. ecalcarata, near Federal, Entre Ríos, Argentina. A mounting attempt occurred when a male climbed onto the dorsum of a female; successful mounting ended in copulation, while an unsuccessful mount ended with the male coming off the female. All field observations involved species that could be reliably distinguished (on the basis of body size, color, or wing spots) from other sympatric species without being collected, and males and females of unknown reproductive history. Vouchers have been deposited in the Colección de Insectos of the Universidad de Costa Rica, and the U. S. National Museum.
Results
Behavior preceding and during copulation
Male and female behavior in Archisepsis and Microsepsis gave clear indications of female resistance, and of male inability to force intromission. Female resistance behavior was common when males mounted females, both in the lab and in the field. In captivity, virgin females shook from side to side in at least 41% of 70 pairs of A. diversiformis during the initial stages of these mounts that eventually led to copulation (lack of female resistance in other pairs was confirmed by direct observation in 34%; the initial stages of the mount that resulted in copulation were not seen in the other 25%). Of these 70 copulations, 21.4% occurred after at least one previous mount that failed after the female had resisted by shaking. In many of these pairs I was able to see that the female also bent her abdomen ventrally while she shook so that her genitalia were out of reach of those of the mounted male. Similar shaking by the female when the male mounted also occurred in the other six species observed in captivity.
The most extensive field observations, of A. ecalcarata, also documented frequent female resistance. All of 136 observed mounting attempts failed; in 82% of these the female shook or otherwise resisted, while in the other 18% the male dismounted (usually immediately) without any sign of forceful resistance from the female. Of 34 cases in which a mounted male apparently attempted to intromit (the male pressed his genitalia against those of the female), 91% failed; the longest unsuccessful genital contact was 100 s. Lack of intromission was deduced by the subsequent immediate and smooth separation of the tips of the flies abdomens, in contrast with the prolonged tugging that occurs at the end of copulation (above). Corresponding failure rates of mounting attempts in A. diversiformis and A. pleuralis were 88% (N = 113) and 100% (N = 6), while failure rates for intromission attempts by mounted males of these two species were, respectively, 92% (N = 52) and 97% (N = 35). Failed mounting attempts in A. diversiformis occurred both with and without overt female resistance behavior.
Details of the movements of the males genitalia while they were pressed against those of the female during an intromission attempt were possible in both the field and captivity when pairs were oriented to afford a view of their genitalia. Males of A. pleuralis made two types of movement: the entire tip of the males abdomen vibrated rapidly from side to side, rubbing against the tip of the females abdomen; and then his surstyli repeatedly opened and closed rapidly, apparently rubbing against or pinching the tip of the females abdomen (possibly her proctiger). These behavior patterns were not seen in the other species. In one of four pairs of captive A. diversiformis (Pan) in which the flies were favorably oriented just as intromission occurred, the male surstyli did not begin to grasp the female sternite until the distal portion
of the males intromittent genitalia was inside the female. In the other three pairs the male briefly grabbed the female tergite VII just prior to intromission; one individual also briefly grasped her proctiger just prior to intromission. In several captive pairs of A. armata the inner and outer processes of the surstyli repeatedly gripped the female briefly near the base of her proctiger or, in some cases, the dorsal tip of her proctiger, rocking briefly from side to side about once/sec as if prying; but in no case did this produce any obvious result such as obtaining a stronger hold or displacing the proctiger, and none of these interactions resulted in intromission. In one A. armata (Pan) pair, the male did not grasp the female at all with his surstyli prior to intromission, while in another the male pinched her near sternite VI (not on the proctiger) several times just prior to intromission, but then spread his surstyli and kept them open during the first several seconds of intromission. No male of any species grasped and then pulled the tip of the females abdomen posteriorly or dorsally, as if to extend it or to lift her proctiger.
Females of A. ecalcarata in the field and A. diversiformis in captivity sometimes also shook periodically, though usually less energetically, during the first several minutes after copulation began. This behavior is paradoxical in terms of rejection of intromission, because the males intromittent genitalia quickly penetrate deep into the female, and are braced tightly inside her vagina by structures which securely pinch the vagina wall (Eberhard and Huber 1998).
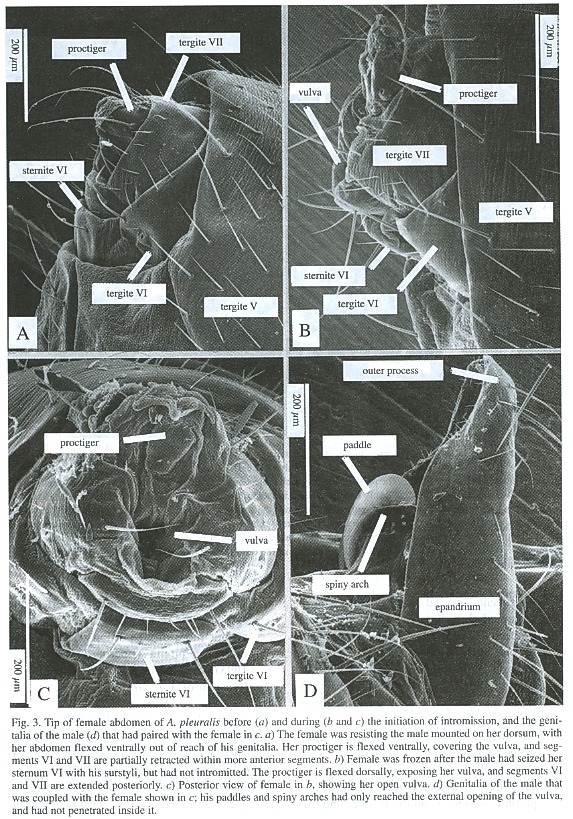
Morphological aspects of intromission and copulation
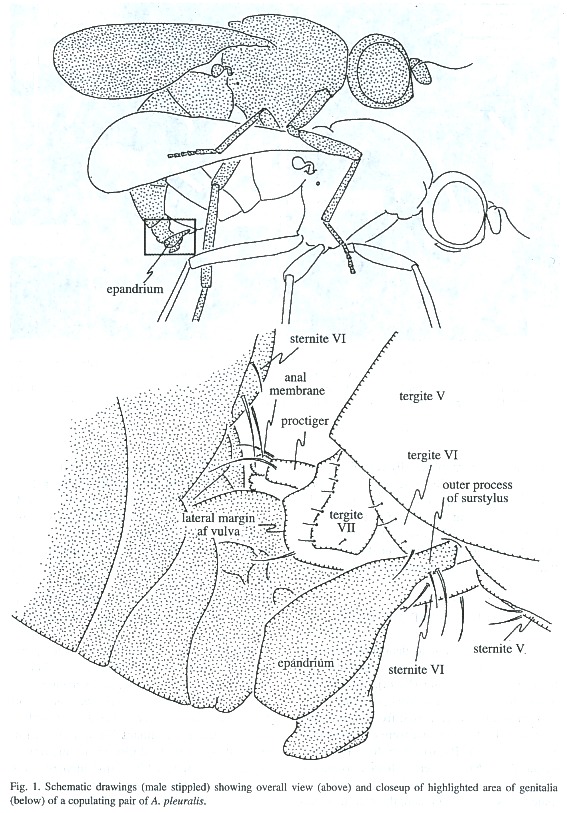
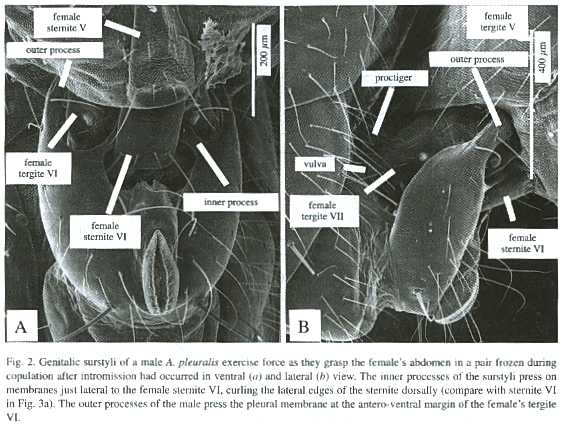
In all frozen pairs of A. discolor and A.pleuralis in which the males genitalia did not touch those of the female, the tip of the females abdomen was in the "resting position" that it consistently assumed in other contexts except oviposition: the proctiger was directed more or less ventro-posteriorly so that its base covered the females external genital opening, and segments VI - VIII were partially retracted and covered by the sclerites of segment V (Fig. 3a). In those pairs in which intromission had occurred, the females segments VI - VIII were more extended and the males surstyli grasped the exposed sternite VI of the female; the females protiger was deflected dorsally, and the males intromittent genitalia were inserted into her vulva (Figs. 1, 2; see also Fig. 1 of A. diversiformis in Eberhard and Pereira 1996). One pair each of S. neocynipsea, T. minor, and Sepsidimorpha sp. frozen while copulating in the field were in this same position.
In four pairs of A. pleuralis and one of A.discolor, the flies were at a preliminary stage of intromission when they were frozen. Three different positions of the females abdomen and the male genitalia were seen. Taking the resting position of the females abdomen (Fig. 3a) as a probable starting point, the probable order of these positions was as follows:
1. The tip of females abdomen was in the resting position, with segments VI and VII partially retracted. The tips of the outer processes of the males surstyli pinched the sides of her proctiger (seen in one pair of A. pleuralis).
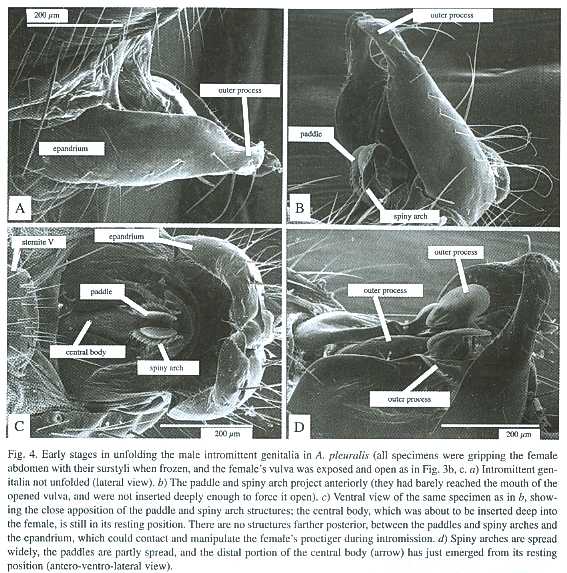
2. The female extended her sixth and seventh abdominal segments posteriorly, and deflected her proctiger dorsally. The vulva was thus exposed, and the outer walls of the females genital chamber were widely separated (Fig. 3c). The inner and outer processes of the males surstyli were nearby but had not grasped females sternite VI, and his intromittent genitalia had not been unfolded from their resting position (Fig. 4a) (two pairs of A. pleuralis, one pair of A. discolor).
3. The males surstyli grasped the females sternite VI, and her proctiger was deflected dorsally (but less so than in stage 2). The males intromittent genitalia were partially unfolded and the paddles and the spiny arches, which would be inserted and spread in the rear portion of her vagina to anchor more distal portions of his intromittent genitalia (Eberhard and Huber 1998), were partially extended (Fig. 4) (one pair of A. pleuralis).
Direct observation of the behavior of M.armillata just before and during intromission gave direct confirmation of female behavior similar to that described in step 2. During pre-intromission genitalic contacts the females proctiger was directed posteriorly, in the resting position. In one pair an especially favorable angle of view allowed me to observe movement of the females proctiger during a series of four genitalic contacts that culminated in intromission. Following the third from final contact before intromission, she had flexed her proctiger sharply dorsally (as in Fig. 3b), and it remained immobile in this "acceptance" posture during the subsequent genitalic contacts and the periods between them. The tips of the males genitalic surstyli never touched her proctiger, and probably did not touch her abdomen at all during this period. Thus, these direct observations confirmed that the dorsal flexion of the females proctiger, which exposed her vulva for intromission, occurred without any contact by the male that forced the proctiger to move dorsally.Examination of pairs of A. pleuralis, A.discolor, S. neocynipsea, Sepsidimorpha sp., and T. minor frozen later during copulation showed that the male surstyli grasp the female abdominal sternite VI and the surrounding membranes in very much the same way as documented in A. diversiformis and A. ecalcarata (Eberhard and Pereira 1996) and M. armillata and M. eberhardi (Eberhard 2001b) (Figs. 1, 2). Sharp indentations in the female membranes, and the bending of the lateral edges of her sternite VI where the surstyli contacted them (Fig. 2), showed that in Archisepsis, Microsepsis, and Themira both the inner and the outer processes of the males surstyli squeezed the female forcefully.
Discussion
Possible intromission by genitalic force in sepsids
Both morphological and behavioral evidence indicates that females of Archisepsis and Microsepsis can and do actively facilitate intromission, and suggest that male coercion by intromission using genitalic force is not feasible in these flies. It might be argued that such female "cooperation" is sometimes inadvertant, and not in fact designed to allow intromission. Perhaps, for instance, she tires of flexing her abdomen ventrally to prevent genitalic contact, or is sometimes surprised by the male before she can begin to defend against intromission. But the normal, "passive" resting positions of the structures at the tip of the females abdomen preclude intromission. That is, without active responses by the female, copulation is impossible. At least two and perhaps three active responses by the female precede intromission: eversion of segments VI -VIII; dorsal deflection of the proctiger to expose the vulva; and possibly opening of the external portion of the genital chamber. The male is not morphologically equipped to force the first two of these processes, and active cooperation by the female in both was confirmed by observations of both processes in living and frozen flies. The male did not bring any structure to bear on the female proctiger that could forcefully raise it while his genitalia were positioned to intromit (and in fact, he has no structure that is equipped to act in this waysee Fig. 4c). Nor did the male make any movements that would forcefully extend the females abdomen to expose her sternite VI so it could be grasped by his surstyli. Thus, neither male brute force, nor male surprise of the female before she could defend against intromission, nor male persistence that eventually tired out the females resistance and lowered her defenses are likely to result in intromission in these flies. In short, intromission by genitalic force seems impossible.
The male genitalia of many other sepsids resemble those of Archisepsis in their lack structures that could forcefully deflect the females proctiger, but the females proctiger is nevertheless deflected dorsally during copulation in A. diversiformis and A. ecalcarata (Eberhard and Pereira 1996), P. dentatiformis (Eberhard in press b), T. minor (Eberhard in prep.), and in P. pusio, A. armata, and M. furcata (Melander and Spuler) (W. Eberhard unpub.).
It might be argued that evidence from the behavior of virgin females of Archisepsis and Microsepsis should not be generalized to non-virgins, since virgins may be especially receptive or not yet mature enough to effectively resist, or that male and female behavior preceding and during intromission would be different and more conflictive if the female were in a less receptive stage. However, the male failed to copulate in many of the pairs observed, yet did not move his genitalia in ways appropriate to forcefully extend the distal portion of the females abdomen while he was being rejected; and males do not have intromittent genitalic structures appropriately designed to forcefully deflect the females proctiger dorsally and expose her vulva.
The behavioral observations reported here, as well as the temporary genitalic contact observed in S. cynipsea (Parker 1972a, Ward et al. 1992), also suggest that female sepsids can and do prevent intromission even after a male has succeeded in bringing his clasping genitalic surstyli into contact with her genitalia, again indicating that males are unable to use their genitalia to force intromission. In A. pleuralis, pre-intromission male contacts with the females proctiger suggested persuasion rather than coercion, as they involved very rapid vibration of his abdomen and his surstyli, possibly combined with brief nips. In A. diversiformis, A. armata, and M. armillata males consistently failed to contact the females proctiger. The prying movements of male A.armata, in contrast, appeared to be forceful; but they consistently failed to result in increased access to the females vulva, again suggesting female ability to prevent intromission. Parker (1972b) stated that male S.cynipsea grasps the female with his genitalia prior to copulation. But these observations were made in the field, so detailed observations of genitalic behavior were presumably not possible. Further observations are needed to clarify this point.
Possible intromission by indirect coercion in sepsids
Indirect male coercion could occur if the male imposes fitness costs on females that refuse to mate. Such indirect coercion seems unlikely, however, in Archisepsis, Microsepsis and Sepsis flies because the cost of copulation, in terms of time spent carrying the mounted male (which may entail increased danger of predation as well as energetic costssee Eberhard 2001a), is probably greater than the cost of rejection. In those species which have been studied, mounting will last longer if the female copulates than if she rejects the male. In S. cynipea, Parker (1972a) and Ward et al. (1992) found that unsuccessful mounts lasted on average 7 and 8 min after oviposition ended. In contrast, successful mounts lasted on average 7 and 19 min after oviposition ended and prior to copulation, and then an additional 20 - 22 min during copulation (Parker 1972a; see also Fig. 4 of Ward et al. 1992). The time needed by a female A. diversiformis or M. armillata to walk into the grass or leaf litter near a dung pat and thus induce a riding male to dismount is on the order of only 30 - 60 s (Eberhard 2001a), much less than the 15 – 20 min duration of copulation. These costs of copulation are conservative because they ignore the possibility that female shaking and other resistance behavior increases her susceptibility to predation, and also the possibly damaging effects of male seminal products on females that occur in other flies (Chapman et al. 1995, Rice 1996, Holland and Rice 1999).
These considerations cast new light on the resistance behavior of female sepsids such as the shaking that is performed when a male mounts. Shaking is not necessary to prevent intromission. Nor is it convincingly explained on the basis of possible costs of carrying a mounted male, especially the very energetic, sustained female resistance seen in some pairs (in a sample of 28 virgin female A. diversiformis, females shook up to more than 3 800 times during mounts that lasted up to 52 min of a 1 hr observation periodM. Baena in prep.). Thus, the resistance as screening hypothesis seems more likely to explain female shaking than resistance as avoidance. In fact, such screening of males does indeed occur in both S. cynipsea and A. diversiformis. Mounts by S. cynipsea males with more symmetrical legs are more likely to result in copulation (Allen and Simmons 1996). Mounts by larger A. diversiformis males and by males with normal as opposed to altered front leg morphology are more likely to result in copulation (M. Baena, in prep., W. Eberhard in press a).
Genitalic evolution in sepsids
The conclusion of Eberhard and Pereira (1996) that one function of the male surstyli of A. diversiformis is to physically restrain the female to allow intromission is not supported by this study. The species-specific morphology of the surstyli was not associated with intromission by genitalic force. Nor is there any sign of species-specific female "defensive" structures that would have selectively favored the species-specific aspects of male surstylus morphology (Eberhard and Pereira 1996), which would have supported the male-female conflict of interest hypothesis to explain rapid divergent genitalic evolution (Lloyd 1979, Alexander et al. 1997).
The pinches by the tips of the outer processes of the male surstyli on the females proctiger and the rapid vibrations of the tip of the males abdomen in A. pleuralis suggest, instead, a courtship function for the surstyli. Observations of the surstyli of two Microsepsis species during copulation showed that they squeeze the female rhythmically, and that the rhythm of squeezing varies between congeneric species (Eberhard 2001b), further favoring the alternative hypothesis of rapid divergence due to sexual selection by cryptic female choice (Eberhard 1985). There are no behavior patterns or structures on the proctiger of female A. pleuralis or on the ventral surface of the female abdomen in Microsepsis that show any hint of resisting the males pinches and rubs, as might be expected under the conflict of interests hypothesis. It could be that females use the force of the males genitalic grip as an indicator stimulus of male vigor (Andersson 1994), although the relatively small amplitude of many squeezing movements (Eberhard 2001b) and the strong negative allometry of surstylus size (Eberhard et al. 1998) argue against this. In sum, the conflict of interest hypothesis fails to explain the species-specific morphology of the male genitalic surstyli in Archisepsis and Microsepsis.
Intomission by genitalic force in other insects?
There is a substantial literature on apparently forced copulations in insects. However, careful examination of the original accounts and of genitalic morphology reveals few if any demonstrations of intromission by genitalic force. I will discuss the most commonly cited examples one by one.
The papers by Thornhill and collaborators on Panorpa scorpionflies probably constitute the most careful and widely cited studies of forced copulation in any insect. It appears, however, that the argument being made was for intromission by indirect coercion rather than intromission by genitalic force: "females... can probably refuse to permit the male ever to copulate with them by preventing entry of his aedeagus" (Thornhill and Alcock 1983, p. 469). Even intromission by indirect coercion is not certain in Panorpa. Disabling the male clasping organ in P. latipennis by covering it with beeswax resulted in males failing to achieve copulations, despite persistent attempts, and control males were often successful despite active female resistance (Thornhill 1980). These results are in accord with male coercion and female resistance to avoid, but they do not eliminate the screening hypothesis; females may screen males for instance, on the basis of these clasping structures (see Krieger and Krieger-Loibl 1958, Belk 1984, and Eberhard in press a for experimental demonstrations of such screening on the basis of the morphology of male clasping structures). This alternative is favored by the elaborate, species-specific clasper forms in males combined with the lack of obvious, species-specific anti-clasper structures of any sort in females (G. Byers pers. comm., Eberhard 1985). Thornhill and Alcock (1983) argued against resistance as screening, noting that the "appearance of forced copulation" (p. 272) by those male Panorpa scorpionflies that lack nuptial gifts represents true forced copulation because of "the clear disadvantage of not receiving a food present in return for mating" (p. 404). But the female is free to mate (and receive food gifts) as many more times as she wishes, and females have been seen to mate up to at least five times in a week (Thornhill 1980), so this argument is weak. Thornhill and Sauer (1991) argued that their observations of P. vulgaris eliminated the resistance as screening hypothesis, but they failed to consider many possible female mechanisms of cryptic choice, so again their argument is inconclusive (Eberhard 1996).
The case of the cricket C. strepitans (Sakaluk et al. 1995) resembles that of the scorpionflies in some respects. The male has a structure (a "gin trap") on the 8th and 10th tergites of his abdomen which clamps the ventral surface of the female abdomen. When the gin trap was disabled, the males mating success was reduced if he was unable to provide the female with a nuptial meal, but was unaffected if he could provide a meal (Sakaluk et al. 1995). The site where the female is clamped is not her genitalia, but is nearby; intromission per se, which involves attachment of a spermatophore, was not studied. In contrast to Panorpa, the simple, unadorned and clearly practical design of the gin trap suggests that it may not have evolved under sexual selection by female choice.
In water striders, the female fitness costs of male harassment and mounting are especially clear and well documented. Females often violently resist male mounting attempts (summary in Arnqvist 1997). In one species females even have an apparent anticlasping structure near the genital opening (Arnqvist and Rowe 1995) (the possibility that this is a screening device to assure copulation only with those males that are especially good at clasping has not been tested, however). However, the morphological details of the process of intromission, as opposed to preliminary grasping of the female, have apparently not been studied in these animals. Although in one species the males genitalia routinely entered the females
reproductive tract immediately after he mounted (Weigensberg and Fairbairn 1994), it is nevertheless possible that a female which has been successfully grasped by the males front legs and genitalia can nevertheless prevent intromission. In fact, G. Arnqvist (pers. comm.) suspects that the male is "most probably unable to morphologically penetrate the female genital tract without the female allowing him to do so." Morphologically forced intromission thus seems not to have been demonstrated in water striders. The lack of sperm transfer in copulations that lasted less than about 15 min (Rubenstein 1989), and the frequency of occurrence of such pseudocopulations (Weigensberg and Fairbairn 1994, 1996), suggest that some intromissions may involve only incomplete intromission, or perhaps female dumping of sperm (Daniellson and Askenmo 1999).
Several other insects have been mentioned to in general discussions of forced copulations. These include two tephritid flies (Prokopy and Hendrichs 1979, and Smith and Prokopy 1980, cited in Thornhill and Alcock 1983), the midge Culicoides melleus (Linley and Adams 1972 cited in Thornhill and Alcock 1983), Drosophila (Manning 1967 in Thornhill 1980), "all grasshoppers" (Alexander et al. 1997), and the planthopper Nilaparvata lugens (Oh 1979 cited in Thornhill and Alcock 1983). Intromission by genitalic force is also unlikely, however, in most if not all of these species.
Headrick and Goeden (1994) noted that intromission in tephritid flies can only occur after the female everts the tip of her ovipositor (the aculeus) from its usual hidden resting position, a process over which the male has no direct control. They argued that forceful intromission cannot occur in these flies, a conclusion supported by a detailed study of the process of intromission in the medfly, Ceratitis capitata (Eberhard and Pereira 1995). Smith and Prokopy (1980) suggested that forced copulations with female Rhagoletis which had been ovipositing were possible because the female still had her ovipositor extended to oviposit, and the male could grasp her aculeus before she was able to withdraw it. This interpretation is doubtful, however. Smith and Prokopy did not observe such quick seizures; male tephritids need several seconds to find and engage the female genitalia after they mount (Headrick and Goeden 1994, Eberhard and Pereira 1995); and direct observations show that female C. capitata can withdraw the aculeus relatively rapidly (W. Eberhard unpub.).
The forced nature of intromission in the midge C. melleus is also doubtful, since the male probably cannot force the female into a position in which he can bring his genitalia to bear on hers. In the first stage of male-female interactions leading to copulation, the female must stop (if she was moving), and raise her abdomen in a very distinctive "receptive posture" (Linley and Adams 1972, p. 89), which allows the male to grasp the ventral surface of her abdomen with his genitalic claspers. In some pairs the female resisted and did not raise her abdomen, and the male pushed dorsally at her abdomen with his hind legs. The much smaller size of the male (in Fig. 1b of Linley and Adams 1972, the females body length is about 140 % that of the male), and the females ability to easily avoid the males advances by simply walking away, suggest that a males ability to force intromission is at best very limited.
Similarly, a female Drosophila (both melanogaster and other species) can avoid being mounted by walking away, extruding her genitalia, or simply ignoring the male (Spieth 1947). In addition, female Drosophila are thought to have to open their vaginal plates for intromission to occur (Bubis et al. 1998 on melanogaster, Spieth 1947 on six species in the willistoni group). Male D. melanogaster usually make genitalic contact with the female two to three times before achieving intromission (Tompkins and Hall 1983). Alonso-Pimentel et al. (1995) reported occasional apparent forced copulations in D. mettleri, but gave no details. There are apparently no detailed descriptions of the early stages of intromission in Drosophila (such as those on sepsids presented above) that would allow evaluation of the possibility that males use their genitalia to force the females plates apart. Direct observations of living D.equinoxalis and D. willistoni showed that a receptive female spreads her genitalia before the male has even mounted (Spieth 1947).
Alexander et al. (1997) state that the males of all grasshoppers employ their genitalia to seize and hold the females genitalia in a "coercive act", but do not provide any supporting references. Two possible, widely cited sources of such information on grasshopper copulation (Gregory 1965 on Locusta migratoria, and Whitman and Loher 1984 on Taeniopoda eques) indeed mention grasping and holding by male genitalia preceding insertion of the aedeagus. There is reason to wonder, however, if these descriptions are complete. Few further details were provided in the Locusta study, and there was a nearly exclusive emphasis on successful intromissions in both studies (such "fertilization myopia" is unfortunately typical of many morphological studiesEberhard 1996). In both species the males abdomen must be closely aligned with that of the female for grasping to occur, and the female abdomen in grasshoppers in general is highly mobile and probably capable of moving so as to deny such positioning by the male. The female of another species of grasshopper, Melanoplus sanguinipes, can also "reach back with her hind legs and dislodge the mounted male with a series of vigorous kicks" (Pickford and Gillott 1971). More subtle, apparently internal rejections also may occur in grasshoppers. In Chorthippus curtipennis females mated on the first day of their receptivity did not always receive a spermatophore: "... the male mounted and copulated, but the aedeagus was withdrawn after a few seconds and the male left" (Hartman and Loher 1974, p. 1714- 1715). Some couplings in Schistocerca gregaria also failed to result in spermatophore transfer even after several hours of copulation (Pickford and Padgham 1973).
Finally, the study of Oh (1979) on the delphacid N. lugens was mainly concerned with ecological consequences of multiple copulations; forced copulation was mentioned briefly as being associated with female resistance behavior but no morphological details were given.
In sum, the evidence from insects that has been cited in previous publications as illustrating forced copulations does not in general document intromission by genitalic force. Morphologically forced intromissions are poorly documented in insects, and may be uncommon. For instance, in all of the beetle species whose mating I have observed (species in the families Bruchidae, Buprestidae, Cantharidae, Carabidae, Cerambycidae, Chrysomelidae, Cicindellidae, Coccinellidae, Curculionidae, Dynastidae, Erotylidae, Languriidae, Melolonthidae, Scaphidiidae, Staphylinidae, and Tenebrionidae), the male is clearly unable to intromit until the female opens the tip of her abdomen. This, of course, does not mean that morphologically forced intromission never occurs in insects (this paper is not a review of all published studies). But it does mean that intromission by genitalic force is probably much less common in insects than might be suggested by a superficial glance at the standard set of citations. Further studies of
grasshoppers or water striders, for example, may reveal genitalic structures capable of forceful intromissions. Species with traumatic insemination (see review in Eberhard 1985), and those in which males copulate with soft, apparently defenseless females soon after their final moult (e.g. some Heliconius and papilionid butterfliesBrown 1981, Suzuki and Matsumoto 1990; some fungus gnats Eberhard 1970; some DrosophilaMarkow 2000) are other likely possibilities (G. Arnqvist pers. comm.). Male persistence plus a sharp, needle-like tip of the aedeagus may sometimes allow some chrysomelid beetle males to pass the females internal sphincter at the opening of the bursa when it becomes tired, while others pass the sphincter so rapidly that it is possible that they bypassed female resistence by surprise (D.W. Tallamy pers. comm.). Even here, however, the female must first open the tip of her abdomen to allow the male access to her vulva.
The distinction between intromission by genitalic force and active female cooperation may sometimes be subtle. Males of some species probably achieve intromission by a mixture that combines behavioral and morphological coercion as well as more classical courtship behavior. The male of beetles in the genus Macrodactylus, for instance, combines several strategies: he rubs and strokes the female with the his middle legs and ventral spines, grasps her prothorax forcefully with his front legs, strokes her gently with his genitalia, forcefully inserts a genitalic "foot-in-the-door" device into her external genital opening, and uses a spiny vulva-spreading device to attempt open an internal entrance to her bursa (Eberhard 1993). In addition, the behavioral interactions that precede morphologically forced intromission (i.e. the threat of genitalic force), and a females ultimate inability to resist forceful genitalic penetration in some species, may sometimes be enough to induce a female to give in and allow intromission even when the male does not exercise this force. There is undoubtedly a spectrum of combinations of male behavioral and morphological duress and of female cooperation in different species, in different pairs of a single species, and even in particular females, whose resistance to intromission probably often varies according to her age, previous history, and current ecological context. Despite these complications, however, it is useful to attempt to check male and female genitalic morphology for the ability to preclude forced intromission, because of the logical consequences of the extreme case, in which a males genitalia are morphologically incapable of forced intromission.
The inability of male genitalia to force intromission is not limited to insects. For instance, female fiddler crabs must lower the abdominal flap to expose their gonopores, and males are not morphologically equipped to force this lowering (J. Christy pers. comm.); many male spiders are unable to force the female into positions that permit intromission (Huber 1998); sperm transfer in many other arachnids depends on insertion of a spermatophore by the female rather than by the male (Thomas and Zeh 1984); in many reptiles the males hemipenis is only inflated after it is inside the female; and most male birds lack intromittent organs.
The finding that direct male coercion using intromission by genitalic force is probably unusual in insects has an important consequence for resolving questions that contrast male coercion with male persuasion. It means that only indirect rather than direct male coercion is feasible in many groups. It thus focuses attention on the ecological context of male-female interactions in any attempt to test male coercion vs. male persuasion hypotheses. If the female clearly stands to lose more from being mounted or from copulating than from continued resistance (e.g. when predation rates increase sharply as in gerrids), then it is reasonable to propose that her resistance behavior represents indiscriminate attempts to escape from male attentions, even though the male cannot force intromission (Arnqvist 1992, 1997, Weigensberg and Fairbairn 1994, 1996). If, on the other hand, no clear ecological cost for the female can be found for being mounted, and the mounted male is incapable of forcing intromission (e.g., the sepsids of this study, coelopid flies in the genus CoelopaCrean and Gilburn 1998, Weall and Gilburn 2000), then it seems prudent to carefully consider the possibility that female resistance functions to screen rather than to avoid potential mates.
It should be noted that the focus on intromission in this paper probably results in a serious underestimate of the likely importance of female resistance as screening. Several additional female processes, such as ejection of the males sperm, failure to transport them, or flooding of the reproductive tract with phagocytes immediately after copulation (Eberhard 1996) could have the same final effects on paternity as prevention of intromission (Gowaty 1997, Gowaty and Buschhaus 1998), but they are less likely to have the kind of naturally selected payoffs expected from resistance as avoidance.
Finally, it is interesting to note the somewhat surprising trend for male insects to seldom intimidate or damage females, even when they possess strong mandibles, poisonous bites, or powerful armed legs that serve in predation or as weapons in battles with other males (e.g. Rodríguez 1998, Eberhard and Marín 1996, Eberhard 1998b). Among 131 species of insects and spiders in eight different orders whose copulatory behavior I studied (Eberhard 1994) and a number of additional species in which males attempted to mate but were consistently rejected by females, there was not a single species in which male behavior suggested that potential damage to the female was used to induce cooperation in copulation attempts. Male assassin bugs in the genus Zelus may be an exception to this pattern; the male jumps on the female without preliminaries, and immediately places his predatory beak at the females neck (D. Tallamy pers. com.). The reason for the apparently general failure of male insects to use intimidation (Clutton-Brock and Parker 1995), which is widespread in vertebrate groups ranging from primates (Thornhill and Thornhill 1983, Crawford and Galdikas1986, Smuts and Smuts 1993) to lizards (Olsson 1995), is not clear. Male insects do, in contrast, often harass females to induce copulation (e.g. Thornhill and Alcock 1983, Fincke et al. 1997, Arnqvist 1997, Wilkinson and Dodson 1997, Crean and Gilburn 1998, Cordero 1999).
Acknowledgments
I thank A. Ozerov for unstinting help in identifying specimens, and Maribelle Vargas for outstanding production of SEM images. Martha Lucia Baena assisted in raising flies. The Ortiz-Basualdo family kindly hosted me on their ranch in Argentina, and the Harms family in Ithaca. Göran Arnqvist, Martha Lucia Baena, John Christy, Carlos Cordero, Jessica Eberhard, Andre Gilburn, Kyle Harms, Doug Tallamy, Laurie Tompkins, David Westneat, Mary Jane West-Eberhard, and anonymous referees made useful comments on previous drafts of the manuscript; a discussion with Rex Cocroft was also helpful. Financial support was provided by the Smithsonian Tropical Research Institute, and the Vicerrectoría de Investigación of the Universidad de Costa Rica.
Resumen
Este artículo intenta organizar algunas ideas teóricas sobre posibles conflictos entre machos y hembras en cuanto a la cópula. Las ideas se ilustran con datos nuevos de una familia de moscas (Sepsidae), y con otros datos ya publicados sobre otros insectos. El hecho de que una hembra opone resistencia a la penetración por el macho no implica en sí mismo que la cópula es forzada, ya que la resistencia de la hembra también puede funcionar como una prueba del macho, para así tamizar entre diferentes machos y conseguir hijos superiores. Se contrastan dos hipótesis para el macho, la de la cópula por coerción vs. la de la cópula por persuasión; también se contrastan, desde la perspectiva de la hembra, dos hipóteses relacionadas, la resistencia para evitar a todo macho en forma no selectiva vs. la resistencia para tamizar entre machos. Cuando la morfología de la genitalia de los dos sexos es tal que el macho no es físicamente capaz de forzar la intromisión, como es el caso en las moscas sépsidas y probablemente en muchos otros insectos (a pesar de algunas publicaciones que sugieren lo contrario), las hipótesis contrastantes para cada sexo pueden resolverse con datos sobre el contexto ecológico en el cual ocurre la interacción macho-hembra. Se realizó un análisis de esta clase con los sépsidos. Parece más probable que la resistencia energética que la hembra pone al macho sea un esfuerzo para tamizar entre machos que un esfuerzo para evitar la cópula en forma no selectiva.
References
Alexander, R.D. D.C. Marshall & J.R. Cooley. 1997. Evolutionary perspectives on insect mating, p. 4-31. In J.C. Choe & B. Crespi (eds.). The evolution of mating systems of insects and spiders. Cambridge University, Cambridge. [ Links ]
Allen, G.R. & L.W. Simmons. 1996. Coercive mating, fluctuating asymmetry and male mating success in the dung fly Sepsis cynipsea. Anim. Behav. 52: 737-741. [ Links ]
Alonso-Pimentel, H., H.G. Spangle & W.B. Head. 1995. Courtship sounds and behavior of the two saguarobreeding Drosophila and their relatives. Anim. Behav. 50: 1031-1039. [ Links ]
Andersson, M. 1994. Sexual selection. Princeton University, Princeton. [ Links ]
Arnqvist, G. 1992. Pre-copulatory fighting in a water strider: Intersexual conflict or mate assessment? Anim. Behav. 43: 559-567. [ Links ]
Arnqvist, G. 1997. The evolution of water strider mating systems: Causes and consequences of sexual conflicts, p. 146-163. In J.C. Choe & B. Crespi (eds.). The evolution of mating systems of insects and spiders. Cambridge University, Cambridge. [ Links ]
Arnqvist, G. & L. Rowe. 1995. Sexual conflict and arms races between the sexes: Amorphological adaptation for control of mating in a female insect. Proc. Roy. Soc. Lond. Ser. B 261: 123-127. [ Links ]
Belk, D. 1984. Antennal appendages and reproductive success in the Anostraca. J. Crust. Biol. 4: 66-71. [ Links ]
Berry, J.F. & R. Shine. 1980. Sexual dimorphism and sexual selection in turtles (Order Testudines). Oecologia 44: 185-191. [ Links ]
Blankenhorn, W.U., T. Reusch & C. Mühlhäuser. 1997. Fluctuating asymmetry, body size and sexual selection in the dung fly Sepsis cynipsea - Testing the good genes assumptions and predictions. J. Evol. Biol. 10. [ Links ]
Blankenhorn, W.U., C. Mühlhäuser, C. Morf, T. Reusch & M. Reuter. 2000. Female choice, female reluctance to mate and sexual selection on body size in the dung fly Sepsis cynipsea. Ethology. [ Links ]
Brown, K.S. 1981. The biology of Heliconius and related genera. Ann. Rev. Entomol. 26: 427-456. [ Links ]
Brown, W.D., B.J. Crespi & J.C. Choe. 1997. Sexual conflict and the evolution of mating systems, p. 352- 377. In J.C. Choe & B. Crespi (eds.). The evolution of mating systems of insects and spiders. Cambridge University, Cambridge. [ Links ]
Bubis, J.A., H.P. Degreen, J.L. Unsell & L. Tompkin.. 1998. Temporal manipulation of ejaculate components by newly fertilized Drosophila melanogaster females. Anim. Behav. 55: 1637-1645. [ Links ]
Chapman, T., L.F. Liddle, J.M. Kalb, M.F. Wolfner & L. Partridge. 1995. Cost of mating in Drosophila melanogaster females is mediated by male accessory gland products. Nature 373: 241-244. [ Links ]
Clutton-Brock, T.H. & G.A. Parker. 1995. Sexual coercion in animal societies. Anim. Behav. 49: 1345-1365. [ Links ]
Connolly, K. & R. Cook. 1973. Rejection responses by female Drosophila melanogaster: Their ontogeny, causality and effects upon the behaviour of the courting male. Behaviour 44: 143-165. [ Links ]
Cordero, A. 1999. Forced copulations and female contact guarding at high male density in a calopterygid damselfly. J. Insect Behav. 12: 27-37. [ Links ]
Crawford, C. & B.M. Galdikas. 1986. Rape in non-human animals: An evolutionary perspective. Can. Psychol. 27: 215-229. [ Links ]
Crean, C.S. & A.S. Gilburn. 1998. Sexual selection as a side effect of sexual conflict in the seaweed fly Coelopa ursina (Diptera: Coelopidae). Anim. Behav. 56: 1405-1410. [ Links ]
Daly, M. 1978: The cost of mating. Amer. Natur. 112: 771-774. [ Links ]
Daniellson, I. & C. Askenmo. 1999. Male genital traits and mating interval affect male fertilization success in the water strider Gerris lacustris. Behav. Ecol. Sociobiol. 46: 149-156. [ Links ]
Eberhard, W.G. 1970. The natural history of the fungus gnats Leptomorphus bifasciatus (Say) and L. subcaeruleus(Coquillett) (Diptera: Mycetophilidae). Psyche 77: 361-383. [ Links ]
Eberhard, W.G. 1985. Sexual selection and animal genitalia. Harvard University, Cambridge, MA. [ Links ]
Eberhard, W.G. 1993. Copulatory courtship and genital mechanics of three species of Macrodactylus (Coleoptera, Scarabeidae, Melolonthinae). Ecol. Ethol. Evol. 5: 9-63. [ Links ]
Eberhard, W.G. 1994. Evidence for the widespread occurrence of courtship during copulation in 131 species of insects and spiders, and implications for cryptic female choice. Evolution 48: 711-733. [ Links ]
Eberhard, W.G. 1996. Female control: Sexual selection by cryptic female choice. Princeton University, Princeton, NJ. [ Links ]
Eberhard, W.G. 1998a. Female roles in sperm competition, p. 91-116. In T.H. Birkhead & A.P. Møller (eds.). Sperm competition and sexual selection. Academic, New York. [ Links ]
Eberhard, W.G. 1998b. Sexual behavior of Acanthocephala declivis guatemalana (Hemiptera, Coreidae) and the allometric scaling of their modified hind legs. Ann. Entomol. Soc. Amer. 91: 863-871. [ Links ]
Eberhard, W.G. 2001a. The functional morphology of species-specific clasping structures on the front legs of five species of Archisepsis flies (Diptera, Sepsidae). Zool. J. Linn. Soc. 133: 335-368. [ Links ]
Eberhard, W.G. 2001b. Species-specific genitalic copulatory courtship in sepsid flies (Diptera, Sepsidae, Microsepsis). Evolution 55: 93-102. [ Links ]
Eberhard, W.G. Physical restraint or stimulation? The function(s) of the modified front legs of male Archisepsis diversiformis (Diptera, Sepsidae). J. Insect Behav. In press. [ Links ]
Eberhard, W.G. The relation between aggressive and sexual behavior and allometry in Palaeosepsis dentatiformis flies (Diptera: Sepsidae). J. Kans. Entomol. Soc. In press. [ Links ]
Eberhard, W.G. & B.A. Huber. 1998. Copulation and sperm transfer in Archisepsis flies. Stud. Dipterol. 5: 217-248. [ Links ]
Eberhard, W.G. & M.C. Marín. 1996. Sexual behavior and the enlarged hind legs of male Megalopus aramatus (Coleoptera, Chrysomelidae, Megalopinae). J. Kans. Entomol. Soc. 69: 1-8. [ Links ]
Eberhard, W.G. & F. Pereira. 1995. The process of intromission in the Mediterranean fruit fly, Ceratitis capitata (Diptera: Tephritidae). Psyche 102: 99-120. [ Links ]
Eberhard, W.G. & F. Pereira. 1996. The functional morphology of male genitalic surstyli in the dungflies Achisepsis diversiformis and A. ecalcarata. J. Kans. Entomol. Soc. 69, suppl.: 43-60. [ Links ]
Eberhard, W.G., B.A. Huber, R.L. Rodríguez, R.D. Briceño, V. Rodríguez & I. Salas. 1998. One size fits all? Relationships between the size and degree of variation in genitalia and other body parts in twenty species of insects and spiders. Evolution 52: 415-431. [ Links ]
Fincke, O., J.K. Waage & W.D. Koenig. 1997. Natural and sexual selection components of odonate mating patterns, p. 58-74. In J.C. Choe & B. Crespi (eds.). The evolution of mating systems of insects and spiders. Cambridge University, Cambridge. [ Links ]
Gregory, G.E. 1965. The formation and fate of the spermatophore in the African migratory locust, Locusta migratoria migratoroides Reiche and Fairmaire. Trans. Roy. Entomol. Soc. Lond. 117: 33-66. [ Links ]
Gowaty, P.A. 1997. Principles of females perspectives in avian behavioral ecology. J. Avian Biol. 28: 95-102. [ Links ]
Gowaty, P.A. & N. Buschhaus. 1998. Ultimate causation of forced copulation in birds: Female resistance, the CODE hypothesis, and social monogamy. Amer. Zool. 38: 207-225. [ Links ]
Hartmann, R. & W. Loher. 1974. Control of sexual behaviour patterns secondary defence in the female grasshopper, Chorthippus curtipennis. J. Insect Physiol. 20: 1713-1728. [ Links ]
Headrick, D.H. & R.D. Goeden. 1994. Reproductive behavior of California fruit flies and the classification of Tephritidae (Diptera) mating systems. Stud. Dipterol. 1: 194-252. [ Links ]
Hennig, W. 1949. Sepsidae, p. 1-91. In E. Lindner (ed.). Die Fliegen der Palaearktischen Region. No. 39a. E. Schweizerbertsche Verlagsbuchhandlung (Erwin Nagele), Stuttgart. [ Links ]
Holland, B. & W.R. Rice. 1998. Chase-away selection: Antagonistic seduction vs. resistance. Evolution 52: 1-7. [ Links ]
Holland, B. & W.R. Rice. 1999. Experimental removal of sexual selection reverses intersexual antagonistic coevolution and removes a reproductive load. Proc. Nat. Acad. Sci. U. S. A. 96: 5083-5088. [ Links ]
Huber, B.A. 1998. Spider reproductive behaviour: A review of Gerhardts work from 1911-1933, with implications for sexual selection. Bull. Br. Arachnol. Soc. 11: 81-91. [ Links ]
Jormalainen, V., S. Merilaita & R. Härdling. 2000. Dynamics of inter-sexual conflict over precopulatory mate guarding in two populations of the isopod Idotea baltica. Anim. Behav. 60: 85-93. [ Links ]
Krieger, F. & E. Krieger-Loibl. 1958. Beitrage zum Verhalten von Ischnura elegans und Ischnura pumilio (Odonata). Zeitschr. Tierphychol. 15: 82-93. [ Links ]
Linley, J.R. & G.M. Adams. 1972. A study of the mating behaviour of Culicoides melleus (Coquillett) (Diptera: Ceratopogonidae). Trans. Roy. Entomol. Soc. Lond. 124: 81-121. [ Links ]
Lloyd, J. 1979. Mating behavior and natural selection. Florida Entomol. 62: 17-23. [ Links ]
Manning, A. 1967. The control of sexual receptivity in female Drosophila. Anim. Behav. 15: 239-250. [ Links ]
Markow, T.A. 2000. Forced matings in natural populations of Drosophila. Amer. Natur. 156: 100-103. [ Links ]
McKinney, F. & S. Evarts. 1997. Sexual coercion in water fowl and other birds. Ornithol. Monogr. 49: 163-195. [ Links ]
Oh, R.J. 1979. Repeated copulation in the brown plan-thoper, Nilaparvata lugens Stål (Homoptera; Delphacidae). Ecol. Entomol. 4: 345-353. [ Links ]
Olsson, M. 1995. Forced copulation and costly female resistance behavior in the Lake Eyre dragon, Ctenophorus maculosus. Herpetologica 51: 19-24. [ Links ]
Ozerov, A.L. 1992. New data on the neotropical sepsids (Diptera Sepsidae). Russ. Entomol. J. 1: 81-86. [ Links ]
Ozerov, A.L. 1993. Six new species of the genus Palaeosepsis Duda (Diptera Sepsidae). Russ. Entomol. J. 2: 63-71. [ Links ]
Parker, G.A. 1972a. Reproductive behaviour of Sepsis cynipsea (L.) (Diptera: Sepsidae) I. A preliminary analysis of the reproductive strategy and its associated behaviour patterns. Behaviour 41: 172-206. [ Links ]
Parker, G.A. 1972b. Reproductive behaviour of Sepsis cynipsea (Diptera: Sepsidae) II. The significance of the precopulatory passive phase and emigration. Behaviour 41: 243-250. [ Links ]
Parker, G.A. 1974. Courtship persistance and female-guarding as male time-investment strategies. Behaviour 48: 157-184. [ Links ]
Pickford, R. & D.E. Padgham. 1973. Spermatophore formation and sperm transfer in the desert locust, Schistocera gragaria (Orthoptera: Acrididae). Can. Entomol. 105: 613-618. [ Links ]
Pickford, R. & C. Gillott. 1971. Insemination in the migratory grasshopper Melanoplus sanguinipes (Fabr.). Can. J. Zool. 49: 1583-1588. [ Links ]
Pont, A. 1979. Sepsidae, p. 1-35. In M.G. Fitton (ed.). Handbooks for the identification of British insects. Vol. 10, Part 5(c). Roy. Entomol. Soc. Lond., London. [ Links ]
Rice, W.R. 1996. Sexually antagonistic male adaptation triggered by experimental arrest of female evolution. Nature 281: 232-234. [ Links ]
Rodríguez, R.L. 1998. Mating behavior of two Pseudoxychila beetles (Coleoptera: Cicindelidae). Can. Entomol. 130: 1-16. [ Links ]
Rubenstein, D.I. 1989. Sperm competition in the water strider, Gerris remigis. Anim. Behav. 38: 631-636. [ Links ]
Rutowski, R. 1997. Sexual dimorphism, mating systems and ecology in butterflies, p. 257-272. In (J. Choe & B. Crespi (eds.). The evolution of mating systems in insects and arachnids. Cambridge University, Cambridge. [ Links ]
Sakaluk, S.K., P.J. Bangert, A.-K. Eggert, C. Gack & L.V. Swanson. 1995. The gin trap as a device facilitating coercive mating in sagebrush crickets. Proc. Roy. Soc. Lond. Ser. B 261: 65-71. [ Links ]
Schulz, K. 1999. The evolution of mating systems in black scavenger flies (Diptera: Sepsidae). Ph.D. Thesis, University of Arizona, Tucson, AZ. [ Links ]
Silva, V.C. 1993. Revisão da família Sepsidae na região neotropical. III. Os gêneros Palaeosepsis Duda, 1926, Archisepsis gen. n. e Microsepsis gen. n.; chave para os gêneros neotropicais (Diptera, Schizophora). Ihringia, Ser. Zool. 75: 117-170. [ Links ]
Smith, D.C. & R.J. Prokopy. 1980. Mating behavior of Rhagoletis pomonella (Diptera: Tephritidae) VI. Site of early-season encounters. Can. Entomol. 121: 585- 590. [ Links ]
Smuts, B.B. & R.W. Smuts. 1993. Male aggression and sexual coercion in females in nonhuman primates and other mammals: Evidence and theoretical implications. Adv. Stud. Behav. 22: 1-63. [ Links ]
Spieth, H.T. 1947. Sexual behavior and isolation in Drosophila. I. The mating behavior of species of the willistoni group. Evolution 1: 17-31. [ Links ]
ulc, K. 1928. Biologický význam výzbroje přednich saměích nohou u Sepsid (Muscidae). (Biologische Bedeutung der Bewaffnung der männlichen Vorderfüsse bei den Sepsiden (Muscidae). Biologické Spisy Vysoké koly Zvérolékarské Brno, Čsr. 14: 428-439. [ Links ]
Suzuki, N. & K. Matsumoto. 1990. Pair-clinging behavior by males of Atrophaneura alcinous (Lepidoptera: Papilionidae). J. Ethol. 8: 45-51. [ Links ]
Thomas, R.H. & D.W. Zeh. 1984. Sperm transfer and utilization strategies in arachnids: Ecological and morphological contstraints, p. 180-221. In R.L. Smith (ed.). Sperm competition and the evolution of animal mating systems. Academic, New York. [ Links ]
Thornhill, R. 1980. Rape in Panorpa scorpionflies and a general rape hypothesis. Anim. Behav. 28: 52-59. [ Links ]
Thornhill, R. & J. Alcock. 1983. The evolution of insect mating systems. Harvard University, Cambridge. [ Links ]
Thornhill, R. & K.P. Sauer. 1991. The notal organ of the scorpionfly (Panorpa vulgaris): An adaptation to coerce mating duration. Behav. Ecol. 2: 156-164. [ Links ]
Thornhill, R. & N.W. Thornhill. 1983. Human rape: An evolutionary analysis. Ethol. Sociobiol. 4: 137-173. [ Links ]
Tompkins, L. & J.C. Hall. 1983. Identification of brain sites controlling female receptivity in mosaics of Drosophila melanogaster. Genetics 103: 179-195. [ Links ]
Ward, P.I., H. Hemmi & T. Roösli. 1992. Sexual conflict in the dung fly Sepsis cynipsea. Funct. Ecol. 6: 649- 653. [ Links ]
Weall, C.V. & A.S. Gilburn. 2000. Factors influencing the choice of female mate rejection strategies in the sea-weed fly Coelopa nebularum (Diptera: Coelopidae). J. Insect Behav. 13: 539-552. [ Links ]
Weigensberg, I. & D.J. Fairbairn. 1994. Conflict of interest between the sexes: A study of mating interactions in a semiaquatic bug. Anim. Behav. 48: 893-901. [ Links ]
Weigensberg, I. & D.J. Fairbairn. 1996. The sexual arms race and phenotypic correlates of mating success in the waterstrider, Acquarius remigis. J. Insect Behav. 9: 307-319. [ Links ]
Westneat, D.F., P.W. Sherman & M.L. Morton. 1990. The ecology and evolution of extra-pair copulations in birds, p. 330-369. In D.M. Power (ed.). Current ornithology. Plenum, New York. [ Links ]
Whitman, D.W. & W. Loher. 1984. Morphology of male sex organs and insemination in the grasshopper Taeniopoda eques (Burmeister). J. Morphol. 179: 1-12. [ Links ]
Wilkinson, G.S. & G. Dodson. 1997. Function and evolution of antlers and eye stalks in flies, p. 310-328. In J.C. Choe & B.J. Crespi (eds.). The evolution of mating systems of insects and spiders. Cambridge University, Cambridge. [ Links ]