Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.47 n.4 San José Dec. 1999
(Filibranchia: Ostreidae) from Mazatlan, Sinaloa, Mexico
Martín G. Frías-Espericueta1 , 2 Monica A. Ortiz-Arellano1 J. Isidro Osuna-López1 y J. Angel Ronson-Paulin1
Received 18-VIII-1998. Corrected 18-III-1999. Accepted 10-V-1999.
Abstract
Two populations of Crassostrea iridescens were sampled off Mazatlan and in a zone free of anthropogenic activity. The bivalves were collected from February 1992 to February 1993. Concentrations of Cd, Co, Cr, Cu, Fe, Mn, Ni and Zn were determined by flame atomic absorption spectrophotometry. Annual mean values in the sampling zone off Mazatlan were higher in Co, Cu, Fe, Mn, Ni and Zn than in the sampling zone free of anthropogenic activity; however, differences were not statistically significant at 95% confidence level. Urban discharges in the coastal zone in front of Mazatlan city, have little or no heavy metal concentrations dissolved or particulated.
Key words
Crassostrea iridescens, heavy metals, urban discharges, oyster.
In the Pacific region of Mexico, the rock oyster Crassostrea iridescens (Hanley 1954), is present along the coast, and it is associated with subtidal rocky substrates (Páez-Osuna et al. 1995). The species is geographically distributed from La Paz, Gulf of California, to northern Peru (Keen 1971). Recently, its reproductive cycle was described by Frías-Espericueta et al. (1997): there is a resting phase from November through January; a maturation phase from April to May; and a spawning phase from July to October.
Bivalves are extensively used in monitoring programs in the marine environment due to their ability to concentrate pollutants to several orders of magnitude above ambient levels in sea water (Al-Madfa et al. 1998). According to Phillips and Rainbow (1993) an ideal biomonitor should fulfill several requisites: should be sessile or sedentary in order to be representative of the study area; should be abundant in study areas, easy to identify and sample at all times of year, and should have sufficient tissue for analysis of the contaminant of interest; should be hardy, tolerating wide ranges of contaminant concentration, thereby permitting the design of transplant experiments and laboratory studies of contaminant kinetics; and should be strong accumulators of the relevant trace metal. These characteristics, its wide distribution and commercially valuable seafood species, makes C. iridescens a very suitable species as an indicator of pollution (biomonitor) for heavy metals.
Regarding heavy metals, various processes influenced by anthropogenic activities may contribute to increase concentrations in natural waters (Singh and Steinnes 1994): (1) run-off from agricultural and urban areas, (2) discharges from mining, factories and municipal sewer systems, (3) leaching from dumps and former industrial sites, (4) atmospheric deposition.
The purpose of this study was to evaluate the concentration of eight heavy metals (Cd, Co, Cr, Cu, Fe, Mn, Ni and Zn) in two different sites in Mazatlan, Sinaloa, Mex. One site with little influence and another without urban discharge influence.
Materials and Methods
Individuals of C. iridescens were collected (9-10 samplings) from two sites along the coast of Mazatlan, Sinaloa, Mexico from February 1992 through February 1993. One station was located in front of Mazatlan, which receives little urban wastewater (23° 15 41.5N and 106° 15 W). The second station was located north of the city between 23° 28 and 23° 29 N and 106° 36 and 106° 37 W, an area characterized by the absence of significant anthropogenic activity. Oysters were sampled by scuba diving, collecting organisms of similar size to minimize the effect of body weight (Anonymous 1980; Marina and Enzo 1983). In the laboratory, after taking biometric measurements, the soft tissue of each composite sample of 15-20 organisms was dried at 70 °C. All glassware and plastic devices used in the manipulation of the samples were completely acid-washed (Moody and Lindstrom 1977). Pulverization and homogenization were achieved by grinding the tissue samples in a teflon mortar. Samples and blanks for analysis were prepared by digesting aliquots of dry material with concentrate quartz-distilled nitric acid using the standard addition method and the metal concentrations were determined by flame atomic absorption spectrophotometry (Páez-Osuna et al. 1993a, 1995). The heavy metal concentration confidence intervals were calculated using the method proposed by Miller and Miller (1987). The performance of the method was evaluated by analyzing a reference material mussel homogenate MA-M-2/MT (Anonymous 1985). The concentrations found for Cd, Co, Cr, Cu, Ni and Zn were in the acceptable range reported, only Fe and Mn were underestimated (Páez-Osuna et al. 1995) (Table 1).
Heavy metals concentrations (µg/g, dry weight) in the reference material MA-M-2/TM; mean ± standard deviation (n=6).
Metal Reported concentrations Class of results Concentration found
Cd 1.16-1.54 A 1.27 ± 0.08
Co 0.75-1.07 B 1.01 ± 0.10
Cr 0.95-1.62 B 1.28 ± 0.13
Cu 7.53-8.44 A 6.74 ± 2.24
Fe 229.2-268.2 B 171.3 ± 29.1
Mn 60.7-75.3 B 53.9 ± 4.9
Ni 0.89-2.04 C 2.07 ± 0.19
Zn 152.8-166.7 B 192.2 ± 24.4
Certified with satisfactory (A) or acceptable (B) degree of confidence; values not certified (C) (Anonymous 1985).
Results
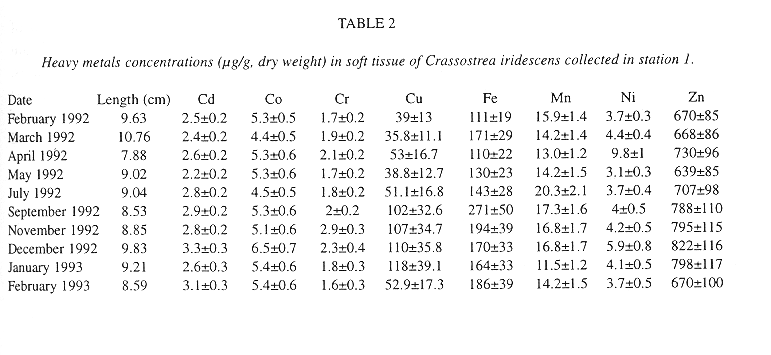
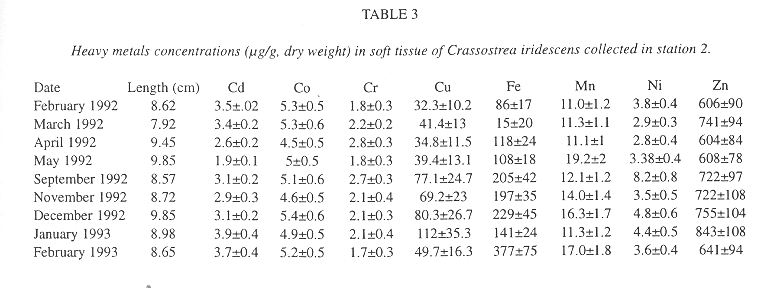
Table 2 and 3 list heavy metal concentrations in the soft tissue of C. iridescens in both stations.
There were no significant correlations between metal concentration and body length (T-students, p > 0.05). Boyden (1974) suggested that for some metals the concentrations in small organisms may be greater than those in large organisms. With regard to the correlation coefficient between metal concentrations, only Cu-Zn revealed a significant coupling with r= 0.95 (p=0.005) and r= 0.88 (p=0.05) for stations 1 and 2, respectively (Fig. 1).
The intervals of concentrations for Cd, Co, Cr, Cu, Fe, Mn, Ni and Zn were: 2.2-3.3, 4.4-6.5, 1.6-2.9, 35.8-118, 110-271, 11.5-20.3, 3.1-9.8 and 639-822 µg/g (dry weight), respectively, for station 1; while for station 2: 1.9-3.9, 4.5-5.4, 1.7-2.8, 32.3-112, 15-377, 11-19.2, 2.8-8.2 and 604-843 µg/g (dry weight), respectively.
In station 1 the highest concentration of Cd, Co, Cr, Cu and Zn was determined in autumn-winter months; while the Ni, Fe and Mn its highest value occurred in the Spring-Summer months (Table 2). Regarding station 2 the highest concentration of Cd, Co, Cu, Fe and Zn was found in autumn-winter; and Cr, Mn and Ni in spring-summer months (Table 3). There were no significant differences between the highest values of spring-summer and autumn-winter. Only Ni, in both stations, differed (means comparison test, p > 0.05, see Miller and Miller 1987).
Mean values of Co, Cu, Fe, Mn, Ni and Zn were higher in station 1 than in station 2, only Cd and Cr mean values were higher in station 2. However, no significant difference was observed (means comparison test, p> 0.05).
In both stations the decreasing order of metals values was the same.
Zn>Fe>Cu>Mn>Co>Ni>Cd>Cr
Discussion
Around the world, the coastal zones are subject to the direct release of urban and industrial discharges, such inputs are known to contain heavy metals, which may increase trace metal concentrations in the coastal zone, some of which are toxic and can endanger human health (Al-Madfa et al. 1998). In the present study, station 1, which receives little urban discharges, apparently showed the highest annual mean values of the majority of metals.
Concerning correlations between heavy metal concentrations in the soft tissue of C. iridescens, only Cu-Zn in both stations were significant. These correlations may be attributable to the fact that C. iridescens metabolically requires Cu and Zn in the same proportion during its life cycle, and similar physical/chemical properties of the metals involved in the metabolic processes (Szefer etal. 1994). Paéz-Osuna and Marmolejo-Rivas (1990a) found the same correlations in Crassostrea corteziensis from the Port of Mazatlán, Sinaloa, Mexico.
Trace metal seasonal variations have been reported for several species of molluscs (Bryan 1973). The causes of seasonal fluctuations have been associated with the food supply, changes in the run-off of particulate metal to the sea, due to high precipitation (Fowler and Oregioni 1976), variations related to the reproductive cycle (Latouche and Mix 1981; Paéz-Osuna and Marmolejo-Rivas 1990 b).
In stations 1 and 2, the peak of Mn was determined in July and May, respectively. According to Frías-Espericueta et al. (1997) in these months C. iridescens was in the maturation phase and close to spawning. Marina and Enzo (1983) found in Donax trunculus that Mn plays an important role in those phases prior to spawning, possibly as an enzymatic catalyst (Galtsoff 1964). Watling and Watling (1976) found in Choromytilus meridionalis that the trace metal distribution changes as a function of growth and maturation.
In the present work, essential metals were accumulated in amounts higher than non-essential metals. This order suggests that C. iridescens accumulates heavy metals according to the biological effect of metals and/or the concentration in the surrounding seawater. Páez-Osuna and Marmolejo-Rivas (1990a) found that, when the levels of trace metals in the oyster Crassostrea corteziensis were correlated with those in the particulate fraction of the month immediately preceding, a significant relationship was obtained for several metals. This is the result of specific mechanisms as uptake, transport, storage and excretion of metals (Simkiss and Mason 1983, Phillips and Rainbow 1989).
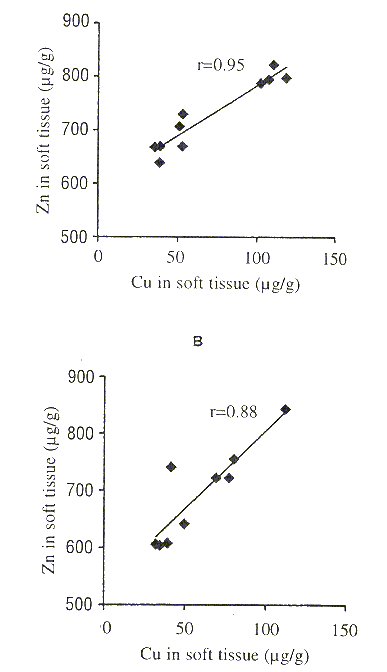
Fig. 1. Relationship between Zn and Cu (µg/g, dry weight) in the soft
tissue of the oyster Crassostrea iridescens along study period. A: station 1; B: station 2.
Zn showed the highest concentrations. Generally, oysters have been considered as strong accumulators of zinc, and these organisms accumulate high concentrations of zinc in detoxified granules (George et al. 1978). With regard to Cu, Eisler (1981) observed that some species could exhibit high Cu concentrations due to the proximity to anthropogenic source or to biotic/abiotic factors capable of modifying Cu uptake and retention. Rainbow (1988), and Depledge and Rainbow (1990), found that heavy metals are taken up and accumulated by marine invertebrates to tissue and body concentrations usually much higher on a wet weight basis than concentrations in the surrounding seawater. Metal accumulation strategies fall along a gradient from the strong accumulation of all metal taken up, to the regulation of the body metal concentration to an approximately constant level; by balancing metal excretion/uptake (Rainbow 1993). Zn, Fe, Cu, and Mn are biologically essential and play an important role as cofactor in enzymatic processes (Singh and Steinnes 1994).
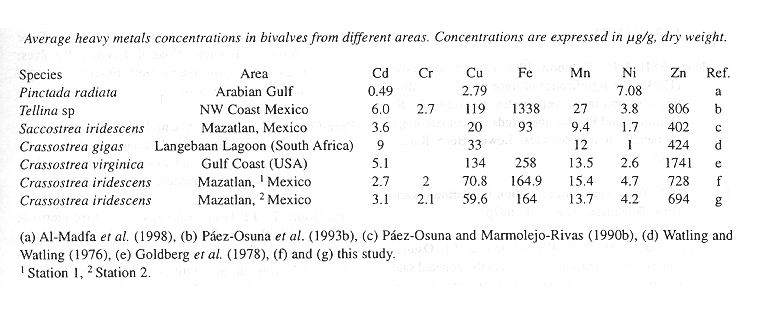
Comparing metal concentrations in oysters from this study with other bivalves collected in other zones of the world (Table 4), our values are lower than those reported by Goldberg et al. (1978) for a polluted zone (except for Ni), besides metal concentrations of this study are slightly higher than those of Langebann lagoon (South Africa), an area relatively unpolluted with respect to trace metals, and according to Watling and Watling (1976), these values represent near-background levels and that metal uptake is "normal". However, and according to Rainbow (1993), defining a background concentration for a metal is too difficult, given the role of biological variables.
A general conclusion is that urban discharges in the coastal zone in front of Mazatlan city, have little or no heavy metal concentrations dissolved or particulated. This work requires future investigations to analyze urban discharges. However, the analysis of total coliform feces (Ortiz-Arellano, in prep.) revealed higher values in station 1 than station 2.
Acknowledgments
The authors thank H.M. Zazueta-Padilla for his help during the analytical work and Marcos Merino for English corrections. We gratefully acknowledge the anonymous reviewers for their thoughtful comments.
Resumen
Dos poblaciones de Crassostrea iridescens fueron muestreadas en la zona costera de la ciudad de Mazatlan y en una zona libre de actividad antropogénica. Los bivalvos fueron colectados de febrero de 1992 a febrero de 1993. Concentraciones de Cd, Co, Cr, Cu, Fe, Mn, Ni y Zn fueron determinados por espectrofotometría de absorción atómica. Los valores medios anuales en la estación de muestreo de la ciudad de Mazatlán fueron mayores en Co, Cu, Fe, Mn, Ni y Zn que los determinados en la zona libre de actividad antropogénica; sin embargo, las diferencias no fueron significativas al nivel de confianza del 95%. Una conclusión es que las descargas urbanas en la zona costera en frente de Mazatlán no tiene, o tiene, bajas concentraciones de metales pesados disueltos o particulados.
References
Al-Mafda, H., M.A.R. Abdel-Moati & F.H. Al-Gimaly. 1998. Pinctada radiata (Pearl Oyster): A Bioindicator for Metal Pollution Monitoring in the Qatari Waters (Arabian Gulf). Bull. Environm. Contam Toxicol. 60: 245-251. [ Links ]
Anonymous, 1980. The International Mussel Watch. National Academy of Science, Washington D.C. [ Links ]
Anonymous, 1985. Intercalibration of analytical methods on marine environmental samples: Trace elements measurements on mussel homogenate. International Atomic Energy Agency, Report No. 26, Monaco. [ Links ]
Boyden, C.R. 1974. Trace metals content and body size in molluscs. Nature 251: 311-314. [ Links ]
Bryan, G.W. 1973. The occurrence and seasonal variation of trace metals in the scallops Pecten maximus (L) and Chlamys opercularis (L). J. Mar. Biol. Ass. U. K. 53: 145-166. [ Links ]
Depledge, M.H. & P.S. Rainbow. 1990. Cited by Rainbow (1993). The significance of trace metal concentrations in marine invertebrates p. 3-24. In R. Dallinger and P.S. Rainbow (eds.). Ecotoxicology of metals in invertebrates. Lewis, Boca Raton, Florida. [ Links ]
Eisler, 1981. Trace metal concentration in marine organisms. Pergamon, New York, 687p. [ Links ]
Frías-Espericueta, M.G., F. Páez-Osuna & J.I. Osuna-López. 1997. Seasonal changes in the gonadal sate of the oysters Crassostrea iridescens and Crassostrea corteziensis (Filibranchia: Ostreidae) in the Northwest coast of Mexico. Rev. Biol. Trop. 45: 1061-1066. [ Links ]
Fowler, S.W. & B. Oregioni. 1976. Trace metals in mussels from the NW Mediterranean. Mar. Pollut. Bull. 7: 26-29. [ Links ]
Galtsoff, P.S. 1964. The American Oyster Crassostrea virginica Gmelin. Fish. Bull. Fish and Wild life Some. Government Printing Office, Washington, D.C. 480p. [ Links ]
George, S.G., B.J.S. Pirie, A.R. Cheyne, T.L. Coombs & P.T. Grant. 1978. Detoxification of metals by marine bivalves: an ultrastructural study of the compartmentation of copper and zinc in the oyster Ostrea edulis. Mar. Biol. 45: 147-156. [ Links ]
Goldberg, E.D., M. Koide, V. Holdge, A.R. Flegal & J. Martin. 1983. U.S. mussel watch: 1977-1978 results on trace metals and radionuclides. Estuar. Coast. Shelf. Sci. 16: 69-93. [ Links ]
Keen, A.M. 1971. Sea Shells of Tropical West America: Marine mollusks from Baja California to Peru. Stanford University, Stanford, California, 356p. [ Links ]
Latouche, Y.D & M.C. Mix. 1981. Seasonal variation in soft tissue weights and trace metal burdens in the Bay Mussel, Mytilus edulis. Bull. Environ. Contam. Toxicol. 27: 821-828. [ Links ]
Marina, M. & O. Enzo. 1983. Variability of zinc and manganese concentrations in relation to sex and season in the bivalve Donax trunculus. Mar. Poll. Bull. 4: 342-346. [ Links ]
Miller, J.C. & J.N. Miller. 1987. Statistics for analytical chemistry. Wiley; New York. [ Links ]
Moody, J.R. & R.N. Lindstrom. 1977. Selection and cleaning of plastic containers for storage trace element samples. Anal. Chem. 49: 2264-2267. [ Links ]
Páez-Osuna, F. & C. Marmolejo-Rivas. 1990 a. Trace Metal in Tropical Coastal Lagoon Bivalves; Crassostrea corteziensis. Bull. Environ. Contam. Toxicol. 45: 538-544. [ Links ]
Páez-Osuna, F. & C. Marmolejo-Rivas. 1990 b. Occurrence and seasonal variation of heavy metals in the oysters Saccrostrea iridescens Bull. Environ. Contam. Toxicol. 44: 129-134. [ Links ]
Páez-Osuna, F., J.I. Osuna-López, G. Izaguirre-Fierro & H.M. Zazueta-Padilla. 1993a. Heavy metals in oysters from a subtropical coastal lagoon associated with an agricultural drainage basin. Bull. Environ. Contam. Toxicol. 50: 696-702. [ Links ]
Páez-Osuna, F., J.I. Osuna-López, G. Izaguirre-Fierro & H.M. Zazueta-Padilla. 1993b. Heavy metals in clams from a subtropical coastal lagoon associated with an agricultural drainage basin. Bull. Environ. Contam. Toxicol. 50: 915-921. [ Links ]
Páez-Osuna, F., M.G. Frías-Espericueta & J.I. Osuna-López. 1995. Trace metals concentrations in relation to season and gonadal maturation in the oyster Crassostrea iridescens. Mar. Environ. Res. 40: 19-32. [ Links ]
Phillips, D.J.H. & P.S. Rainbow. 1989. Strategies of trace metal sequestration in aquatic organisms. Mar. Environ. Res. 28: 207-210. [ Links ]
Phillips, D.J.H. & P.S. Rainbow. 1993. Biomonitoring of trace aquatic contaminants. Elsevier, New York. 371p. [ Links ]
Rainbow, P.S. 1988. The significant of trace metals concentrations in decapods. Symp. Zool. Soc. London, 59: 291-299. [ Links ]
Rainbow, P. S. 1993. The significant of trace metals concentrations in marine invertebrates, p. 3-24. In R. Dallinger & P.S. Rainbow (eds.) Ecotoxicology of metals in invertebrates. Lewis, Boca Raton, Florida. [ Links ]
Simkiss, K. & A.Z. Mason. 1983. Metal ions: metabolic and toxic effects, p. 102-164. In P.W. Hochachka (ed.). The mollusca. Academic, New York. [ Links ]
Singh, B.R. & E. Steinnes. 1994. Soil and water contamination by heavy metals, p. 233-272. In R. Lal & B.A Stewart (eds.) Soil processes and water quality. Lewis, Boca Raton, Florida. [ Links ]
Szefer, P., K. Szefer, J. Pewpkowiak, B. Skwarzec, R. Bojanowski & E. Holm. 1994. Distribution and coassociations of selected metals in seals of the Antartic. Environ. Poll. 83: 341-349. [ Links ]
Watling H.R. and R.J. Watling. 1976. Trace metals in Choromytilus meridionalis. Mar. Pollut. Bull.7: 91-94. [ Links ]
1 Facultad de Ciencias del Mar, Universidad Autónoma de Sinaloa, Apdo. Postal 610, Mazatlán, Sinaloa,
82000, México. Fax: + 69 82-86-56. E-mail: friasm@ola.icmyl.unam.mx
2 Posgrado en Ciencias del Mar y Limnología, ICMyL, Estación Mazatlán, Universidad Nacional Autónoma de
México.