Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.49 n.2 San José Jun. 2001
Received 27-V-1999. Corrected 4-10-2000. Accepted 9-X-2000.
Abstract
During March and April of 1996, I made field observations of the sedentary subspecies of grasshopper sparrow (Ammodramus savannarum cracens), in 600 points of the pine savannas of northeastern Nicaragua. Isolated individuals were found in the humid depressions, but breeding populations were located exclusively in areas that had suffered a recent fire. Territorial behavior varied in intensity apparently as a function of territory size: the most aggressive males were those trying to defend smaller territories in populations close to Miskito villages, where most of the fires occur. In contrast to what is happening in other parts of Central America, the Nicaraguan grasshopper sparrow may be indirectly protected from extinction by the Miskitos traditional fire practices.
Key words
Ammodramus savannarum, grasshopper sparrow, pine savannas, Nicaragua.
The grasshopper sparrow (Ammodramus savannarum) is widely but patchily distributed in the Americas. Some of its subspecies are long distance migrants, some are altitudinal or partial migrants, some are sedentary and others are of unknown status (Monson and Phillips 1981, Bock and Webb 1984, Ridgely and Tudor 1989). The decline of this species in North America prompted an increase in research on its habitat characteristics and life-history requirements (Whitmore 1979, 1981, Askins 1993, Herkert 1994a, 1994b, Knopf 1994, Bollinger 1996), and determined the inclusion of A. s. floridanus in the Federal Endangered Species List in 1986 (Delany et al. 1985).
Little is known about the highly localized populations of the eight recognized subspecies that inhabit tropical and subtropical America (Vickery 1996). The secretive habits of this bird make its observation difficult except during the breeding season, when their territorial singing allows proper detection and identification. The distribution and abundance of grasshopper sparrows (A. s. cracens) in the pine savannas of northeastern Central America were recorded by Monroe (1968) in Honduras and Howell (1971, 1972) in Nicaragua. Monroe found them abundant and widely distributed, the dominant bird in the Honduran lowland pine savannas. In contrast, Howell's surveys located few individuals in his four research plots in the northwestern part of the savanna. Here I report recent information on present distribution, abundance and habitat characteristics of this Central American subspecies (A. s. cracens) in the pine savannas of northeastern Nicaragua, including behavioral parallels with one of the North American migratory subspecies (A. s. pratensis).
Materials and Methods
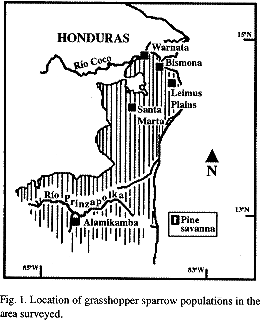
From March 10 to April 28 of 1996, I surveyed all accessible potential areas for grasshopper sparrows in northeastern Nicaragua. This time of year was chosen to coincide with the bird's breeding season as recorded by Howell in 1966. I covered the area south of Río Coco to just south of Río Prinzapolka. Areas to the north east of Puerto Cabezas offer good access during the dry season, because of the many roads built to manage the extensive Pinus caribea plantations. There are very few roads to the south and southwest of Puerto Cabezas.
I surveyed the area using a motorized vehicle and on foot. Once within the defined pine savanna, stops were made every 500-1 000 meters along the roads independent of the particular vegetation characteristics at the point. I entered the land at both sides of the road on foot for approximately 500 meters to scan and listen for grasshopper sparrows. Playbacks of the short (territorial) and long (mated status) songs of A. s. pratensis (Borror Laboratory of Bioacoustics, The Ohio State University) were systematically played at all locations. When birds were located, I used mist-nets to trap them. The individuals were sexed and aged, and color banded to identify recaptures.
Results
Present extent of the pine savanna: The most striking changes in the present distribution of the pine savannas in northeastern Nicaragua from its extent as described by Howell (1972) are man-induced: the existence of relatively old pine forest tracts with dense broadleaf underbrush to the north and northwest which are still live-mined from the 1980's Contra War, and the extensive plantations of the endemic Pinus caribea to the northeast. As noted by Howell (1971) I found tropical forest in the wetter areas with little or no transitional vegetation separating it from the savanna. Howell's "Leicus Creek" type locality, described as "56 miles Northwest of Puerto Cabezas" (Howell 1966), was not recognized by the inhabitants of any of the small villages, and may at present be located within the older pine forests in the heavily land-mined northwest, which I did not survey.
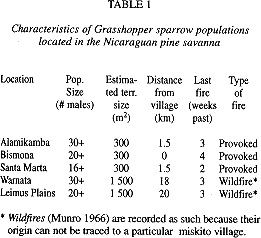
Distribution and abundance: I found grasshopper sparrows only at eleven of the approximately 600 points surveyed. These eleven sites did not include intensively cultivated pine plantations or where brush/pine cover was extensive. I found grasshopper sparrows in large populations, singly, or in groups of a few individuals. Isolated individuals were located in the more humid land depressions, while a group of four first year birds (dispersers?) were mist-netted simultaneously at the edge of a brush area at no less than 35 kilometers from any known breeding population. All sites where I found breeding populations (Fig.1) had suffered at least one fire during the beginning of the dry season (less than two months before this survey). I located only five (Table1) large breeding populations (more than fifteen singing males). The three populations of higher density were near Miskito villages.
The specific vegetation characteristics of the sites varied: scattered, almost exclusively mature P. caribea stands in the Alamikamba area; very low, herbaceous vegetation at Leimus Plains and Warnata; thick grasses with scattered brush in the Bismona area; young, fire-damaged P. caribea plantations spotted with clusters of volcanic rocks around Santa Marta.
Territories and territorial behavior: Defended territories were dominated by low (less than 25 cm high), sparse vegetation. The most abundant were grasses, but mosses, liverworts and particularly forbs were present. All territories included some P. caribea, and a few specimens of Sabal sp. and Byrsonima sp, or less frequently Tripsacum sp. or Miconia sp.
I observed two distinct territory patterns. In the three high density sites (Alamikamba, Bismona, and Santa Marta), territories were less than 300 m2 each, usually with boundaries closely defined by landscape changes (ditches, higher brush). In their absence, I determined territory boundaries based on the behavior of the males. Individuals that crossed a boundary were aggressively expelled. Agonistic behavior between neighboring males consisted of close, fast approach of the territory owner to the invader who would fly off and seek cover. The territory owner would then perch on a low, exposed bush at the edge of the territory and sing the short song repeatedly. The expelled individual did not respond to the singing. After 2-3 minutes of no response the singing would stop.
Isolated individuals or individuals of the fourth and fifth scattered populations (Leimus Plains and Warnata) responded only by infrequent bouts of song to an invader which was simulated by the playback (no natural transgressions were observed). These individuals tended not to approach more than 20 m from the playback. Because of the scarcity of obvious landscape boundaries and the low intensity response to territorial invasion, I could only roughly estimate territory size as at least 1 500 m2, about five times the size of a territory in the high density populations.
When not disturbed the birds sang intensively in the early morning and evening. Both the short and the long songs were heard, as well as chipping, which is used by A. s. pratensis to stay in touch while foraging (Vickery 1996). As I did not locate any nests, I do not know if the long song indicates mated status, as it usually does in A. s. pratensis. No singing was heard when the temperatures soared to 32ºC, usually between noon and mid-afternoon. Evening singing was typical of moonlit nights, lasting sometimes until dawn. The birds responded to playbacks of the songs of A. s. pratensis in a similar way as this northern subspecies during the breeding season.
Interspecific relations: The only potential avian predator I encountered was the crested caracara (Polyborus plancus). No mammals or reptiles were ever observed on the open savanna, although the presence of large felids (Felis concolor and F. onca) was detected by their calls and footprints at the boundary between the savanna and the forest patches. At least during this season no other passerine occupied areas of identical vegetation characteristics. In central North America, savanna sparrows (Passerculus sandwichensis) would compete with grasshopper sparrows for singing perches and respond aggressively to playbacks of their songs (pers. obs.). This type of relationship was not observed between the Nicaragua birds and any other species in the savanna. Chipping sparrows (Spizella passerina pinetorium) were found infrequently, always in small flocks (4-5 individuals) and only on pine trees, not descending to the ground. The more common rusty sparrow (Aimophila rufescens discolor) occupied wet areas with dense brush. Red crossbills (Loxia curvirostra), Botteri's sparrows (Aimophila botterii) and black-headed siskins (Carduelis notata) were each encountered only once, all in dense vegetation not used by grasshopper sparrows.
I observed single birds feeding on the ground apparently on ants. These individuals did not flush when approached, but continued walking and foraging, staying at a distance of 3-4 m. The stomach contents of a single net loss included grass seeds, insect parts and large amounts of grit. By the end of April, small groups (young and adults) were observed feeding together on the pine trees at an average height of 3 m.
Discussion
As Monroe (1968) suggested for the Honduran Miskitia, and based on the number of individuals I encountered, grasshopper sparrows may be the dominant species in the Nicaraguan Miskitia. Though very local in their distribution, they were abundant in the new-growth habitat. The difference between my observations and those of Howell's in the late 1960's may be explained in part by recent environmental changes, especially widespread burning during the Contra War which might have expanded the birds' previous range. It is also possible that Howell's results were an artifact of his research design in which he studied intensively and almost exclusively four particular pine savanna plots, all of them in the northwestern part of the savanna (Howell, 1971).
The permanent presence of grasshopper sparrows near Miskito villages was confirmed by their inhabitants. Fire used to clear up the brush and provide fodder for cattle may also be providing the sparrows with appropriate habitat. Miskitos do not practice fire control but expect the fire to burn itself out, except when the winds drive it toward the village. Fires that spread into more distant areas are responsible for large tracts of burnt savanna as in Warnata and Leimus Plains. In these areas, the large size of grasshopper sparrow territories and their weak territorial behavior may be the result of sudden, extensive opening of new appropriate habitat.
Ridgely and Gwynne (1989) and Ridgely and Tudor (1989) relate the changes in grasshopper sparrow abundance in Central and South America to the introduction of intensive agricultural systems which include frequent mowing and an increase in the use of pesticides. At present, the status of the populations of this bird is predominantly unknown. In Panama they have not been seen since the seventies, and they are considered extinct (N. Smith, pers. comm.). Strict fire protection measures in the Guanacaste National Park of Costa Rica (R. Blanco, pers. comm.) intended to promote the regeneration of the dry forest, may be threatening the persistence of one of the two populations found in the country (Stiles and Skutch 1989).
The Nicaraguan pine savanna is considered faunistically depauperate (Howell 1972). As such it is not listed as an area that requires conservation action (Wege and Long 1995, Mittermeier et al. 1998). The Nicaraguan pine savanna has no agricultural potential except for pine. The expansion of the P. caribea plantations may pose a moderate threat to grasshopper sparrows if fire protection measures are effective. In northeastern Nicaragua most of the pine savanna is formally under the control of traditional Miskito rule. As long as their fire practices continue, the bird's persistence may be ensured in this part of their range.
Acknowledgments
Special thanks to J. C. Martinez for his help with the logistics of travel in northeastern Nicaragua, and P. Ortiz and R. Cruz-Lazo for assistance in the field. Thanks also to the Costa Rica Ambassador in Managua for his timely assistance. Support was provided by two anonymous contributors, and The Ohio State University through The Office of International Education, The Graduate School Alumni Association, and Parker Lab.
Resumen
En marzo y abril de 1996, llevé a cabo observaciones del semillero colicorto (Ammodramus savannarum cracens) en las sabanas de pino del noreste de Nicaragua. Encontré individuos aislados en las depresiones más húmedas, pero las poblaciones en estado reproductivo ocupaban solamente áreas que hubieran sido quemadas recientemente. El comportamiento territorial de las aves parecía estar relacionado al tamaño del territorio: las aves más agresivas defendían territorios relativamente pequeños, cercanos a los poblados miskitos, que es adonde los fuegos se producen con mayor frequencia. Fue notable la ausencia de posibles depredadores en las áreas más abiertas de la savanna. Contrario a lo que sucede en otras partes de Centroamérica, la persistencia de esta ave en las savannas de pino de Nicaragua podría estar asegurada por las tradiciones miskitas en el uso del fuego.
References
Askins, R. A. 1993. Population trends in grassland, shrubland, and forest birds in eastern North America.Curr. Ornithol. 11: 1-34. [ Links ]
Bock, C. E. & B. Webb. 1984. Birds as grazing indicators in southeastern Arizona. J. Wildl. Manage. 48: 1045-1049. [ Links ]
Bollinger, E. K. 1996. Successional changes and habitat selection in hayfield bird communities. Auk 112: 720-730. [ Links ]
Bond, J. 1961. Birds of the West Indies. Houghton Mifflin, Boston, Massachusetts. [ Links ]
Delany, M., H. Stevenson & R. McCracken. 1985. Distribution, abundance and habitat of the Florida grasshopper sparrow. J. Wildl. Manage. 49: 626-631. [ Links ]
Herkert, J. R. 1994a. The effects of habitat fragmentation on Midwestern grassland bird communities. J. Ecol. Appl. 4: 461-471. [ Links ]
Herkert, J. R. 1994b. Breeding bird communities on Midwestern prairie fragments: the effects of prescribed burning and habitat-area. Natur. Areas J. 14: 128-135. [ Links ]
Howell, T. R. 1966. Precise location of Leicus Creek, Nicaragua, as a type locality. Auk 83: 665. [ Links ]
Howell, T. R. 1971. An ecological study of the birds of the lowland pine savanna and adjacent rain forest in northeastern Nicaragua. [ Links ]
Living Bird, Tenth Annual, Cornell University, Ithaca, New York, p. 185-242. [ Links ]
Howell, T. R. 1972. Birds of the lowland pine savanna of northeastern Nicaragua. Condor 74: 316-340. [ Links ]
Monroe, B. L., Jr. 1968. A Distributional Survey of the Birds of Honduras. Ornithol. Monogr. 7. Amer. Ornithol. Union, Washington, D. C. [ Links ]
Monson, G. & A. R. Phillips. 1981. Annotated checklist of the birds of Arizona. University of Arizona, Tucson, Arizona. [ Links ]
Munro, N. 1966. The fire ecology of Caribbean pine in Nicaragua. Proc. 5th Annu. Tall Timbers Fire Ecol. Conf., Seattle, Washington, p. 67-83. [ Links ]
Ridgely, R. S. & J. A. Gwynne, Jr. 1989. A Guide to the Birds of Panama, with Costa Rica, Nicaragua and Honduras. Princeton University, New Jersey. [ Links ]
Ridgely, R. S. & G. Tudor. 1989. The Birds of South America. Volume 1: The Oscine Passerines. University of Texas, Austin, Texas. [ Links ]
Small, A. 1994. California birds: their status and distribution. Ibis, Vista, California. [ Links ]
Stiles, F. G. & A. F. Skutch 1989. A guide to the birds of Costa Rica. Cornell University, Ithaca, New York. [ Links ]
Vickery, P. 1996. Grasshopper sparrow (Ammodramus savannarum). The birds of North America, 239. Amer. Ornithol. Union and Acad. Natur. Sci. Philadelphia,Philadelphia, Pennsylvania. [ Links ]
Wege, D. C. & A. J. Long. 1995. Key areas for threatened birds in the Neotropics. BirdLife Conservation Series 5, BirdLife International, Burlington, Cambridge. [ Links ]
Whitmore, R. C. 1979. Short-term change in vegetation structure and its effects on grasshopper sparrows in West Virginia. Auk 96: 621-625. [ Links ]
Whitmore, RC. 1981. Structural characteristics of grasshopper sparrow habitat. J. Wildl. Manage. 45: 811-814. [ Links ]
1Department of Evolution, Ecology and Organismal Biology, The Ohio State University, 104 Botany and Zoology Building, 1735 Neil Avenue, Columbus, Ohio 43210. Fax: 614-292-2030; arguedas.2@osu.edu