Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.49 n.1 San José Mar. 2001
Nélida J. Carnevale 1 and Juan Pablo Lewis 1.
Recibido 19-I-2000. Corregido 14-VIII-2000. Aceptado 19-IX-2000.
Abstract
Amazonia and the Gran Chaco are the largest phytogeographic units of the Neotropical Region. The Forest Wedge of the Santa Fe province of Argentina is the southernmost part of the Eastern Chaco, and there are three main types of forest, 1) the mixed dense forests, 2) the Schinopsis balansae quebrachal and 3) the Prosopis nigra var. ragonesei forests, distributed along an environmental gradient correlated with topographic elevation. In this paper litterfall and organic matter decomposition of four species were studied in two different microsites in a Schinopsis balansae forest during three years. Litter fall varied along the year following seasonal environmental variation but litterfall variations between years were not significant. Litterfall was not uniform over the whole surface, under close canopy monthly average litterfall amounts 32.67 g/m2, in open gaps 4.47 g/m2 and 15.07 g/m2 under medium density canopy. Organic matter decomposition, like in other forests, was a negative exponential function of time, and the decomposition rate is independent from the microsites where it falls, 0.15 and 0.12 in sunny and shadow microsites respectively for Maytenus vitis-idaea, 0.10 and 0.11 in sunny and shadow microsites respectively for Achatocarpus praecox, 0.04 and 0.03 for Acacia praecox, 0.04 and 0.06 for Schinopsis balansae in sunny and shadow microsites respectively. Shurbs litter was decomposed faster than the tree litter, and the shurbs litter nutrientes level was also higher. Therefore the rate of organic matter decomposition is more correlated with leaf characteristics than with environmental variables.
Key words: Chaco, forest communities, litter fall, litter decomposition, organic matter circulation, Schinopsis balansae forests.
Organic matter circulation has been studied in tropical rainforests, boreal deciduous forests, and other forests but very little is known about carbon cycle in chaquenian forests. Litterfall and decomposition as well as seasonal changes of bioelements in four species of chaquenian forests have been recently studied (Palma et al. 1998, 2000) but the problem has never been analysed on Schinopsis balansae Engl. forests (quebrachal) of the Cuña Boscosa of Santa Fe, Argentina. The quebrachal is the most widespread type of forest in the southernmost portion of the Eastern Chaco. It is a xerophitic forest where Schinopsis balansae is abundant together with Acacia praecox Gris., Aspidosperma quebracho blanco Schlecht., Prosospis sp. (P. alba Gris., P. nigra (Gris) Hieron. and their hybrids) Myrcianthes cisplatensis (Camb.) Berg. and many other tree species and shrubs (Lewis & Pire 1981). They have been heavily fell for tanin industry in the past, followed by grazing cattle and timber extraction for fuel and charcoal production.
Litterfall is the most important way of energy and organic matter transfer from the forest canopy to decomposer organisms of the soil surface (Lousier & Parkinson 1976, Hirabuki 1991). If litterfall is analysed, it is possible to asses its variation, storage, composition and decomposition (Hart 1995), organic matter production rate, and to quantify nutrient availability and its recycling capacity, which is different among species (Montagnini et al. 1993) Nutrient liberation from organic matter is determined by the environment and the physico-chemical properties of the litter that falls (Facelli & Pickett 1991).
The most important factors that affect litter decomposition are on one hand temperature, humidity and aeration which affect soil biota activity (Santa Regina et al. 1989, Martin et al. 1994), and on the other hand, litter composition and quality (Velazco 1992-1993, Montagnini et al. 1993, Gallardo & Merino 1993, Couteaux et al. 1995). Litter quality is related to leaf characteristics such as lignine/nitrogen ratio (Facelli & Pickett 1991, Cornelissen 1996). Nutrient content of litter is determined by mineral content of foliar tissues and time and mode of deposition (Lousier & Parkinson 1976). Organic matter deposition and decomposition rate may not be constant along the year and over the surface, so they can contribute to the creation of different microhabitats and therefore they may have an effect on the floristic composition, productivity and diversity of the community (Carson & Peterson 1990, Molofsky & Augspurger 1992, Facelli & Facelli 1993). The same type canopy disturb can have different effects on different communities of similar architecture, but with different amount of litter on the ground (Carson & Peterson 1990). These together with other factors contribute that mature forests became a mosaic of several different units (Hirabuki 1991).
The object of this paper is to analyse litterfall, its amount and temporal and spatial distribution, and litter decomposition on different microsites on a S. balansae forests of the Cuña Boscosa (Forest Wedge) of Santa Fe (Argentina). The hypotheses are that: 1) litterfall varies along the year and between years and it is not uniform over the whole surface. 2) the organic matter decomposition rate (k) is different on different soil microsites. 3) the organic matter decomposition rate depends on the chemical composition and leaf characteristics.
Materials and methods
The region and the study site: Amazonia and the Gran Chaco are the most important phytogeographyc units of South America. The Cuña Boscosa (Forest Wedge) of Santa Fe is the southernmost portion of the Eastern Chaco and runs from 28ºS to 30ºS paralell, between the River Paraná and the Submeridional Lowlands, covering more than 1,000,000 hectares (Lewis y Pire 1981). Climate is humid warm with Summer rainfalls and a relatively long Winter drought, average annual rainfall ranges from 800 to 1100 mm. (Burgos 1970). Soils are a complex mosaic of different types which often have a halo-hydromorphic character (Espino 1981). There are three fundamental kinds of forest distributed along soil gradients correlated with topographic elevation (Ragonese y Covas 1940, Lewis 1991). At the highest part of the gradient are the mixed dense forests (Lewis et al. 1994), then the quebrachal of S. balansae (Lewis et al. 1997) and at the lowest part the Prosopis nigra var ragonesei Burk. Forests. The study area was on a relatively well recovered stand of S.balansae, quebrachal placed in the Provincial Ministry of Agriculture estate, 20 km west of Vera, in Santa Fe Province, Argentina.
Data collection : on a transect 22 funnels were layed 1 m above the ground every 10 m, 11 funnels were under closed canopy, 6 on open gaps and 5 under medium density canopy. Each was made of glass fiber 0,70 m x 0,70 m. Every month from January 1995 up to December 1997, fallen litter from the trees was collected from every funnel. Leaves from the litter were oven dried for 48 h at 70°C and then weighted.
Decomposition rate of organic matter, (k), of four species, 2 trees (S. balansae, Acacia praecox), and 2 shrubs (M. vitis idaea and Achatocarpus praecox) was analysed. These species were chosen for their relative importance in terms of cover and abundance in the quebrachal. Leaves were collected from at least three individuals of each species, in late Autumn previous to their fall, oven dried, and then 10 g were put in 20 x 20 cm sacs of 1 mm mesh, and left on the ground in two different microsites; 1) in open gaps exposed to the sun and 2) in the shadow of closed canopy forest. Every month during a whole year, three sacs of each species and microsites were taken, oven dried and weighted.
As litter decomposition is a negative exponential function of time, the rate (k) of decomposition can be expressed with Olson (1963) equation for organic matter decomposition with no production:
k= - [ln(W/Wo)]/t
where W: is the dry weight, Wo: initial dry weight and t: time.
Leaves were collected from three individuals of each of the four species and analysed to determine nutrient content and C/N ratio, in Autumn 1996 and again on Spring 1997 in order to find out if differences were intrinsic to the species or were due to translocation. Organic matter was determined by weight loss after ignition in a furnace. Mineral nutrients were determined by dissolving the ashes in hydrochloric acid, Potassium by flame photometry, and Calcium and Magnesium with the Varsene titration procedure. Nitrogen was determined by Kjeldahl method (Jackson 1958). Two replicas of all analysis were done.
Data analysis : litterfall data were analysed by Kruskal-Wallis test. Organic matter left over after a year of decomposition of the four species and on the two microsites were analysed using one and two way ANOVA test. Mean values of organic matter decomposition (k) of each species and microsites were compared using Friedman rank test. Differences in nutrient content among the leaves of the four species were analysed by Kruskal-Wallis test.
Results
Litterfall varied along the year and between years (Fig. 1). Although, leaves from the canopy fall to the ground the whole year around, litterfall is more important during Winter than during Summer. Most leaves fall from April to September, with a maximum from June to August; August in 1995, July in 1996 and June in 1997. Litterfall annual average was 20.89 g/m2 in 1995, 21.26 g/m2 in 1996 and 22.44 g/m2 in 1997, however differences between years are not significant. Nevertheless the litterfall is not constant over the whole surface, funnels in an open canopy collect far less leaves and debris than those under a close canopy. As an average, litterfall from the tree layer is 32.67 g/m2 under a close canopy, 4.47 g/m2 in large canopy gaps and 15.07 g/m2 under a medium density canopy and these differences between microsites are significant (p<.05) (Table 1).
In all cases Olson (1963) model fit well decomposition data, r2 was high in all species and in both microsites except for S. balansae in open gaps and Acacia praecox under dense canopy, however deviations from the model were not significant. Decomposition rates are significantlty different between species when analysed with one way ANOVA (p<0.05, and r2 = 0.98). When analysed with two way ANOVA 93.67% of total variation correspond to species which is significant, and 10.06% to microsites, which is not significant.

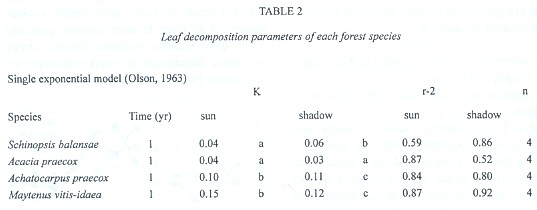
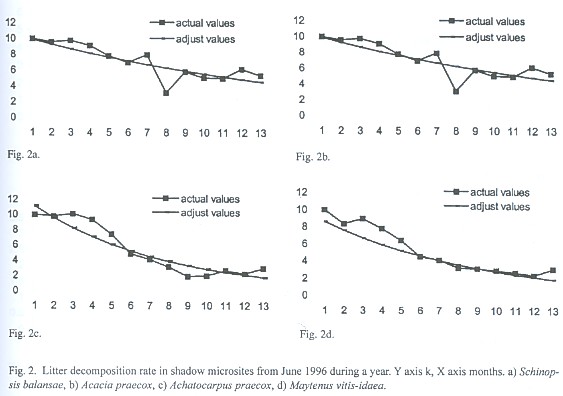
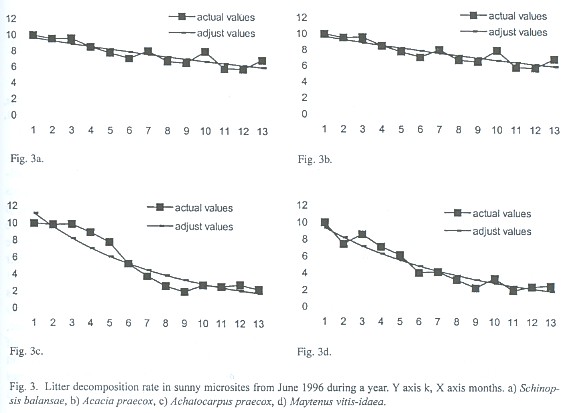
Decomposition rates of S. balansae and Acacia praecox are significantly different (p<.05) from those of M. vitis-idaea and Achatocarpus praecox on the sun microsite while under shadow microsite are significantly different between S. balansae and M vitis-idaea, between Acacia praecox and M. vitis-idaea and between Acacia praecox and Achatocarpus praecox (Table 2). The decomposition rate in both microsites of S. balansae and Acacia praecox trees is similar between them, as well as it is between M. vitis-idaea and Achatocarpus praecox shrubs. But there are significant differences of organic matter decomposition rates between the tree species and shrubs; organic matter decomposition in shrubs is faster than in trees (Fig. 2 and 3).
Decomposition values were always fitted to the negative exponential model, therefore according to the decomposition rates found, the half life for accumulation or decay (0.693/k) of tree litter more than doubled half life shrub litter, and the time required to reach 99% of accumulation or decay (5/k) is shown in Table 3 (Olson 1963).
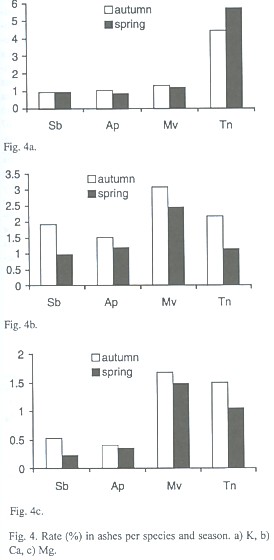
Although there may be significant differences in the amount of organic matter and nutrient content of leaves of the four species (Table 4), the most significant differences (p=.05) were in the ammount of ashes and, Potassium, Calcium and Magnesium. Achatocarpus praecox and M. vitis idaea had a higher Calcium and Magnesium content than the big trees and Achatocarpus praecox had a higher Potassium content than any other species (Fig. 4 a, b, c).


Litterfall from the canopy occurs throughout a long time during the year, however it is not very important until May and comes almost to and end in September. Most trees, such as S. balansae, Acacia praecox and Prosopis sp are deciduous, but a few tree species like A. quebracho blanco and M. cisplatensis and some shrubs like M. vitis-idaea keep part of their foliage the whole year. Strong frosts accelerate the whole process but they affect in a different way each individual according to its position in the forest. Those trees protected by other ones, on more humid ground or at the edge of internal ponds, keep part of their leaves for a longer period during Winter, even until the Spring sprout. Nevertheless, leaves fall continuously, especially after strong frosts and winds. During Summer some leaves fall if there is water shortage, strong winds or if they are old remaining leaves from a former season.
The amount of litter fall in the quebrachal, even under close canopy microsites (32 g/m2), is within the range considered very low by Olson (1963) for montane forests of California, what means that they are relatively unproductive forests. We assume that the herbaceous layer contribution to soil organic matter is important in the big canopy gaps and may be important in other microsites too, but we have no data and the problem needs further research.
Litterfall below 100-300 g/m2, increases yield and diversity in herbaceous communities, probably because it preserves moisture, while in woody communities less amount of tree leaves litter may intercept more light and may work as formidable barrier for germination (Carson & Peterson 1990).
However, in tropical forests of Costa Rica there were more abundance of individuals in forest plantations where was found more accumulation of litter (Horn & Montagnini 1999, Carnevale & Montagnini in press).
Our results strongly support the hypothesis that litterfall varies along the year and that it is not homogeneous over the whole surface because the forest canopy is not uniform and there are big gaps without any trees at all. However, leaves are carried to gaps with no trees by the wind from the tall surrounding trees, and they are trapped by the grass mesh. Variation along the year follow seasonal variations, droughts and frosts, but between years differences were not significant in the analysed years.
Our results do not support the hypothesis that organic matter decomposition rate (k) is different on different soil microsites. As these forests are deciduous, during Winter there is little light interception by the forest and it is also the dry season, so there is a possibility that soil surface temperature and moisture are relatively homogeneous. In the same way, on Summer as the air is almost still inside the forest, temperature is high in the understory as well as under the grass canopy. Therefore, at this scale, environmental conditions for organic matter decomposition may be similar in both microsites.
Decomposition rates range from large negative values (e.g., k < -1.0) in the tropics to very small negative values (e.g., k = -0.001) at high elevations, high latitudes, and other areas of impeded decomposition (Olson 1963, Schelesinger 1977, 1981). Our results (k = -0.03 a k = -0.15) are within the range of values observed by other authors fot other species (k = -0.04 a k = -0.28) of the Chaco (Palma et al. 1998).
Litter decomposition rate of shrubs is higher than that of trees, that means a more rapid nutrient reposition to the system of shurbs rather than of trees. These differences in decomposition rate between trees and shurbs may be related to the higher nutrient level of M. vitis-idaea and Achatocarpus praecox, as in many other litter sources, higher levels of Ca, Mg, and K make litter more palatable, favorouring microorganisms activity, therefore litter decomposition (Nicolai 1998, Berg 1998). Our data agree with other authors conclusions that litter decomposition rate is more related to leaves chemical composition than to environmental conditions (Day 1983, Facelli 1991, Alvarez Sánchez & Becerra Enríquez 1996, Scott & Binkley, 1997), and the results support the hypothesis that litter decomposition rate depends on leaves chemical composition.
The different half life and decomposition rates between trees and shrubs discussed above show that nutrient releasing time may be very different according to specific composition of litter, and this can compensate temporary nutrient losses such as litter sweeped from surface by heavy rains during Autumn or Spring.
Nevertheless, there are other factors that affect litter decomposition such as the composition of organic matter, celulose, hemicelulose, lignin, phenols, Si content (Keyser et al. 1978, Horner et al. 1987, Berg 1998), structural characteristics of the leaves, which are in need of further research. Also there should be analysed with more detail environmental conditions, moisture, flood length, temperature, etc. inside the quebrachal in order to have a better understanding of this complex process in this type of forest.
Acknowledgment
Financial aid from CONICET (Consejo Nacional de Investigaciones Científicas), CIUNR (Consejo de Investigaciones de la Universidad Nacional de Rosario) and FONCYT (Fondo para la investigación científica y tecnológica) is gratefuly acknowledged. We are grateful to Patricia Torres for the statistical support, to Rodolfo Comuzzi and Luis Schamburg of the Ministry of Agriculture estate for their logistic support. Ignacio Barberis and Eduardo Pire help us with data collection, Esteban Brnich with the graphs edition, Valeria Bortollotti and Viviana Martínez with chemical analysis and Adriana Pérez with final English version.
Resumen
Amazonia y el Gran Chaco son las unidades fitogeográficas más importantes de la región Neotropical. La Cuña Boscosa de Santa Fe es el extremo austral del Chaco Oriental. Está cubierta por tres tipos fundamentales de bosques, 1) los bosques densos mixtos, 2) los quebrachales de Schinopsis balansae y 3) los bosques de Prosopis nigra var. ragonesei, los que están ordenados siguiendo un gradiente correlacionado con la topografía. En este trabajo se analiza la circulación de materia orgánica, la caída de la hojarasca durante tres años y la descomposición de la hojarasca de cuatro especies en dos micrositios diferentes del mismo bosque, durante un año. La caída de hojarasca es diferente a lo largo del año, siguiendo variaciones ambientales estacionales, y las diferencias interanuales no son significativas. La caída de hojarasca no es uniforme sobre la superficie, en las abras se registró un promedio de 4.47 g/m2, bajo canopeo cerrado 32.67 g/m2 y bajo canopeo semi cerrado 15.07 g/m2. La descomposición de materia orgánica es independiente del micrositio donde se deposita. La hojarasca de los arbustos se descompone mas rápido que la de los árboles. La tasa de descomposición es de 0.15 al sol y 0.12 a la sombra para M. vitis-idaea y de 0.10 al sol y 0.11 a la sombra para Achatocarpus praecox, en los árboles disminuye a 0.04 y 0.03 para Acacia praecox y 0.04 y 0.06 para S. balansae en micrositio soleado y umbrío respectivamente. El contenido de nutrientes también es mayor en los arbustos. Por lo tanto y aparentemente, la tasa de descomposición de la materia orgánica está más correlacionada con las características de las hojas que con las variables ambientales.
Referencias
Alvarez-Sanchez J. & R. Becerra Enríquez. 1996. Leaf decomposition in a Mexican Rain Forest. Biotropica 28: 657-667. [ Links ]
Berg B. 1998. Organic matter quality and C/N ratio as controlling factors of RSOM turnover. Mittelungen der Deutschen Bodenkundlichen Gesellschaft 87: 79-91. [ Links ]
Burgos J. J. 1970. El clima de la región noreste de la República Argentina. Bol. Soc. Arg. Bot. II (Suplemento): 37-102. [ Links ]
Day F.P. Jr. 1983. Effects of flooding on leaf litter decomposition in microcosms. Oecología 56: 180-184. [ Links ]
Carson W. P. & C. J. Peterson. 1990. The role of litter in an old-field community: impact of litter quantity in different seasons on plant species richness and abundance. Oecologia 85: 8-13. [ Links ]
Cornelissen J. H. C. 1996. An experimental comparison of leaf decomposition rates in a wide range of temperate plant species and types. J. Ecology 84: 573-582. [ Links ]
Couteaux W. W., P. Boetner & B. Berg. 1995. Litter decomposition, climate and litter quality. TREE 10: 63-65. [ Links ]
Espino L. M., M. A. Seveso & M. A. Sabatier. 1981. Mapa de suelos de la Provincia de Santa Fe e Instituto Nacional de Tecnología Agropecuaria Tomo II, Rafaela. Argentina. [ Links ]
Facelli J. M. & S T. A. Pickett. 1991. Plant litter : Its dynamics and effects on plant community structure. Bot. Rev. 57: 1-33. [ Links ]
Facelli J. M. & E. Facelli. 1993. Interactions after death: plant litter controls priority effects in a successional plant community. Oecologia 95: 277-282. [ Links ]
Facelli J.M. 1994. Multiple indirect effects of plant litter affect the establishment of woody seedlings in old fields. Ecology 75: 1727-1735. [ Links ]
Gallardo A. & J. Merino. 1993. Leaf decomposition in two mediterranean ecosystems of southwest Spain: influence of substrate quality. Ecology 74: 152-161. [ Links ]
Hart D.M. 1995. Litterfall and decomposition in the Pilliga State Forests, New South Wales, Australia. Australian J. Ecol. 20: 266-272. [ Links ]
Hirabuki Y. 1991. Heterogeneous dispersal of tree litterfall corresponding with patchy canoy structure in a temperate mixed forest. Vegetatio 94: 69-79.
Horn N. & F. Montagnini. 1999. Litterfall, litter decomposition and maize bioassay of mulches from four indigenous tree species in mixed and monospecific plantations in Costa Rica. International Tree Crops Journal 10: 37-50.
Horner, J.D.; Gates, R.G. and Gostz, J.R. 1987. Tannin, nitrogen, and cell wall composition of green vs. senecent Douglas-fir foliage. Oecologia. 72: 515-519. [ Links ]
Jackson M. L. 1958. Soil chemical Analysis. Constable, London. 498 pp. [ Links ]
Keyser, P; Kirk T.K y Zeikus, I.G. 1978. Ligninolytic enzyme of Phanerochatet chrysosporum synthesized in the absence of lignin in response to nitrogen starvation. J. Bacteriol 135: 790-797. [ Links ]
Lewis J. P. & E F. Pire. 1981. Reseña sobre la vegetación del Chaco santafesino. Serie fitogeográfica Nº18. Instituto Nacional de Tecnología Agropecuaria, Buenos Aires. 42 pp.
Lewis J.P. 1991. Three levels of floristical variation in the forests of Chaco, Argentina. J. Veg. Sci. 2: 125-130. [ Links ]
Lewis J.P., E. F. Pire & J. L. Vesprini. 1994. The mixed dense forest of the Southern Chaco. Contribution to the study of flora and vegetation of the Chaco VIII. Candollea 49: 159-168. [ Links ]
Lewis J. P., E. F. Pire & I. M. Barberis. 1997. Structure, physiognomy and floristic composition of a Schinopsis balansae (Anacardiaceae) forest in the Southern Chaco, Argentina. Rev. Biol. Trop. 45: 1013-1020. [ Links ]
Lousier J.D. & D. Parkinson. 1976. Litter decomposition in a cool temperate deciduous forest. Can. J. Bot. 54: 419-436. [ Links ]
Martín A., M Rapp, I. Santa Regina & J. F. Gallardo. 1994. Leaf litter decomposition dynamics in some mediterranean deciduous oaks. Eur. J. Soil Biol. 30: 119-124.
Molofsky J & C. K. Augspurger. 1992. The effect of leaf litter on early seedling establishment in a tropical forest. Ecology 73: 68-77.
Montagnini F., K. Ramstad & F. Sancho. 1993. Litterfall, litter decomposition and the use of mulch of four indigenous tree species in the Atlantic lowlands of Costa Rica. Agrofor. Syst. 23: 39-61. [ Links ]
Montagnini F., E. González. & F. Sancho. 1996. Relación entre área basal arbórea y propiedades químicas del suelo en un bosque tropical secundario de 10 años de edad. Ivyraretá 7: 47-56. [ Links ]
Nicolai V. 1998. Phenolic and mineral content of leaves influences decomposition in European forest ecosystems. Oecología 75: 575-579. [ Links ]
Olson J.S. 1963. Energy storage and the balance of producers and decomposers in ecological systems. Ecology 44: 322-331. [ Links ]
Palma R. M., J. Prause, A. V. Fontanive & M. P. Jiménez 1998. Litter fall and litter decomposition in a forest of the Parque Chaqueño Argentino. Forest Ecol. Manag. 106: 211-222. [ Links ]
Palma R. M., R. L. Defrieri., M. F. Tortarolo, J. Prause & J. F. Gallardo. 2000. Seasonal changes of bioelements in the litter and their potential return to green leaves in four species of the Argentine subtropical forest. Ann. Bot. 85: 181-186.
Santa Regina I., J. F. Gallardo & I. San Miguel. 1989. Ciclos biogeoquímicos en bosques de la Sierra de Béjar (Salamanca, España). Descomposición de la hojarasca. Rev. Ecol. Biol. Sol. 26: 407-416. [ Links ]
Ragonese A. E. & G. Covas. 1940. La distribución geográfica de los quebrachales en la provincia de Santa Fe. Rev. Arg. Agron. 7: 176-184. [ Links ]
Scott N. A. & D. Binkley. 1997. Foliage litter quality and annual net N mineralization: comparison across North American forest sites. Oecologia 111: 151-159. [ Links ]
Schlesinger W. H. 1977. Carbon balance in terrestrial detritus. Annual Review of Ecology and Systematics 8: 51-81. [ Links ]
Schlesinger W. H. & M. M. Hasey. 1981. Decomposition of chaparral shrub foliage: losses of organic and inorganic constituents from deciduous and evergreen leaves. Ecology 62: 762-774. [ Links ]
Swift M. J., O. W. Heal & J. M. Anderson. 1979. Decomposition in terrestrial ecosystems. Studies in Ecology. Volume 5. University of California, Berkeley and Los Angeles 372 pp. [ Links ]
Velazco F., N. García Calderón & N. Aguilera. 1992-1993. Evolución de la composición química y propiedades de la materia orgánica del suelo por la transformación de áreas forestales en agrosistemas en Veracruz (México). Ciencia del Suelo 10-11: 98-103.
Wedin W. 1994. Species, nitrogen and grassland dynamics: the constraints of stuff. pp 253-264. In: Jones, C.H. and Lawton, J.H. (eds.) Linking species and ecosystems. Chapman and Hall, London.
1 Cátedra de Ecología. Facultad de Ciencias Agrarias C.C. 14. 2123 Zavalla. Santa Fe. Argentina.E-mail: ncarneva@citynet.net.ar














