Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.49 n.3-4 San José Dec. 2001
on the population growth of rotifers Brachionus calyciflorus
and Brachionus patulus (Rotifera)
1 Laboratorio de Zoología Acuática, División de Investigación y Posgrado, Edificio UMF, Universidad Nacional Autónoma de México, Campus Iztacala, AP 314, CP 54090, Tlalnepantla, Edo. de México. México. FAX: +52 5 623 1256. corel: sarma@servidor.unam.mx
2 División de Investigación y Posgrado, Proyecto CyMA, Universidad Nacional Autónoma de México, Campus Iztacala, AP 314, CP 54090, Tlalnepantla, Edo. de México.
*Corresponding author
Recibido: 12-VI-2000 Corregido: 31-I-2001 Aceptado: 25-II-2001
Abstract
In order to maintain rotifer populations during periods of low algal production, it is necessary to offer alternate diets, some of which include forms of preserved algae. The present work is based on the effect of live and dead Chlorella vulgaris on the population growth of Brachionus calyciflorus and Brachionus patulus. The experimental design consisted of 3 algal levels (0.5x106, 1.5x106 and 4.5x106 cells ml-1) offered in 3 forms (living, frozen and heat-killed). The maximal population density values for B. calyciflorus ranged from 55±1 ind. ml-1 (at 0.5x106 cells ml-1) to 471±72 ind. ml-1 (at 4.5x106 cells ml-1) with live Chlorella, but was much lower (6±1 to 26±6 ind. ml-1) with frozen or heat-killed alga under comparable food levels. However, the maximum population density of B. patulus under live or or heat-killed Chlorella was similar at comparable algal levels but when offered frozen algae it was four times less. The highest mean peak population density was 1227±83 ind. ml-1 under 4.5x106 cells ml-1. The rate of population increase for B. calyciflorus varied from 0.50 to 0.79 using live Chlorella, but under comparable conditions, this range was lower (0.21 to 0.31) for B. patulus. Results have been discussed in light of possible application for aquaculture.
Key words: Rotifera, Chlorella, Population growth, Live-alga, Dead-alga.
Rotiers of the genus Bracchionus are widely used as live food in rearing larval fish, crabs, prawns, shrimps and molluscs under both marine and freshwater culture conditions (Sarma 1991). For mass production of B. plicatilis various types of food, both natural (e.g., algae) and artificial diets (microencaspulated pallets) have been used. Hirata and Mori (1967) replaced green algae with baker's yeast for mass production of B. plicatilis. However, due to low nutritional value of normal yeast-fed rotifers, a number of enrichment procedures have to be followed (Hirayama and Funamoto 1983). Althuogh some of these enrichment procedures are easily done, the use of microalgae in rotifer cultures has been largely practiced (Pourriot 1991).
Since large- scale algal production is relatively cheap, both under field and laboratory conditions, various types of algae are routinely being produced for feeding planktonic rotifers (Groeneweg and Schuter 1981). However, some times algal cultures crash (Suminto and Hirayama 1997) which may, in turn, lead to problems of finding adequate food for rotifers. Therefore, stored algae could be used as substitute. On the other hand, at times there may be excessive production of microalgal which could be stored for future use (Millamena et al. 1990, Martínez-Jeronimo ansd Espinosa-Chavez 1994). The nutrional quality as well as the digestibility of the stored-microbal may vary considerably and consequently the growth responses of zooplankton feeding on them (Gastesoupe and Robin 1981, Dobberfuhl and Elser 1999). In this context, population level responses of rotifers to stored microalgae has t been well document (Starkweather and Bogdan 1980, Lubzens et al. 1995).
The main of this study was not compare the population growth of tow freshwater rotifers, B. calciflorus and B. patulus using Chorella vulgaris in live, frozen and heat-killed forms under three food levels.
Materials and methods
The test rotifer species Brachionus calyciflorus and Brachionus patulus were isolated from lake Chapultepec (Mexico City) and the waterbody Presa Santa Elena (State of Mexico), respectively. Clonal populations of both these species were established using the single celled green algae Chlorella vulgaris. For mass cultures as well as for experiments, we used C. vulgaris, raised on Bold's basal medium (Borowitzka and Borowitzka 1988). Algae in log phase of growth were harvested, centrifuged at 3000 rpm for 5 minutes, rinsed with distilled water and resuspended in reconstituted moderately hardwater, (the EPA medium), which was prepared by dissolving 96 mg NaHCO3, 60 mg CaSO4, 60 mg MgSO4 and 4 mg KCl in one litre of distilled water (Anonymous 1985). The stock algal density was estimated using a haemocytometer. Test rotifers were offered C. vulgaris in three forms: live, cold-killed (freshly frozen) and heat-killed (with boiling water). For experiments, we used algae of no more than two days old, following harvest.
The experimental design for B. calyciflorus consisted a total of 27 transparent test jars (50 ml capacity) containing 50 ml EPA medium. We used three food levels namely, 0.5x106, 1.5x106 and 4.5x106 cells ml-1 in EPA medium. For each food density and form, we used 3 jars per treatment. Into each of the test jars we inoculated individuals of B. calyciflorus at an initial density of 5 ind. ml-1 using finely drawn Pasteur pipette. The test jars were maintained at 22±2 ºC, pH 7.2-7.5 and continous but diffused flourescent illumination (1000 lux). Following inoculation the density of B. calyciflorus was estimated daily using total counts or 2-3 aliquots of 1 ml each. We counted the number live rotifers (females). Due to uncertainity of the time of death, dead individuals were not estimated. After estimating the density, rotifers were transferred using 50mm mesh to new jars containing appropriate algal food density and form (i.e. live, frozen or heat-killed state). The experiment was discontinued after two weeks by which time B. calyciflorus in most replicates began to decline. Concurrently but separately, experiments were also conducted using the other brachionid rotifer B. patulus. For this species also, the experimental design and the test conditions were similar to those B. calyciflorus, except for the duration, which was 40 days.
Based on the data collected, we calculated the rate of population increase (r) using the exponential growth equation: r = (ln Nt- ln No)/t, where, No = initial population density, Nt = density of population after time t (days) (Krebs, 1985). The r was obtained from a mean of 4-5 values during the exponential phase of the population growth for each species.
Results
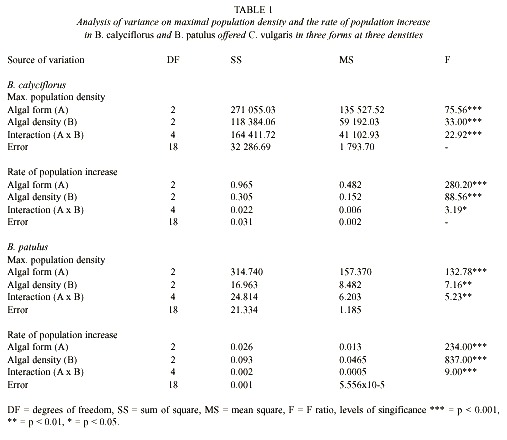
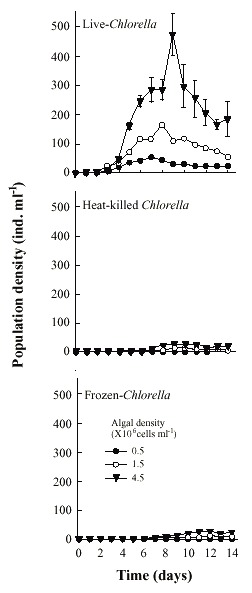
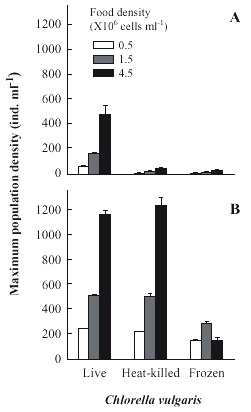
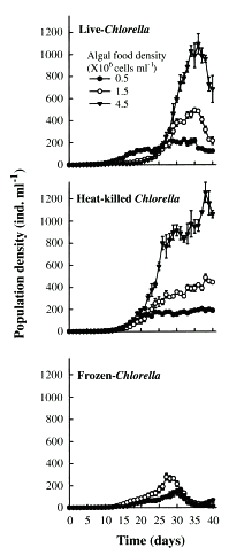
Population growth curves of B. calyciflorus in relation to different Chlorella levels and forms are presented in the Fig. 1. Regardless of form in which Chlorella was offered, B. calyciflorus showed increased population abundance with increasing food level. Within the three forms of Chlorella, live algae supported the best growth of B. calyciflorus. The maximal population density values for B. calyciflorus ranged from 55±1 ind. ml-1 (under 0.5x106 cells ml-1) to 471±72 ind. ml-1 (under 4.5x106 cells ml-1) with live Chlorella, but was much lower (6±1 to 26±6 ind. ml-1) with frozen alga under comparable food levels (Fig. 2A). The maximum population density of rotifers was significantly affected by both the algal level and the form (Table 1, p<0.001, ANOVA, Sokal and Rohlf 1985). The rate of population increase for B. calyciflorus varied from 0.50 to 0.79 on live algae, depending on the food level. However, the values were much lower (0.10 to 0.41) when heat-killed or frozen Chlorella was used as food (Fig. 3A). Thus, both food level and form as well as their interaction had a significant effect on the rate of population increase of B. calyciflorus (Table 1).
Fig. 1. Population growth curves of B. calyciflorus in relation to Chlorella density offered as live, heat-killed and
frozen forms. Shown are the mean±SE values based on three replicate recordings.
Fig. 2. Maximum population density (ind. ml -1 ) of B. calyciflorus (A) and B. patulus (B) in relation
to Chlorella density offered as live, heat-killed and frozen forms. Shown are the mean±SE
values based on three replicate recordings.
Brachionus patulus needed from 27 to 40 days to show a declining trend in the population densities (Fig. 4). The maximum population density of B. patulus under live or or heat-killed Chlorella was similar at comparable algal levels (Fig. 2B), with the trend of increasing population abundance with increasing availability of food. Thus the highest mean peak population density was 1 227±83 ind. ml-1 under 4.5x106 cells ml-1. B. patulus offered frozen algae increased only one-fourth of this. The rate of population increase varied from 0.21±0.003 to 0.31±0.002 under 0.5x106 cells ml-1 and 4.5x106 cells ml-1 of live Chlorella, respectively (Fig. 3B). Both form and level of Chlorella, as well as their interactions signficantly (p<0.05, ANOVA, Table 1) influenced the mean maximal population density and rate of increase.
density offered as live, heat-killed and frozen forms. Shown are the mean±SE values based on three replicate
recordings.
Fig. 4. Population growth curves of B. patulus in relation to Chlorella density offered as live,
heat-killed and frozen forms. Shown are the mean±SE values based on three replicate recordings.
Discussion
Various studies have shown the importance of food density on the population growth of zooplankton, both from field collections and laboratory studies (Edmondson 1960, Halbach and Halbach-Keup 1974). Rotifers being opportunistic species, respond more rapidly to changes in food levels (Nogrady et al. 1993). It is known that rotifers show an almost linear numerical increase with increasing food levels (Dumont et al. 1995). Chlorella is one the most widely used food for culturing planktonic rotifers (Pourriot and Rougier 1997). Studies on the use of preserved algae for zooplankton growth are rare because of a general idea that non-living algae do not support their growth (Kumlu 1997, Baer and Goulden 1998). Our study showed that this is true for B. calyciflorus but not for B. patulus. For example, B. calyciflorus did not grow well on either heat-killed or frozen Chlorella while, B. patulus was able to utilize heat-killed (but not to the same extent on frozen) algae more effectively and reached densities comparable to those of live Chlorella at all tested levels.
Data available in literature on the use of various forms of algae such as frozen, dry powder and freeze-dried so far have concentrated on Brachionus plicatilis and usually under a single food density (Yamasaki et al. 1989, Yúfera and Navarro 1995, Navarro 1999). Although these results form the basis, they cannot be extrapolated to other commonly co-occuring species of the same genus. Thus, the present study provides the possibility of comparing two rotifer species belonging to the same genus, not only under the same food levels, but also under identical test conditions. It is evident from the data on population growth that B. calyciflorus (Fig. 1) could reach peak abundances much earlier than B. patulus (Fig. 4). This was also reflected in the rates of population increase. Among the planktonic rotifers, B. calyciflorus is known to have the highest growth rates (Bennett and Boraas 1989). In the present study also, under comparable live Chlorella levels, B. calyciflorus had higher growth rates than those of B. patulus (Fig. 3). The mean maximal population density reached by a given rotifer species is dependent on its body-size. Generally, large rotifer species have lower numerical abundance per unit volume of medium compared to smaller species (Sarma et al. 1999). For example, the rotifer Anuraeopsis fissa with a body lenght of about 70 µm, could reach an abundance of > 9500 ind. ml-1 but under comparable food levels, a large species (B. calyciflorus, lenght about 255 µm) could reach only one-tenth of this density (Sarma et al. 1996). In the present study also, B. patulus being smaller than B. calyciflorus reached much higher abundances. The r values obtained in this study are comparable to those reported in a review of growth rates of selected rotifer species by Sarma et al. (2000).
The differential responses of B. calyciflorus and B. patulus to non-living Chlorella is probably related to their natural feeding habits. For example, B. calyciflorus is typically planktonic, while B. patulus is normally associated with vegetation, probably feeding on detritus and epiphytic algae (Ruttner-Kolisko 1972). This is reflected in the higher maximal population abundances of B. patulus fed on heat-killed Chlorella compared to B. calyciflorus. However, both the rotifer species could not utilize frozen algae effectively. This suggests that frozen Chlorella is probably difficult to digest and/or the tested rotifers might have the capacity to discreminate different forms of the same alga (DeMott, 1986). Lubzens et al. (1995) have also shown that the nutritonal quality of frozen alga is not significantly different from live alga. When heat-killed, algal digestibility probably would increase but the nutritional quality may decrease (Hohman et al. 1982, Brown 1995). However, the fact that B. patulus could grow well on the heat-killed Chlorella suggests that the response of brachionids could vary considerably based on their adaptations to the natural conditions they inhabit. This could also be due to the discriminatory capacity of rotifer species to live versus dead food particles (Starkweather and Bogdan 1980, DeMott 1986).
In conclusion, some implications of the present work are that although frozen Chlorella could not support growth of the test species, it could be used just for maintaining the high densities of rotifers, until live alga is available. Also, if the nutritional quality of rotifers fed on heat-killed algae is found comparable to those fed on live alga, then it could be possible to heat-kill and preserve algae for later use to maintain rotifer cultures.
Acknowledgements
This investigation was supported by a Project from PAPIIT (IN223799). Additional support from a grant awarded for SNI members by ENEP-Iztacala to SSSS is gratefully acknowledged. ELP thanks CONACyT (Ref. 67991017) for a scholarship. SN thanks the National System of Investigators (SNI-20520).
Resumen
Para mantener poblaciones de rotíferos durante periodos con escasez de microalgas, es necesario ofrecer dietas alternativas, incluyendo algunas formas de microalgas preservadas. El presente trabajo analiza el efecto de Chlorella vulgaris viva y muerta sobre el crecimiento poblacional de Brachionus calyciflorus y Brachonus patulus. El diseño experimental consistió en tres niveles de algas (0.5x106, 1.5x106 y 4.5x106 células ml-1) ofrecidas en tres formas (viva, congelada y muerta con agua caliente). Las abundancias máximas de población de B. calyciflorus variaron desde 55±1 ind. ml-1 (en 0.5x106 células ml-1) a 471±72 ind. ml-1 (en 4.5x106 células ml-1) con Chlorella viva, mientras que, cuando se utilizó alga congelada o muerta con agua caliente, la abundancia fue mucho menor (6±1 a 26±6 ind. ml-1). Sin embargo, la máxima densidad de población de B. patulus con Chlorella viva o muerta con calor fue similar bajo niveles de algas comparables, mientras que cuando se ofreció alga congelada, la abundancia fue cuatro veces menor. La abundancia máxima de B. patulus fue 1 227±83 ind. ml-1 bajo 4.5x106 células ml-1. La tasa de crecimiento poblacional de B. calyciflorus varió desde 0.50 hasta 0.79 usando Chlorella viva, pero bajo las mismas condiciones, el rango es menor (0.21 a 0.31) para B. patulus. Los resultados se discuten para su posible aplicación en la acuacultura.
References
Anonymous. 1985. Methods of measuring the acute toxicity of effluents to freshwater and marine organisms. US Environment Protection Agency EPA/600/4-85/013, Washington. [ Links ]Baer, K.N. & C.E. Goulden. 1998. Evaluation of a highhardness COMBO medium and frozen algae for Daphnia magna. Ecotoxicol. Environ. Saf. 39: 201- 206. [ Links ]
Bennett, W.N. & M.E. Boraas. 1989. A demographic profile of the fastest growing metazoan a strain of Brachionus calyciflorus (Rotifera). Oikos 55: 365-369. [ Links ]
Borowitzka, M.A.& L.J. Borowitzka. 1988. Microalgal biotechnology. Cambridge University, London. 480 p. [ Links ] Brown, M.R. 1995. Effects of storage and processing on the ascorbic acid content of concentrates prepared from Chaetoceros calcitrans. J. Appl. Phycol. 7: 495-500. [ Links ]
DeMott, W.R. 1986. The role of taste in food selection by freshwater zooplankton. Oecologia 69: 334-340. [ Links ]
Dobberfuhl, D.R. & J.J. Elser. 1999. Use of dried algae as a food source for zooplankton growth and nutrient release experiments. J. Plankton Res. 21: 957-970. [ Links ]
Dumont, H.J., S.S.S. Sarma & A.J. Ali. 1995. Laboratory studies on the population dynamics of Anuraeopsis fissa (Rotifera) in relation to food density. Freshwater Biol. 33: 39-46. [ Links ]
Edmondson, W.T. 1960. Reproductive rates of rotifers in natural populations. Mem. Ist. Ital. Idrobiol. 12: 21-77. [ Links ]
Gatesoupe, F. J. & J. H. Robin. 1981. Commercial singlecell proteins either as sole food source or in formulated diets for intensive and continuous production of rotifers (Brachionus plicatilis). Aquaculture 25: 1-15. [ Links ]
Groeneweg, J. & M. Schluter. 1981. Mass production of freshwater rotifers on liquid wastes II. Mass production of Brachionus rubens Ehrenberg, 1838 in the effluent of high-rate algal ponds used for the treatment of piggery waste. Aquaculture 25: 25-33. [ Links ]
Halbach, U. & G. Halbach-Keup. 1974. Quantitative beziehungen zwischen Phytoplankton und der Populationsdynamik des Rotators Brachionus calyciflorus Pallas. Befunde aus Laboratoriumsexperimenten und Freilanduntersuchungen. Arch. Hydrobiol. 73: 273- 309. [ Links ]Hirata, H. & Y. Mori. 1967. Cultivation of the rotifer Brachionus plicatilis fed on a mixed diet of marine Chlorella and bakers yeast. Saibai Gyigyo 5: 36-40. [ Links ]
Hirayama, K. & H. Funamoto. 1983. Supplementary effect of several nutrients on nutritive deficiency of bakers yeast for population growth of the rotifer Brachionus plicatilis Bull. Jap. Soc. Sci. Fish. 49: 505-510. [ Links ]
Hohman, T.C., P.L. McNeil & L. Muscatine. 1982. Phagosome-lysosome fusion inhibited by algal symbionts of Hydra viridis. J. Cell Biol. 94: 56-61. [ Links ]
Krebs, C.J. 1985. Ecology. The experimental analysis of distribution and abundance. Harper and Row, New York. 789 p. [ Links ]
Kumlu, M. 1997. The effect of feed types on survival and trypsin activity in Temora longicornis (Crustacea: Copepoda). Isr. J. Aquacult. 49: 199-204. [ Links ]Lubzens, E., O. Gibson, O. Zmora & A. Sukenik. 1995. Potential advantages of frozen algae (Nannochloropsis sp.) for rotifer (Brachionus plicatilis) culture. Aquaculture 133: 295-309. [ Links ]
Martínez-Jeronimo, F. & F. Espinosa-Chavez. 1994. Alaboratory-scale system for mass culture of freshwater microalgae in polyethylene bags. J. Appl. Phycol. 6: 423-425. [ Links ]
Millamena, O.M., E. J. Aujero & I. G. Borlongan. 1990. Techniques on algae harvesting and preservation for use in culture and as larval food. Aquacult. Engineer. 9: 295-304. [ Links ]Navarro, N. 1999. Feeding behaviour of the rotifers Brachionus plicatilis and Brachionus rotundiformis with two types of food: Live and freeze-dried microalgae. J. Exp. Mar. Biol. Ecol. 237: 75-87. [ Links ]
Nogrady, T., R.L. Wallace & T. W. Snell. 1993. Rotifera. Vol. 1. Biology, ecology and systematics. SBP Academic Pub., The Hague. 142 p. [ Links ]
Pourriot, R. 1991. Los rotiferos. Biología, p. 171-187. In B. Gilbert (ed.). Acuicultura. Omega, Barcelona. [ Links ]Pourriot, R. & C. Rougier. 1997. Reproduction rates in relation to food concentration and temperature in three species of the genus Brachionus (Rotifera). Ann. Limnol. 33: 23-31. [ Links ]
Ruttner-Kolisko, A.1972. Rotatorien. Die Binnen-gewässer Bd. 26: 99-234. [ Links ]
Sarma, S.S.S. 1991. Rotifers and aquaculture (Review). Environ. Ecol. 9: 414-428. [ Links ]Sarma, S.S.S., N. Iyer & H. J. Dumont. 1996. Competitive interactions between herbivorous rotifers: Importance of food concentration and initial population density. Hydrobiologia 331: 1-7. [ Links ]
Sarma, S.S.S., M.A. Fernández-Araiza & S. Nandini. 1999. Competition between Brachionus calyciflorus Pallas and Brachionus patulus (Müller) (Rotifera) in relation to algal food concentration and initial population density. Aquat. Ecol. 33: 339-345. [ Links ]
Sarma, S.S.S., P.S. Larios-Jurado & S. Nandini. 2001. Effect of three food types on the population growth of the rotifers Brachionus calyciflorus and Brachionus patulus (Rotifera: Brachionidae). Rev. Biol. Trop. 49: 75-82. [ Links ]
Sokal, R.R. & F.J. Rohlf. 1981. Biometry. W. H. Freeman, San Francisco. 859 p. [ Links ]
Starkweather, P.L. & K.G. Bogdan. 1980. Detrital feeding in natural zooplankton communities: discrimination between live and dead algal foods. Hydrobiologia 73: 83-85. [ Links ]Suminto, K. & K. Hirayama. 1997. Application of a growth-promoting bacteria for stable mass culture of three marine microalgae. Hydrobiologia 358: 223-230. [ Links ]
Yamasaki, S., K. Tanabe & H. Hirata. 1989. Efficiency of chilled and frozen Nannochloropsis sp. (marine Chlorella) for culture of rotifer. Mem. Fac. Fish. Kagoshima Univ. 38: 77-82. [ Links ]
Yúfera, M. & N. Navarro. 1995. Population growth dynamics of the rotifer Brachionus plicatilis cultured in non-limiting food condition. Hydrobiologia 313/314: 399-405. [ Links ]