Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.48 n.2-3 San José Jun. 2000
(Hymenoptera: Sphecidae) in southeastern Brazil
Evandro Camillo1 and Antonio D. Brescovit2
Abstract
Fifty five nests and 216 cells of Trypoxylon (Trypargilum) rogenhoferi were obtained from trap-nests (cut bamboo canes) in Santa Carlota Farm (Itaoca Section=IS and Santana Section=SS), Cajuru and on the São Paulo University Campus, Ribeirão Preto (=RP), both in the State of São Paulo, Brazil (Sept. 1993 – Oct. 1995). The prey (spiders) of 40 cells from IS, 58 from SS and 39 from RP were identified. The greatest nesting frequency occurred during the hot and wet season (September to April). T. rogenhoferi preyed upon individuals of five spider families, with Araneidae (orb-weaver spiders) being the most frequent (99.6%). Alpaida aff. negro (58%) was the most frequently collected species in IS, followed by A. alto (24.8%); in SS (59.6%) and RP (64.7%) the most frequent species was A. veniliae, followed in SS by A. aff. negro (14.9%) and in RP by A. leucogramma (13.5%). The size of reproductive niches, H=1.25 (IS), H=1.30 (SS) and H=1.29 (RP) were not significantly different. There was a positive correlation between the reproductive niche width (H) and evenness.
Key words
Araneae, evenness, reproductive niche, trap-nests, Sphecidae, Trypoxylon, wasps.
Species of the genus Trypoxylon, subgenus Trypargilum, are restricted to the Western Hemisphere from Canada to Argentina. The Neotropical region contains most of the species (Bohart and Menke 1976, Coville 1982). These wasps are solitary. Each female constructs and provisions the nest with paralyzed spiders. Some species construct mud nests, whereas others utilize pre-existing cavities. Nests consist of linear series of cells that are subdivided by mud partitions, usually followed by an empty vestibular cell and an end plug made of mud (Bohart and Menke 1976, Coville and Coville 1980, Coville 1982).
One important species characteristic in Sphecidae is the species of prey that females captured. This is important from both evolutionary and taxonomic perspectives. These wasps collect prey in places where people do not normally look (Genaro et al. 1989); thus, nests of these wasps can provide vast collections of spiders, including rarely collected species (Coville 1987).
The purpose of this paper is to present data on prey species collected by Trypoxylon (Trypargilum) rogenhoferi Kohl, 1884. In addition observations obtained from trap-nests placed in three sites in southeastern Brazil are presented to reveal some parameters of their reproductive niches.
Materials and Methods
Study areas: Area 1- Santa Carlota Farm (SCF), Cajuru, SP (21°18-27 S and 47°12-17 W). The vegetation consists of semideciduous mesophyll and riparian forests, cerrados (s.l.), cerradões (Rizzini 1979) and various types of plantations. There are also some areas showing several stages of secondary growth of the main vegetation types. Farm altitudes vary from 540 to 944 m. The study sites in the farm, Itaoca Section (=IS) and Santana Section (=SS) (approximately six km from each other), is characterized by abandoned orchards surrounded by sugar cane plantations, pasture and cerrados (s.l.) with various abandoned buildings. Semideciduous mesophyll and riparian forests are located approximately 300 m from the orchards. Area 2. Campus of São Paulo University, Ribeirão Preto (=RP), SP (21°11 S and 47°13 W). The vegetation is formed by small areas of semideciduous forests, grasses, shrubs, ornamental plants, fruit trees and reforested areas with arboreous angiosperms. Altitudes vary from 580 to 620 m.
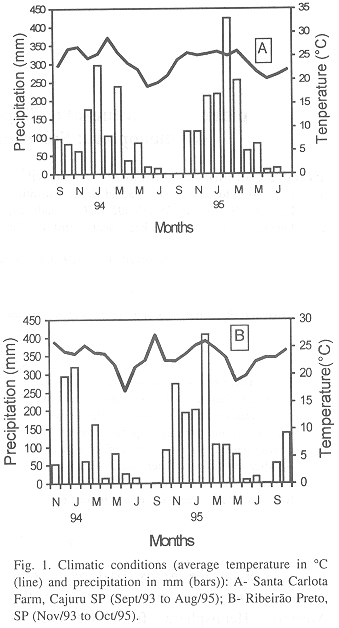
Two distinct seasons are recognized in this region: 1- Cold and dry season (May to August) with monthly average temperatures ranging from 18.5 to 22.2 °C (SCF) and from 17 to 23 °C (RP), precipitation from 0 to 84.4 mm (SCF) and from 0 to 82.9 mm (RP); 2- Hot and wet season (September to April) with monthly average temperatures ranging from 23 to 28.8 °C (SCF) and from 22.4 to 27.2 °C (RP), precipitation from 0 to 422.6 mm (SCF) and from 1.4 to 409.4 mm (RP) (Fig. 1).
Trap-nests: Bamboo stems (B) were used as trap-nests (TN) with a nodal septum closing one end. The internal diameter of nests ranged from 7 to 25 mm, and nest lengths ranged from 70 to 250 mm. These TN (450 in RP, 450 in SS and 200 in IS) were tied together in batches of six to eight units and placed on the shelves of shelters built at the study locations.
The TN were inspected at least once a month during the period from September/93 to August/95 at SCF and from November/93 to October/95 in RP. During the inspections, which were performed with the help of an otoscope, the nests that had been recently completed or were being provisioned were collected and transported to the laboratory. Each nest was replaced by a new TN. The nests were opened in the laboratory and the prey (spiders) from cells, which contained eggs, or recently hatched larvae were removed and preserved in 80% alcohol. At least one immature wasp was left in all of these nests so that it could develop fully, thus allowing for determination of the nesting species. All the wasp and spider specimens were deposited in the Entomology Collection of Biology Department-FFCLRP-USP and a portion of the spiders samples was deposited in the arachnological collection of Butantan Institute.
Data analysis: The reproductive niche (following Álvarez et al. 1988) width was calculated using the number of prey species as well as the Shannon-Weaver Diversity Index H= - ph. ln ph, where ph is the proportion of individuals belonging to the hth species in the total sample (Pielou 1975).
The evenness index was calculated according to Pielou (1966): J= H / Hmax, where H is the Shannon-Weaver Index and Hmax is the logarithm (ln) of the total number of prey species in the sample. This index varies from zero to one.
The similarity in species composition among the studied habitats was calculated by the Sörensen Similarity Quotient (Sörensen 1948): S.Q. = 2J/ a+b, where a and b are the numbers of families, genera and species of collected prey in each of the habitats and J is the number of families, genera and species of collected prey common to all samples. The similarity among the studied communities, considering the number of collected spiders of each sampled species, was calculated by the method of percentage similarity (Hanski and Koskela 1977) : PSij = min. (pih, pjh), where pih and pjh are proportions of the h families, genera or species of collected prey. The Test for difference between two indices was used in the statistical analysis of indices (Zar 1984).
Results
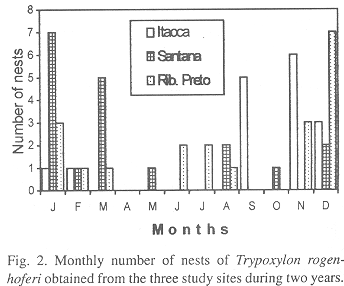
Number of nests, cells and seasonal abundance: 216 cells of T. rogenhoferi were obtained, 69 were from IS (16 nests), 84 from SS (19 nests) and 63 from RP (20 nests). Prey was collected and identified from 40, 58 and 39 cells, respectively. The nests (19 completed ones and 36 being provisioned) were collected throughout the year, except in April, with the greatest nesting frequency occurring in the hot and wet season (September to April), during which 85.4% of the sampled nests were collected. The months with the greatest nesting frequencies were September and November in IS, January and March in SS and December in RP (Fig. 2).
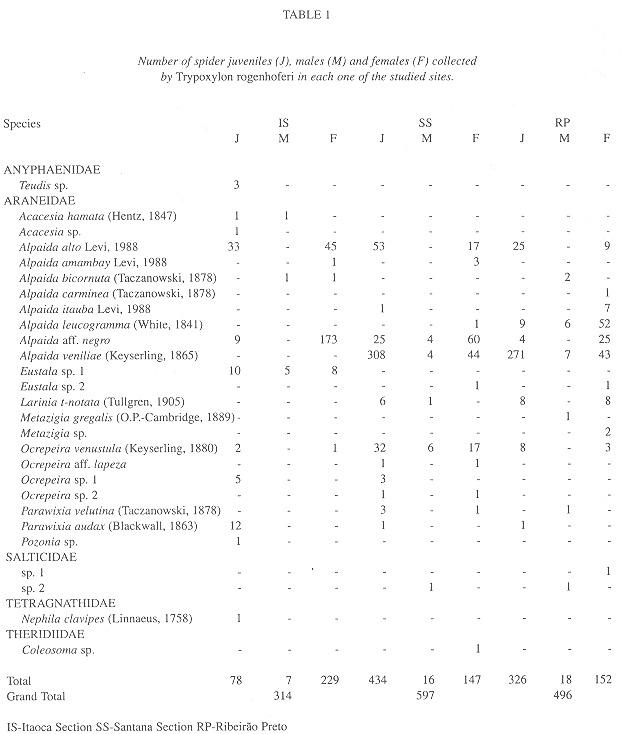
Collected prey: Five families of spiders were found with Araneidae being the most frequent in all of the sites in number of genera (61.5%), species (81.5%) and individuals (99.4%). Only Araneidae was found at all three sites while Salticidae was found in two (SS and RP), Anyphaenidae and Tetragnathidae were only collected in IS and Theridiidae was collected only in SS (Table 1).
Araneidae was the most frequent family in the samples. This suggests a strong preference (99.6%) of T. rogenhoferi for species of orb-weaver spiders, Alpaida (88.4%) was the most frequent genus (Table 1).
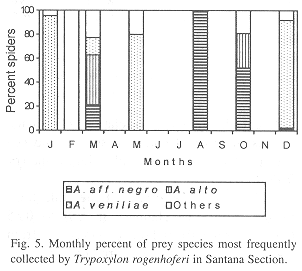
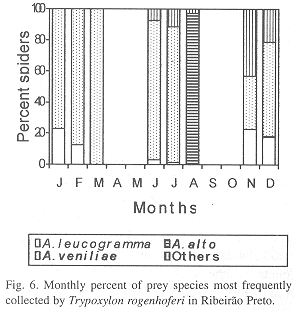
Alpaida aff. negro (58%) was the most frequent species of prey in IS, followed by Alpaida alto Levi, 1988 (24.8%) and Eustala sp.1 (7.3%); in SS the most frequent was Alpaida veniliae (Keyserling, 1865) (59.6%), followed by A. aff. negro (14.9%) and A. alto (11.7%); in RP A. veniliae was also the most common (64.7%), followed by Alpaida leucogramma (White, 1841) (13.5%) and A. alto (6.8%). Of the 27 prey species collected by T. rogenhoferi, four were found in all three study areas, six only in IS, three in SS and four in RP; two species were common in IS and SS, six in SS and RP and one in IS and RP (Table 1).
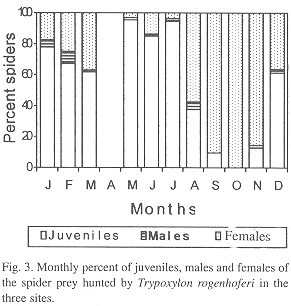
T. rogenhoferi captured spider juveniles, males and females of seven prey species, only juveniles of eight, only males of two and only females of six species. Juveniles and females were collected from four species and males and females from one species (Table 1). Juveniles, males and females were collected during nine months of the year, while in October only females were present in the samples. In September males were not found. The percent of females was greater than that of juveniles in August through November, while in the others months, the percentage of juveniles was greater than that of females. The percentage of both, juveniles and females were greater than that of males. The greatest frequencies of juveniles occurred in May (95.4% of the collected spiders), June (84.9%) and July (94.4%); for males the greatest frequency was found in February (7.8%) and August (5%) and for females in September (90.3%), October (100%) and November (85.8%) (Fig. 3). In all locations the juveniles correspond to 59.6%, females to 37.5% and males to 2.9%.
Phenology of three most frequently collected prey species: In IS, A. aff. negro (collected during three months) was the most frequent species in September, November and December, A. alto (collected during four months) in February and Eustala sp.1 (collected during two months) in January (Fig. 4). In SS, A. veniliae (collected during four months) was the most frequent species in January, May and December, A. aff. negro (collected during four months) in August and October, with A. alto (collected in three months) in March (Fig. 5). In RP, A. veniliae (collected during seven months) was preferred in January through March, June, July, November and December, with A. alto (collected in one month) in August (Fig. 6).
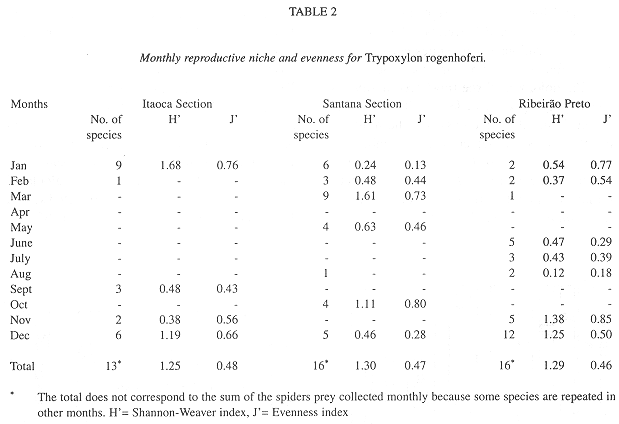
Parameters of the reproductive niche: The monthly reproductive niche width calculated by the number of prey species collected varied from one (February in IS, August in SS and March in RP) to 12 (December in RP), while the total niche width was 13 (IS) and 16 (SS and RP). The monthly reproductive niche width calculated by the Shannon-Weaver Index (H) varied from 0.12 (August in RP) to 1.68 (January in IS) (Table 2). The total reproductive niche widths, H= 1.25 (IS), H= 1.30 (SS) and H= 1,29 (RP) were not significantly different (P>0.05). The correlation between the indices (number of collected species and H) was positive and statistically significant (r= 0.73, P<0.01).
The monthly evenness of the collections ranged from 0.13 (January in SS) to 0.85 (November in RP), and the total evenness from 0.46 (RP) to 0.48 (IS). The correlation between the monthly reproductive niche width calculated using the number of collected prey species and evenness (J) was positive and non-significant (r= 0.18, P>0.05), while the correlation between H and J was positive and significant (r= 0.75, P<0.01).
The greatest similarity value at the family level among the sites was observed when comparing SS to RP (SQ= 0.80). At the genus level, the values were similar, and at the species level, the value was greatest when comparing SS to RP (SQ= 0.75). The percentage similarity was markedly high at the family and genus level, moderate at the species level when comparing SS to RP and low when comparing SS to IS and RP to IS (Table 3).
Discussion
The great preference of T. rogenhoferi for prey of the Araneidae family was also observed in the same three habitats by Camillo and Brescovit (1999a) in relation to T. lactitarse Saussure, 1867 (96.6%). In these habitats Camillo et al. (1994) observed in 14 T. rogenhoferi cells that 99.4% of prey were from the family Araneidae. In the Central Amazon, Garcia and Adis (1995) observed that the females of T. rogenhoferi captured one species of this spider family.
Some species of the subgenus Trypargilum preyed preferentially upon spiders of the Araneidae family: T. mexicanum Saussure, 1867 (Rau 1943), T. politum Say, 1837 (Muma and Jeffers 1945, Cross et al. 1975, Barber and Matthews 1979, Rehnberg 1987), T. texense Saussure, 1867 (Kurczewski 1963), T. collinum F. Smith, 1856 (Krombein 1967, Medler 1967), T. lactitarse (Krombein 1967, Coville 1981, Camillo et al. 1993, Camillo and Brescovit 1999a), T. tridentatum Packard, 1867 (Krombein 1967, Jiménez and Tejas 1994), T. orizabense Richards 1934 (Coville 1979), T. tenoctitlan Richards, 1934 (Coville and Coville 1980), T. nitidum schultessi Richards, 1936 T. saussurei Rohwer, 1912 (Coville 1981), T. evansiCoville, 1982 T. monteverdaeCoville, 1982 T. occidentalis Coville, 1982 (Coville 1982), T. subimpressum F. Smith, 1856 (Genaro et al. 1989, Genaro and Alayón 1994).
A. aff. negro (58% in IS) and A. veniliae (59.6% in SS and 64.7% in RP) were the most frequently captured prey species, whereas T. lactitarse (Camillo and Brescovit 1999a) preferentially captured Eustala sp1 (31.6% in IS, 20.1% in SS and 48.7% in RP). Thus, T. lactitarse (Camillo and Brescovit 1999a) did not capture A. veniliae, and A. aff. negro was represented in the samples by some individuals (15), and T. rogenhoferi preyed upon Eustala sp1 only in IS (23 individuals). Garcia and Adis (1995) observed, in Central Amazon, that T. rogenhoferi provisioned cells only with A. veniliae.
T. rogenhoferi captured more juvenile prey (50.3%) than adult females (34.3%) and males (15.4%). The same fact was observed by Camillo and Brescovit (1999a) for T. lactitarse but in different percentages (59.6%, 37.5% and 2.9%, respectively). Collection of large percentage of juveniles was also observed for T. texense (Kurczewsky 1963), T. politum (Barber and Matthews 1979), T. orizabense (Coville 1980), T. tenoctitlan (Coville and Coville 1980), T. nitidum schultessi, T. saussurei (Coville 1981), T. monteverdae (Coville 1982, Brockmann 1992), T. occidentalis (Coville 1982), T. subimpressum (Genaro and Alayón 1994) and T. tridentatum (Jiménez and Tejas 1994).
The reproductive niche widths (H) of T. rogenhoferi in different localities were not significantly different. But when comparing reproductive niche widths with those of T. lactitarse (Camillo and Brescovit 1999a) (H=2.24 in IS, H= 2.72 in SS and H=1.76 in RP) they were smaller. Significant differences were also observed in relation to evenness (J) which was smaller for T. rogenhoferi.
Reproductive niche overlap between T. rogenhoferi and T. lactitarse (Camillo and Brescovit 1999a), calculated using the percentage similarity of Hanski and Koskela (1977) was very low, 0.16 for SI, 0.06 for SS and 0.04 for RP. These results indicate that these species do not compete in any significant manner for food in the three habitats studied, a fact that is easily observed when comparing the most common prey species.
An estimate of the weight of each prey collected was obtained by dividing the total biomass of the cell group by the total number of prey. Thus, we calculated that the prey collected by T. lactitarse had a lower biomass (22.8 mg, n=1241) than those collected by T. rogenhoferi (33.3 mg, n=1407). This difference is more evident when we consider only the most common prey species. In this case, we observe that the mean individual weight was 48.5 mg (n=108) for A. aff. negro and 26.6 mg (n=212) for A. veniliae, values greater than 14.6 mg (n=93), the weight calculated for Eustala sp1 which was collected by T. lactitarse. The difference in mean weight between A. aff. negro and A. veniliae is related to the different number of prey provisioned per cell (6.7 and 13.2, respectively).
T. rogenhoferi collected heavier prey than T. lactitarse wich is directly related to the size of these species. According to Camillo and Brescovit (1999b), T. rogenhoferi females are larger than T. lactitarse, and since the transport of prey is carried out in flight, larger individuals are able to carry larger prey. This result clearly shows why there is no overlap of the reproductive niches.
Acknowledgments
Special thanks to the owners of Santa Carlota Farm, Cajuru, SP, for allowing us free access to their land. We are especially grateful to Carlos Alberto Garófalo for his comments and suggestions and José Carlos Serrano for technical help.
References
Álvarez, V.B., J.A. Genaro & C.S.S. Alonso. 1988. Nuevas considerations acerca del nicho ecológico. Ciencias Biológicas 19/20: 3-8. [ Links ]
Barber, M.C. & R.W. Matthews. 1979. Utilization of trap nests by the pipe-organ mud-dauber, Trypargilum politum (Hymenoptera: Sphecidae). Ann. Entomol. Soc. Am. 72: 260-262. [ Links ]
Bohart, R.M. & A.S. Menke. 1976. Sphecid wasps of the world. University of California, Berkeley. 695 p. [ Links ]
Brockmann, H.J. 1992. Male behavior, courtship and nesting in Trypoxylon (Trypargilum) monteverdae (Hymenoptera: Sphecidae). J. Kans. Entomol. Soc. 65: 66-84. [ Links ]
Camillo, E., C.A. Garófalo, G. Muccillo, & J.C. Serrano. 1993. Biological observations on Trypoxylon (Trypargilum) lactitarse Saussure in southeastern Brazil (Hymenoptera: Sphecidae). Rev. Bras. Entomol. 37: 769-778. [ Links ]
Camillo, E., C.A. Garófalo & J.C. Serrano. 1994. Observações sobre a biologia de Trypoxylon (Trypargilum) rogenhoferi Kohl (Hymenoptera: Sphecidae). An. Soc. Entomol. Brasil 23: 299-310. [ Links ]
Camillo, E. & A.D. Brescovit. 1999a. Spider (Araneae) captured by Trypoxylon (Trypargilum) lactitarse (Hymenoptera: Sphecidae) in southeastern Brazil. Rev. Biol. Trop. 47: 149-160. [ Links ]
Camillo, E. & A.D. Brescovit. 1999b. Aspectos biológicos de Trypoxylon (Trypargilum) lactitarse Saussure e Trypoxylon (Trypargilum) rogenhoferi Kohl (Hymenoptera: Sphecidae) em ninhos armadilhas, com especial referência a suas presas. An. Soc. Entomol. Brasil 28: 251-262. [ Links ]
Coville, R.E. 1979. Biological observations on Trypoxylon (Trypargilum) orizabense Richards in Arizona (Hymenoptera: Sphecidae). J. Kans. Entomol. Soc. 52: 613-620. [ Links ]
Coville, R.E. 1981. Biological observations on three Trypoxylon wasps in the subgenus Trypargilum from Costa Rica: T. nitidum schultessi, T. saussurei and T. lactitarse (Hymenoptera: Sphecidae). Pan-Pacif. Ent. 57: 332-340. [ Links ]
Coville, R.E. 1982. Wasps of the genus Trypoxylon subgenus Trypargilum in North America. University of California, Berkeley. 147 p. [ Links ]
Coville, R.E. 1987. Spider-hunting sphecid wasps, p. 309-318. In W. Nentwig (ed.). Ecophysiology of spiders. Springer, Berlin. 448 p. [ Links ]
Coville, R.E. & P.L. Coville. 1980. Nesting biology and male behavior of Trypoxylon (Trypargilum) tenoctitlan in Costa Rica (Hymenoptera: Sphecidae). Ann. Entomol. Soc. Am. 73: 110-119. [ Links ]
Cross, E.A., M.G. Stith & T.R. Bauman. 1975. Bionomics of the organ-pipe mud-dauber, Trypoxylon politum (Hymenoptera: Sphecidae). Ann. Entomol. Soc. Am. 68: 901-916. [ Links ]
Garcia, M.V.B. & J. Adis. 1995. Comportamento de nidificação de Trypoxylon (Trypargilum) rogenhoferi Kohl (Hymenoptera, Sphecidae) em uma floresta inundável de várzea na Amazônia Central. Amazoniana 13: 259-282. [ Links ]
Genaro, J.A., C.S. Sanchez & G. Alayón. 1989. Notas sobre la conducta de nidificación de Trypoxylon (Trypargilum) subimpressum Smith (Hymenoptera: Sphecidae). Caribbean J. Sci. 25: 228-229. [ Links ]
Genaro, J.A. & G. Alayón. 1994. Las presas (Araneae) de Trypoxylon (Trypargilum) subimpressum (Hymenoptera: Sphecidae) en Cuba. Rev. Biol. Trop. 42: 353-356. [ Links ]
Hanski, I. & H. Koskela. 1977. Niche relations among dung inhabiting beetles. Oecologia 28: 203-231. [ Links ]
Jiménez, M.L. & A. Tejas. 1994. Las arañas presa de la avispa lodera Trypoxylon (Trypargilum) tridentatum tridentatum en Baja California Sur, Mexico. Southwest. Entomol. 19: 173-180. [ Links ]
Krombein, K.V. 1967. Trap-nesting wasps and bees. Life histories, and associates. Smithsonian Institution, Washington, D.C. 570 p. [ Links ]
Kurczewski, F.E. 1963. A first Florida record and note on the nesting of Trypoxylon (Trypargilum) texense Saussure (Hymenoptera: Sphecidae). Fla. Entomol. 46: 243-245. [ Links ]
Muma, M.H. & W.F. Jeffers. 1945. Studies on the spider prey of several mud-dauber wasps. Ann. Entomol. Soc. Am. 38: 245-255. [ Links ]
Pielou, E.C. 1966. An introduction to Mathematical Ecology. John Wiley, New York. 286 p. [ Links ]
Pielou, E.C. 1975. Ecological diversity. John Wiley, New York. 165 p. [ Links ]
Rau, P. 1943. The nesting habits of certain sphecid wasps of Mexico, with notes on their parasites. Ann. Entomol. Soc. Am. 36: 647-653. [ Links ]
Rehnberg, B.G. 1987. Selection of spider prey by Trypoxylon politum (Say) (Hymenoptera: Sphecidae). Can. Ent. 119: 189-194. [ Links ]
Rizzini, C.D. 1979. Tratado de fitogeografia do Brasil, Vol. 2. Hucitec, São Paulo. 374 p. [ Links ]
Sörensen, T. 1948. A method of establishing group of equal amplitude in plant sociobiology based on similarity of species content and its application to analyses of the vegetation on Danish commons. Kong. Danske Vidensk. Selsk. Biol. Skr. 5: 1-34. [ Links ]
Zar, J.H. 1984. Biostatistical analysis. Prentice-Hall, Inglewood Cliffs, New Jersey. 718 p. [ Links ]
1Departamento de Biologia, Faculdade de Filosofia Ciências e Letras de Ribeirão Preto-USP, 14040-901, Ribeirão Preto, SP, Brasil. Fax: 55 016 602 3666. E-mail: ecamillo@ffclrp.usp.br
2 Laboratório de Artrópodes Peçonhentos, Instituto Butantan, Av. Vital Brasil 1500, 05503-900, São Paulo, SP, Brasil.