Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.46 n.4 San José Dec. 1998
Abstract
A comparison was made of the gametophytic development of Phlebodium araneosum, P. pseudoaureum and P. decumanum. Spores were collected from several Mexican localities ranging in altitude from 30 to 2300 m and sown on agar with mineral salts. Similarities in germination type and prothallial development were observed. Adult gametophytes are cordate-spathulate to cordate-reniform; gametangia are of the tipical type of the common leptosporangiate ferns. All three species share similar morphogenetic features of the gametophytes, and these do not lend additional characteristics to support the distinction of Phlebodium from Polypodium, Microgramma and Niphidium, as regard the sexual phase.
Key words
Gametophyte, México, Microgramma, morphogenesis, Niphidium, Phlebodium, Polypodiaceae, Pteridophyta.
Phlebodium differs from areolated species of Polypodium by the sori ocurring on the junction of double included veins in the areoles, whereas the sori in Polypodium are terminal on single included vein.
The genus Phlebodium comprises epiphytic to terrestrial plants (Mickel and Beitel 1988), with three species in the tropical and subtropical areas of Mexico: Phlebodium araneosum (M. Martens and Galeotti) Mickel and Beitel, P. pseudoaureum (Cav.) Lellinger and P. decumanum (Willd.) J. Sm. The three species are widely distributed and grow in Pinus-Liquidambar-Quercus forests, Pinus-Quercus forests, tropical evergreen forests and mountain rain forests. Phlebodium araneosum and P. pseudoaureum are epiphytic or terrestrial, in altitudes ranging from 600 to 2,500 m; P. decumanum is a typical epiphytic plant on palms, at altitudes from 200 to 500 m (Mickel and Beitel 1988).The morphological characteristics distinguishing the three species are the sori present in either one or several rows and the absence or presence of scales and trichomes in the leaf blade. P. decumanum has 3-7 series of sori between the costa and the margin, while P. araneosum and P. pseudoaureum (P. areolatum fide Mickel and Beitel 1988) have one series. P. araneosum has scales in the abaxial surface of the lamina, and the lamina of P. decumanum and P. pseudoaureum is glabrous.
The morphological features of development from the spore to the young sporophyte in the three species are presented, in order to assess whether there are interspecific differences in the sexual phases of the studied species.
Previous studies have been conducted on the sexual phase of Phlebodium aureum (Nayar and Kaur 1971). There are also studies on fertilization and embryo development of that species (Ward 1954a, 1954b, Ward and Wetmore 1954). Other studies on Indian taxa allied to Phlebodium are those of Nayar (1954, 1962), Stokey (1959), Atkinson and Stokey (1970). Studies on development of antheridia and antheridial dehiscence have been conducted by Davie (1951) and Hartman (1931).
Materials and Methods
Spores were collected from different individuals at the localities shown in Table 1. Leaves with mature sporangia were placed in paper envelopes and sealed to prevent contamination and to allow for the sporangia to open. Spores were obtained by sifting the envelopes contents through a sieve (# 200) with a 0.074 mm mesh (Reyes and Pérez-García 1994, Mendoza et al. 1996-1997).
| | | | | |
| Phlebodium araneosum (M. Martens & Galeotti) Mickel & Beitel | AMR-107 | Km 504 México-Oaxaca Hgy., Oaxaca | BE, 2010 m | 11-IX-1995
|
| IRJ-1B | Rincón de Ugarte, Tejupilco de Hidalgo, México | BE, 1500 m | 08-VIII-1993 | |
| BPG-1030, 1031 | Tuxtepec, Oaxaca | SAP, 930 m | 09-IX-1995 | |
| Phlebodium pseudoaureum (Cav.) Lellinger | AMR-57 | Cerro de Iztapalapa, Mpio. Tepoztlán, Morelos | BPE, 2225 m | 13-IX-1993 |
| BPG-1012 | Pedregal de de San angel, Cd. Universitaria, Distrito Federal | SBC, 2300 m | 14-IX-1993 | |
| IRJ-68 | Cañon de Iscahuasco, Tetela de Ocampo, Puebla | BE, 1580 m
| 11-XI-1994 | |
| IRJ-82
| Behind Ometepetl, Tetela de Ocampo., Puebla. | BPE, 1830 m | 12-XI-1994
| |
| Phlebodium decumanum (Willd.) J. Smith | BPG-1032, 1033 | Tuxtepec, Oaxaca | SAP, 100 m | 09-IX-1995 |
| BPG-1036 | Tuxtepec, Oaxaca | SAP, 30 m | 09-IX-1995 | |
| IRJ-101, 102 | Tuxtepec, Oaxaca | SAP, 85 m | 10-IX-1995 |
- Abbreviations: AMR, Aniceto Mendoza Ruíz; BPG, Blanca Pérez-García; IRJ, Irma.
Reyes Jaramillo; BE, oak forest; BPE, pine-oak forest; SAP, tropical evergreen forest;
SBC, tropical deciduous forest.
Spores for each species were sown without previous sterilization in 35 petri dishes (5 cm across) in Thompson medium (Klekowski 1969, Pérez-García 1989) to a density of 80-150 spores/cm2. Cultures were grown at room temperature (25-30 C); and 75 watt (Luz de Día Solar brand t 38/ A 1D, Slimline 75 v) lamps were used to provide light with a 12 h/12 h photoperiod. Photomicrographs were taken from live specimens.
Result
Spores: In all three species, spores are monolete, ellipsoid, and golden in appearance. The exospore is bilayered and the perispore is usually thin (Erdtman and Sorsa 1971, Tryon and Lugardon 1991) and type 1 (Lloyd 1981). The spores of P. araneosum and P. pseudoaureum are 40-(45)-48 µm in diameter, with a roughly papillated surface (Fig. 1). The papillae are blunt and irregular in size (2-5) and the laesura ranges 30-35 µm; the spores of P. decumanum are 36-(38)-40 µm in diameter, and the surface is verrucose; the verrucae are 2.5 µm high, and have an irregular distribution pattern; laesura ranges from 25-30 µm in length (Fig. 2).
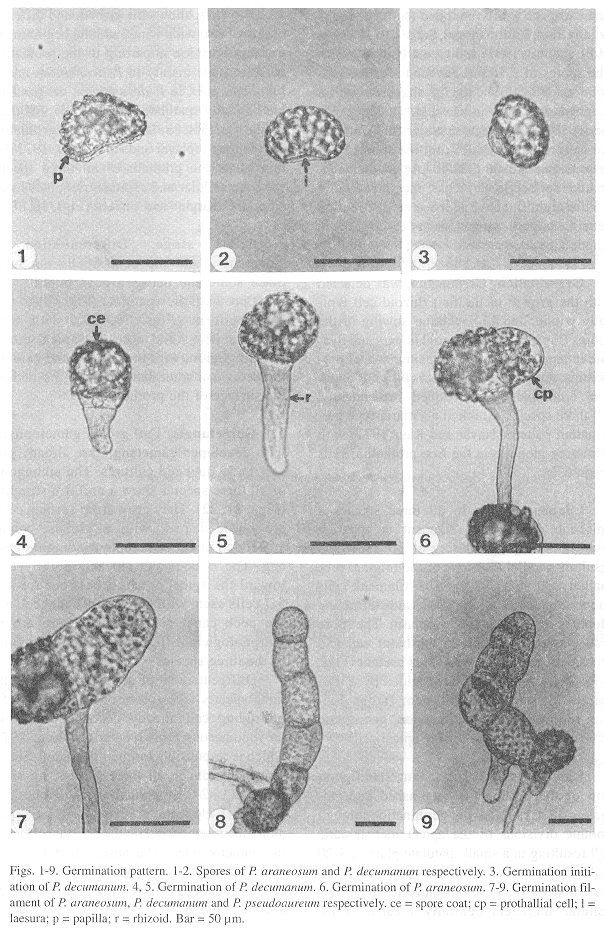
Germination: Germination was detected with the growth of the first rhizoid cell typically within 7 to 15 days after sowing of the spores (Figs. 3-5). The rhizoid is long and hyaline in appearance with a large number of proplastids. A globose prothallial cell with abundant chloroplasts was formed subsequently. All three species exhibited a Vittaria-type germination pattern (Nayar and Kaur 1971) with refracting globules in the first prothallial cells (Figs. 6,7).
Filament stage: In all three species a uniseriate germination filament is apparent after 12-30 days, consisting of 2-7 cells as a result of parallel divisions of the original prothallial cell (Figs. 7-9). The filament cells show abundant chloroplasts and refracting globules scattered in the cytoplasm. The spore wall was still attached to the basal cell and there were one to three rhizoids present (Figs. 7-9). The terminal cell was divided by a wall parallel to the axis of the filament; further division follows an oblique direction, giving rise to an obconic or meristematic apical cell.
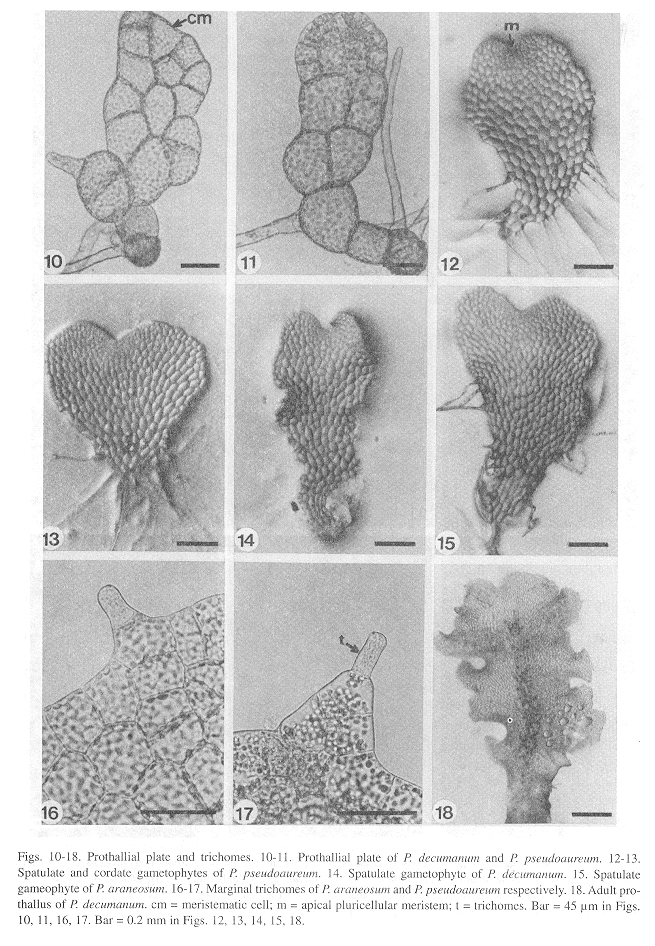
Laminar stage: The laminar configuration of the prothalli is established approximately 30 days following germination through oblique divisions of the apical meristematic cell resulting in a small spatulate plate of 8-20 cells (Figs. 10, 11). Within 40-60 days, the meristematic cell develops into an apical pluricellular meristem centrally located (Figs. 12,13), which allows the growth and differentiation of two wide wings and the beginning of a central cushion imparting to the prothalli a cordate shape mainly in P. araneosum and P. pseudoaureum; in P. decumanum the prothalli are basically spatulate (Figs. 14,15). All three species exhibit a Drynaria-type prothallial development (Nayar and Kaur 1969, 1971). At this stage, the prothalli of all three species show unicellular and capitate trichomes located on the margins and surface (Figs. 16,17).
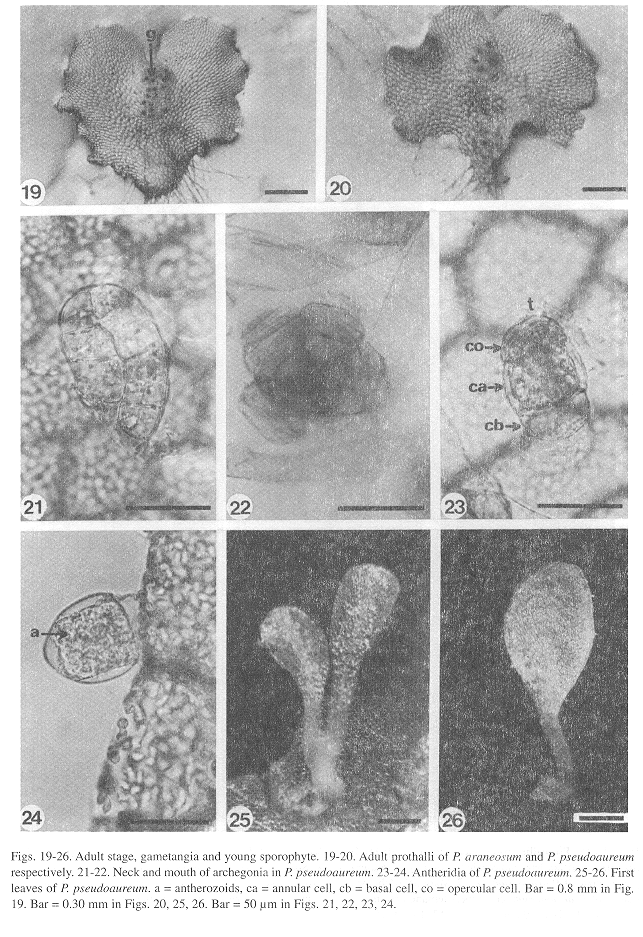
Adult stage: Differentiation of gametangia and the central cushion takes place sometime between days 60-120. The adult prothalli are spatulate (Fig. 18) to cordate-reniform (Figs. 19, 20), with wide undulate wings and a central apical meristematic region, with unicellular and capitate secretory trichomes at the margins and on the surface of the prothalli.
Gametangia: Full grown gametophytes with developed gametangia are already present in 55 days old cultures. The archegonia of all three species show a uniform structure (Figs. 21, 22). They grow from surface cells, are bottle-shaped, with a globose venter embedded in the gametophyte containing the egg. The neck is straight and slightly bent toward the apical notch, it has four rows of 5-7 cells each, with the ventral canal cell and the neck canal cells inside. There are no morphological differences in the archegonia of the three species.
Differentiation of the antheridia in the three species takes place on the surface of a protruding cell already described for other polypodiaceous ferns (Nayar and Kaur 1971) which is responsible for their pendulous shape. The antheridia in all three species are small (Figs. 23, 24), spherical or subglobose to somewhat elongate in shape and are found along the margins and on the ventral surface of the gametophytes. The basic structure of the antheridia consists of a basal cell with a flat to concave shape, a ring cell lining the antheridial cavity, and an opercular cell.
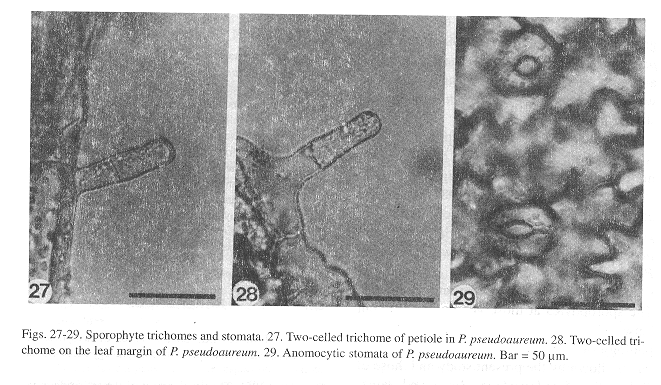
Sporophyte: The embryo development is similar to that of Phlebodium aureum (Ward 1954b). The first leaves, which appear within 150-400 days after sowing, in all three species (Figs. 25, 26), are simple, spatulate, and have a dichotomous vascular bundle. As in the gametophytes, the young sporophyte shows unicellular, short, capitate trichomes along the margin and on the leaf surface, although two-celled trichomes can also be found on the petiole (Fig. 27) and on the margin of the lamina (Fig. 28). Stomata are anomocytic (Van Cotthem 1973), with two occlusive cells surrounded by epidermal cells with wavy lateral walls (Fig. 29), and are present on the abaxial surface of the lamina, but absent on the adaxial surface.
Discussion
Comparative analysis of the gametophyte morphogenesis of these three species of Phlebodium showed similarities (Fig. 30). Spores are all monolete, ellipsoid, and golden although spores from P. decumanum are smaller (by about 10 µm), and show a less conspicuous ornamentation, with underdeveloped papillae as compared to the spores of P. araneosum and P. pseudoaureum.
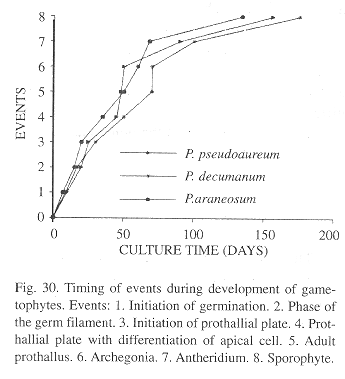
Differences in shape were evident in the gametophytic phase. In P. araneosum and P. pseudoaureum prothalli are mainly cordate, although sometimes reniform and with wide wings, whereas in P. decumanum, wings are shorter and spatulate.
Gametangia are of the leptosporangiate type in all three species, with no significant morphological differences among them.
According with Stolze (1981), there are several ideas about the circumscription of the genera segregated from Polypodium. Recent papers recognize the following genera: Polypodium, Microgramma, Niphidium, Campyloneurum, Phlebodium and Pleopeltis. The sporophytes at those genera are different between them, but the development and morphology of their gametophytes are very similar, as shown by the present study and those of Polypodium lepidotrichum (Reyes and Perez-García 1994), Niphidium crassifolium (Reyes et al. 1996) and Microgramma nitida (Ramírez and Pérez-García 1998).
The sexual phase of the species mentioned above and the species studied of Phlebodium share several characters as monolete ellipsoidal spores, with oil globules typical of Polypodiaceae s.str., germination and pattern of prothallial development, archegonia and antheridia typical of the leptosporangiate ferns and unicellular trichomes in margins and surfaces of the adult gametophytes.
Although there are differences in the shape of the adult gametophytes and the time apparition of the sporophytes those are not significative and do not lend additional support for the circumscription of the genera as regard their sexual phase.
Acknowledgements
The authors thank Laura E. Riba for the English translation and Robbin C. Moran for extremely valuable comments to the manuscript.
Resumen
Una comparación del desarrollo del gametofito de Phlebodium araneosum, P. pseudoaureum y P. decumanum fue hecha. Las esporas fueron sembradas en agar con sales minerales. Similitudes en el tipo de germinación y desarrollo protálico fueron observadas. Los gametofitos adultos fueron cordiforme-espatulados a cordiforme-reniformes; los gametangios son del tipo común de los helechos leptosporangiados. Las tres especies muestran caracteristicas morfogenéticas del gametofito similares y no a aden características adicionales que apoyen la distinción entre Phlebodium, Polypodium, Microgramma y Niphidium, por lo que se refiere a la fase sexual.
References
Atkinson, L.R. & A.G. Stokey. 1970. Gametophyte of Polypodium chnoodes. Phytomorphology 20: 364-367. [ Links ]
Davie, J.H.1951. The development of the antheridium in the Polypodiaceae. Amer.J. Bot. 38: 621-628. [ Links ]
Erdtman, G. & P. Sorsa. 1971. Pollen and spore morphology / Plant taxonomy, Pteridophyta, 302 pp, Almqvist and Wiksell, Stockholm. [ Links ]
Hartman, N.E. 1931. Antheridial dehiscence in the Polypodiaceae. Bot. Gaz. (Londres) 91: 252-276. [ Links ]
Klekowski, E. J., Jr. 1969. Reproductive biology of the Pteridophyta. III. A study of the Blechnaceae. J. Linn. Soc., Bot. 62: 361-377. [ Links ]
Lloyd, R.M. 1981. The perispore in Polypodium and related genera (Polypodiaceae). Canad. J. Bot. 59: 175-189. [ Links ]
Mendoza, A., B. Pérez-García, I. Reyes Jaramillo & M. Ricci. 1996-1997. Desarrollo del gametofito de Pteris berteroana (Pteridaceae: Pterideae). Rev. Biol. Trop. 44/45: 51-57. [ Links ]
Mickel, J.T. & J.M. Beitel. 1988. Pteridophyte Flora Of Oaxaca, Mexico. Mem. New York Bot. Gard. 46: 1-568. [ Links ]
Pérez-García, B. 1989. Morfogénes de gemetofitos de Cyatheaceae (Pterophyta: Filicales). tesis Doctoral. Fac. Ciencias. Universidad Nacional Autónoma de México, México, D.F. [ Links ]
Nayar, B.K. 1954. Studies in Polypodiaceae-II. Contributions to the morphology of Pseudodrynaria coronans (Wall.) C. Chr. Phytomorphology 4: 379-390. [ Links ]
Nayar, B.K. 1962. Morphology of spores and prothalli of some species of Polypodiaceae. Bot. Gaz. (Crawfordsville) 123: 223-232. [ Links ]
Nayar, B.K. & S. Kaur. 1969. Types of prothallial development in homosporous ferns. Phytomorphology 19: 179-188. [ Links ]
Nayar, B.K., & S. Kaur. 1971. Gametophytes of homosporous ferns. Bot. Rev. (Lancaster) 37: 350-351. [ Links ]
Pérez-García, B. 1989. Morfogénesis de gametofitos de Cyatheaceae (Pterophyta: Filicales). Tesis Doctoral. Fac. Ciencias, Universidad Nacional Autónoma de México, México, D. F. [ Links ]
Ramírez T. R. & B. Pérez-Garcia. 1998. Fase gametofítica de Microgramma nitida (J.Sm.) A. R. Sm. (Polypodiaceae s.str.). Rev. Biol. Trop. 43: 000-000- [ Links ]
Reyes, I. & B. Pérez-García. 1994. Morfología y estrategias reproductivas del gametofito de Polypodium lepidotrichum (Fée) Maxon (Polypodiaceae). Acta Bot. Mex. 28: 71-78. [ Links ]
Reyes Jaramillo, I., B. Pérez-García & A. Mendoza. 1996. Desarrollo del gametofito y del esporofito joven de Niphidium crassifolium (Filicales: Polypodiaceae s.str.). Rev. Biol. Trop. 44: 485-490. [ Links ]
Stolze, R. G. 1981. Ferns and fern allies of Guatemala. Fieldi. Bot., N. S., No.6: 358-359. [ Links ]
Stokey, A.G. 1959. Polypodium pectinatum and P. plumula – Polypodiaceae or Grammitidaceae?. Amer. Fern J. 49: 142-146. [ Links ]
Tryon, A.F. & B. Lugardon. 1991. Spores of the Pteridophyta: surface, wall structure, and diversity based on electron microscope studies. Springer, New York. 648 p. [ Links ]
Van Cotthem, W.R.J. 1973. Stomatal types and systematics. In: A. C. Jermy, J. A. Crabbe and B. A. Thomas (Eds.). The Phylogeny and classification of the ferns. Academic, New York. p. 59-71. [ Links ]
Ward, M. 1954a. Fertilization in Phlebodium aureum J. Sm. Phytomorphology 4: 1-17. [ Links ]
Ward, M. 1954b. The development of the embryo of Phlebodium aureum J. Sm. Phytomorphology 4: 18-26. [ Links ]
Ward, M. & R.H. Wetmore. 1954. Experimental control of development in the embryo of the fern Phlebodium aureum. Amer. J. Bot. 41: 428-434. [ Links ]
1Departamento de Biología, C. B. S., Universidad Autónoma Metropolitana. Ap. Postal 55-535. México, D. F. 09340. Fax: 7-24-46-88, e-mail: bpg@xanum.uam.mx














