Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Acta Médica Costarricense
On-line version ISSN 0001-6002Print version ISSN 0001-6012
Acta méd. costarric vol.60 n.2 San José Apr./Jun. 2018
Articles
New Delhi Metallo-β-lactamase in Enterobacteriaceae species isolated from hospitalized patients, Managua Nicaragua
1Trabajo realizado en Laboratorio de Biología Molecular “MA. Elmer Cisneros in memoriam”, Instituto Politécnico de la Salud, Universidad Nacional Autónoma de Nicaragua.Nicaragua
Bacterial infections due to Enterobacteriaceae and non-fermenting Gram negative are very frequent in patients hospitalized in underdeveloped countries, and they behave as multiresistant, worsening the health conditions of the patients. Antimicrobial resistance should be considered a public health problem, reports are alarming and on the rise in different countries; the therapeutic options have been reduced, due to the increase of mechanisms that microorganisms have developed to defend themselves against the “constant attack” to which they have been subjected by the antibiotics. The carbapenemases are enzymes that have developed the bacteria to hydrolyze the carbapenems, which are the last line of antibiotics for clinical use at this time, which has produced therapeutic failures. These have emerged as a potential problem for the health of the patients, so that controlling the infectious processes produced by the bacteria with this type of resistance is a challenge for any health unit, because they are highly disseminative.
With the appearance of the New Delhi metallo-β-lactamase, the situation is even more alarming, because microorganisms with this type of gene, are becoming more frequent, which leads to think that the mechanism by which they share this gene, are mobile genes that are easy to transfer or share between very close Gram negative bacteria.1-3
Carbapenems have the broadest spectrum of activity against Gram-negative microorganisms and are the most widely used in recent years as a result of the emergence of ESBL strains (extended-spectrum beta-lactamases); unfortunately, the bacteria have developed a new mechanism that neutralizes the action of carbapenems, becoming a public health problem, because they are characterized by being multiresistant.4
The New Delhi metallo-β-lactamase belongs to group B classification of Ambler, these bacteria are known to have a zinc ion cofactor in their structure, which is characterized by neutralizing all beta-lactams, except Aztreonam; is inhibited only by divalent cation chelating agents such as EDTA.4 Since its discovery in 2008 in India, in Klebsiella, the reports in different countries are constant, both in Klebsiella and in other Enterobacteriaceae; have been isolated in the European continent, also in the United States, Japan, Brazil, Canada and in Central America in 2011 Guatemala reports the finding of Klebsiella, 5 and in 2014, Costa Rica refers its first case of New Delhi.6 The reports are alarming in different journals, which has generated concern to the scientific community and organizations such as PAHO and WHO, for the impact on the clinical outcome of the patient. The strains that produce carbapenemase are a danger, and the treatment of these infections is a challenge for the doctor, which is why microbiological tests are of great help in evaluating the patient’s therapy. 5,7-12
Methods
A cross-sectional descriptive investigation was conducted from August 2015 to October 2016, where 249 strains were studied in hospitalized patients of the Nicaraguan German Hospital. Samples were taken from the Neonatal Intensive Care and Pediatric Intensive Care units, neonatology, medicine and surgery. The identification and resistance profile was performed in Vitek2 compact; resistance to carbapenems was defined when the minimum inhibitory concentrations of imipenem and meropenem were 2-4 μg / ml and ertapenem of 2 μg / ml. The phenotypic characterization was performed by Kirby Bauer using the triple disc synergy test, EDTA (10μg), imipenem (10μg) and meropenem (10μg). Quality control was established with the use of reference strains, Pseudomonas aeruginosa ATCC 27853, Escherichia coli ATCC 25922, Escherichia coli ATCC 35218, Klebsiella pneumoniae ATCC700603.5,13-15
DNA extraction. The strains analyzed and controls were grown on MacConkey agar, incubated for 18-24 hours at 37°C; three CFUs were taken from the culture and inoculated into cryovials containing 100μl of nuclease-free water; it was placed in a boiling water bath for 10 minutes; samples were allowed to cool on ice for 5 minutes, then centrifuged at 12,000 rpm for 5 minutes, and 80μl of the supernatant was removed; the concentration of DNA extracted in NanoDrop lite 2763 was determined.4
Genotypic detection of the New Delhi metallo-βlactamase. It was carried out by means of a polymerase chain reaction, using the nucleotide sequence: NDMF5-3 AGC ACA CTT CCT ATC TCG AC, NDMR5-3 GGC GTA GTG CTC AGT GTC, in which DNA was used 2,5μl, buffer 10X (2,5ul), Enhancer solution 5x (0,7μl), dNTP’s mix (40 mM) 0,5μl, Taq Polymerase 5U/ul (0,5 ul), Primer Forward 10nM (0,5ul), Primer Reverse 10nM (0,5ul), nuclease-free water (17,6μl), for a final volume 25μl.16
The amplification. The following amplification program was used: denaturation 94o C, for 5 minutes, followed by 35 cycles, 94o C for 30sec, hybridization 50o C for 30sec, amplification 72o C for 60seg, final extension 72o C for 10 minutes and final temperature of 4o C; the samples were analyzed in a Master Cycler, Eppendorf brand, model number 5341.5 Electrophoresis. The PCR product was evaluated on a 1.5% agarose gel with 0.5 μg/mL ethidium bromide; Electrophoresis was run at 120 volts for 50 minutes; the DNA bands were visualized in a camera with ultraviolet light and photographed. The above corresponded to a molecular weight of 512 bp. Figure 1.
Results
249 strains were analyzed, of which only 45 were resistant to carbapenems, for 18%. Of these, 43 were positive for the EDTA synergy test; 20 of the strains tested positive for metalloenzyme genes (IMP, VIM, SPM, SIM, GIM). However, 21 strains were not identified with any gene, although the synergy was positive, and from there emerges the idea of looking for the New Delhi gene.
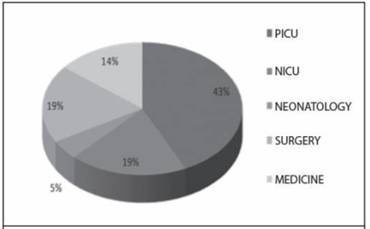
The New Delhi gene was found in 22 of the 43 positive samples for the synergy test. Of the 22 samples with New Delhi, most were isolates from Klebsiella pneumoniae (15 samples, 66%), followed by Escherichia vulneris with 3 samples (14%), and had isolation of the following bacteria: Escherichia coli, Providencia rettgeri, Pantoea agglomerans and Kluyvera cryocrescens. The frequency and distribution by service of New Delhi are reflected in figure 2, where it is observed that 62% of the isolations came from intensive care units of children, 19% of surgical services and 14% of medical services.
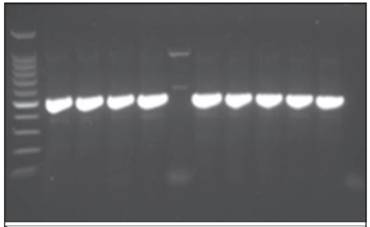
Figure 1 Electrophoresis in 1.5”agarose gel, New Delhi gene, Molecular Weight of 512 bp. Well 2 positive control, wells 3, 4, 5, 7, 8, 9, 10, 11, patient, Well 6 negative internal control, well 12 negative control PCR grade water.
Discussion
In Nicaragua, NDM is reported for the first time, and with an alarming percentage. Enterobacteria are often isolated with positive synergy test with EDTA, which means that metalloenzymes are mostly isolated; many publications report blaKPC. This behavior is particular to our country, perhaps due to the high percentage of NDM. Since its discovery in India, in Klebsiella, this gene has been identified in several countries, with rapid dispersion; in Nicaragua, the percentage of NDM is also alarming.
The NDM producing bacteria are capable of rapidly disseminating, becoming a serious problem for any health unit, due to nosocomial diseases and limited therapeutic options; the reports are very common in Klebsiella and Escherichia coli, as the most carriers, which can lead to having them in the extra-hospital environment or in various infectious processes.
Neonatal septicemia continues to be one of the main causes of nosocomial disease in our hospitals, and one of the main causes that could produce a tragic outcome. In our study 6 types of microorganisms carrying the NDM gene were found. Klebsiella was the main cause of sepsis with this gene, according to what is reported in the international literature. The 21 strains were multiresistant, with the only options of Colistin and Tigecycline.17,18
The findings of this study are a clear warning about the circulation of New Delhi strains that encode the resistance to carbapenems in hospitals in Nicaragua. It is essential to take this into consideration in clinical practice, given the drastic reduction of therapeutic options for patients with infections due to these strains. From our results, some containment measures to avoid dissemination is recommended, and provides relevant data to the units where the isolations are made, in terms of the resistance profile and the resistance gene that circulates.
Gratitude: the staff of the bacteriology laboratory of the Nicaraguan German Hospital, the teaching and research team. The study was carried out with funds for research projects (FRP) of the UNAN-Managua, granted to the main author MSc. Oscar Arbizú Medina, and executed by the Office of the Vice-Rector for Research and Graduate Studies.
REFERENCES
1. Poirel L, Bonnin RA, Boulanger A, Schrenzel J, Kaase M, Nordmann P. Tn125- Related Acquisition of blaNDM-Like Genes in Acinetobacter baumannii. Antimicrob Agents and Chemother. 2012; 56:1087-1089. [ Links ]
2. Amaya E, Cáceres M, Fang H, Torres Ramirez A, Palmgren A. C, Nord C. E, Weintraub A. Antibiotic Resistance Patterns in Gram-Negative and Gram- Positive Bacteria Causing Septicemia in Newborns in León, Nicaragua: Correlation with Environmental Samples. J Chemother. 2010; 22:25-29 [ Links ]
3. González-Escalante E, Vicente-Taboada, Champi-Merino, Soto-Pastrana, Flores-Paredes W, Lovera-García M, et al. Metalo-β-Lactamasas en aislamientos clínicos de pseudomona aeruginosa en Lima, Perú. Rev Peru Med Exp Salud Publica. 2013; 30:241-245. [ Links ]
4. Bush K, Jacoby GA. Updated functional classification of β-lactamases Antimicrobi Agents and Chemother. 2010; 54: 969-976. [ Links ]
5. Pasteran F, Albornoz E, Faccone D, Gomez S, Valenzuela C, Morales M, et al. Emergence of NDM-1-producing Klebsiella pneumoniae in Guatemala. J Antimicrobi Chemother. 2012; 67:1795-1797. [ Links ]
6. Muñoz-Acuña R, Méndez-Rodríguez JD, Villalobos-Vindas J. Bacteriemia por Kluyvera cryocrescens. Acta méd costarric. 2016; 58:38-39. [ Links ]
7. Struelens MJ, Monnet DL, Magiorakos AP, O’Connor FS, Giesecke J. New Delhi metallo-beta-lactamase 1-producing. Euro Surveill. 2010; 15: pii=19716. [ Links ]
8. Smyth , Kahlmeter G, Olsson Liljequist B, Hoffman B. Methods for identifying methicillin resistance in Staphylococcus aureus. J Hosp Infect. 2001;48:103- 107. [ Links ]
9. Na Kim M, Yong D, An D, Chung HS, Woo JH, Lee, K, et al. Nosocomial Clustering of NDM-1-Producing Klebsiella pneumoniae Sequence Type 340 Strains in Four Patients at a South Korean Tertiary Care Hospital. J Clin Microb. 2012; 50:1433-1436. [ Links ]
10. Dortet L, Poirel L, Nordmann P. Worldwide Dissemination of the NDM-Type Carbapenemases in Gram-Negative Bacteria. BioMed Research International. 2014. 12p doi.org/10.1155/2014/249856. [ Links ]
11. Padgett D, Luque MT, Rivera DM, Galindo C, Zepeda LM, Hernandez AL. Resistencia antimicrobiana en bacterias aisladas en el Instituto Hondureño de Seguridad Social. Rev Med Hondur. 2011; 79:117-121. [ Links ]
12. Labarca LJ, Araos BR. Resistencia antimicrobiana: Problema en aumento y soluciones escasas. Rev Chil Infect. 2009; 26(Supl 1): 8-9. [ Links ]
13. Smyth, Kahlmeter G, Olsson Liljequist B, Hoffman B. Methods for identifying methicillin resistance in Staphylococcus aureus. J Hosp Infect. 2001;48:103- 107. [ Links ]
14. CLSI. Performance Standards for Antimicrobial Susceptibility Testing. 27th ed. CLSI supplement M100. Wayne, PA: Clinical and Laboratory Standards Institute; 2017. [ Links ]
15. Nicola FG, Nievas J, Smayevsky J. Evaluación de diversos métodos fenotípicos para la detección de carbapenemasas KPC en Klebsiella pneumoniae. Rev. Argent. Microbiol. 2012; 44:290-302. [ Links ]
16. Smyth, Kahlmeter G, Olsson Liljequist B, Hoffman B. Methods for identifying methicillin resistance in Staphylococcus aureus. J Hosp Infect. 2001;48:103- 107. [ Links ]
17. Rahman M, Shukla SK, Prasada K, Ovejeroc C, Patia, B , Tripathia, et al. Prevalence and molecular characterisation of New Delhi metallo-β- lactamases NDM-1, NDM-5, NDM-6 and NDM-7 in multidrug-resistant Enterobacteriaceae from India. Int J Antimicrob Agents. 2014;44:30-37. [ Links ]
18. Amaya E, Cáceres M, Fanga H, Torres Ramirez A, Cathrin Palmgrena A, Norda E, Weintraub Andrej. Extended-spectrum β-lactamase producing Klebsiella pneumoniae in a Neonatal Intensive Care Unit in León, Nicaragua. Int J Antimicrob Agents. 2009; 33:386-387. [ Links ]
Work done in
3The Molecular Biology Laboratory “MA. Elmer Cisneros in memoriam,” Polytechnic Institute of Health, National Autonomous University of Nicaragua.
Abbreviations:
4NDM, New Delhi Metallo-β-lactamase; NICU, Neonatal Intensive Care Unit; PICU, Pediatric Intensive Care Unit.
Conflict of interest:
Received: August 17, 2017; Accepted: January 31, 2018











 text in
text in