Introduction
Colombia is one of the most orchiddiverse countries in the world, particularly across the three mountain ranges of the northern Andes (Karremans et al., 2023; Luer & Thoerle, 2012; Pérez-Escobar et al., 2022). However, deforestation, primarily driven by agricultural expansion, has diminished the natural habitat cover of Andean ecosystems. The remaining habitat exhibits signs of significant human intervention, with 78% of forest recovery being disrupted, arrested, or unsuccessful (Christmann et al., 2023; Rodríguez Eraso et al., 2013). This trend poses a threat to the rich diversity of many Andean orchid species (Parra-Sánchez et al., 2016, 2023).
Restrepia Kunth (Orchidaceae: Pleurothallidinae) is one of the most taxonomically challenging genera due to high intra-specific variation within species (e.g., color patterns) and extremely uniform interspecific floral morphology. These factors complicate species circumscription and identification in both herbaria and living specimens (Luer, 1996). This taxonomic difficulty has persisted since the earliest collections of the genus in the late 18th and early 19th centuries (Gutiérrez Morales et al., 2023; Luer, 1996). The genus currently comprises approximately 60 species, distributed from Mexico to the tropical Andes (Gutiérrez Morales et al., 2023; Karremans et al., 2023). It is monophyletic (Chumová et al., 2021; Pérez-Escobar et al., 2017) and can be recognized by its ramicauls, which are surrounded by imbricating, papery, distichous, laterally flattened sheaths, as well as the dorsal sepal and petals that are thickened at the apex by osmophores (Vogel, 1990). Additionally, the lip’s hypochile features a pair of uncinate, capillary processes, a characteristic that is absent in Restrepia aberrans Luer, the sole member of the Restrepia subgen. Ecmeles Luer (Luer, 1996).
Phylogenetic studies place Restrepia within the Restrepia affinity clade (sensu Chumová et al., 2021; Karremans, 2016), alongside related genera such as Barbosella Schltr., Chamelophyton Garay, Dresslerella Luer, Echinosepala Pridgeon & M.W.Chase, Myoxanthus Poepp. & Endl., Pleurothallopsis Porto & Brade, and Restrepiella Garay & Dunst. All these genera are characterized by exhibiting single-flowered co-florescences (Rojas-Alvarado & Karremans, 2024). According to the IUCN criteria (2020), 75% (35 species) of assessed Restrepia species are threatened with global extinction due to deforestation and illegal collection (BGC, 2024; Calderón-Sáenz, 2007). This issue is exacerbated by the narrow endemism of many species, some of which are historically rare (e.g., Restrepia chocoensis Garay, R. cuprea Luer & R.Escobar, R. chrysoglossa Luer & R.Escobar, R. howei Luer). The Colombian Andes serve as the center of diversity for the genus, with 31 species, of which 19 are endemic to the country (Gutiérrez Morales et al., 2023; (Karremans et al., 2023; (Luer, 1996). However, less than 40% of the natural cover remains across the three cordilleras in Colombia (Rodríguez Eraso et al., 2013), and the transformation of natural habitats further endangers the future of many orchid species (Ospina-Calderón et al., 2023).
In this study, we describe and illustrate a newly discovered Restrepia species found during recent botanical explorations in the montane rainforests of the Department of Valle del Cauca, Colombia. This region has drawn the attention of botanists because it harbors a high level of plant species endemism, and several novel orchids from various genera have been documented in the area (e.g. Ortiz et al., 2011; Reina-Rodríguez et al., 2019, 2022). The new species, R. restrepoi, appears to be restricted to the western Andes of Colombia and is morphologically similar to the Valle del Cauca endemic R. chrysoglossa. Natural populations of this species have been identified in both highly fragmented and conserved forest patches. We provide a color illustration, a taxonomic description, a comparison with its morphologically similar species, and a distribution map.
Materials and methods
We sampled natural and human-modified habitats using a randomized design in the Departments of Valle del Cauca in Colombia. Following Parra-Sánchez et al. (2023), we established 50 sampling points in natural habitats (37 plots) and pastures (13 plots) across an 800 m elevational range (1800-2600 m, Tadono et al., 2014). Natural habitats generally support native species and represent baseline ecosystems, serving as references for understanding ecological dynamics. In contrast, human-modified habitats often display altered species compositions, which can provide valuable insights into the effects of environmental change. After the conversion of natural habitats, about 80% of orchid species are lost, while driving 90% of orchid turnover, indicating that most orchid species are confined to pristine environments (Parra-Sánchez et al., 2016, 2023). Each plot covered 300 m2 (10 × 30 m), maintaining a minimum distance of 200 m apart, and we sampled all possible substrates up to 2 m in height (i.e., phorophytes, ground, fallen branches). In addition to the randomized plot sampling, we conducted over 56 hours of random walks to enhance our sampling effort. Plants were collected under collection permits issued by the Agencia Nacional de Licencias Ambientales (ANLA n. 791), and vouchers were preserved as dried and spirit specimens for future reference at Jardín Botánico de Bogotá (JBB sensuThiers, 2024).
Field images were captured using a Canon ® T3i, equipped with a Canon 100 mm f/2.8 USM macro lens. Sketches of living and preserved specimens were digitized, and the images were utilized to create a draft composite plate in Adobe Photoshop® 2020. Living and preserved specimens were examined for morphological and taxonomic comparisons in accordance with the latest taxonomic treatment (Luer, 1996) and subsequent new species descriptions (e.g., Gutiérrez Morales, 2023). Botanical terminology adhered to the standards set by Luer (1996) , Gutiérrez Morales et al. (2023) , and Rojas-Alvarado and Karremans (2024). Environmental variables were obtained from ClimateCharts.net (et al., 2020) and the Holdridge life zones (Holdridge, 1987). Maps were produced using QGis 3.16 (QGIS 2021). Plant names and authors are based on standard databases (Epidendra, 2025; IPNI, 2025; (Tropicos, 2025).
Taxonomic treatment
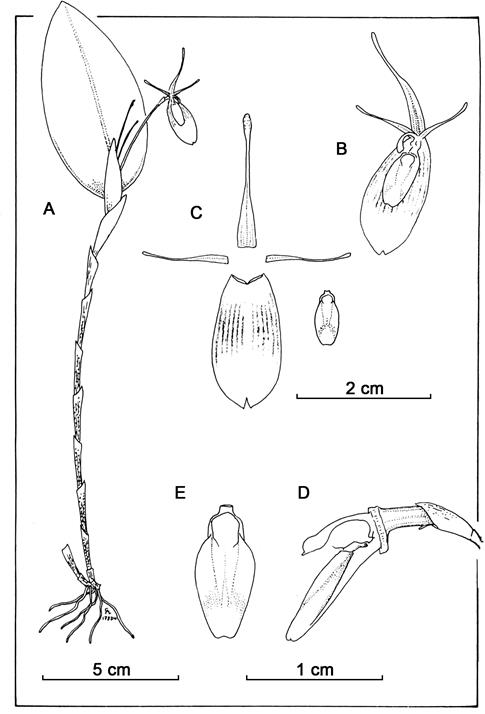
Restrepia restrepoi E.Restrepo & E.Parra sp. nov.(Fig. 1, 4A).
TYPE: COLOMBIA. Valle del Cauca: Yumbo, Dapa. Vía a los cultivos de Té, 2200 m, 15 December 2024 (fl.), E.Restrepo & D. Edwards 319 (holotype JBB!).
Diagnosis: reminiscent of Restrepia chrysoglossa (Fig. 2, 4B) in plant shape, size and flower colors, but easily distinguished by its brown-spotted sheaths (vs. unspotted, except the lowermost, dotted with brown), the cleistogamous, non-spreading flowers (vs. non-cleistogamous, spreading), the oblong, attenuated, non-clavate dorsal sepal (vs. narrowly ovate-triangular, clavate) and its ovate lip, pyriform when expanded, 4.8 × 2.9 cm (vs. oblong-ovate, 9.00 × 4.25 cm).
Plant epiphytic, caespitose, erect to sub-erect, up to 35 cm tall; roots slender. Ramicauls erect, 12-24 cm long, enclosed by 5-10 thin, whitish, brown-spotted, papery, loose, imbricating, oblique, laterally compressed, acute sheaths, the uppermost prominent, elongate, acuminate, extended to near the middle of the pseudopeduncle, acuminate. Leaf erect, coriaceous, ovate-elliptical, 7.2-8.1 × 2.8-3.4 cm, the apex emarginate, with the mid vein extending beneath and ending in a short mucro, the base slightly cuneate to rounded, contracted into a twisted petiole ca. 3 mm long. Inflorescence with a reduced peduncle ca. 2 mm long, enclosed by a 3 mm long spathe, producing single-flowered coflorescenses in succession. Pseudopeduncle 3.2-3.6 cm long, glabrous, subtended by a 1.0-2.5 mm long bracts, 2-3 at a time; floral bract thin, tubular, 3 mm long, pedicel 1.5 mm long, the remnant of the axis a filament 1 mm long, free; ovary 2.63 mm long; Flower yellow, often cleistogamous, sepals membranous, glabrous, not wide-spreading, the dorsal sepal free, oblong, attenuated at the apex, slightly thickened near the apex, 7.5 × 2.1 mm, 5-veined, the lateral sepals connate to each other forming a synsepal, bifid at the apex when expanded, oblong, attenuated, 7.5 × 5.4 mm, 13-veined; petals membranous, translucent, suffused with yellow along the margins, with a notorious midvein discontinuously suffused with purple, narrowly linear triangular, the apex minimally clavate-thickened, 7 × 0.8 mm at the base; lip magenta-yellow, oblong and concave in natural position, pyriform when expanded, 4.8 × 2.2 mm in natural position, 2.9 mm wide across the lateral processes expanded, 3-veined, the epichile indistinctly demarcated from the hypochile, broadly triangular, concave, smooth, rounded at the apex, the hypochile subquadrate, concave, each side with a microscopic, capillary, uncinate process, the disc featureless, the base connected to the column-foot by a short, thick, cylindrical neck; column greenishwhite, the basal half slender, clavate, the margins of the apical half irregular 4.92 mm long, the base pedestal-like. Pollinia and anther cap not seen. Capsule oblong, strongly ridged, about 7.9 cm long.
Eponymy: Named after Eugenio Restrepo Hoyos, Colombian entrepreneur and father of the first author, who has enormously supported him in all his passions and personal aims. This species is dedicated to him as an act of gratitude.
Discussion
The infrageneric placement of Restrepia restrepoi (Fig. 1, 4A) remains uncertain. According to Carl Luer's infrageneric system (Luer, 1996), the new species displays diagnostic features from both Restrepia subgen. Restrepia and the monospecific Restrepia subgen. Ecmeles. The former includes most of the species and is characterized by elliptical to ovate, petiolate leaves; clavate dorsal sepals; lateral sepals without thickened apices; and a lip hypochile that features microscopic, capillary, uncinate processes on each side (also present in Restrepia subgen. pachymeles). The latter, Restrepia subgen. Ecmeles, is distinguished by the non-clavate dorsal sepals, free lateral sepals, and a lip with obtuse, marginal, lateral lobes on the hypochile (Luer, 1996).
However, free lateral sepals cannot be used to diagnose the subgenus itself, as they have proven inconsistent across populations of its only member, R. aberrans (Fernández et al., 2014; Luer, 1996). Since the new species presents a non-clavate dorsal sepal, lateral sepals connate into a synsepal, and the typical uncinate, capillary processes on the lip hypochile, we believe this species should be classified as “incertae sedis” until further phylogenetic studies on the infra-generic relationships of the genus are conducted. Thus, the new species further provides evidence that groups within the genus need to be reconsidered.
Restrepia restrepoi (Fig. 1, 4A) can be distinguished from other species in the genus by its ramicauls, which are enclosed by dark-spotted sheaths, entirely light yellow, unspotted cleistogamous flowers, the oblong, non-clavate dorsal sepal that is attenuated and slightly thickened at the apex, the 13-veined synsepal, and its lip, which is intense rose at the base and pyriform when expanded, featuring basal, capillary, uncinate processes.
The species that are morphologically most similar to R. restrepoi are R. chrysoglossa (Fig. 2, 4B) and R. flosculata Luer (Fig. 3, 4C). Both are yellow-flowered species from the Western Colombian Andes in Valle del Cauca’s department. The former is known only from its type locality and is endemic to the department, while the latter, which is also found in northwestern Ecuador, exhibits various color forms across populations, ranging from purple-dotted or striped to entirely yellow (as shown in Fig. 3; Luer, 1996). All three species exhibit similar overall plant size, short pseudopedunculate coflorescences, and almost entirely yellow flowers.
As discussed in the diagnosis, R. restrepoi is distinguished from R. chrysoglossa, by its brown-spotted sheaths (vs. unspotted, except for the lowermost, dotted with brown), the cleistogamous, non-spreading flowers (vs. non-cleistogamous, spreading), the oblong, attenuated, non-clavate dorsal sepal (vs. narrowly ovate-triangular, clavate) and its ovate lip, pyriform when expanded, 4.8 × 2.9 (vs. oblong-ovate, 9.0 × 4.25 cm). In addition to those characteristics, the new species exhibits shorter sepals, ca. 7.5 mm long (vs. up to 20 mm long), and unstripped synsepal (vs. striped with red below the middle).
The new species can be easily distinguished from Restrepia flosculata by its brown-spotted sheaths all along the ramicaul (vs. the lowermost brown spotted), the uppermost sheath elongate, acuminate (vs. oblique, acute), the cleistogamous, non-spreading flowers (vs. non-cleistogamous, spreading), the shorter sepals, ca. 7.5 mm long (vs. to 14 mm long), the oblong, attenauted, non-clavate dorsal sepal (vs. narrowly triangular below the middle, attenuate above, clavate), straight petals (vs. decurved), and attenuated lip, pyriform when expanded, with rounded apex, 4.8 × 2.9 (vs. broadly oblong lip with retuse apex, 7 × 3.75 cm).
Finally, the new species resembles R. aberrans (Fig. 4D), which is restricted to Costa Rica and Panama. Both species show autogamous plants with non-clavate dorsal sepals, but the new species presents larger ramicauls, 12.3-23.4 cm long (vs. 7.5-11.5 cm long), enclosed by brown-spotted sheaths (vs. 1.5-3.5 cm long, unspotted sheaths), lateral sepals connate to each other forming a synsepal, bifid at the apex when expanded, oblong, attenuate, 7.5 × 5.4 mm, 6-7 veined each (vs. often free or slightly adherent, oblique, oblong-acute, 10.0 × 1.5 mm, 5-veined each) and the lip shape and dimensions, pyriform when expanded, 4.8 × 2.9 mm, rounded at the apex, the hypochile with two lateral, capillary, uncinate processes (vs. oblong-trilobed when expanded, 3.0 × 2.0 mm; the hypochile with two lateral, oblique, erect lobes, and two inner erect blades). The differences among the new species and its morphological affinities are shown in Table 1.

A1. Plant habit.
A2. Floral bract.
A3. Close-up to the peduncle of the inflorescence bearing multiple single flowered co-florescences.
B. Adaxial view of the leaf, showing the multiple inflorescences at a time.
C. Mature bud, lateral view.
D. Dissected perianth.
E. Column plus lip, lateral view.
F. Column, – view.
G. Expanded lip.
H. Capsule, lateral view.
Prepared by Eugenio Restrepo from the holotype.
Figure 1 Dissection plate of Restrepia restrepoi E.Restrepo & E.Parra.
A. Plant habit.
B. Flower, – view.
C. Dissected perianth.
D. Column and lip, lateral view.
E. Lip, frontal view.
Illustration by Carlyle A. Luer, featured in the Monographs in Systematic Botany from the Missouri Botanical Garden, Vol. 59, p. 43, Plate 10 (Luer, 1996). Reproduced with permission from the Missouri Botanical Garden Press.
Figure 2 Type illustration of Restrepia chrysoglossa Luer & R.Escobar.

A. Plant habit.
B. Close-up to the peduncle of the inflorescence bearing multiple single flowered coflorescences.
C. Flower, – view.
D. Dissected perianth.
E. Column and lip, lateral view.
F. Expanded lip.
G. Column, ventral and – views.
Prepared by Eugenio Restrepo.
Figure 3 Dissection plate of Restrepia flosculata f. xanthina.

A. Restrepia restrepoi E.Restrepo & E.Parra.
B. R. chrysoglossa Luer.
C. R. flosculata Luer, type form.
D. R. aberrans Luer.
Photographs by Eugenio Restrepo (A), Wiel Driessen (B, D), and Andreas Kay (C).
Figure 4 Species comparison.
Habitat and ecology: The remaining forests on the eastern flank of Colombia’s western cordillera are often highly fragmented, primarily due to extensive conversion of forested areas to pastures (Rodríguez Eraso et al., 2013). Restrepia restrepoi is exclusively known from three localities located 9.2 km apart (Fig. 5). Individuals grow epiphytically on branches of tree canopies and young trees, near roads or trails (Fig. 6), alongside other Restrepia species (R. brachypus Rchb.f.) and several members of the Orchidaceae family, including Dracula chimaera (Rchb.f.) Luer, Dichaea andina Alomía & Sambin, Lepanthes trimerinx Luer, L. sanjuanensis Bogarín & Karremans, L. stellaris Luer & Hirtz, and Telipogon mayoi Reina-Rodr. & C.Martel, among others.
Flowering has been observed in the wild from February to May (Haelterman, pers comm.). Several individuals were found with capsules at different stages of development from flowers, leading us to hypothesize, a priori, that this species may be autogamous. However, further field observations and experimental treatments are needed to better understand its pollination syndrome and ecological interactions.
Conservation status: The species may be classified as Data Deficient (DD) according to IUCN criteria (IUCN, 2020). However, our data suggests that conservation actions are necessary for the populations. The species is currently known from three sites that are located 9.2 km apart within a narrow elevational band (between 1900 and 2200 m in elevation). We encountered only four mature individuals and two juveniles, which indicates a small population size. Additionally, no individuals were found in 50 randomized sampling plots, despite significant sampling efforts involving two experienced orchidologists working eight hours a day for nine days near the type locality of R. restrepoi. Therefore, we will refrain from collecting any more individuals until further populations are discovered. The species inhabits a fragmented landscape where population dynamics are disrupted (Ospina-Calderón et al., 2023), and natural forests experience edge effects that may reduce habitat availability (Parra Sánchez et al., 2016).
Our study provides evidence that the species is geographically rare, characterized by a small population size and highly specialized habitat requirements (Rabinowitz, 1981). Two key conservation actions are necessary. First, additional field surveys should be conducted to search for the species in adjacent areas, potentially expanding its known range. Second, the protection of the species’ habitat and surrounding forests must be prioritized to ensure habitat conservation. This approach would likely facilitate seed dispersal from existing mother plants and promote the regeneration of more extensive secondary forest areas within the species’ distribution.
Table 1 Comparison among Restrepia restrepoi E.Restrepo & E.Parra and its closest morphological affinities.
| Traits | R. aberrans | R. chrysoglossa | R. flosculata | R. restrepoi |
|---|---|---|---|---|
| Distributional and abiotic conditions | ||||
| Distributional range | Atlantic basin, premontane wet forest | Cauca river basin, montane wet forest | Cauca river basin, premontane rain forest | Cauca river basin, Montane rain forest |
| Habitat | Primary cloud forest | Secondary cloud forest | Primary cloud forest | Primary and secondary cloud forest |
| Elevation range (m a.s.l.) | 350-790 | 2164 | 2122 | 1910-2100 |
| Mean annual rainfall (mm) | 3040 | 2934.5 | 1682.3 | 1900-2100 |
| Annual mean temperature (°C) | 23 | 23.7 | 21 | 15-17 |
| Morphological and phenological conditions | ||||
| Ramicauls length (cm) | 1.5-3.5 | 9-20 | 15-20 | 12.3-23.4 |
| Leaf blade size (cm) | 6.0-8.0 × 3.0-4.0 | 6-8 × 3-4 | 8-10 × 3.5-4.5 | 7.9-8.1 × 3.2-3.4 |
| Lateral sepals | 5-veined | 6-veined | 7-8-veined | 6-7-veined |
| Dorsal sepal | Narrowly lineartriangular | Narrowly ovatetriangular, clavate, thickened at the apex | Narrowly ovatetriangular, clavate, thickened at the apex | Oblong, attenuated, slightly thickened at the apex |
| Dorsal sepal lenght (mm) | 10 | 20 | 14 | 7.5 |
| Lip shape (expanded) | Oblong-trilobed | Oblong-ovate | Oblong | Ovate, Pyriform |
| Expanded lip dimensions (mm) | 3.0 × 2.0 | 9.00 × 4.25 | 7.00 × 3.75 | 4.8 × 2.9 |
| Lip hypochile | Broadly concave above the subtruncate base, bearing two lateral, oblique, erect lobes, and two inner, erect blades | Subquadrate, concave, with a microscopic, capillary, uncinate processes | Rounded, concave with thin, erect margins, each side with a capillary, uncinate process | Subquadrate, concave, with a microscopic, capillary, uncinate processes |
| Lip epichile (mm) | Oblong, 3-veined, rounded at the apex | Ovate, thickened, smooth, subtruncate | Oblong, smooth, the apex subtruncate-retuse | Broadly triangular, concave, smooth, rounded at the apex |
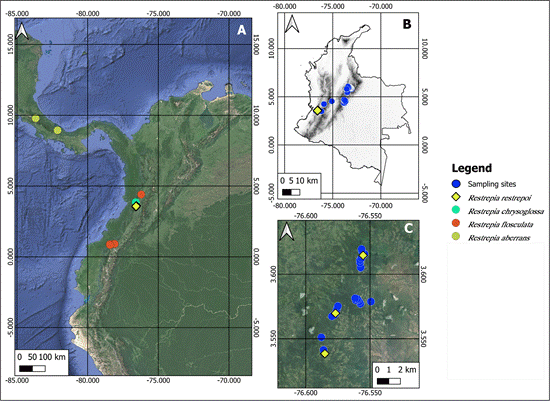
A. Distribution of R. restrepoi E.Restrepo & E.Parra. (yellow triangles), R. chrysoglossa Luer & R.Escobar (cyan dots), R. flosculata Luer (red dots), and R. aberrans Luer (green dots).
B. Study area within Colombia (black silhouette), along with sampling plots (blue dots) and elevation data (digital elevation model from Tadono et al., 2014).
C. Landscape across sampling sites and the localities where R. restrepoi was found.
Map created by Edicson Parra using QGIS 3.24.1.
Figure 5 Distribution map of Restrepia restrepoi E.Restrepo & E.Parra, related species, and the study area in the Western, Central and Eastern Cordilleras of the Colombian Andes.

A. Mature plant growing on medium-sized branches of a fallen tree.
B. A juvenile plant growing on thin branches.
C. Mature individual seen from the underside, along with other Lepanthes spp.
Photographs by Eugenio Restrepo (A, B) and David Edwards (C). Prepared by Eugenio Restrepo.
Figure 6 Mature and juvenile plants of Restrepia restrepoi E.Restrepo & E.Parra growing in situ.












 uBio
uBio 


