Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
Acta Médica Costarricense
On-line version ISSN 0001-6002Print version ISSN 0001-6012
Acta méd. costarric vol.58 n.1 San José Jan./Mar. 2016 Epub Mar 01, 2016
Original
Candidemia and it’s Epidemiology at Hospital México
1,3,4Infectious Diseases Departmente, Hospital Mexico, CCSS
2Pharmacoepidemiology Department, CCSS
5.6Division of Bacteriology, Clinical Laboratory, Hospital Mexico, CCSS
Invasive infections by Candida species have become a major cause of nosocomial infection, particularly in Intensive Care Units.1-3 Overall, Candida species account for almost 80% of all pathogenic fungi causing nosocomial infections. Candidemia is the most common form of invasive disease by this species, representing about 15% to 20% of all bloodstream infections in hospitals in developed countries.4-7
In the last two decades significant changes have occurred in the epidemiology of candidemia, especially related to the frequency of the species involved. There has been a decrease in the incidence of Candida albicans from 90% in the 70, up to 50% or less at present, because of an increase of other species of non-albicans Candida, such as C. glabrata, C. tropicalis, C. parapsilosis and C. krusei.7 The incidence of candidemia by C. parapsilosis has grown progressively and recent studies place it as the third most common cause of candidemia in the world.8 In addition, the infection leads to high attributable mortality, prolonged hospital stay and increased costs of care.9
Despite the growing importance of this disease, there are only two national publications; both from the Hospital San Juan de Dios. The first analyzed the period from 1996 to 1998 and showed that C. albicans was the most frequent, 10, but in the second study, conducted five years later, C. parapsilosis 11 was ranked first.
The purpose of this study was to characterize the epidemiology of candidemia in the Hospital Mexico, another center of the Costa Rican social security, in order to expand the existing knowledge about the behavior and dynamics of this infection in the country.
Methods
This is a retrospective observational study conducted at the Hospital Mexico, part of the Social Security System in San Jose, Costa Rica. It include all patients over 17 yearsold during the period between January 2007 and December 2010, that had, at least one blood culture positive for Candida spp.; only the first episode of candidemia detected per patient was analyzed. The investigation began getting a list of all blood cultures reported as positive by any Candida species during this period, the database Observa®, from the bacteriology identification system VITEK®2 (bioMérieux), of the Bacteriology Division of the Clinical Laboratory.
From this information, the medical records were reviewed and the following variables were studied: sex, age, date of admission to hospital, hospitalization area, use of central venous catheter (CVC), catheter-days until the time of blood culture, parenteral nutrition, mechanical assisted ventilation at diagnosis of candidemia, antifungal treatment, and mortality in the first 30 days of candidemia. The Charlson comorbidity index 12.13 (ICC), adjusted for age was determined for each patient. Patients excluded from the study were those where he could not get all of the clinical variables studied, or the clinical record was not found.
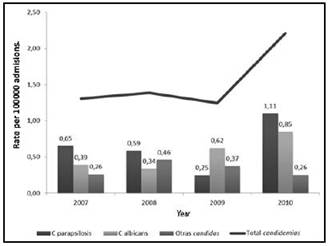
Figure 1: Cumulative incidence of candidemia for 1000 admissions, according to year and species. Mexico Hospital, 2007-2010.
Quantitative variables were described using measures of central tendency and dispersion. The comparison of averages was performed by analysis of variance or the Kruskal - Wallis test, when it was not valid to apply the first test. The qualitative variables were described using frequency distributions; and, confidence intervals at 95% for the relative frequencies were calculated, and the proportions were compared using hypothesis tests (Student t-test) by normal approximation. A significance level of 0.05 was established. The cumulative incidence per year was calculated per 1000 discharges and its behavior over time was plotted. The associated factors were analyzed for the presence of C. parapsilosis relating to other Candida species, using for these the calculation of odds ratio (OR) with their respective 95% confidence intervals (CI 95%). In addition, a Kaplan-Meier analysis was performed and Cox regression was used to assess survival at 30 days of patients with C. parapsilosis, compared with other species of Candida and other variables.
Microsoft Office Excel 2010 and SPSS version Version 18 were used for the statistical analysis.
The study was reviewed and approved by the Local Ethics Committee of the Hospital Mexico.
Results
145 cases of candidemia were identified, of which 136 different episodes of candidemia were analyzed and 9 cases were excluded because it was not possible to obtain all the variables studied.
Candida spp. was the sixth more frequent among the recovered microorganisms in blood during this period, corresponding to 5% of all germs isolated from blood cultures. All episodes were nosocomial candidemia.
An average of 34 different episodes of candidemia per year (range 27-51) and a cumulative incidence of candidemia for the period, from 1.1 cases per 1000 discharged patients was obtained; year cumulative incidence was 0.9, 1.0, 0.9 and 1.7, respectively (Figure 1). The increased incidence of 2010 compared to previous years, was statistically significant (p = 0.01).
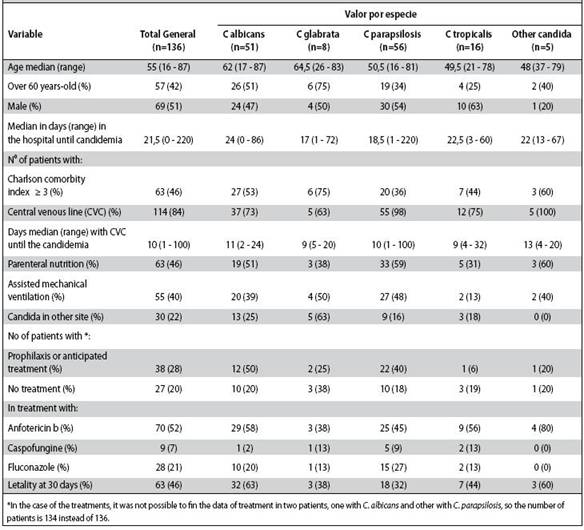
The non-albicans Candida species accounted for 62% of the yeasts isolated in the episodes of candidemia studied. C. parapsilosis was the most frequently isolated species (41%), followed by C. albicans (38%), C. tropicalis (12%), C. glabrata (6%); other species accounted for 3% (Table 1).
The individual analysis of the information collected per year (Figure 1) revealed that C. parapsilosis was the most common species in 3 of the 4 years of the study (44% in 2007, 40% in 2008, 51% in 2010). In 2009 this was reversed, C. albicans frequency increased to 50%, and C. parapsilopsis was reduced to 21%. The difference observed between 2009 and 2010 in the case of C. parapsilosis was statistically significant (p = 0.023).
Table 1: Demographic, clinical and therapeutic characteristics of patients with candidemia. Hospital México. 2007 - 2010
The distribution per month showed a decrease of candidemia cases of C. parapsilosis throughout 2009, with a rise from January 2010; on the contrary, the other species had a stable behavior over the 4 years (Figure 1).
42% of individuals aged 60 years or older, with a median age of 55 years (range 16-87 years) there was no statistically significant difference between the age of patients and Candida species (Table 1).
Half of the patients (51%) was hospitalized in different surgical specialties; medical specialties accounted for 38% of cases and intensive care 8%, the latter having a candidemia rate of 5.4 cases per 1000 discharged patients; the remaining 3% of patients was detected in emergency departments and obstetrics and gynecology.
When evaluating the comorbidity of patients, it was determined that 46% of them had an ICC of 3 or more points (estimated 10-year survival of 77% or less). 75% of patients with C. glabrata had an ICC greater or equal to 3, while in patients with C. parapsilosis this percentage was 36% (Table 1). Patients had a diverse variety of pathological conditions, but the most frequent were cancer (32%), postoperative intestinal fistula (29%), diabetes mellitus (21%) and multiple trauma (16%). No statistically significant association between these conditions and the species of Candida isolated in blood was found.
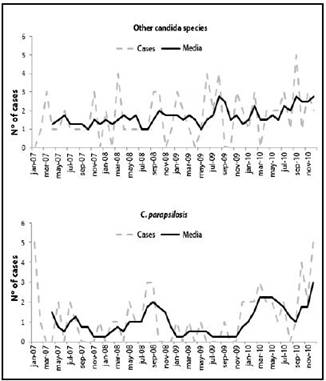
Figura 2: Temporal distribution of the candidemia cases for C. parapsilosis and other species. Hospital México. 2007-2010.
The median number of days of hospitalization until candidemia was 21.5 days, with no statistically significant difference between Candida species (Table 1). When analyzing this variable according to the hospitalization department, the median stay in the surgical specialties was higher (25 days) than in the medical (15.5 days) or in intensive care (10 days), a difference that was statistically significant (p <0.001).
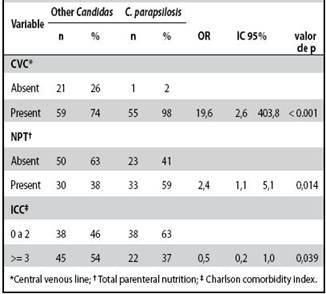
84% of patients had a CVC when candidemia occurred; in individuals with C. parapsilosis this occurred in 98%, while in patients with C. albicans this percentage was 73%.
A strong association between the presence of candidemia and CVC was observed for C. parapsilosis, compared with other species of Candida (OR = 19.6, 95% CI 2.6 to 403.8, p <0.001) (Table 2). By studying the number of days between the placement of the CVC and the diagnosis of candidemia, a significant difference between Candida species (Table 1) was found.
46% of patients were receiving parenteral nutrition at the time of candidemia (Table 2) and a greater likelihood that the species associated was C. parapsilosis (OR = 2.4, 95% CI 1.1 to 5.1, p = 0.014).
In relation to the treatment of candidemia, 20% of patients did not receive antifungal treatment, 28% had prophylaxis or treatment was initiated due to clinical suspicion without blood culture results (Table 1), and the remaining 52% received treatment once the report of the blood culture was obtained. The most commonly used drug was amphotericin B followed by fluconazole and caspofungin, there was no difference in the choice for each species (Table 1) or associated comorbidity (ICC). Those who did not receive antifungal therapy had a significantly higher risk of death at 3, 7, 30 days, when compared with patients who did receive antifungal (HR = 34.7, p <0.001; HR = 17.7, p < 0.001; HR = 3.1, p <0.026, respectively). Of the 18 patients who received no treatment and died, 13 they died before or the day of the blood culture report with Candida.
The 30-day mortality was 0.52 per 1,000 discharges, equivalent to 46% of patients. Candida species with longer survival were C. parapsilosis, with 70.4% (95% CI 56.3 to 80.7%) (Figure 3); conversely, C. albicans had the lowest survival rate, with 38.8% (95% CI 25.3 to 52.0%). By Cox regression we found that patients with C. parapsilosis had a lower chance of dying in relation to other species of Candida (HR = 0.5, 95% CI 0.3 to 0.8, p = 0.01) (Figure 2). People over age 60 had a lower survival (HR = 2.2, 95% CI 1.3 to 3.7, p = 0.004). Factors such as the ICC, department in which they were hospitalized, CVC, parenteral nutrition, mechanical ventilation and presence of Candida spp. in another anatomical site, did not demostrate to have a statistically significant association with mortality at 30 days.
Discussion
In a similar way to other centers, the Candida species are among the most frequently isolated microorganisms from blood cultures14 The rate of candidemia in the hospital is within the range reported in the literature (0.2 -. 2.8 cases / 1000 expense)15 and is similar to other Latin American hospitals,16,17 but higher than described in hospitals in developed countries.16
Contrary to the findings of other researchers18,19 most cases came from surgical specialties, not intensive care. This situation could be due to factors specific to the Hospital, such as the large number of high-risk patients who are cared outside of this unit and with the size and complexity of the Hospital (2% of the beds).
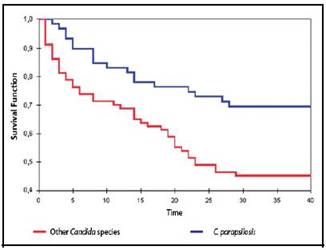
Figure 3: Kaplan - Meier estimation on survival of patients with candidemia for C.parapsilosis and other species. Hospital Mexico. 2007-2010.
Table 2: Univariate analysis of risk factors for candidemia presented by C. parapsilosis. Hospital México. 2007 - 2010
Candida species other than C. albicans accounted for 62% of cases of candidemia in the Hospital Mexico, which is consistent with that reported in other centers in Latin America and the world16,17. 20-22 However, when analyzing the incidence by species, surprisingly, C. parapsilosis was the most common cause of candidemia, and their predominance in 3 of the 4 years analyzed, displayed as an endemic problem in the Hospital. This phenomenon differs from that described in most reports in the literature, including Latin America,16,17,21,25 where C. albicans is the most common species and C. parapsilosis takes second or third place, with an incidence ranging between 10% and 25%.11,23,24 There are isolated reports in the international literature where C. parapsilosis is the most common species,from 8,26 to 29 with isolated outbreaks in specific departments of a hospital, or in pediatric hospitals.
The significant decrease in cases of candidemia by C. parapsilosis in 2009, requires a detailed analysis in the future the factors involved in this phenomenon, which would help determine the reasons why the species has been the most common in the Hospital Mexico, and could indicate the actions to be implemented to modify the endemic nature of this infection.
Our data indicate that the major gateway to the bloodstream of C. parapsilosis is the presence of a CVC, as a strong association between both was demonstrated, a hypotheses that coincides with those published by other investigators.26,30,31 This is partly explained by the ability of this species to adhere to synthetic materials and form a growth plate (biofilm) in the CVC.32-35 In addition, the composition of parenteral nutrition solutions that are infused by CVC, favor the growth of this species and thus facilitate blood invasion,35- 37 association that was proved in the study.
It should be noted, in relation to what happened in 2009, no changes occurred in the frequency of use of CVC, or parenteral nutrition during the year, which could explain the change in the distribution of Candida species.
The 30-day mortality found in the study is described in the literature,38 but higher than that reported in most developed countries,18 although in the serie C parapsilosis was the most frequent and the lowest mortality species. This high mortality may be due to one-fifth of the patients, which received no antifungal treatment. In addition, only 3 out of 10 patients received antifungal empirically or anticipated, therefore, most had to wait until the results of blood culture, which on average was higher than two days, reason why valuable time was lost to initiate antifungal therapy, and it has been demonstrated that the delay in starting treatment negatively influences survival.39-42
A weakness of the study is that a period of four years was analyzed alone, the ability to better assess temporal changes and the behavior of this infectious disease is limited.
A larger number of patients would have been better.
In conclusion, the study shows that in the Hospital Mexico there is a predominance of the species C. parapsilosis as a cause of candidemia, unlike described in the rest of the world. The relationship established between CVC and C. parapsilosis indicates the need to implement local guides for installation, use and optimal handling of the CVC, and this could be a key element in reducing the high incidence of infection. In addition, the rational use of parenteral nutrition may also influence on the frequency of this species, although there are other critical factors that should be involved in this dynamic, as suggested by the change in the incidence of C. parapsilosis in 2009. Better understand the dynamics of candidemia, identifying patients at high risk of developing it as well as an early start of antifungal treatment, represent the best opportunities to change the high mortality of this infection in this hospital.
This work was done in the Bacteriology and the Infectious Diseases Department at Hospital Mexico, Costa Rican Social Security, San José, Costa Rica.
Referencias
1. Bouza E, Muñoz P. Epidemiology of candidemia in intensive care units. International J Antimicrob Agents 2008; 32:S87-S91. [ Links ]
2. Darouiche RO. Candida in the ICU. Clin Chest Med 2009; 30:287-93. [ Links ]
3. Guery BP, Arendrup MC, Auzinger G, Azoulay E, Borges Sá M, Johnson EM, et al. Management of invasive candidiasis and candidemia in adult non-neutropenic intensive care unit patients: Part I. Epidemiology and diagnosis. Intensive Care Med 2009; 35:55-62. [ Links ]
4. Wisplinghoff H, Bischoff T, Tallent SM, Seifert H, Wenzel RP, Edmond MB. Nosocomial bloodstream infections in US hospitals: analysis of 24,179 cases from a prospective nationwide surveillance study. Clin Infect Dis 2004, 39:309-17. [ Links ]
5. Pfaller MA, Jones RN, Doern GV, Sader HS, Holli RJ, Messer SA. For the SENTRY Participant Group. International surveillance of blood stream infection due to Candida species: frequency of occurrence and antifungal susceptibility of isolates collected in 1997 in the United States, Canada, and South America for the SENTRY Program. J Clin Microbiol 1998; 36:1886-9. [ Links ]
6. Pfaller MA, Jones RN, Doren GV, Sader HS, Hollis RJ, Messer SA, et al. International surveillance of blood stream infections due to Candida species in the European SENTRY Program: species distribution and antifungal susceptibility including the investigational triazole and echinocandin agents. Diag Microbiol Infect Dis 1999; 35:19-25. [ Links ]
7. Pfaller MA, Diekema DJ. Role of Sentinel Surveillance of Candidemia: Trends in Species Distribution and Antifungal susceptibility. J Clin Microbiol 2002; 40:3551-57. [ Links ]
8. Trofa D, Gácser A, Nosanchuk JD. Candida parapsilosis, an Emerging Fungal Pathogen. Clin Microbiol Rev 2008; 21: 606-25. [ Links ]
9. Gagne JJ, Goldfarb NI. Candidemia in the in-patient setting: treatment options and economics. Expert Opin Pharmachother 2007; 8:1643-50. [ Links ]
10. Caballero E, Boza R, González K. Candidiasis sistémica - Experiencia en el Hospital San Juan de Dios 1996 - 1998. Rev Costarric Cienc Méd 1999; 20:153-65. [ Links ]
11. Mora N, Carrillo P. Candidemia en el Hospital San Juan de Dios: casos del 2003 al 2005. Rev. Colegio de Microbiólogos y Químicos Clínicos de Costa Rica 2005; 11:17-19. [ Links ]
12. Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. See comment in PubMed Commons belowJ Chronic Dis 1987; 4:373-83. [ Links ]
13. Quan H, Li B, Couris CM, Fushimi K, Graham P, Hider P, et al. Updating and validating the Charlson comorbidity index and score for risk adjustment in hospital discharge abstracts using data from 6 countries. Am J Epidemiol 2011; 173:676-82. [ Links ]
14. Almirante B, Rodríguez D, Park BJ, Cuenca-Estrella M, Planes AN, Almela M, et al. Epidemiology and predictors of mortality in cases of Candida bloodstream infection: results from population-based surveillance, Barcelona, Spain, from to 2002 to 2003. J Clin Microbiol 2005; 43:1829-35. [ Links ]
15. Bennet JE. Echinocandins for Candidemia in Adults without Neutropenia. N Engl J Med 2006; 355:1154-59. [ Links ]
16. Nucci M, Queiroz-Telles F, Alvarado-Matute T, Tiraboschi I, Cortes J, Zurita J, et al. Epidemiology of candidemia in Latin America: a laboratory-based survey. PLoS ONE 2013; 8: e59373. [ Links ]
17. Colombo AL, Nucci M, Park BJ, Nouer SA, Arthington-Skaggs B, da Matta DA, et al. Epidemiology of candidemia in Brazil: a nationwide sentinel surveillance of candidemia in eleven medical centers. J Clin Microbiol 2006; 44:2816-23. [ Links ]
18. Tortorano AM, Kibbler C, Peman J, Bernhardt H, Klingspor L, Grillot R. Candidemia in Europe: epidemiology and resistance. International J Antimicrob Agents 2006; 27:359-66. [ Links ]
19. Diekema DJ, Messer SA, Hollis DJ, Boyken L, Tendolkarb S, Kroeger J, et al. A global evaluation of voriconazole activity tested against recent clinical isolates of Candida spp. Diagn Microbiol Infect Dis 2009; 63:233-36. [ Links ]
20. Colombo AL, Nucci M, Salomao R, Branchini ML, Richtmann R, Derossi A, Wey SB. High rate of nonalbicans candidemia in Brazilian tertiary care hospitals. Diagn Microbiol Infect Dis 1999; 34:281-86. [ Links ]
21. Borges M, Zaragoza R. Critical overview of clinical guidelines relating to invasive fungal infections. International J Antimicrob Agents 2008; 32:S155-S159. [ Links ]
22. Calandra T, Marchetti O. Clinical trials of antifungal prophylaxis among patients undergoing surgery. Clin Infect Dis 2004; 39:S185-S192. [ Links ]
23. Nakamura T, Takahashi H. Epidemiological study of Candida infections in blood: susceptibilities of Candida spp. to antifungal agents, and clinical features associated with the candidemia. J Infect Chemother 2006; 12:132-38. [ Links ]
24. Asmundsdottir LR, Erlendsdottir H, Gottfredsson M. Increasing incidence of candidemia: results from a 20-year nationwide study in Iceland. J Clin Microbiol 2002; 40: 3489-92. [ Links ]
25. Colombo AL, Nucci M, Park BJ, Nouér SA, Arthington-Skaggs B, da Matta DA, et al. Epidemiology of Candidemia in Brazil: a Nationwide Sentinel Surveillance of Candidemia in Eleven Medical Centers. J Clin Microbiol 2006; 44:2816-23. [ Links ]
26. Brito LR, Guimaraes T, Nucci M, Rosas RC, Almeida LP, da Matta DA, et al. Clinical and microbiological aspects of candidemia due to Candida parapsilosis in Brazilian tertiary care hospitals. Med Mycol 2006; 44: 261-66. [ Links ]
27. Diekema DJ, Pfaller MA. Nosocomial candidemia: an ounce of prevention is better than a pound of cure. Infect Control Hosp Epidemiol 2004; 25:624-26. [ Links ]
28. Dizbay M, Fidan I, Kalkanci A, Sari N, Yalcin B, Kustimur S, et al. High incidence of Candida parapsilosis Candidaemia in non-neutropenic critically ill patients: Epidemiology and antifungal susceptibility. Scand J Infect Dis 2010; 42:114-20. [ Links ]
29. Huang YC, Lin TY, Leu HS, Peng HL, Wu JH, Chang HY. Outbreak of Candida parapsilosis fungemia in neonatal intensive care units: clinical implications and genotyping analysis. Infection 1999; 27:97-102. [ Links ]
30. Levin AS, Costa SF, Mussi NS, Basso M, Sinto SI, Machado C, et al. Candida parapsilosis fungemia associated with implantable and semi-implantable central venous catheters and the hands of healthcare workers. Diagn Microbiol Infect Dis1998; 30: 243-9. [ Links ]
31. Medrano DJ, Brilhante RS, Cordeiro RA, Rocha MF, Rabenhorst SH, Sidrim JJ. Candidemia in a Brazilian hospital: the importance of Candida parapsilosis. Rev Inst Med Trop Sao Paulo 2006; 48:17-20. [ Links ]
32. Hawser SP, Douglas LJ. Biofilm formation by Candida species on the surface of catheter materials in vitro. Infect Immun 1994; 62:915-21. [ Links ]
33. Branchini, ML, Pfaller MA, Rhine-Chalberg J, Frempong T, Isenberg HD. Genotypic variation and slime production among blood and catheter isolates of Candida parapsilosis. J Clin Microbiol 1994; 32:452-6. [ Links ]
34. Ramage G, Saville SP, Thomas DP, Lopez-Ribot JL. Candida biofilms: an update. Eukaryot Cell 2005; 4: 633-8. [ Links ]
35. Silva S, Negri M, Henriques M, Oliveira R, Williams DW, Azeredo J. Adherence and biofilm formation of non-Candida albicans Candida species. Trends in Microbiology 2011; 19: 241-47. [ Links ]
36. Weems JJ, Chamberland ME, Ward J, Willy M, Padhye AA, Solomon SL. Candida parapsilosis fungemia associated with parenteral nutrition and contaminated blood pressure transducers. J Clin Microbiol 1987; 25: 1029-32. [ Links ]
37. Pfaller MA. Nosocomial candidiasis: emerging species, reservoirs, and modes of transmission. Clin Infect Dis 1996; 22: S89-S94. [ Links ]
38. Eggimann P, Garbino J, Pittet D. Epidemiology of Candida species infections in critically ill non-immunosuppressed patients. Lancet Infect Dis 2003; 3: 685-702. [ Links ]
39. Kollef M, Micek S, Hampton N, Doherty JA, Kumar A. Septic shock attributed to Candida infection: importance of empiric therapy and source control. Clin Infect Dis 2012; 54: 1739-46. [ Links ]
40. Morrell M, Fraser VJ, Kollef MH. Delaying the empiric treatment of Candida bloodstream infection until obtaining positive blood culture results: A potential risk factor for hospital mortality. Antimicrob Agents Chemother 2005; 49:3640-45. [ Links ]
41. Garey KW, Rege M, Pai MP. Time to initiation of fluconazole therapy impacts mortality in patients with candidemia: A multicenter study. Clin Infect Dis 2006; 43:25-31. [ Links ]
42. Grim SA, Berger K, Teng C, Gupta S, Layden JE, Janda WM, et al. Timing of susceptibility-based antifungal drug administration in patients with Candida bloodstream infection: correlation with outcomes. J Antimicrob Chemother 2012; 67: 707-14. [ Links ]
Received: April 23, 2015; Accepted: November 05, 2015











 text in
text in 


