Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.49 n.1 San José Mar. 2001
Eric N. Smith 1 and Brice P. Noonan 1
Received 6-X-1999. Corrected 6-VI-2000. Accepted 19-IX-2000.
Abstract
A new member of the genus Osteocephalus is described from the Pakaraima mountains of western Guyana. This species is the smallest known member of the genus and is probably closely related to O. subtilis. Both share a small size (less than 40 mm snout-vent length), large and bulgy eyes directed somewhat rostrally, green bones, smooth and brownish dorsal skin, relatively short and truncate snout, small tympanum, subgular and laterally expanded vocal sac, poorly developed subarticular and supernumerary tubercles, a supra-anal glandular ridge, and cream-white venter and subocular region. The new species can be distinguished from O. subtilis by the Buff iris (vs black), smaller overall size (32.7 vs 35.8–38.8 mm snout-vent length), relatively larger toe disks, and less developed foot webbing. The cranium of the new species is well ossified, relatively reduced in width between the orbits, without an exposed frontoparietal fontanelle and with the anterior arm of the squamosal extending to about half the distance to the maxillary. The vocal sac is subgularly poorly developed and possess lateral extensions to the area behind the jaw angles. Well developed supraocular and suprasquamosal cartilages give support to the enlarged eyes of this species.
Key words: Anura, Hylidae, Osteocephalus, New species, Guyana, Osteology, Myology.
Recent herpetological exploration into the interior mountains of Guyana by the junior author has lead to the collection of several species of Osteocephalus, namely O. buckleyi, O. cabrerai, O. leprieurii, O. taurinus, and a very distinctive undescribed species. The description of the later species is the main purpose of this paper. These represent all species of Osteocephalus currently known to us to inhabit Guyana, and with O. oophagus all of those presently known from the Guiana Shield (O. ayarzaguenai Gorsula and Señaris (1996) from the Gran Sabana, Venezuela is considered a junior synonym of O. leprieurii for reasons stated by E. N. Smith et al. , unpublished). We report for the first time O. cabrerai from Guyana. O. cabrerai was placed in the synonymy of O. buckleyi by Trueb and Duellman (1971), later O. cabrerai was resurrected by Duellman and Mendelson (1995). Originally, O. cabrerai was described based on two specimens from Colombia and one from Amapá Territory, eastern Brazil (Cochran and Goin 1970). Gorzula and Señaris (1998) report this species from the Amacuro Delta in Venezuela. Based on these distributional data it is not surprising to find this species in Guyana, where it was probably confused with O. buckleyi.
Although the junior author only collected a single specimen of the undescribed species, the fact that other trips to the collecting locality have yielded no more specimens and the uniqueness of this species when compared to other Osteocephalus leds us to describe it herein. This species is presumably closely related to the large eyed and small bodied O. subtilis Martins and Cardoso (1987) from Acre, Brazil, approximately 2 000 km to the southwest. We also describe the cranium and vocal sac structure of the new species.
Materials and methods
Measurements of tympanum and eye diameter, head, foot, tibia, and snout-vent lengths (SVL) follow Duellman (1970); measurements of head width and inter-orbital distance follow Lynch and Duellman (1980), webbing notation follows Savage and Heyer (1967), as modified by Myers and Duellman (1982), and toe and finger terminology is that of Savage (1987). The following measurements were also included: internarial distancedistance between medial borders of nares; snout lengthdistance from tip of snout to anterior margin of eye; orbit-jaw distanceshortest distance between deepest region below eye and margin of upper jaw; naris-jaw distanceshortest distance between naris and margin of upper jaw; choana widthmaximum width of choana; femur lengthdistance from tip of urostyle to distal end of femur; arm lengthdistance from proximal end of radio-ulna to proximal base of pollex; hand lengthlength from proximal base of pollex to tip of third finger; interfascicular distanceshortest distance between dentigerous fascicles; and tarsal lengthdistance from proximal border of inner metatarsal tubercle to proximal end of tibiale. The widths of the pads of the third finger and the fourth toe were also measured. Lengths were recorded from the right side of each specimen, unless the specimen was damaged or abnormal on that side. Measurements greater than 10 mm were taken to the nearest 0.1 mm with digital calipers. Those less than 10 mm were taken using a stereo microscope with an ocular micrometer.
The skin on the head of the holotype herein described was carefully peeled back in order to examine the underlying musculature. After jaw musculature examination the skull was removed by cutting at the level between the occipital condyles and the atlas. The head skin was then returned to its original position. The removed cranium was cleared and double stained according to the Kansas University Museum of Herpetology modification of the procedure of Dingerkus and Uhler [1977] revised by Taylor and Van Dyke (1985). Jaw musculature terminology is that of Tyler (1971). Cranial descriptions follow the format of Trueb and Duellman (1970) and the terminology of Trueb (1970).
Specimens examined are listed in Appendix I. Museum abbreviations follow Leviton et al. (1985) or are defined in the text, referred specimens are presented with their field catalogue number.
Color descriptions of live specimens were made based on Kodachrome color transparencies deposited at The University of Texas at Arlington (UTA) and the Kansas University (KU) herpetological slide collections (Appendix I). Colors are taken from the Naturalist's Color Guide (Smithe 1975). Colors taken from this color guide, in the case of slide-descriptions, are presented in capital letters, others are not. Color descriptions in the diagnosis refer to live specimens. The number in parentheses after a color first appears in a paragraph refers to that of the color swatch in the guide.
All specimens examined were stored in 70% ethanol. Most were probably fixed in the field with a 10% formalin solution and later transferred to ethanol, as was done for the holotype of the species described herein. The sex of preserved specimens was determined by the presence of prepollical nuptial excrescences in adult males and their absence in adult females. Sex of young individuals was determined by examining gonads. Gonad development was observed to verify maturity in seemingly small adults.
DESCRIPTION
Osteocephalus exophthalmus sp. nov.
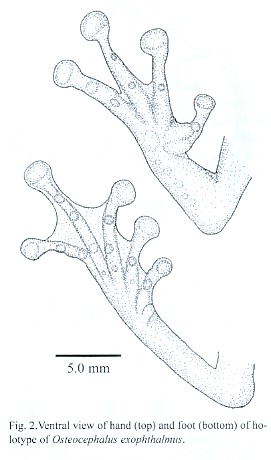
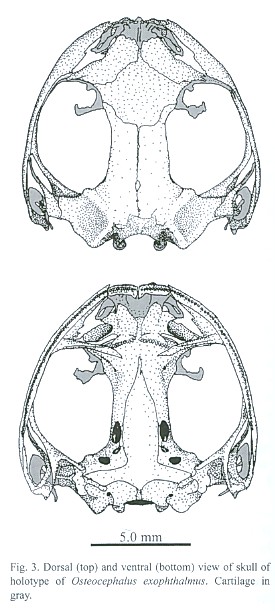
(Figs. 1, 2, 3)
Holotype: The University of Guyana Center for the Study of Biological Diversity UG/CSBD HA-722, (cranium, The University of Texas at Arlington UTA A-51620) (Original number BPN 166), adult male collected at ca 30 km SE Imbaimadai, Mazaruni-Potaro District, Guyana, ca. 585 m, on 10 June 1997 by Brice P. Noonan and Daniel W. Carpenter, approximately 5°37'30'' N, 60°14'42'' W.
Diagnosis: The smallest known member of the genus Osteocephalus (32.7 mm, SVL of only known specimen). Eyes large and bulgy, directed rostrally. Iris Buff (24) with black markings, somewhat forming a cross with the pupil as the center. Male with poorly developed vocal sac that extends as a tube of thin muscle and fascia from the throat to the area behind and slightly above the angle of the jaw, where it is more muscular. The skin associated with the vocal sac is unmodified and the well developed vocal slits are located near the angle of the jaw. Osteocephalus exophthalmus is probably closely related to O. subtilis, with whom it shares a small size (less than 40 mm SVL), large and bulgy eyes directed somewhat rostrally, green bones, smooth and brownish dorsal skin, relatively short and truncate snout, small tympanum, subgular and laterally expanded vocal sac, poorly developed subarticular and supernumerary tubercles, a supra-anal glandular ridge, and cream-white venter and subocular region. The new species can be distinguished from O. subtilis (comparison in parentheses, pad ratio and webbing formulae taken from illustration in Martins and Cardoso, 1987) by its Buff iris with a black cross (vs. black), smaller overall size (32.7 vs. 35.8–38.8 mm SVL), relatively larger toe pads (0.9 vs. 0.6, pad width of Toe IV/pad width of Finger III), and less developed hand and foot webbing (I 21/3–21/2 II 12/3-3 III 22-3–22-3 IV, I 11/2–2 II 11/2–2 III 11/2–2 IV 2–11/2 V vs. I 2–21/3 II 11/2–21/3 III 21/2–2 IV, I 1–12/3 II 1–11/2 III 1–11/2 IV 11/3–1 V). Male O. exophthalmus can easily be distinguished from all other adult members of the genus by its smaller size. Among the smaller bodied Osteocephalus, the SVL of the smallest adult O. buckleyi known to us is 34.3 mm, the smallest O. cabrerai 38.9 mm, the smallest O. subtilis 35.8 mm (Martins and Cardoso 1987), the smallest O. oophagus 35.7 (Jungfer and Schiesari 1995), and the smallest O. sp. from Colombia 34.1 mm (member of the verruciger group; E. N. Smith and W. W. Lamar, unpublished). From all other species of Osteocephalus, except O. subtilis, the new species can also be distinguished by its large and bulgy eyes directed rostrally and by the black posterior area of its thighs. Osteocephalus exophthalmus is unique within the genus in possessing a Buff colored iris with a dark cross. In the mountains of Western Guyana O. exophthalmus could be confused with Tepuihyla talbergae, from which it differs in possessing large and bulgy eyes, more vomerine teeth (21 vs. 12, total), ulnar tubercles, and lacking spiculate tubercles on dorsum, a bifid subarticular tubercle on Finger IV, a bifid and small palmar tubercle, large and numerous cloacal tubercles, and a pale subocular area instead of a well developed labial stripe.

Description: Measurements in mm: SVL 32.7, head length 12.5, head width 12.0, eye diameter 4.6, snout length 5.5, internarial distance 2.9, naris-jaw distance 2.0, orbit-jaw distance 0.8, choana width 1.2, tympanum diameter 2.3, interfascicular distance 0.0, tarsal length 9.2, hand length 11.0, arm length 6.7, tibia length 17.3, foot length 14.3, femur length 16.3, IOD 3.0, pad width of Finger III 2.0, pad width of Toe IV 1.8.
Head broader than body, about as long as wide; head length 104 % head width; head width 37 % SVL; snout length 46 % head width, somewhat pointed in dorsal view, nearly truncate in lateral profile; no rostral keel; exostosed dermal roofing bones on skull absent; canthus rostralis somewhat straight, elevated, curved anteriorly; loreal area straight, with scattered small tubercles; inter-orbital distance 25 % head width; internarial distance 24% head width; nostrils slightly protuberant laterally; internarial area depressed; lips thin, straight, not flared; upper eyelid bearing few small tubercles; tympanum distinct, 51 % eye diameter, with a moderately developed and tuberculate supratympanic fold covering its upper edge; no cranial crests; dentigerous processes bearing 11/10 (left/right) teeth, curved and in contact, between small elliptical choanae; vocal slits relatively large and opened, towards angle of jaw; vocal sac beginning medially on throat, continuing laterally as a tube, and expanding behind angle of jaw, associated skin unmodified; tongue lanceolated, wider anteriorly, longer than wide, anteriorly notched.
Skin of dorsal surfaces smooth with very few, scattered, tiny tubercles; skin on throat, belly, and proximal posteroventral surfaces of thighs granular, other ventral areas smooth; flanks smooth, slightly areolate anteriorly and with a few rounded tubercles on upper area; anal opening on upper level of thighs, directed posteroventraly, with short flap and dorsal low tubercular ridge.
Arm length 20 % SVL; hand length 34 % SVL; upper arm slender; forearm moderately robust; no axillary membrane; fingers bearing large, expanded disks; several large and small rounded tubercles present in ulnar region, aligned; pollical tubercle oval; prepollex enlarged, with keratinized spinous nuptial excrescence; subarticular tubercles large, globular or very slightly obtuse in lateral profile, rounded to ovoid in basal outline, Fingers I and II with one, Fingers III and IV with two; palmar tubercle indistinct; accessory palmar tubercles small, globular in lateral profile, rounded in basal outline, in rows, few, Finger I with none, Finger II with 1, Finger III with 1 or 2, Finger IV with 2; Finger III pad width 86 % tympanum diameter. Hand webbing formula I 21/3–21/2 II 12/3–3 III 22/3–22/3 IV. (Fig. 2)
Hind limbs slender, moderately long; femur length 50 % SVL; tibia length 53 % SVL; tarsal length 28 % SVL; foot length 44 % SVL; heels with few very low and rounded tubercles or no tubercles, overlapping when flexed; with few ventrolateral tarsal tubercles; inner metatarsal tubercle elongate and much larger than rounded outer metatarsal tubercle; subarticular tubercles globular in lateral profile, round in basal outline, Toes I and II with one, Toes III and V with two, Toe IV with three; fingers elongated; plantar tubercles globular in lateral profile, rounded in basal outline, very small, Toe I and V with none, Toe II and IV with 1, Toe III with 2; Toe IV disk width 79 % tympanum diameter; toe disks slightly smaller than those of fingers. Foot webbing formula I 11/2–2 II 11/2–2 III 11/2–2 IV 2–11/2 V. (Fig. 2)
In preservative, dorsum Drab-Gray (119D) with Prout's Brown (121A) suffusions and Vandyke Brown (121) blotches, "Y" shaped blotch with arms above eyes and base above insertion of head, one above scapular area, one above mid-trunk, and one or two above upper sides of lower-trunk; area below eye, Drab-Gray with few and small Vandyke Brown markings; whitish flank areolation and tubercles (behind tympanum, on lower arm, along outer tarsum, and on heels); tympanum transparent with dark peppering; Dusky Brown (19) coloration in area below canthus rostralis and from posterior corner of eye, bordering tympanum, along flanks, to area before insertion of hind limbs; dorsal bars Vandyke Brown, two on lower arm, three on upper leg, three on lower leg, two on tarsal area and a few diffused on outer foot; posterior and anterior femoral areas unicolor, Fuscous (21) anteriorly, Vandyke Brown posteriorly; Dusky Brown anal patch; throat Drab-Gray (119D) with Vandyke Brown spotting; Pale Horn Color (92) on chest, belly, and venter of arms and upper leg; lower leg Pearl Gray (81) below; some fine Vandyke Brown pebbling on venter of legs and on chest area near insertion of limbs.
Color in life: Light Buff (124) dorsum with suffusions of Raw Sienna (136); loreal area Raw Sienna; area below canthus rostralis and dorsal blotches and bars Vandyke Brown, somewhat dark edged; flanks Jet Black (89) with light Buff areolations; anterior area of insertion of limbs Jet Black; iris dark Buff with Jet Black cross radiating from black pupil; area below eye cream-white with small Vandyke Brown markings; Jet Black coloration behind posterior corner of eye, bordering transparent tympanum, and continuing to area of insertion of forelimbs; upper area of lower arms dark Buff (24); hand webbing transparent; anterior area of thighs, foot webbing, and anal patch Sepia (119), almost black; white coloration surrounding anal patch, including tubercles, and extending as lines of posterodorsal reticulations to about mid thigh; posterior area of thighs Jet Black; axillary and groin areas Pearl Gray (81); throat white with Sepia (119) spotting, stronger on sides; chest, belly, and arm ventral areas Drab-Gray (119D); chest with fine Natal Brown (219A) spotting on sides, near insertion of limbs; palms Olive Brown (28); ventral surface of thighs Olive Brown (28); ventral shank and tarsal areas Pearl Gray; sole of feet Hair Brown (119A); jaw and hind limb bones Emerald Green (163).
Cranium: (Fig. 3) Skull slightly broader than long; snout somewhat rounded in dorsal view; dorsal surfaces of skull mostly smooth; dorsolateral surface of nasals very slightly exostosed with an irregular reticulate pattern; overlying skin on dorsum of head not co-ossified. Prenasal, internasal, and dermal sphenethmoid absent. Septomaxillaries type II (Trueb 1970). Anterior supraorbital margins of frontoparietals barely upturned, forming an indistinct crest; posterior supraorbital margin somewhat upturned; frontoparietals extending over dorsal area of prootic to level of epiotic eminence; anterior arm of squamosal extending about half distance to maxillary.
Premaxillaries laterally and medially separated by connective tissue from other bones; pars palatina weakly developed and smooth; alary processes slightly rounded and directed somewhat posterodorsaly, slightly more than three and a half times depth of pars dentalis; palatine process conspicuous, slightly longer than width of pars dentalis. Prevomers not converging medially; ossified area just behind palatine process; anterior ends somewhat rounded and bluntly serrated; dentigerous processes very slightly rounded and directed posterolaterally, slanted backwards in relation to main axis of cranium; lateral wings moderately developed, bordering choanae, lateral rami pointed and smooth anterolaterally, posterolateral rami slender, four fifths length to neopalatines (Trueb and Cloutier 1991, Trueb 1993), and synchondrotic to sphenethmoid and neopalatines. Neopalatines narrow, thin, forming posterior margins of choanae, each with an elongate and conspicuous ventral ridge situated anteriorly and roundly directed posteroventraly; proximal ends pointed and lying on anterior ventrolateral extensions of sphenethmoid; distal ends somewhat expanded and rounded, lying dorsomedial to pars dentalis and pars palatina of maxillary.
Nasals moderate in size; anteriorly well behind premaxillaries; almost touching medially; one fourth overlapping and contacting sphenethmoid; canthal area rounded; maxillary process directed posterolateral to neopalatines and caudal to posterior process of pars facialis of maxillary. Maxillary with a high and well developed pars facialis anterior to orbit; pars facialis with two or three serrations at mid-length; pars facialis articulating dorsomedially only with neopalatines; pars palatina small throughout its length; maxillary firmly articulating with quadratojugal posteriorly and to pterygoid medially.
Sphenethmoid well ossified; anterior terminus lying well behind anterior margins of nasals; margins of orbitonasal foramina bony; posterior border of bony sphenethmoid laterally and ventrally reaching level of anterior third of optic foramina, dorsomedially reaching area above foramen magnum; overlain by frontoparietals dorsally; bony anterodorsal and lateral surfaces exposed; cartilaginous ventral element without alae over parasphenoid alae and extending caudally horseshoe-shaped with its rounded apex reaching level between columellae. Frontoparietals thin; anterior margins extending laterally over dorsolateral optic margin of sphenethmoid, but well behind posterior margin of nasals; in contact medially; posterolaterally in contact with epiotic eminences; not elevated posteriorly to form crest.
Parasphenoid without odontoids at level of optic foramen; parasphenoid alae directed posterolaterally, distal truncation almost at 60° angle from main axis of cranium; posterior end pointed and ending anterior to jugular foramina. Squamosal delicate; anterior arm slender; posterior arm three fifths length of anterior arm, medial ala overlying dorsolateral surface of crista parotica; ventral arm well developed, distally articulating with quadratojugal laterally and posterior ramus of pterygoid medially. Pterygoid relatively robust; anterior ramus long, anterior terminus lying at about midlevel of orbit; medial ramus long, synchondrotic to anteroventral corner of otic capsule; posterior ramus articulating with ventral half of ventral arm of squamosal.
Prootics and exoccipitals fused; entire posterior end of skull well ossified; optic foramina ventrally marginated by cartilage; oculomotor foramina marginated anteroventraly by cartilage; prootic and jugular foramina with bony margins; with small bony knob formed below epiotic eminences above occipital condyles. Crista parotica well developed, dorsally in bony sutural contact with squamosal, laterally synchondrotic; pars externa plectri and pars ascendens plectri cartilaginous; pars media plectri bony.
Superficial throat musculature and vocal sac structure: As in other Osteocephalus, the submentalis is of moderate size and araphic. The intermandibularis is not differentiated but is divided medially by an elongate aponeurosis. The intermandibularis and submentalis are in contact medially; the medial aponeurosis of the intermandibularis occupies about one-fourth of the area at mid-throat and tappers medially towards the submentalis and caudally towards the area between the jaw angles; the interhyoideus, which forms the vocal sac, is poorly developed ventrally, forms a very small but muscularly developed sac supramandibularly, and expands somewhat before it attaches to the dorsal fasciae. The supramandibular skin associated with the interhyoideus is not modified. This skin possesses no direct muscular attachments of the interhyoideus. The geniohyoideus medialis is differentiated from the geniohyoideus lateralis; the two muscles are joined by a slender, short, and triangular aponeurosis anteriorly and a raphe medially and posteriorly. The geniohyoideus medialis is, throughout its length, about half the width of the geniohyoideus lateralis at mid throat, at the level of the episternum it is one-third the width of this structure; the geniohyoideus lateralis arises from the mandible next to the attachment of the submentalis and at mid-throat occupies almost one-half of the area between the geniohyoideus medialis and the mentomeckelian bone, it widens from rostrum towards intermandibular region.
Etymology: The specific epithet alludes to the huge and protruding nature of the eyes of this novel species.
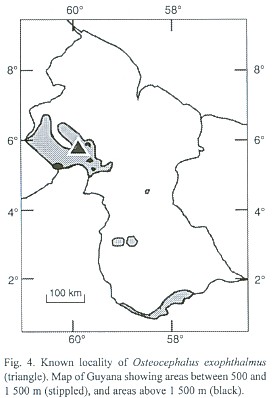
Distribution and natural history: Osteocephalus exophthalmus is known only from approximately 585 m in the mountain slope of the Tepui S of Imbaimadai, Guyana (Fig. 4). This slope of the Pakaraima Mountains is drained by the Mazaruni River. The vegetation formation of the area is classified as Tall/medium, evergreen, lower montane sclerophyllous forest (Huber et al. 1995). The only known specimen, an adult male with well developed testes and nuptial pads, was found active at night, 20:00 hr, on a small plant at approximately 1 m above a pool of water.
Relationships: Osteocephalus exophthalmus is apparently closely related to O. subtilis. The characters that best support this relationship are mentioned in the diagnosis. From the members of the genus Tepuihyla, a recently described genus probably closely related to Osteocephalus (Ayarzagueña et al. 1992a, 1992b, Duellman and Yoshpa 1996), it differs in possessing more webbing on hands and feet, large and protruding eyes, a longer anterior arm of the squamosal (extending about half distance to maxillary), and lacking an exposed frontoparietal fontanelle. The large and bulgy eyes of O. exophthalmus possess some cranial support that is more prominent than in other Osteocephalus, except probably O. subtilis. The longer anterior arm of the squamosal apparently gives extra support to the posterior area of the eye, dorsaly aided in part by a more developed associated cartilage. There is also a well developed supraocular cartilage with a relatively large and protruding extra process anterior to the main supraocular cartilaginous process (Fig. 3). The width of the interorbital bone area is also relatively reduced.
Acknowledgments
Special thanks to M. Tammessar, J. Singh and D. Nariane of the Centre for the Study of Biodiversity, University of Guyana, Georgetown, for their assistance in obtaining the necessary permits to conduct scientific explorations in Guyana, and to the North Texas Herpetological Society for providing partial funding for this expedition. We are grateful to Daniel W. Carpenter for his invaluable field assistance, and W. W Lamar for his helpful comments on color variation on frogs of the genus Osteocephalus. We thank several museum curators and directors who have made available several Osteocephalus specimens to us, namely, L. Trueb and J. Simmons (Kansas University), S. O. Kullander (Naturhistoriska Riksmuseet, Stockholm), J. V. Vindum (California Academy of Sciences), A. Resetar (Field Museum of Natural History), T. Daeschler (Academy of Natural Sciences, Philadelphia), D. Frost (American Museum of Natural History), R. P. Reynolds and R. W. McDiarmid (United States National Museum), J. E. Cadle (Museum of Comparative Zoology), and G. Nilson (Naturhistoriska Museet, Göteborg). William E. Duellman, C. F. Franklin, M. B. Harvey, and W. W. Lamar generously provided photographs of live specimens. Carol Stewart (The University of Texas at Arlington) kindly helped receiving and returning loaned material. Karin S. Castañeda (The University of Texas at Arlington) expertly created several of the figures.
Resumen
Un nuevo miembro del género Osteocephalus se describe de las montañas Pakaraima del oeste de Guyana. Es la especie más pequeña del género y probablemente esté estrechamente relacionada con O. subtilis. Ambas especies comparten un tamaño pequeño (menos de 40 mm SVL), ojos grandes y sobresalientes, dirigidos hacia el rostro, huesos verdes, piel dorsal lisa, de tono castaño, hocico relativamente truncado y corto, tímpano pequeño, saco vocal subgular y expandido lateralmente, tubérculos subarticulares y supernumerarios poco desarrollados, pliegue supra-cloacal glandular, vientre y área subocular de color blanco-crema. La nueva especie se distingue de O. subtilis por su iris castaño-mostaza (negro en O. subtilis), tamaño menor (32.7 contra 35.8–38.8 mm SVL), discos de los dedos del pie relativamente más grandes, y membranas del pie menos desarrolladas. El cráneo de la nueva especie está bién osificado, relativamente reducido en ancho entre las orbitas de los ojos, sin una ventana frontoparietal expuesta, y con el brazo anterior del escuamoso extendiendose hasta cerca de la mitad de la distancia al maxilar. El saco vocal está poco desarrollado en el área subgular, y posee extensiones laterales hacia el área detrás del ángulo mandibular. Cartílagos supraoculares y supraescuamosales dan soporte a los grandes ojos de esta especie.
References
Ayarzagüeña, J., J. C. Señaris & S. Gorzula. 1992a. El grupo Osteocephalus rodriguezi de las tierras altas de la Guyana Venezolana: Descripción de cinco nuevas especies. Mem. Soc. Cien. Nat. La Salle 52: 113–142. [ Links ]
Ayarzagüeña, J., J. C. Señaris & S. Gorzula. 1992b. Un nuevo género para las especies del "grupo Osteocephalus rodriguezi" (Anura: Hylidae). Mem. Soc. Cien. Nat. La Salle 52: 213–221. [ Links ]
Cochran, D. M. & C. J. Goin. 1970. Frogs of Colombia. Bull. U.S. Natl. Mus. 288: xii+655. [ Links ]
Dingerkus, G. & L. D. Uhler. 1977. Enzyme clearing of alcian blue stained whole small vertebrates for demonstration of cartilage. J. Stain Technology 52: 227–32. [ Links ]
Duellman, W. E. 1970. Hylid frogs of Middle America. Monog. Mus. Nat. Hist. Univ. Kansas 1: 1–753. [ Links ]
Duellman, W. E. & J. R. Mendelson III. 1995. Amphibians and reptiles from northern Departamento Loreto, Peru: Taxonomy and biogeography. Univ. Kansas Sci. Bull. 55: 329–376. [ Links ]
Duellman, W. E. & M. Yoshpa. 1996. A new species of Tepuihyla (Anura: Hylidae) from Guyana. Herpetologica 52: 275–281. [ Links ]
Gorsula, S. & J. C. Señaris. 1996. Una nueva especie del género Osteocephalus (Anura: Hylidae) de la Gran Sabana, Venezuela. Acta Biol. Venez. 16: 19–22. [ Links ]
Gorzula, S. & J. C. Señaris. 1998. Contribution to the herpetofauna of the Venezuelan Guayana I. A data base. Scientia Guaianae 8: xviii+1–270+32. [ Links ]
Huber, O., G. Gharbarran & V. Funk. 1995. Vegetation Map of Guyana (Preliminary Version). Centre for the Study of Biological Diversity, University of Guyana, Georgetown. Lanman Co., Washington D. C.
Jungfer, K. -H. & L. C. Schiesari. 1995. Description of a central Amazonian and Guianan tree frog, genus Osteocephalus (Anura: Hylidae), with oophagus tadpoles. Alytes 13: 1–13. [ Links ]
Leviton, A. E., R. H. Gibbs, Junior, E. Heal & C. E. Dawson. 1985. Standards in herpetology and ichthyology: Part I. Standard symbolic codes for institutional resource collections in herpetology and ichthyology. Copeia 1985: 802–832.
Lynch, J. D. & W. E. Duellman. 1980. The Eleutherodactylus of the Amazonian slopes of the Ecuadorian Andes (Anura: Leptodactylidae). Misc. Publ. Univ. Kansas Mus. Nat. Hist. 69: 1–86. [ Links ]
Martins, M. & A. J. Cardoso. 1987. Novas espécies de hilídeos do Estado do Acre (Amphibia: Anura). Rev. Brasil. Biol. 47: 549558. [ Links ]
Myers, C. W. & W. E. Duellman. 1982. A new species of Hyla from Cerro Colorado, and other tree frog records and geographical notes from western Panama. Am. Mus. Novit. 2752: 1–32. [ Links ]
Savage, J. M. 1987. Systematics and distribution of the Mexican and Central American rainfrogs of the Eleutherodactylus gollmeri group (Amphibia: Leptodactylidae). Fieldiana Zool., New. Ser. 33: 1–57.
Savage, J. M. & W. R. Heyer. 1967. Variation and distribution in the tree-frog genus Phyllomedusa in Costa Rica, Central America. Beitr. Neotrop. Fauna 5: 111–131. [ Links ]
Smithe, F. B. 1975. Naturalist's color guide. The American Museum of Natural History. New York. 16 p. [ Links ]
Taylor, W. R. & G. C. Van Dyke. 1985. Revised procedures for staining and clearing small fishes and other vertebrates for bone and cartilage study. Cybium 9: 107–119. [ Links ]
Trueb, L. 1970. Evolutionary relationships of casque-headed treefrogs with co-ossified skulls (family Hylidae). Univ. Kansas Publ. Mus. Nat. Hist. 18: 547716. [ Links ]
Trueb, L. 1993. Patterns of cranial diversity among the Lissamphibia, p. 255–343. In J. Hanken & B. K. Hall (eds). The Skull. Vol. 2. Patterns of Structural and Systematic Diversity. University of Chicago Press, Chicago & London.
Trueb, L. & R. Cloutier. 1991. A phylogenetic investigation of the inter- and intrarelationships of the Lissamphibia (Amphibia: Temnospondyli), p. 175–188. In H. -P. Schultze & L. Trueb (eds.). Origins of the Higher Groups of Tetrapods: Controversy and Consensus. Cornell University Press, Ithaca.
Trueb, L. & W. E. Duellman. 1970. The systematic status and life history of Hyla verrucigera Werner. Copeia 1970: 601–610. [ Links ]
Trueb, L. & W. E. Duellman. 1971. A synopsis of Neotropical hylid frogs, genus Osteocephalus. Occas. Papers Mus. Nat. Hist. Univ. Kansas 1: 1–47. [ Links ]
Tyler, M. J. 1971. The phylogenetic significance of vocal sac structure in hylid frogs. Univ. Kansas Publ. Mus. Nat. Hist. 19: 319–360. [ Links ]
1 Department of Biology, The University of Texas at Arlington, Arlington, Texas 76019, USA. Fax: (817) 272-2855. E-mail: mailto: e.smith@uta.edu; bpn@exchange.uta.edu
Specimens Examined
The following is a list of specimens examined during the course of this project, many of these are members of the verrucigerous group of Osteocephalus or were used to diagnose its members, some were reidentified, or their identification verified. Photographic slides used throughout the text are incorporated to this list, where stated, these form part of The University of Texas at Arlington and Kansas University slide collections. If both specimens and slides were available to examination, the former is followed by a slide number in parenthesis. Specimens from which skulls were dissected and cleared and stained are indicated by "(C & S)". For some taxonomic rearrangements see E. N. Smith et al. (unpublished) and E. N. Smith and W. W. Lamar (unpublished).
Osteocephalus buckleyi (9).BOLIVIA: Santa Cruz: Andres Ibañes, El Potrerillo, UTA A-51633. ECUADOR: Napo: Santa Cecilia, KU 105208–9. GUYANA: Essequibo: Kartabo, AMNH A-70971. Mazaruni-Potaro: Bartica, 9.7 km S Bartica (by road), near landing strip, UTA A-51630 (C & S, UTA slide No. 23810, 23967–68), 51631 (UTA slide No. 23969–71), 51632 (UTA slide No. 23812, 23969–71). Upper Takutu: Kuyuwini Landing, AMNH A-46283, Marudi Creek, AMNH A-46233. PERU: Loreto: 1.5 km N Teniente López, KU 221926.
Osteocephalus cabrerai (8).COLOMBIA: Meta: ca 28.0 km WSW Vista Hermosa, UTA A-3883. Putumayo: Santa Rosa de Sucumbíos (Kofan Indian village), upper Río San Miguel, AMNH A-116290. ECUADOR: Pastaza: Río Pastaza: Andoas, AMNH A-79984–85. ECUADOR/PERU: Peru-Ecuador frontier: Río Pastaza, Gauche, AMNH A-79986. GUYANA: Mazaruni-Potaro: Imbaimadai, ca 0.8 km NW Mazaruni camp, UTA A-51621 (C & S, UTA slide No. 23965–66), 51622–23 (UTA slide No. 23965–66; 51623UTA slide No. 23818, 23821).
Osteocephalus carri (7).COLOMBIA: Huila: Acevedo, Río Suaza, Río Aguas Claras, near San Adolfo, 467 m, FMNH 69702 (holotype), 69709–10. Putumayo: 10.3 km W El Pepino, 1440 m, KU 169586 (KU slide No. 5813), 169587 (KU slide No. 5814–16), 169589 (KU slide No. 5817), UTA A-50690 (C & S).
Osteocephalus exophthalmus (1).GUYANA: Mazaruni-Potaro: Imbaimadai, ca 0.8 km N Mazaruni camp, UG/CSBD HA-722 (UTA A-51620, C & S skull; UTA slide No. 23814, 23965–70).
Osteocephalus leprieurii (46).COLOMBIA: No specific locality: UTA A-6715 (skeleton). Amazonas: Puerto Nariño, KU 153390. Meta: 37.6 mi WNW Mapiripán, UTA A-8449; Boca del Cabra, UTA A-8443, 8447–48; Boca del Caño Cabra, UTA A-8430, 8432–40; Caño Cabra, UTA A-3251–53 (skeletons), 8444–46; Río Guayabero and Caño Cabra, UTA A-3264 (skeleton). Vaupés: ca. Yapima, UTA A-4293; Yapima, UTA A-8457–60, 8462–64, 8465 (C & S), 8466, 8468–70. BRASIL: Acre: Tarauaca, FMNH 83247. ECUADOR: Napo: S slope, Cordillera del Due above Rio Coca, 1150 m, KU 123170. GUYANA: Essequibo: Kartabo, AMNH A-70967–68. Mazaruni-Potaro: Imbaimadai, ca 100 m W Mazaruni camp, UTA A-51618 (UTA slide No. 23837–38); Imbaimadai, Mt. NE Imbaimadai, UTA A-51619 (UTA slide No. 23831, 23972–74). PERU: Loreto: 1.5 km N Teniente López, 310 m, KU 221928–32; Quebrada Orán, ca. 5.0 km N Río Amazonas, 85 km NE Iquitos, UTA A-20923; Río Curanja, Balta, UTA A-20924.
Osteocephalus mimeticus (16).PERU: Ayacucho: La Mar, Sivia, Río Apurimac FMNH 39853. Huanuco: Río Pachitea, midway between Puerto Victoria and Puerto Inca, CAS-SU 17745. Pasco: Yaupi, KU 136312. San Martín: Lamas, W slope Abra Tangarana, 7 km NE San Juan de Pacaysapa, 1080 m, KU 212189 (KU slide No. 8367), 212190 (KU slide No. 8368); Moyobamba, Moyobamba, 860 m, KU 212188; Rioja, Río Cerranayacu, 76 km NW Rioja, 1200 m, KU 212195 (KU slide No. 8372); Rioja, Venceremos, 89 km NW Rioja, 1630 m, KU 212186 (KU slide No. 8366), 212187; Roque, NHMG Ba.ex. 469a (holotype); San Martin, Cataratas Ahuashiyacu, 14 km NE Tarapoto, 730 m, KU 212191, 212192 (KU slide No. 8369), 215699 (2, KU slide No. 8370–71); San Martin, 14 km ESE Shapaja, 360 m, KU 212193, UTA A-50692 (C & S).
Osteocephalus pearsoni (4).BOLIVIA: El Beni: upper Río Beni, below mouth of Río Mapiri, MCZ 15565, UMMZ 67464–65; Rurrenbaque, UMMZ 57533.
Osteocephalus planiceps (24).COLOMBIA: Amazonas: Puerto Nariño, KU 153387–88, 153391; Petuña, Río Loreto-yacu, KU 153389. Putumayo: Santa Rosa de Sucumbíos (Kofan Indian village), upper Río San Miguel, AMNH A-116292. Vaupés: Wacará, UTA A-4063, 8023 (C & S), 8024, 8453; Timbó, UTA A-3848, 4459, 8451; Boca del Río Ariari and Río Guaviare, Casa de Piedras, Serranía San José, UTA A-8019–20; Yapima, UTA A-8455. ECUADOR: Napo: Lago Agrio, KU 126647, 126649–51, 126655; Limón Cocha, Río Napo, KU 99208. PERU: Loreto: Madre Selva II, Río Orosa, (UTA slide No. 24040–41); Nauta, ANSP 11399 (holotype); San Jacinto, 175 m, KU 221933–36; 1.5 km N Teniente López, 310 m, KU 221937–39.
Osteocephalus taurinus (47).BOLIVIA: Santa Cruz: Buena Vista, AMNH A-33951–52, 33958; Velasco, Estancia El Refugio, UTA A-45537–38 (45537UTA slide No. 18606–07). BRASIL: Pará: Belém, KU 129866. COLOMBIA: Amazonas: Santa Lucia on Río Cotuhe, ca 1 1/2 hrs upriver from Tarapacá on Río Putumayo, AMNH A-90326–27. Meta: 37.6 km WSW Maperipán, UTA A-8441; 110.0 mi E Puerto Gaitán, UTA A-3266 (skeleton), 3288 (skeleton), 6099; 111.0 mi E Puerto Gaitán, UTA A-6093–98; Boca del Cabra, UTA A-6100; Caño Sardinata, ca. 30 km WNW (WSW) Vistahermosa, UTA A-6101; Lomalinda, UTA A-4239, 4248, 7931, 8026; Mt. Macarena, Río Duda (Río Duita), 1000 m, AMNH A-79914; Sierra de la Macarena, ca. 35.0 km WSW Vista Hermosa, peak ca. Caño Sardinata, UTA A-3516. Vaupés: Camanaus, UTA A-8475; Pirá, UTA A-3842; Timbó, UTA A-3846, 6090–92; Wacará, UTA A-3840–41, 3843; Yapima, UTA A-8476. Vichada: Corocito, UTA A-7932, 7938, 8027. ECUADOR: Napo: Lago Agrio, KU 126648, 126652. Pastaza: Region of Puyo, UTA A-45541–42. GUYANA: No specific locality: AMNH A-39637, 10665. Cuyuni: Kamakusa, AMNH A-21418–19. Mazaruni-Potaro: Imbaimadai, ca 1.0 km N Imbaimadai, Peters Mt., UTA A-51628, 51629 (C & S); Imbaimadai, ca 3.4 km N Imbaimadai, Peters Mt., UTA A-51625–27 (51625UTA slide No. 23827–28; 51626UTA slide No. 23830, 23977–79); Kartabu, 1.6 km W Kartabu Point (by road), UTA A-51624 (UTA slide No. 23823, 23975–76).
Osteocephalus verruciger (30).ECUADOR: Napo: Avila, UMMZ 90413; S slope Cordillera del Due, 1150 m, KU 123176 (KU slide No. 3158), 123177 (KU slide No. 3160), 123178–81, 123184–85, 123186 (KU slide No. 3162), 123188, UTA A-50691 (C & S); Río Azuela, 1740 m, KU 143545 (4, two photographed, KU slide No. 4222–23); Río Pacayacu, tributary of Río Cotapino, CAS-SU 13150 (holotype of O. orcesi); 16.5 km NNE Santa Rosa, 1700 m, KU 143208 (KU slide No. 4220); southeast slope Volcán Sumaco, CAS-SU 11442. Pastaza: Abitagua, CAS-SU 5067, FMNH 25791, 27619, UMMZ 90414, 92095; Mera, UMMZ 90412(4). Tungurahua: 11 km E Río Negro, 1170 m, KU 146469 (KU slide No. 4440).
Osteocephalus sp. 1 (10).BOLIVIA: Santa Cruz: Velasco, Estancia El Refugio, UTA A-45536 (UTA slide No. 18605); Velasco, Parque Noel Kempff Mercado, Serranía de Huanchaca, near Lago Caimán, ca. 250 m, UTA A-51638 (UTA slide No. 23992), 51639 (UTA slide No. 23991), 51641 (C & S), 51642; Velasco, Parque Noel Kempff Mercado, Serranía de Huanchaca, 6.4 km from Estancia Flor de Oro on trail to Lago Caimán, near Río Itenez, UTA A-51643–45; Velasco, Parque Noel Kempff Mercado, Serranía de Huanchaca, Lago Caimán, UTA A-51646.
Osteocephalus sp. 2. (28 + color notes and transparencies on 13 additional specimens).COLOMBIA: Boyacá: Sutatenza, USNM 152054–56. Cundinamarca: Medina, USNM 152089, 152090,152092–97, 152757. Meta: Serrania de la Macarena, 30 km WSW Vista Hermosa, Caño Sardinata, KU 288581, UTA A-8479–83, 8486–91, 8493–94, 8496, 8500; Villavicencio, La Reforma, Finca Las Orquídeas, 600–650 m, UTA slide No. 23987–90 (slides of specimens collected by W. W. Lamar [WWL], field catalogue numbers, 1256–57); Villavicencio, Caño El Buque, WWL color notes and slides available for 8 specimens (WWL 1200, 1291–92, 1314–17 [UTA slide No. 23984, first specimen], 1665 [UTA slide No. 23985–86]); Villavicencio, Quebrada Colorado, 1000–1200 m, UTA slide No. 23983 (WWL 441) and color notes on WWL 449; near Restrepo, ca. 500 m, UTA slide No. 23982 (WWL 476).














