Introduction
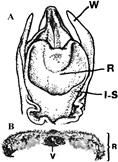
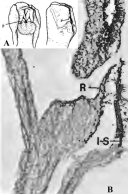
Monandrous orchids are distinguished not only by the beauty and intricacy of their flowers but also by the modifications of floral segments, organs, and structures. In these orchids, the style, stigma, and stamens (including anther and filaments) are fused into a single structure known as the gynostemium (Fig. 1: A1-3; B1-3; C1-2; D1-2; F1-3). A key structure within the gynostemium is the rostellum (Table 1). In some species, the rostella can be challenging to observe or recognize (Fig. 1: A1-4, B1-4, C2-4, D1-2, F1-6; 2A-E; 3A-E; 4A-E; 5A-D; 6A-Q; 7: 2, 3, 5; 8A-E, G1; Table 1). The rostellum, located at the base of the pollinarium (which includes the pollinium or pollinia, stipes, and viscidium) and positioned above the stigma, acts as the “roof” of the stigma and the “floor” of the anther, effectively separating the sigma from the pollinia. During the ontogeny of Bulbophyllum ecornutum J.J.Sm., the rostellum curves and develops a hook-like shape (Rasmussen, 1986b). In some members of the Pleurothallidinae, the rostellum is prominent and curves downward toward the stigma (Cardoso-Gustavson et al., 2017). Although rostella are generally believed to function similarly across all orchid flowers, exceptions do exist (Cheng et al., 2009; Darwin, 1904; Nazarov & Gerlach, 1997).
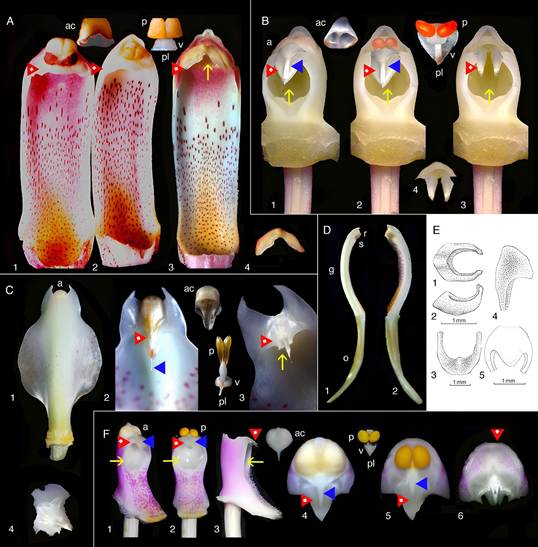
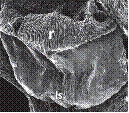
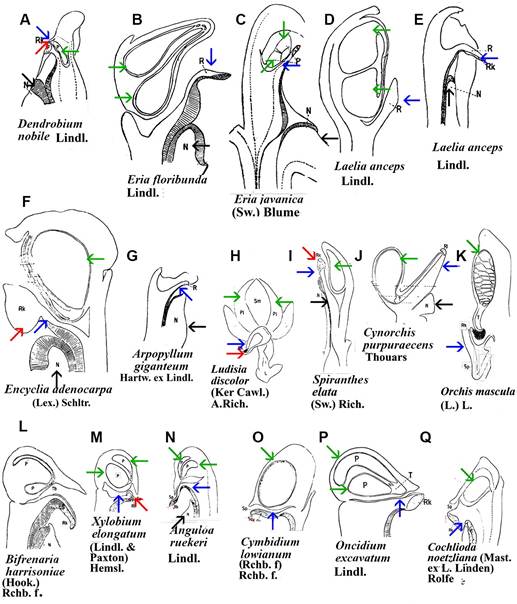
A. Cymbidium Sw. hybrid.
1. Intact gynostemium.
2. Anther cap removed showing pollinia in place on the rostellum.
3. Pollinarium removed showing rostellum and stigma.
4. Rostellum.
On top, the anther cap and pollinarium (with pollinia and viscidium).
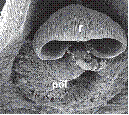
B. Phalaenopsis Blume hybrid.
1. Intact gynostemium showing parts of viscidium, rostellum, and stigma.
2. Anther cap removed showing two pollinia, stipe and viscidium in place å the rostellum.
3. Pollinarium removed showing rostellum and stigma.
4. Rostellum.
On top, anther cap and pollinarium (pollinia, stipe and viscidium).
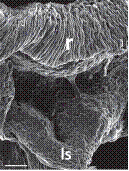
C. Stanhopea J.Frost ex Hook.
1. Dorsal view with yellowish anther cap visible on top.
2. Ventral view with yellowish anther cap in place showing rostellum and viscidium.
3. Ventral view with anther cap and pollinaria removed showing rostellum and stigma.
On top, anther cap and pollinarium (pollinia, stipe and viscidium).
D. Cymbidium. Longitudinal section of gynostemium showing side views of rostellum and stigma.
E. Brazilian Habenaria Willd. species showing very small rostella. Habenaria hipporepica J.A.N.Bat. & Blanch.
1. Upper view.
2. Lateral view. Habenaria espinhacensis
3. Upper view.
4. Lateral view. Habenaria quadriferricola J.A.N.Bat. & B.M.Carvalo
5. Upper view.
F. Vanda R.Br. Miss Joaquim, a natural hybrid, which is now the National Flower of Singapore.
1. Gynostemium intact showing anther cap in place and stigma.
2. Anther cap removed showing pollinia, rostellum and stigma.
3. Pollinia removed showing tip of rostellum and side view of stigma.
4. View from above showing anther cap in place, rostellum, and viscidium.
5. View from above after removal of anther cap showing pollinia, rostellum and part of viscidium.
6. View from above with anther cap and pollinaria removed, showing the rostellum.
On top, anther cap and pollinarium (pollinia, stipe and viscidium).
Explanation of symbols: a, anther; ac, anther cap; g, gynostemium (column); o, ovary; p. pollinia; pl, pollinarium; r, rostellum; s, stigma; v, viscidium; blue wedge, viscidium; red wedge with white dot, rostellum; white or yellow arrow, stigma; Sources: Joseph Arditti (A-D), courtesy of Dr. João A. N. Batista (E), courtesy of Dr. Tim Wing Yam (F).
Figure 1 Parts of gynostemia (columns).
Rostella vary in size and shape (Fig. 1-7; 8A, F, G; 10; 12-16; Table 1), but they are usually small and delicate. For example, a flower of Cymbidium Sw. Jungfrau hybrid weighs approximately 8.25 g, measures around 12.5 cm in width, and contains a rostellum weighing 0.02 g, which accounts for 0.24% of the flower’s weight and 1.43% of the gynostemium’s weight. The rostellum itself measures approximately 1 cm in width (side to side when facing the flower), 1.2 cm in depth (front to back), and 0.2-0.3 cm in height (thickness).
The rostella of Habenaria Willd. are even smaller (Fig. 1E: 1-5). In Pterygodium vermiferum E.G.H.Oliv. & Liltved, a species native to The Western Cape of South Africa, the rostellum bears “several remarkable, long twisted appendages arising from the rostellum” (Oliver et al., 2008). The specific epithet vermiferum refers to these wormlike outgrowths.
Some orchids either lack a rostellum or have one that is not fully developed. In self-pollinating Spiranthinae taxa, a rostellum is absent, allowing pollinia to come into contact with the stigmatic fluid before the flowers open, leading to self-pollination (for a review, see Salazar et al., 2016). For example, the selfpollinating Greenwoodiella micrantha (Lex.) Salazar & R.Jiménez var. micrantha often lacks a rostellum (Salazar et al., 2016). However, this trait is not consistent even within a genus in the Spiranthinae. A good example is the cross-pollinated Greenwoodiella micrantha var. garayana (R.González) Salazar & R.Jiménez which shows a well-developed rostellum (Salazar et al., 2016).
Bletiinae are often characterized as a subtribe of orchids lacking rostella, but Bletia purpurea (Lam.) DC., does possess one (Kurzweil, 1987a; Valencia-Nieto et al., 2018). In Chysis laevis Lindl., the rostellum may be vestigial or absent, whereas Chysis bractescens Lindl., and Chysis limminghei Linden & Rchb.f., have fully developed rostella (Valencia-Nieto et al., 2018). These examples suggest that the rostellum may be lost in self-pollinating forms, though this trend is not always consistent.
History and nomenclature
The term “rostellum” (meaning “little beak” in Latin; plural “rostella,” not “rostellums”) was coined by Louis Claude Marie Richard (1754-1821; Fig. 9A), a French botanist, illustrator, and the father of another orchid scientist, Achille Richard (1794-1852). He used the term to describe a small structure found in monandrous orchid flowers. Richard’s original definition was likely inspired by the genus Orchis L., is “Supermus stigmatis prossesus ultra gynizum” a “projection above the stigmas (and) beyond the receptive surface,” (translation from Dressler, 1989). Richard was also the first to describe and illustrate the rostella of several orchids (Fig. 3).
John Lindley (1799-1865; Fig. 9C) and Francis (Franz) Bauer (1758-1840; Fig. 9I) described the orchid stigma as “in most cases it is quite simple, merely terminating in a glandular dilatation of the upper margin, called the rostellum. The glandular dilatation separates from the stigma and adheres to the pollen masses” (Bauer & Lindley, 1830-1838). Interestingly, this is the only instance in which the term “rostellum” appears in their book (Bauer & Lindley, 1830-1838), despite several illustrations depicting structures that can be interpreted, or at least be speculated upon, as rostella (Fig. 10A-D).
Table 1 Morphological features of the gynostemium and rostellum in monandrous orchids.
| Species name | Details | Image | Reference |
|---|---|---|---|
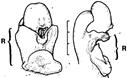
| Acianthera aphthosa (Lindl.) Pridgeon & M.W.Chase | Rostella of two species in one genus differ. Symbols: ls, lateral stigma; r, rostellum. |
|
Cardoso-Gustavon et al. (2017) |
| Acianthera fenestrata (Barb. Rodr.) Pridgeon & M.W.Chase | Rostella of two species in one genus differ. Symbols: pol, pollinia; r, rostellum. |
|
Cardoso-Gustavon et al. (2017) |
| Anathallis obovata (Lindl.) Pridgeon & M.W.Chase | Symbols: ls, lateral stigma; r, rostellum. |
|
Cardoso-Gustavon et al. (2017) |
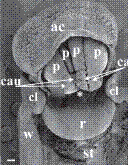
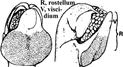
| Bletia purpurea (Lam.) DC. | Rostellum is tongue or semi linate shaped. Shield separates pollinarium from stigma. Symbols: *, anther apex; ac, anther cap; cau, caudicle; cl, clinandrium; p, pollinium; r, rostelum; st, stigma; w, column wing. Scale bar = 100 µm. |
|
Valencia-Nieto et al. (2018) |
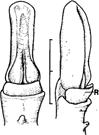
| Bletia striata Druce | Median lobe becomes rostellum. Left: Front view. Right: Side view. Symbol: R, rostellum. |
|
Rasmussen (1986b) |
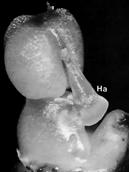
| Bulbophyllum ecornutum J.J.Sm. | Rostellum swells ventrally, apex reflexes, median stigma becomes the rostellum. Symbol: Ha, hamulus. |
|
Rasmussen (1985) |
| Bulbophyllum lomsakense J.J.Verm., Schuit. & de Vogel | Rostellar projection is deeply bifid. Symbol: R, rostellum |
|
Rasmussen (1986a) as Sunipia thailandica (Seienf. & Smitinand) P.F.Hunt. |
| Bulbophyllum rimannii (Rchb.f.) J.J.Verm. Schuit. & de Vogel | A deeply bifid rostellar projection. Symbol: R, rostellum. |
|
Rasmussen (1986a) as Sunipia rimannii (Rchb.f.) Seidenf. |
| Photographs: Cephalanthera longifolia (L.) Fritsch. Line drawings: Cephalanthera damasonium Druce | Rostellum is located above lateral stigmatic lobes. Symbols: R, rostellum; S, stigma. Bar at upper right of drawing: 1 mm. |
|
Rasmussen (1986b) |
| Cymbidium aloifolium (L.) Sw. | Rostellar projection elongates. Symbol: R, rostellum. |
|
Rasmussen (1982) |
| A. Diuris punctata Sm. B. Diuris emarginata var. pauciflora (R.Br.) A.S.George | A. Front view of top of column. B. Cross section of rostellum and viscidium. Symbols: I-S, lateral stigma lobe; R, rostellum; V, viscidium; W, wing of column. |
|
Rasmussen (1982) |
| Epipogium aphyllum Sw. | Front and side view of top of the column showing anther cap, stigma and elongated rostellum. Symbol (only the rostellum is labeled in the original): R, rostellum. |
|
Rasmussen (1982) |
| Goodyera repens (L.) R.Br. | Rostellum is long, pointed, erect, triangular with large broadly elliptic detachable viscidium. |
|
Kurzweil (1989) |
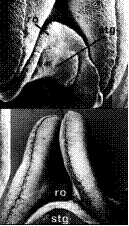
| Huttonaea pulchra Harv. | Central lobe of median carpel becomes broadly hemispherical. Symbols: ro, rostellum; stg, stigma. |
|
Rasmussen (1982) |
| Lecanorchis malaccensis Ridl. | A. Front and side views of column tip showing pollinia. Symbols: r, rostellum; s, stigma; p, pollinia. B. Cross section of rostellum and lateral stigma lobe. Rostellum is hidden behind plate formed by I-S. Symbols: l-S, lateral stigma lobe; r, rostellum. |
|
Dressler (1989); Rasmussen (1982). |
| Neottia Guett. (no species listed in original) | Unique . . . described as a gland (Dressler, 1989). Symbols: A, anther cap, R, rotellum; P, pollinia; I-S, lateral stigma lobe |
|
Rasmussen (1989b) |
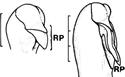
| Phalaenopsis pulcherrima (Lindl.) J.J.Sm. | Rostellar projection (RP) is elongated. Scale bar = 3 mm |
|
Rasmussen (1982) |
| Stereosandra javanica Blume | Rostelum is shorter than the anther. It projects beyond the lateral lobes. Has no viscidium. Symbol: R, rostellum. |
|
Rasmussen (1982) |
| Tropidia curculigoides Lindl. | Erect rostellum is as long as anher. Removal of viscidium leaves a V-shaped scar. Symbols: R, rostellum; V, viscidium. Scale bar = 2 mm |
|
Rasmussen (1982) |
| Vanilla imperialis Kraenzl. | Column and labellum are fused. Rostellum is extended. |
|
Rasmussen (1986b) |
| Zeuxine affinis (Lindl.) Benth. ex Hook.f. | Rostellum errect, triangular, retuse apex with ridge. |
|
Rasmussen (1982) |
The British botanist Robert Brown (1773-1858; Fig. 9B), who discovered the cell nucleus while working with orchids, also suggested a stigmatic origin of the rostellum. He proposed that it arose from the median stigma lobe, stating, “The result of my examination of this point satisfies me that Orchideae have in reality three stigmata, generally more or less confluent, but in some cases, manifestly distinct, and two of which are in several instances furnished with styles of considerable length” (Brown, 1833). The third, anterior lobe, “manifestly differs from the other two. To this. . . lobe. . . the glands (now referred to as viscidium or viscidia) always belong to which the pollen-masses become attached” (Brown, 1833; Fig. 1: A1-4, B1-4, D1-2, F1-3; 10: A1, 3, 5, B6, 7). The statement that the viscidium (a sticky pad) “originates from a part of the rostellum” (Rasmussen, 1985) further supports Brown’s hypothesis regarding the origin of the viscidium.
These interpretations were widely accepted by many subsequent researchers, including renowned orchid scientists and illustrators such as Francis (Franz) Bauer, George Bentham (1800-1884; Fig. 9J), Olaf Hagerup (1889-1961; Fig. 9K), John Stevens Henslow (1796-1861; Fig. 9L), Joseph Dalton Hooker (1817-1911; Fig. 11G), and Charles Darwin (Fig. 1D) (Bauer & Lindley, 1830-1838, Bentham, 1881; Darwin, 1904; Hagerup, 1952; Henslow, 1858; Hooker, 1854, 1855; for reviews see Arditti & Flick, 1974; Cardoso-Gustavson et al., 2017; Strauss & Arditti, 1984).
Pioneering studies of orchid flower vascularization by Bengaluru Gundappa Lakshminarayana Swamy (1916-1980; Fig. 11I) provided further anatomical support for the stigmatic origin of the rostellum. In Habenaria Willd., for instance, the compound stigma has three vascular strands, though one fails to develop when the dorsal stigma fails to form (Garay, 1960; Swamy, 1948).
The Reverend Professor John Stevens Henslow (1796-1861), Darwin’s mentor and friend, suggested that the rostellum produces an adhesive substance and speculated that it might function as an anterior stigma (Henslow, 1858). This was later confirmed by George Bentham (1881), Joseph Dalton Hooker (1854, 1855), and Olaf Hagerup (1952). Heinrich Gustav Reichenbach (1823-1889; Fig. 11H) observed that this adhesive (viscid) substance accumulates in Cattleya Lindl. and Laelia Lindl. (Reichenbach, 1885).
Charles Darwin (1809-1882; Fig. 9D) studied the rostella of several orchids, including Catasetum Rich. ex Kunth (Fig. 2A-D, black and white; captions in the figures are those in the original) and Vanda R.Br. (Fig. 2E). He provided a clear description, detailed illustrations (Fig. 2), and a convincing hypothesis (Darwin, 1888, 1904): “The rostellum strictly is a single organ, formed by the modification of the dorsal stigma and pistil. . . In. . . the Orchideae there are three confluent pistils; of these the dorsal one forms the rostellum. . . ” and “there is every reason to believe that the whole of this upper stigma, and not merely a part, has been converted into the rostellum; for there are plenty of cases of two stigmas, but not one of three stigmatic surfaces being present in these Orchids which have a rostellum.” This concept has since gained general acceptance (Vermeulen, 1959).
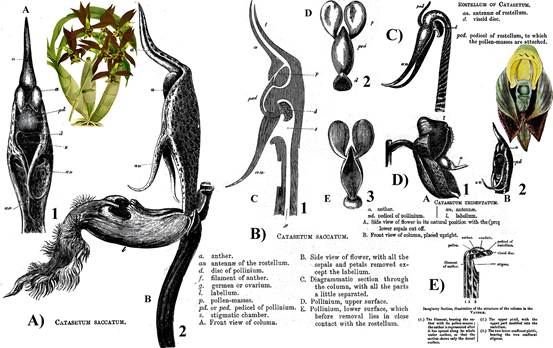
The captions and labels are those in the original publication, except for the bold numbers and letters, which were added for clarity.
What Darwin labeled as pedicel of the pollinium is now called stipe.
The stipe is connected to the viscidium (Darwin’s term is viscid disk).
Black and white drawings by Charles Darwin (Darwin, 1904).
Color illustrations: A) Painting by John Nugent Fitch of Catasetum saccatum, plate 83 in the Orchid Album (1882-1897).
D) Catasetum tridentatum, currently C. macrocarpum, plate 3329, volume 61 (1834) of Curtis’ Botanical Magazine.
Figure 2 Drawings and illustrations of Catasetum saccatum Lindl. (A-B), Catasetum tridentatum Hook., currently Catasetum macrocarpum Rich. ex Kunth (C-D), and Vanda Jones ex R.Br. (E).
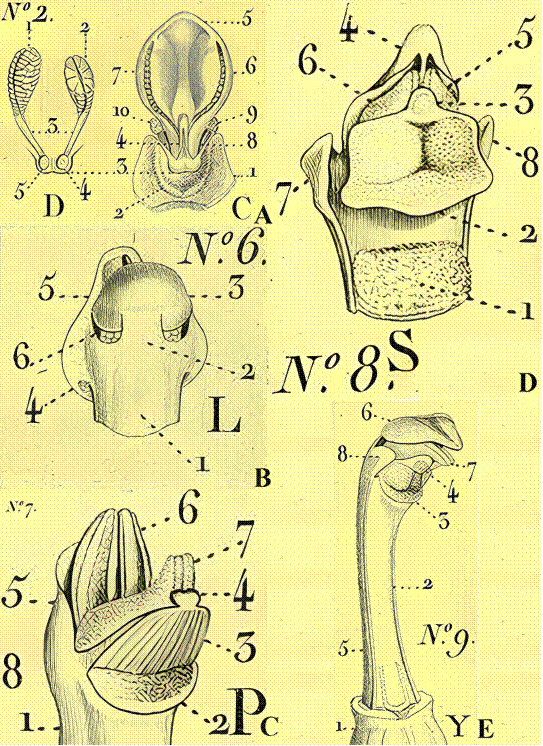
Only some of his drawings are presented here.
A. Loroglossum Rich.
B. Nigritella Rich.
C. Gymnadenia R.Br.
D. Platanthera Rich.
E. Herminium L. Generic names are those used by Richard. The numbers (N°2, N°6, N°7, N°8, N°9 and those pertaining to every structure as well as letters C, D, L, P, S, Y), in black or blue are as in the original.
Letters in modern bold Times Roman font (A, B, C, D, and E) at bottom right of the figures were added.
Only numbers pertaining to rostella in the figures are noted here.
A. In N°2 the rostellum is labeled as 3 in C and D.
B. The rostellum in N°6, L is labeled as 5.
C. Number for the rostellum in N°7, P is 3.
D. Designation for the rostellum in N°8, S is 3.
E. Rostellum is 4 in N°9, Y.
Numbering and lettering is somewhat confusing in the original, and therefore, also here.
The sepia cast is due to aging of the original.
Source: Richard (1818).
Figure 3 Orchidearum Genitalia (term used in the original source) as drawn by L. C. Richard.
Darwin’s special interest in the rostellum of Catasetum led him to suggest that the antennae (Fig. 2A1, B1, C, D2), which trigger the ejection of pollinaria (Fig. 2B1, B2) are part of this structure. He described them as “are prolongations of the sides of the anterior face of the rostellum” (Darwin, 1904). His drawings show this clearly (Fig. 2A1, B1, C, D2).
One of the preeminent orchid systematists during the last half century, Robert Louis Dressler (1927- 2019; Fig. 9H) noted that since Darwin, “most authors. . . defined the rostellum. . . as the median stigma lobe” (Dressler, 1989). However, since “the boundaries between the stigma lobes are often unclear, . . . authors used the term as Richard did” (Dressler, 1989). Dressler’s well-reasoned perspective defined the rostellum as the “modified portion of the median stigma lobe” (Dressler, 1961). Developmental studies on Bletia purpurea (Lam.) DC., Malaxis commelinifolia (Zoll. & Moritzi) Kuntze, Calanthe × veitchii R.H.Torr and other Epidendroid species “conformed that the rostellum is the upper part of the median stigma” and originates entirely from the primordium of the median carpel (Kurzweil, 1987a). In Orchidoid species, the rostellum is three-lobed and originates “entirely from the median carpel” (Kurzweil, 1987b). The rostellum in Neottioideae develops completely from the upper part of the median stigma lobe (Kurzweil, 1988). In the Australian Genoplesium fimbriatum (R.Br.) D.L.Jones & M.A.Clem. (Prasophyllum fimbriatum R.Br.), the rostellum is likewise formed from the median carpel (Kurzweil et al., 2005).
Leslie Andrew Garay (1924-2016; Fig. 11C), a distinguished orchid systematist of the 20th century, described the rostellum as “the median stigma (which) during reorganization of the flower has evolved into a new organ. . . with a specific function. It is the controlling and ensuring device for fertilization” (Garay, 1960). Although he did not address the antennae of Catasetum as Darwin did, his definition appears broad enough to include them.
It has been suggested that “the term ‘rostellum’ is used in various senses, sometimes referring to the entire median stigma-lobe and sometimes (emphasis added) to the extension only” (Rasmussen, 1985). In fact, the term “rostellum” is generally applied for all parts of this structure.
The rostella of Himantoglossum hircinum (L.) Spreng. (Loroglossum hircinum (L.) Rich.; Heusser, 1914) and Cattleya labiata Lindl. (Rotor & MacDaniels, 1951) are formed by the invagination of part of the stigma and subsequent forward growth. Because of these and similar observations, the rostella of these and other orchids, such as Cymbidium, were thought to represent a third stigma (Gellert, 1923; Heusser, 1914; Hirmer, 1920; Rotor & MacDaniels, 1951; Wolf 1865-1866). In Herminium monorchis (L.) R.Br., the rostellum originates from “the middle lobe (apex) of the median carpel lobe” (Rudall et al., 2013). In the Pleurothallidinae, the median stigmatic lobe of the carpel serves as the origin of the rostella, with this lobe differentiating before the lateral lobes (Cardoso-Gustavson et al., 2017). Thus, “the rostellum is accepted as a modified. . . portion of the median stigma lobe” (Cardoso-Gustavson et al., 2017).
There is a general agreement that rostella produces a viscid substance (Cardoso-Gustavson et al., 2017), which supports the theory of their stigmatic origin since stigmas typically produce sticky substances. However, other glands in plants produce similar secretions. Spiranthes Rich. provides evidence supporting the stigmatic origin of this sticky substance. In these orchids, two functional stigmas are located below an elongated third stigma, the rostellum, whose underside has been described as a functional, presumably sticky, stigma (Garay, 1960).
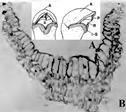
Theodor Franz Wolf (1841-1924; Fig. 11B), a theologian, geologist, and botanist, studied the rostella of European, Asian, and American orchids in the late 1800s (Fig. 4) and recognized the taxonomic significance of the rostellum. He observed that rostella can vary in size and form, consist of parenchyma cells along with spiral vessels, and produce adhesive in pads of various shapes.
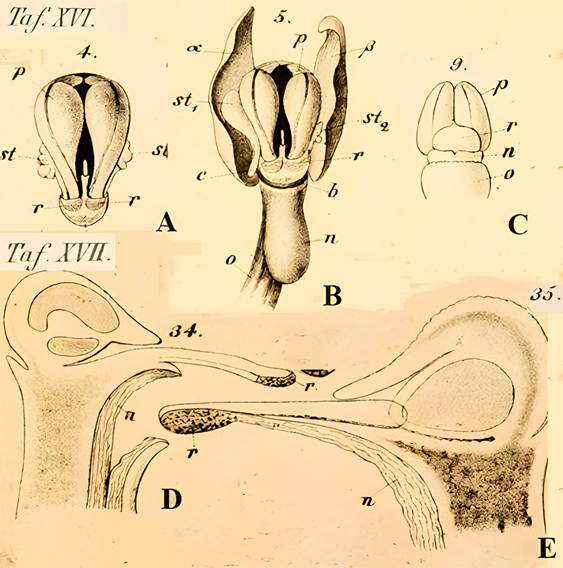
A. Taf. XVI, 4. Mature pollinia of Orchis mascula (L.) L.
B. Taf. XVI, 5. Position of rostellum in the column of O. mascula.
C. Taf. XVI, 9. Pollinia of O. mascula, on the rostellum.
D. Taf. XVII, 34. Positions of stamen, pollinia, stigma and rostellum in Lycaste aromatica (Graham) Lindl. in a longitudinal section.
E. Taf. XVII, 35. Longitudinal section through the gynostemium of L. aromatica.
Names and lettering used here are those in the original.
Only letters pertaining to the rostellum and associated structures are noted here: p, pollinia; n, stigma; r. rostellum; rounded shaded areas on r in A and B, viscidium; st, stamen.
Source: Wolf (1865-1866).
Sepia cast and low contrast are due to aging of the original.
Figure 4 Early detailed drawings of the rostellum by Theodor Franz Wolf (1841-1925) in his Taf. XVI and Taf XVII.
Ernst Hugo Heinrich Pfitzer (1846-1906; Fig. 9F), Professor of Botany and Director of the Botanical Garden at the University of Heidelberg, and a leading German orchid biologist of the late 1800s, was the first to use the rostellum as a taxonomically descriptive feature extensively. In his contributions to Adolf Engler (1844-1930) and Karl Anton Eugen Prantl’s (1849-1893) Die Natürlichen Pflanzenfamilien (published from 1844 until 1930), Pfitzer produced 605 illustrations and 192 figures of the Orchidaceae, many of which depict the rostellum’s variability in size and shape (Fig. 5).
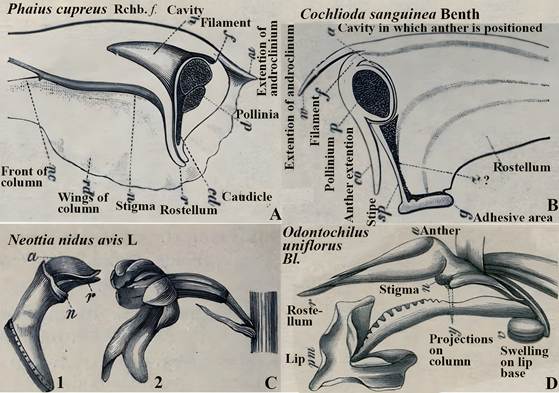
Symbols: Most labeling is on the original illustration except in C1 were a, anther; n, stigma; r, rostellum.
In A and B, androclinium is a collective name for stamens, which is hard to explain because orchid flowers do not have separate stamens.
Also, in A and B, stipe and caudiculum seem to be used for the same structure.
The question mark (?) In B refers to a symbol, which could be a sideways Z, but not deciphered with certainty.
Source: Pfitzer, 1889.
The grey cast is due to the aging of the original.
Figure 5 Drawings of orchid gynostemia (columns) showing different sizes and shapes of rostella in the first major taxonomic treatment of the Orchidaceae, which considered the rostellum.
The most detailed and impressive early study of the rostellum was conducted by Max Hirmer (1893- 1981; Fig. 9E), a German botanist, photographer, publisher, archaeologist, historian, and art student, who was dismissed as a professor by the Nazis in 1936 for political reasons. Hirmer studied the rostellum because, as he noted, “Darwin left open the organographic connection between the rostellum and the anther (and he studied them) in greenhouses in England and not in (the natural habitat) of the orchids . . . and the visiting (pollinators)” (Hirmer, 1920). He created longitudinal sections of numerous flowers, illustrating the differences in size and shape among rostella (Fig. 6). His drawings confirmed both the morphological variability of rostella and their consistent location within the gynostemium. Hirmer was also the first to include photomicrographs of the gynostemium in a publication, offering an unparalleled study of these intriguing yet somewhat mysterious organs in orchid flowers.
Source: Hirmer, 1920, courtesy of Thomas Zuhr of Hirmer Verlag and the Hirmer family.
Figure 6 Drawings of parts of the gynostemium, stigmas and rostella in one the best structural studies of the rostellum.
Hirmer reviewed previous research and statements on the function and taxonomy of the rostellum, reaching conclusions (beyond physiological aspects) that remain valid regarding the rostellum’s role, its functions, and its relationship with the pollinia and their adhesive pad, the viscidium. His comprehensive discussion spans anatomy, morphology, microscopy, pollen, pollinia, pollination, taxonomy, and evolution. One of his key conclusions was that the rostellum originates from the median stigma, which “no longer retains the function of pollen reception, but at least in its apical part functions in pollen transmission through the formation of an adhesive mass” (Hirmer, 1920 and see above). Hirmer’s meticulous and insightful work deserves more recognition than it has received.
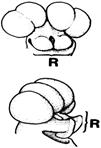
In his study of rostella in the Ophrydeae (now Orchideae) and Neottiae (now Neottieae), Dutch botanist Peter Vermeulen (1899-1981; Fig. 9G) observed structural differences in the rostellum between these groups (Vermeulen, 1959). He also noted that “in many Ophrydeae (and) several Neottiae,” the stigma has three lobes (Fig. 12), which is “contrary (to) the opinion of Darwin (Vermeulen, 1959).” Vermeulen believed that in Neottieae with three stigmas, the rostellum forms “in connection with, or above the median stigma lobe.” At the same time, in the more primitive Orchideae, it originates from the lateral stigma lobes. He suggested that rostella may not have a single origin.
Existing rostellum nomenclatural and origins questions
Despite extensive discussions (Arditti, 1992; Arditti & Flick, 1974; Cardoso-Gustavson et al., 2017; Dressler, 1981, 1993; Rasmussen, 1982, 1985, 1986a, 1986b, Valencia-Nieto et al., 2018; van der Pijl & Dodson, 1966; Vermeulen, 1955; Yam et al., 2009), questions remain surrounding the rostellum’s structure and classification due to insufficient information (Cardoso-Gustavson et al., 2017). For example, in species with three stigmas (Fig. 12A-D), such as Coeloglossum Hartm. (a synonym of Dactylorhiza Neck. ex Nevski), Dactylorchis (Klinge) Verm. (a synonym of Dactylorhiza), Galeorchis Rydb. (a synonym of Galearis Raf.), Himantoglossum Spreng., Ophrys L., Orchis, and Platanthera Rich., what is assumed to be the rostellum may, in fact, represent a different organ.
According to Garay (1960 and personal communication to J. Arditti around 1973), this structure “must be given a different name or it is merely connective tissue between the two thecae of the anthers.” These interpretations suggest that the definition of the rostellum might need broadening to encompass variations in its position, origins, and functions, indicating a need for further research.
In any case, if the rostellum or rostellar structures originated strictly from stigmas, then no orchids would possess both a rostellum and three stigma lobes. However, contrary to Darwin’s view, there are cases where such lobes exist (Fig. 12), as seen in Orchis, Galeorchis, Ophrys, Himantoglossum, Dactylorchis, Coeloglossum, and Platanthera (Vermeulen, 1955, 1959). This suggests two possibilities. The first is an independent origin for some rostellar structures, as Vermeulen (1959) proposed. The second is that the origin of the rostellum in orchidoids may be the same as that in epidendroids, as suggested by Kurzweil (1987a, b).
Leslie A. Garay also suggested that if the term “rostellum” is used strictly according to its original definition, a different term might be required for these rostellar structures (personal communication, ca. 1973, to J. Arditti from the late L.A. Garay, then at the Botanical Museum, Harvard University). Alternatively, what appears to be a rostellum could be connective tissue between the anthers. These interpretations highlight the complexity of the rostellum’s origins and functions and suggest that the traditional definitions may need to be broadened to reflect its diverse structural forms and potential variations in origin.
Rostellum and taxonomy
As previously stated, the rostellum has long been used in the description and/ or revision of orchid species (Batista et al., 2016, Eftimov, 2022; Jakubska-Busse et al., 2020; Pfitzer, 1889). Significant morphological differences in rostellum structure can be observed even between species within the same genus, such as Acianthera aphthosa (Lindl.) Pridgeon & M.W.Chase and Acianthera obovata (Lindl.) Pridgeon & M.W.Chase (see Table 1; Cardoso-Gustavson et al., 2017), justifying its use in taxonomic classification. In the predominantly African genus Satyrium Sw., the rostellum structure was deemed taxonomically and phylogenetically significant enough to aid in establishing phylogenetic relationships (Johnson & Kurzweil, 1998; Kurzweil & Linder, 1999); however, this was later contradicted by molecular data (personal communication to J. Arditti, 2023, from Hubert Kurzweil, now retired from the Singapore Botanic Gardens).
At one time, subtribe Calypsoinae (= Corallorhizinae) was considered potentially paraphyletic, partly due to the presence of hamulus or tegula (Freudenstein, 1994). The hamulus (Fig. 13B, D) is a stalk formed by the distal section of the rostellum, while the tegula (Fig. 13A, C) is a cuticular strap originating from the dorsal epidermis of the rostellum. Hamuli are present in Aplectrum (presumably Aplectrum Nutt., not Applectrum Blume), Corallorhiza Gagnebin, Cremastra Lindl., and Oreorchis Lindl. Based on a cladistic analysis of morphological features, it was suggested that these genera form the core of Calypsoinae and are closely related to a clade that includes Tipularia Nutt., Calypso (presumably Calypso Salisb., not Calypso Thouars), and Yoania Maxim. Govenia Lindl. is likely most closely related to Cymbidieae, such as Eulophia R.Br., due to its cellular epidermal pollinium stalk and Cymbidium-type velamen. Dactylostalix Rchb.f. and Ephippianthus Rchb.f. appear to represent earlier diverging lineages within the Epidendroideae.
In summary, the size and morphology of rostella are sufficiently variable (Fig. 1-6, 10, 12) to be useful in orchid taxonomy, as has been discussed by Valencia-Nieto et al. (2018).
Development and formation of the rostellum
Extensive studies of the development and formation of the rostellum have been undertaken only more recently (Cardoso-Gustavson et al., 2017, Freudenstein et al., 2002; Rasmussen 1982, 1985, 1986a, 1986b; Rudall et al., 2013, Valencia-Nieto et al., 2018).
Extensive studies on the development and formation of the rostellum have only recently been conducted (Cardoso-Gustavson et al., 2017; Freudenstein et al., 2002; Rasmussen, 1982, 1985, 1986a, 1986b; Rudall et al., 2013; Valencia-Nieto et al., 2018). In the early stages of rostellum ontogeny in Bulbophyllum lomsakense J.J.Verm., Schuit. & de Vogel (Table 1), whose synonymy with Sunipia thailandica (Seidenf. & Smitinand) P.F.Hunt is debated, the rostellum’s lobes appear as mere knobs, later becoming hammershaped at the apex (Rasmussen, 1986a). Just before anthesis, the rostellum lobe differentiates into a pollinium stalk and a “supporting tray” (Rasmussen, 1986a).
Pollinium stalks that arise from the rostellum are referred to as stipes (singular: stipe; Rasmussen, 1986a). Gene-derived stalks from the rostellum have been termed tegulae (singular: tegula; Rasmussen, 1986a; Fig. 13). Tegulae are found in epidendroid (formerly vandoid) orchids and in the Australian genus Prasophyllum R.Br. (see Rasmussen 1986a for a review). Upon removal, tegulae leave a scar on the rostellum, which is physiologically significant but likely not taxonomically relevant. In Phalaenopsis pulcherrima (Lindl.) J.J.Sm. (Doritis pulcherrima Lindl.), the rostellum is a long, pointed projection (Rasmussen, 1986a; Table 1).
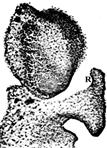
Observations indicate a lack of strict uniformity in the development, shape, and precise positioning of rostella. In the musk orchid, Herminium monorchis R.Br., a European terrestrial species, the rostellum differentiates through “deep trilobing of the apex of the median carpel, which is highly reminiscent of the trilobing of the labellum. Such carpel lobing also suggests differentialperhaps prolonged-timing of gene expression during floral ontogeny” (Rudall et al., 2013; Fig. 14).
Significant diversity in floral morphology and pollination systems is evident in the Epidendreae (Valencia-Nieto et al., 2018). A comparative study of rostellar development and other floral organs in six subtribes of Epidendreae at the late anther stage recorded notable differences. For example, the developing rostellum in Coelia triptera (Sm.) Mutel (subtribe: Calypsoinae) is transverse, with rectangular cells that form many intercellular connections in the lower tissue layer. A layer formed by the mature rostellum separates the stigmatic surface (Valencia-Nieto et al., 2018).
Despite mentioning a “lack of rostellum in Bletiinae” (Valencia-Nieto et al., 2018), the development of the rostellum in Bletia purpurea (Lam.) DC., is described as beginning with a transverse ridge, which forms a “defined rostellum” that projects “as a well-developed tongue shaped structure with rectangular epidermal cells” (Valencia-Nieto et al., 2018; Table 1). Underneath it “is the stigmatic surface covered by the well-developed rostellum” (Valencia-Nieto et al., 2018).
In Chysis bractescens Lindl. (Fig. 15) and Chysis limminghei Linden & Rchb.f., the developing rostellum at the anther’s base is semi-lunate in shape and points upward, a position retained through later stages. In contrast, Chysis laevis Lindl. shows only two underdeveloped, inconspicuous projections (Valencia-Nieto et al., 2018).
In Ponera juncifolia Lindl., a species of Ponerinae, the obtriangular rostellum consists of two cell types and projects forward. One cell type is slender and elongated longitudinally, extending from the front to the middle part, while the other is glandular and produces abundant secretions (Valencia-Nieto et al., 2018).
Exudate production by the rostellum
In unopened flowers, the rostellum produces a mucilaginous exudate, described as visually resembling stigmatic exudate (Arditti & Flick, 1974; Cardoso-Gustavson et al., 2017). This exudate is “copious and viscous” similar in appearance to the stigmatic exudate of Zootrophion atropurpureum (Lindl.) Luer and Acianthera fenestrata (Barb.Rodr.) Pridgeon & M.W.Chase (Cardoso-Gustavson et al., 2017), although its precise chemical composition and physical state remains unknown. In many orchid taxa, the pollinaria contain an “adhesive secreted by the. . . rostellum” (Freudenstein et al., 2002). In the Pleurothallidinae, the rostellum has a palisade epidermis that secretes a mucilaginous exudate resembling that of the stigma, released through cell walls and the cuticle (Cardoso-Gustavson et al., 2017).
In Acianthera fenestrata, pollinia and pollen tubes adhere to the rostellum, which has been interpreted to suggest that the rostellar exudate “assists adherence of the pollinia and the germination and growth of pollen tubes” (Cardoso-Gustavson et al., 2017). This adherence may be due to the physical properties of the exudate. At the same time, the germination and growth of pollen tubes suggest that the chemical compositions of rostellar and stigmatic exudates are likely similar or identical. However, while Cardoso-Gustavson et al. (2017) study was of high quality, it is important to note that it: a) included only a few orchid species, and b) did not involve detailed chemical and physical analyses of rostellar and stigmatic exudates. Consequently, its findings may not be representative of all taxa. Given the diversity within orchids, additional research could reveal findings that either support, expand upon, or challenge current knowledge.
In many orchids, the rostellum contains “a sharply defined body made up of rostellar glue and usually some cellular tissue” (Dressler, 1989). Early literature often refers to this area as a gland or glandular structure, even though it is not strictly a gland. Other terms include retinaculum, proscollum, viscid disc, and viscidium, with Dressler (1989) favoring “proscolla”. At anthesis, pollinia attach to this area “directly or by a stipe” (Dressler, 1989). To avoid confusion, Dressler and Salazar (1991) proposed the term “viscarium” to denote “the glue and glue-producing area in those orchids that do not have a distinct (removable) viscidium” because “it is better to use a special term in place of rostellum for a structure that is only part of the rostellum” (Dressler & Salazar, 1991).
Rostellum and pollination
Darwin’s studies, along with those of other researchers (for a review, see Arditti, 1992), led to the conclusion that the rostellum plays a major role in orchid pollination. This conclusion is well supported, as indicated by the examples below.
As previously mentioned, Charles Darwin developed a keen interest in the rostellum (for a review, see Yam et al., 2009), studied it extensively (Darwin, 1904), and wrote: A) “Pollen masses are never retained on the rostellum except by accident.”, B) in some orchids, such as Vanda (with some species now under Papilionanthe Schltr.), pollinaria are attached to the rostellum (Fig. 2A). C) the rostellum of Catasetum ((presumably Catasetum Rich ex Kunth, not Catasetum Blind.; (Fig. 2B) has “lost the normal (stigmatic) function of being fertilized” and “its shape is most singular with the upper end thickened, bent over and produced into two long. . . antennae”, D) additionally, variations in the structure of rostella can be found across different orchid species.
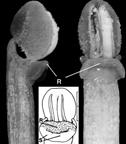
In Cyrtorchis chailluana (Hook.f.) Schltr., the column is very short, the rostellum is long and extended, and the pollinia are positioned on two separate, elongated stalks that merge into a single stipe near the rostellum (Fig. 8F). When pollinaria (i.e., pollinia, stipe, and viscidium) are removed from the rostellum, a long tear or slit forms in its extension, facilitating detachment (Gellert, 1923; Fig. 8F).
In Oncidium wentworthianum Bateman ex Lindl. and Oncidium otogaya (an unverified taxon not listed in IPNI, Plants of the World Online, or the RHS List of Orchid Hybrids), pollinaria removal involves a two-stage process. The mechanical resistance of this detachment process has been measured (Thielen et al., 2021). The first stage involves the removal of the anther cap, which protects the pollinia and prevents the premature detachment of young, immature pollinaria. Once the pollinaria mature, the anther cap sheds, initiating the second stage: a detachable bond forms between the pollinarium and the rostellum. Pollinators can overcome this bond by applying a force of at least 10.8 mN (in O. otogaya) or 12.6 mN (in O. wentworthianum) to the viscidium, thereby breaking the connection and allowing for pollinarium removal.
Some orchids, such as species of Catasetum (Darwin, 1904), do not display stigmatic functionality in their rostella. However, certain autogamous species retain or reacquire the rostellum. In an auto-pollinating variant of Bulbophyllum bicoloratum Schltr., rostellum penetration and pollen tube growth have been observed, suggesting that the non-vascularized parts of its rostellum may contain stigmatic tissue (Gamisch et al., 2013). Rostellum penetration has also been documented in Eulophia nyasae Rendle, though this may not result from self-pollination (Gamisch et al., 2013; Williamson, 1984). Despite these observations, some rostella are considered unsuitable for pollen germination (Cardoso-Gustavson et al., 2017), potentially due to orchids’ specific pollen germination requirements, which appear more stringent than those of other plants (Kemeç-Hürkan et al., 2015; Pritchard & Prendergast, 1989; Rao & Lee, 1973; Sanford et al., 1963).
This inability of the rostellum to support pollen germination has evolutionary advantages. In orchid pollination, pollen germinates on the stigma in a position that allows the pollen tubes to reach the ovary and fertilize ovules, leading to seed formation. In contrast, pollen tubes germinating on the rostellum may be unable to reach the ovary, thus preventing successful fertilization and reproduction.
In Neottia ovata (L.) Bluff & Fingerh. (Listera ovata (L.) R.Br.), pollinia adhere to the pollinator’s face with the “help of the explosive mechanism of the rostellum” (Nazarov & Gerlach, 1997). When the pollinator visits another flower, the pollinia are inserted into the stigma (Nilsson, 1981). In Cephalanthera longifolia (L.) Fritsch., pollinia attach to the thorax of the pollinating vector, Halictus Latreille. Upon a subsequent visit to a flower, the rostellum scrapes the pollinia of the pollinator (Nazarov & Gerlach, 1997). Species of Coryanthes Hook. are pollinated when the pollinator passes through a narrow tunnel between the labellum and the gynostemium while escaping from a liquid-filled bucket. As the pollinator moves through the tunnel, the rostellum removes the pollinia (Nazarov & Gerlach, 1997; Fig. 16).
An intriguing and unique function of the rostellum has been observed in the self-incompatible, food-mimicking orchid Coelogyne fimbriata Lindl. in China. This orchid has a large rostellum that “projects beyond and between the stigma and the anther” (Cheng et al., 2009) and forms a flap over the stigma (Fig. 8G1). Within 24 hours after pollination, the flap bends down, covering the stigma (Fig. 8G2). This prevents additional pollen deposition and causes visiting wasps, which might otherwise attempt to pollinate, to back away (Cheng et al., 2009). Orchids have evolved various mechanisms that prevent, or at least discourage, repeat visits by pollinators to flowers that have already been pollinated.
Pollinia typically adhere to pollinators via the viscidium, which is named for the viscid substance produced by the rostellum. In most orchids, pollinia are attached to a stalk bearing the viscidium at its base, an important feature in orchid taxonomy (for discussions, see Rasmussen 1985, 1986a). One type of these stalks, the caudicula, originates from the tapetum and pollen mother cells (Rasmussen, 1985, 1986a). Orchids lacking caudiculae have viscidia originating from the rostellum (Freudenstein et al., 2002). Another type of stalk, the stipe, is formed from the rostellum (Freudenstein et al., 2002). Stipes are found in epidendroid and other orchid subdivisions and are formed by a strap of rostellar epidermis in epidendroid orchids (Rasmussen 1985, 1986a; Fig. 13).
Rostellum and post pollination phenomena
It is important to emphasize that the removal of pollinaria causes wounding of the rostellum (Fig. 8D-F). While this wound has no role in the transfer of pollen, it plays a significant physiological role in inducing post-pollination phenomena (discussed below). For a long time, pollination was thought to be the primary, perhaps sole, biological function of the rostellum. Oakes Ames (1874-1950; Fig. 11D) poetically described the rostellum as having “resigned itself to the altruistic function of bringing to its fertile sisters the fertilizing pollen of an anther” (Ames, 1946), a statement that is both teleological and anthropomorphic. While the rostellum indeed supports reproduction, it also initiates the senescence and eventual death of floral segments that have completed their functions once a flower is pollinated or emasculated (for reviews, see Arditti, 1979, 1992, Avadhani et al., 1994).
For over a century after its naming, the rostellum was studied primarily for its structural, taxonomic, and pollination-related characteristics. During this period, no one-not even Darwin-suspected it had an important physiological function. Given the state of plant physiology knowledge at the time (the discovery of the first plant hormone, auxin, was still about 65 years away) and the limited understanding of orchid physiology in particular, this oversight is neither surprising nor scientifically disappointing.
The definitions and descriptions discussed above do not address the physiological function of the rostellum (for reviews, see Arditti, 1992; Arditti & Flick, 1974; Avadhani et al., 1994). Although it was known that orchid flowers produce ethylene by the time Vermeulen, Dressler, and Garay formulated their definitions, the physiological role of the rostellum and its involvement in producing this gaseous plant hormone had yet to be discovered.
The effects of ethylene on plants were first identified in 1901 by Russian scientist Dimitry Nikolayevich Neljubov (1879-1926). In 1934, Richard Gane demonstrated that plants biosynthesize ethylene, but research on this gas as a plant hormone remained limited between 1930 and 1960, and none of it involved the rostellum. Extensive knowledge about ethylene as a plant hormone-including its effects on orchids-accumulated between 1960 and 1980 and has continued to expand since (Abeles, 1973; Abeles et al., 1992; Burg & Dijkman, 1967; Dijkman & Burg, 1970).
Dr. John Thomas Curtis (1913-1961; Fig. 11A), Professor of Botany at the University of Wisconsin, Madison, was the first to suggest that rostella might have a physiological function. In his study of fruit development in Phalaenopsis Blume, Curtis observed that pollination caused wilting of the perianth in Phalaenopsis Pamela (Fig. 7: A1; Curtis 1943 wrote “Pamala” but the correct name is Pamela (Phalaenopsis Elisabethae × P. Fontainebleau)) and swelling of the column (Fig. 7: A2, A3). Curtis found that removal of the pollinia (Fig. 7: A4), emasculation (Fig. 7: A5), and even “merely stroking the rostellum with a needle” (likely causing a wound) also triggered perianth wilting (Curtis, 1943). Although Curtis could not provide a “satisfactory explanation of the physiological reactions” he hypothesized that these effects involved a substance similar to the plant hormone auxin, specifically indoleacetic acid (IAA) (Curtis, 1943).
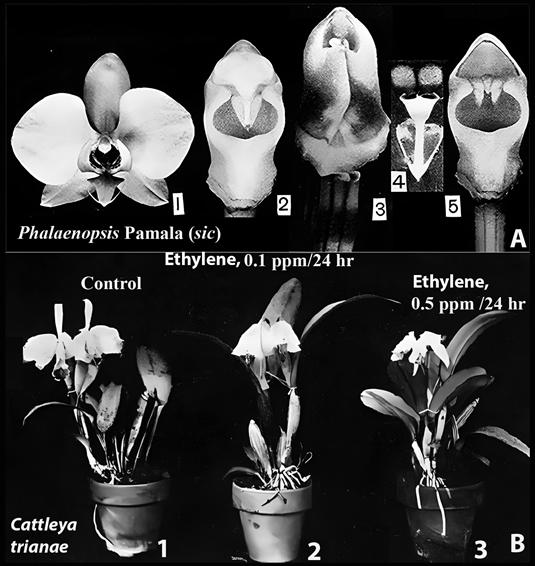
A. Phalaenopsis Blume Pamala (sic).
1. Unpollinated flower.
2. Gynostemium (column) of unpollinated flower.
Light grey horizontally elliptical area is the stigma.
3. Gynostemium following pollination.
4. Pollinarium.
5. Gynostemium after removal of the pollinia.
Light grey triangular area on top is where the pollinarium was located.
The barely visible thin dark line at the base of the light grey area is part of the rostellum; two horn like structures pointing down are also parts of the rostellum; dark grey horizontally elliptical area is the stigma.
This figure duplicates parts of Fig. 1B.
It is included here intact with no additional labels because of its historical value.
The black numerals on white background are as in the original.
B. Effects on ethylene on Cattleya flowers.
Exposure to the gas causes wilting.
Sources: A. Curtis (1943).
He spelled the grex name as “Pamala,” but the correct name is “Pamela.”
B. Davidson (1949) with notations added.
Figure 7 Pollination and ethylene effects on orchid flowers.
Curtis’s suggestions about the involvement of the rostellum and a regulatory substance were prescient, as the effects of IAA (indoleacetic acid) and ethylene on orchid flowers are similar. IAA, a plant hormone, can initiate ethylene production. However, at the time, little was known about the connection between IAA and ethylene or the roles of these plant hormones in orchid flower physiology. Additionally, ethylene was not yet considered a plant hormone, and its effects on orchid flowers were unknown. Curtis continued his research on post-pollination phenomena, but not specifically on the rostellum. One of his students later published the first substantial report on post-pollination phenomena in orchid flowers (see Avadhani et al., 1994 for a review and reminiscences by the student).
Six years after Curtis’s findings on the role of the rostellum in post-pollination phenomena in Phalaenopsis, Dr. O. Wesley Davidson (ca. 1904-1991) demonstrated at Rutgers University that exposure of Cattleya Lindl. flowers to atmospheric ethylene caused injury and wilting (Davidson, 1949; Fig. 7B2, B3). At that time, ethylene was not yet recognized as a plant hormone, and no connection was made between ethylene and the rostellum.
Today, it is well established that the rostellum is involved in inducing post-pollination phenomena in Angraecum Bory (Strauss, 1976, Strauss & Koopowitz, 1973), Cymbidium (Arditti & Flick, 1974, Chadwick et al., 1980; 1986; Duncan & Schubert, 1947), Dendrobium Sw. (Nair et al., 1991), Phalaenopsis (Curtis, 1943; Duncan & Schubert, 1943), Vanda (Burg & Dijkman, 1967; Dijkman & Burg, 1970), and other orchids (for reviews, see Arditti, 1979, 1992; Avadhani et al., 1994). In Cymbidium, rostellum injury induces anthocyanin synthesis-a known effect of ethylene (Fig. 17F)-but does not trigger stigmatic closure, which is instead induced by auxins. Ethylene production by the rostellum, a modified stigma, aligns with reports that stigmas in other plants produce ethylene as well (for a brief review, see Strauss & Arditti, 1984).
Research on the interactions between ethylene, the gaseous plant hormone (Fig. 12F), and orchid flowers began in the 1940s (for reviews, see Akamine, 1963; Arditti, 1979, 1992; Avadhani et al., 1994; Burg & Dijkman, 1967; Chadwick et al., 1986; Davidson, 1949, Dijkman & Burg 1970; Duncan & Schubert, 1943; Hew & Yong, 1997, 2004; Hew et al., 2002). This research led to the discovery that the following factors trigger the autocatalytic production of ethylene (Fig. 19B): pollination (Fig. 17A, B), emasculation (Fig. 17A, B, D), application of auxin (IAA or its analog NAA) (Fig. 17B, E, H), aging of flowers (Fig. 17C, fading of blossoms (Fig. 17G), injury to blooms (Fig. 17H), and application of the ethylene precursor 1-ami-nocyclopropane-1-carboxylic acid (ACC).
Ethylene production triggered by auxin application and pollination follows the same pattern: it begins, peaks, and then declines after a similar period (Fig. 17B). This parallel timing is likely due to post-pollination ethylene synthesis being initiated by auxin from the pollinia.
In contrast, ethylene production following emasculation takes longer to start and reach its peak than ethylene induced by pollination (Fig. 17A, B, D), possibly because emasculation triggers the production of wound-induced ethylene, which has a delayed onset. Emasculated flowers also tend to have a longer lifespan than pollinated ones, which is evolutionarily advantageous since they remain receptive to pollen and can produce seeds if pollinated later. In nature, emasculation often occurs when pollinators remove the pollinaria.
A suggestion that rostellum desiccation is “responsible for post-emasculation phenomena in orchid flowers” is supported by data showing higher levels of ethylene production in emasculated Cymbidium and Phalaenopsis flowers at 60% relative humidity (RH) compared to 100% RH (Woltering & Harren, 1989). However, further studies have not been conducted on Cymbidium, Phalaenopsis, or other orchids. While this suggests a role for desiccation, it seems intuitively unlikely to play as significant a role as plant hormones like ethylene and auxin.
Structural, color, and vegetative differences between different clones within the same grex may result from mutations or genetic variation within seedling populations. Papilionanthe Schltr. (formerly Vanda) ‘Rose Marie’ (Fig. 17A) and Vanda (now Papilionanthe) ‘Miss Joaquim’ (Fig. 17G), the National Flower of Singapore and a natural hybrid (Papilionanthe teres (Roxb.) Schltr. var. andersonii × Papilionanthe hookeriana (Rchb.f.) Schltr.), share the same parentage (Arditti & Hew, 2007). As a result, the patterns of ethylene production following emasculation in these orchids are similar, though not identical (Fig. 17A vs. Fig. 17G). Any differences likely stem from varietal characteristics or different cultivation conditions in Florida (Fig. 17A) and Hawaii (Fig. 17G).
Papilionanda R.E.Schult. & Pease ‘Petamboeran’ (Fig. 17B) was originally hybridized and cultivated in Indonesia. Its parentage includes Vanda foetida J.J.Sm. × Papilionanthe ‘Miss van Deun’ (Papilionanthe Schltr. ‘Miss Joaquim’ (Papilionanthe teres (Roxb.) Schltr. var. andersonii × Papilionanthe hookeriana)) × Papilionanthe teres (Roxb.) Schltr. The overall plant architecture, with its thin upright stems and terete leaves, is strongly influenced by Papilionanthe teres (Roxb.) Schltr., which is also evident in the similarity of the initial peak of ethylene production after pollination in Papilionanthe ‘Rose Marie’ (Fig. 17A) and Papilionanda ‘Petamboeran’ (Fig. 17B).
Altogether, it is reasonable to assume that the similarities and differences in ethylene production, timing, levels, and patterns in the rostellum, stigmas, and other parts of the gynostemium are genetically determined. More research is needed to clarify these aspects of rostellum and orchid flower physiology, as the rostellum is clearly a more complex organ than its small size suggests. Ethylene evolution begins: A) within an hour or less after pollination (Fig. 17A, B, D). B) Following emasculation, between one and 96 hours later (Fig. 17A, B, D, G, 18). C) Two to five hours after the application of auxin (indoleor naphthalene-acetic acid) (Fig. 17B, 18). D) Twenty-two days after the opening of intact, uninjured, unpollinated, and untreated flowers in Dendrobium ‘Pompadour’ (Fig. 17D) and earlier in Cymbidium ‘Jungfrau’ (Fig. 17H).
Despite speculation that ethylene is produced and released by rostella and stigmas (for reviews, see Arditti 1979, 1992; Arditti & Flick 1974; Avadhani et al., 1994), experimental evidence was lacking until Dr. Arthur V. Chadwick (1943- Fig. 11E, then at Loma Linda University in California), the late Dr. Leslie P. Nyman (a postdoctoral fellow in Joseph Arditti’s laboratory; for a photograph, see Orchid Biology, Reviews and Perspectives Vol. VI), Dr. Helen Nair (Fig. 11F, now retired from the University of Malaya in Kuala Lumpur), and Dr. Joseph Arditti conducted the necessary experiments (Chadwick et al., 1986; Nair et al., 1991). Emasculation and surgical experiments, combined with applications of naphthalene acetic acid (NAA), demonstrated that in Cymbidium ‘Jungfrau’ (Chadwick et al., 1986): A) regardless of any other treatment, NAA (an IAA analog) induces or enhances ethylene evolution (Fig. 18A1-3, B4-6, C7-9, D10- 12). B) The stigma and rostellum are the primary sites of ethylene evolution (Fig. 18A3, B6, C7-9, D10-11). C) An excised rostellum produces the highest levels of ethylene per-weight basis (Fig. 18D10). D) The gynostemium can still produce ethylene even after the stigma and rostellum are removed (Fig. 18B4, B5).
Besides pollination, another event that induces ethylene evolution by the rostellum in nature is emasculation (i.e., pollinia removal by a pollinator; Fig. 18B5). To investigate how pollinia removal triggers ethylene evolution, Dr. Michael S. Strauss (then a graduate student in JA’s laboratory) used light and electron microscopy (Strauss & Arditti, 1984, Strauss & Koopowitz, 1973). The structure of the Phalaenopsis rostellum made the experimental process challenging, as fixation, embedding, sectioning, and staining were difficult for both light and electron microscopy. Images of the Phalaenopsis rostellum revealed elongated, prosenchymatous (soft and succulent) vacuolated cells (Fig. 8B, C), with a strand of vascular tissue extending through the rostellum. These cells are rich in mitochondria with easily visible cristae (Fig. 8C), which may be associated with the metabolic regulation of xylem and/or phloem functions and could provide the energy required for ethylene synthesis.
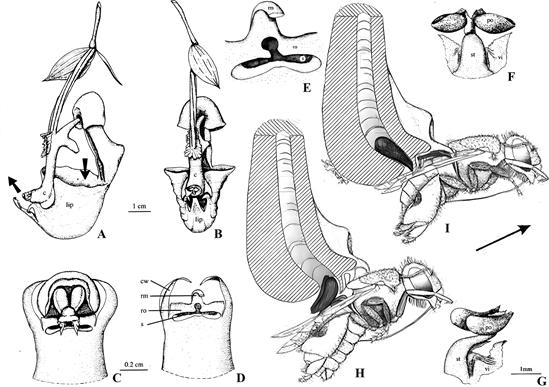
Pollinia removal causes wounding along a tear in the rostellum (Fig. 8D, arrows; Fig. 8E, F). Previous studies, which were mainly structural, reported similar wounding in other orchids (Gellert, 1923; Table 1). As is typical in plants, tissue damage induces wound ethylene production. Because ethylene production is autocatalytic, the wound ethylene initiates a subsequent increase in gas production.
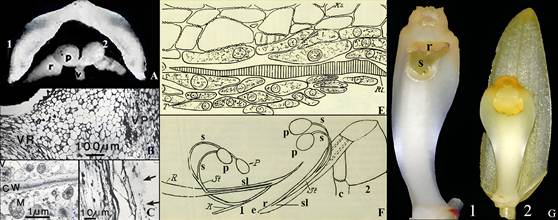
A. Excised rostellum of Cymbidium Jungfrau.
1. Rostellum only. About 9-10 mm wide (side to side), 12 mm wide (front to back), 2-3 mm thick.
2. Rostellum with pollinaria in place. Lettering: p, pollinia; r, rostellum; v, viscidium.
B. Light microscope photograph of Phalaenopsis Blume gynostemium showing vascular strands leading to the rostellum (VR) and pollinia (VP).
C. Electron microscope photograph of rostellum cells showing numerous mitochondria (M), cell wall (CW), and vacuole (V).
D. Light microscope photograph of the wounded outer edge of the rostellum where the viscidium was attached, showing wound where cells are degenerating (arrows).
E. Separation layer between the viscidium and rostellum in Vandopsis lissochiloides Pfitzer.
Lettering as in the original: Ks, viscidium; Rt, separation area, break or crack where wounding occurs; T, separation tissue consisting of loosely packed cells.
F. Column, pollinarium and rostellum of Cyrtorchis chailluana Schltr.
1. Lower end of rostellum with detached pollinarium.
The stipe is bifurcated about half the distance above the viscidium, with every branch carrying a single pollinium.
2. Column following the removal of the anther cap.
The stipe is detached from the rostellum.
Dotted line indicates he original position of the stipe.
When the pollinarium is removed a slit (wound) forms on the rostellum.
Lettering: Cursive letters are on the original.
Modern Times Roman font letters were added.
c, column; e, tip of the extension of the rostellum; K, viscidium; p, pollinium; R and r, rostellum; s, branch of the stipe; sl, slit; St, unbranched part of stipe.
Light grey cast in E and F is due to aging of the original.
G. Coelogyne fimbriata Lindl.
1. Column showing stigma and rostellum before pollination.
2. Rostellum (yellow, nearly oval) covering stigma after pollination.
Sources: Joseph Arditti (A), Dr. Michael S. Strauss (B-D), a graduate student in JA’s laboratory at the time; Gellert, 1923 (E-F), and courtesy of J.-C. Cheng and Y.-B. Luo (G).
The yellowish and greyish cast of E and F are due to the aging of the original.
Figure 8 Rostella: intact and microscopy.

A. Ludovico (Louis) Claudio (Claude) Marie Richard (1754-1821).
B. Robert Brown (1773-1858).
C. John Lindley (1799-1858).
D. Charles Robert Darwin (1809-1882).
E. Max Hirmer (1893-1981).
F. Ernst Hugo Heinrich Pfitzer (1846-1806).
G. Peter Vermeulen (1899-1981).
H. Robert Dressler (1927-2019).
I. Francis (Franz) Andreas Bauer (1758-1840).
J. George Bentham (1800-1884).
K. Olaf Hagerup (1889-1961).
L. John Stephens Henslow (1796-1861), friend and mentor to Darwin.
Sources: A, https://images.shoutwiki.com/roses/6/6c/Richard%2C_Louis_Claude_Marie.jpg and https://archives.somme.fr/https://archives.somme.fr/ark:/58483/j39p8wr04cq6.
B-D, F, I-L, Wikipedia; E. Courtesy Thomas Zuhr, Hirmer Verlag and the Hirmer family; G, Joseph Arditti; H, Orchids.
Figure 9 Students of the rostellum.
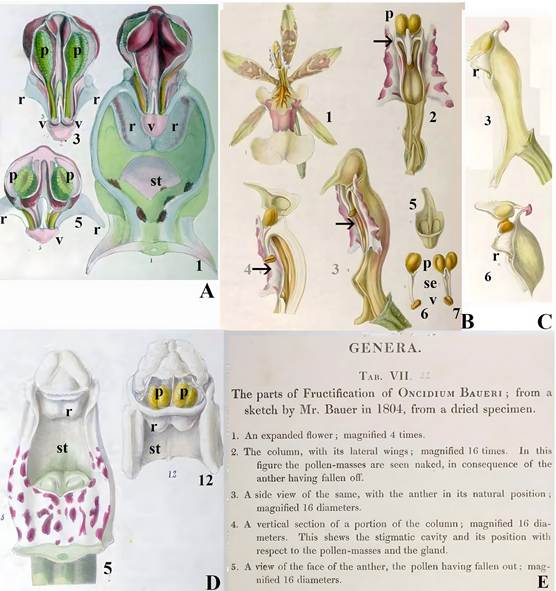
A. Fructification. Plate 3.
Illustration 1, original caption, “A front view of the column of Orchis mascula with the base of the labellum, shewing the position of the anther with its pouch, the cells of the former being partially open, so as to shew the pollen within; on each side of the anther are the two glandular tumors, which are the rudiments of stamens; magnified 15 times.”
Illustration 3, original caption. “A front view of the anther with the cell opened, so as to exhibit the pollen masses inside in their natural position; magnified 15 times. The pouch in the glands of the pollen-masses are confined is forced a little down.”
Illustration 5. “A transverse section of fig 3; magnified 15 times. In this the inner structure of the cells of the anther is exhibited, and the pouch is in its natural position.”
Illustrations numbers (except for modern bold face letters and numbers, which were added for clarity), spellings, lack of italics for species names, and spaces before semi colons are as in the original.
“Gland” seems to refer in illustration 3 to what is currently called “viscidium.”
It is not clear to what “glandular tumors” refers to.
Both the location of the “pouch” and the attachment of the “gland” (viscidium) to it in illustrations 1, 3 and 5 indicate that it is the rostellum.
Numbers 1, 3 and 5 refer to illustration numbers in the original plate 3 (which contains 16 illustrations).
Lettering: ac, anther cap; p, pollinia; r, rostellum; st, stigma; v, viscidium.
B, E. Genera Plate 7.
Parts of the fructification of Oncidium baueri Lindl.
Illustration 1. Expanded flower.
Illustrations 2-6. See caption in E.
In all illustrations anther refers to the anther cap, caudicula is the stipe (se), gland is the viscidium (v) and pollen-masses is the pollinia (p).
In 2, 3 and 4 the arrows point to what seems to be the rostellum.
Both B and E are fully reproduced to show the language, terminology, descriptions and spellings as they were used by Lindley.
C. Genera plate 8. Fructification of Galeandra baueri Lindl.
Illustration 3. Side view of column.
Illustration 6. Side view of the apex of the column with the anther cap raised up to show pollinia, their stipes and the viscidium.
What seems to be the rostellum (r) was painted by Bauer, but not identified.
D. Genera Plate 6.
Parts of fructification of Dendrobium speciosum Sm.
Illustration 5. Front view of the column.
Illustration 12. Apex of the column with the anther cap removed to show pollinia, the rostellum is shown, but not identified.
E. Original captions for B.
Lettering: p, pollinia; r, what seems to be the rostellum; st, stigma.
Source: A-E, Bauer and Lindley, 1830-1838.
Some of the images are not sharp due to the aging of the original.
Figure 10 Rostella painted by Francis (Franz) Bauer with captions by John Lindley.

A. John Thomas Curtis (1913-1961).
B. Theodor Franz Wolf (1841-1924).
C. Leslie Andrew Garay (1924-2016). The original photograph was out of focus and required sharpening with Photoshop. No other photograph was available.
D. Oakes Ames (1874-1950).
E. Arthur V. Chadwick (1943).
F. Helen Nair (Mrs. James Bonney) (No year here?).
G. Joseph Dalton Hooker (1817-1911).
H. Heinrich Gustav Reichenbach (1823-1889).
I. Bangalore Gundappa Lakshminarayana Swamy (1918-1980).
Sources: A, courtesy Dr. Kenneth Cameron; B, D, G-I, (1874-1950) Wikipedia; C, courtesy Bill Thoms; E, Courtesy Dr. A. V, Chadwick; F, Joseph Arditti.
Figure 11 Rostellum investigators.
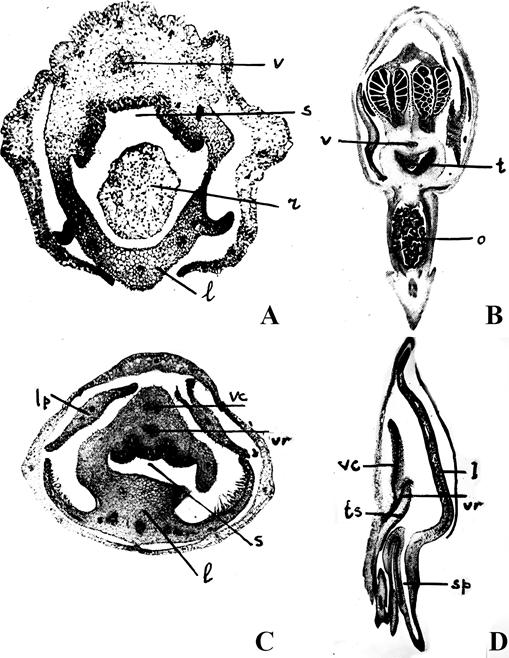
A. Transverse section of Himantoglossum Spreng., flower.
B. Ophrys L., no species listed in the original.
C. Transverse section of Ophrys L., flower.
D. Galeeis spectabilis (L.) Raf. (Galeorchis spectabilis (L.) Rydb.)
Lettering, handwritten as in the original: l, lip; lp, lateral petal in C; r, part of the rostellum in A; s, sigma with three separate lobes in A, C; sp, spur in D; t, third tongue shaped stigmatic lobe in B; ts, third stigmatic lobe in D; v, vascular bundle in A; vc, vascular bundle of anther in C; vr, vascular bundle of rostellum in C, no explanation given in the original for D; vs, vascular bundle for anther in D.
Source: Vermeulen, 1955.
Figure 12 Stigmas with three lobes.
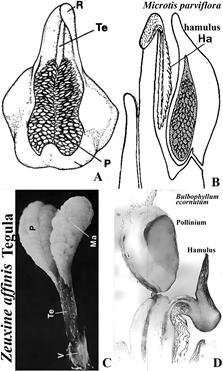
A. Line drawing of Zeuxine affinis (Lindl.) Benth. ex Hook.f., tegula.
B. Line drawing of Microtis parviflora R.Br. hamulus.
C. Photograph of Zeuxine affinis (Lindl.) Benth. ex Hook.f., tegula.
D. Longitudinal section of Bulbophyllum ecornutum (J.J.Sm.) J.J.Sm., hamulus.
Explanation of symbols: Ha, hamulus; Ma, massula; P, pollinium; R, rostellum; Te, tegula; V, Viscidium.
Sources: A, B, Rasmusen (1982); C, D, Rasmussen (1986a).
Figure 13 Tegula and hamulus.
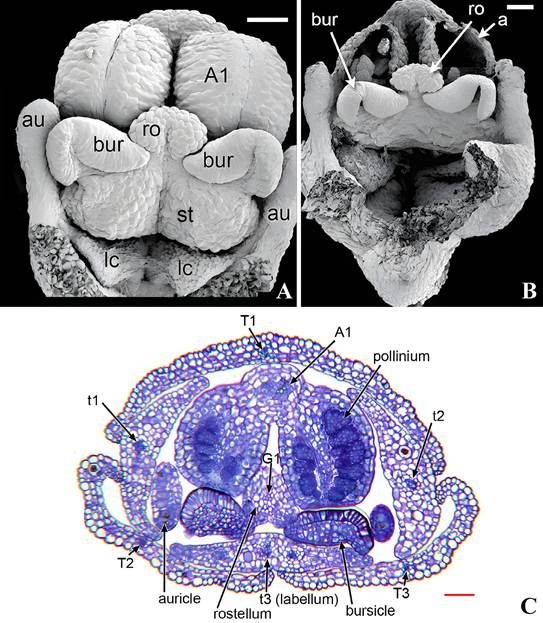
A. Scanning electron microscope of a stage in rostellum and bursicle development.
B. Scanning electron microscope of dissected gynostemium of anthetic flower showing developed rostellum.
C. Light microscope photograph of a serial transverse section of a pre-anthetic bud early state of rostellum development and vasculature.
Explanation of symbols: A1, putative vascular bundle of outer whorl stamens; a, fertile anther; au, auricle; bur, bursicle; lc, lateral carpel; ro, rostellum; st, stigmatic surface, T1, T2, stamens; t1, t2, petals; t3, labellum; Scales, bar in A, B, C, 100 μm.
Source: Rudall et al. 2013.
Figure 14 Development of rostellum in Herminium monorchis (L.) R.Br.
Experiments conducted by others (Burg & Dijkman, 1967; Fig. 17B) and in J. Arditti’s and Chadwick’s laboratories (Chadwick et al., 1986; Fig. 17) confirmed that pollen induces ethylene evolution by providing auxin, which is known to be present in high concentrations in orchid pollen (see Avadhani et al., 1994 for a review). Additionally, the enzyme responsible for ethylene production is present in orchid flowers before pollination, and its levels increase after pollination or auxin treatment (Bui & O’Neil, 1998).
An unanswered question at the time was why ethylene production drops sharply after reaching its peak (Fig. 17A-E, G, H; 18A-D; 19A, B). To investigate this phenomenon, the auxin naphthalene acetic acid (NAA; Fig. 19A) and the ethylene precursor 1-aminocyclopro-pane-1-carboxylic acid (ACC; Fig. 19B) were applied to the stigmas of Dendrobium ‘Tommie Tokyo’ (Nair et al., 1991; Fig. 15A, B). For the first six hours following treatment, ethylene evolution was similar for both NAA and ACC applications, although ethylene production was higher after NAA application. Both treatments peaked at 28 hours, after which ethylene production decreased substantially. Similar patterns were observed in other orchids (Fig. 17), though with differences in timing, ethylene levels, and curve shapes.
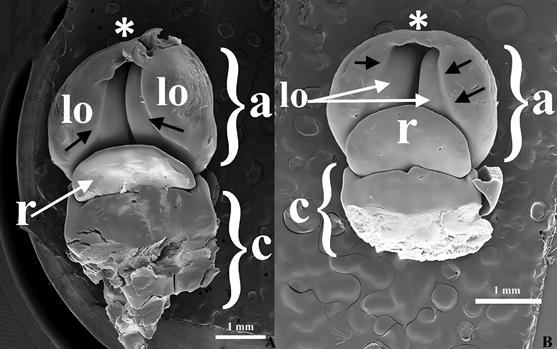
A. Chysis bractescens Lindl.
B. Chysis limminghei Linden & Rchb.f.
Explanation of symbols: *, apex of anther; black arrows, longitudinal lines in epidermal tissue; a, anther; c, column; lo, lobule; r, rostellum.
Source: Valencia-Nieto et al. 2018.
Figure 15 Scanning electron microscope photographs of early stages of rostellum and anther development in Chysis.
A. Lateral view of flower with one column wing, petals and sepals removed.
B. Dorsal view of flower with one column wing, petals and sepals removed.
C. View from above of apex of column before first visit by pollinator.
D. View from above of apex of column after first visit by pollinator, which removed the pollinarium.
E, F, G. Rostellum and pollinarium.
H, I. Pollination, scraping of pollinia from pollination by the rostellum.
Explanation of symbols: arrows in a, path of pollinator, which falls into the bucket (arrow on right) and escapes through narrow tunnel (arrow on left); c, column; cw, column wing; po, pollinarium; rm, rostellum mid lobe; ro, rostellum; s, stigma; st stipe; vi, wing.
Source: Nazarov and Gerlach, 1997.
Figure 16 Coryanthes senghasiana G.Gerlach, flower and its pollination.
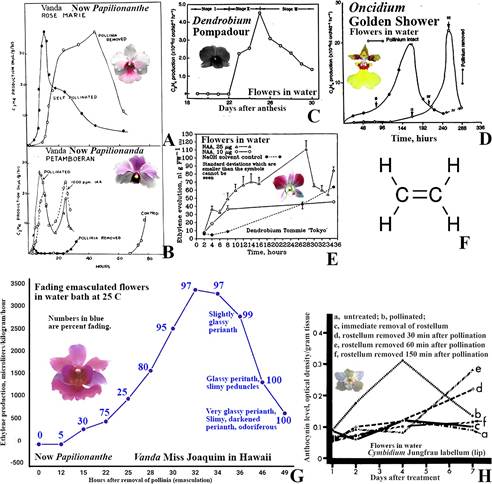
A, B. First graphic presentation of ethylene evolution by orchid (Vanda) flowers following pollination, emasculation (removal of pollinia), and auxin (indoleacetic acid, IAA) treatment.
C. Ethylene evolution by senescing untreated cut Dendrobium, flowers in water.
D. Ethylene evolution by intact and emasculated Gomesa R.Br. Golden Shower flowers.
E. Ethylene evolution by flowers of Dendrobium. Tommie ‘Tokyo’ after auxin application (naphthaleneacetic acid, NAA) to stigmas.
F. Ethylene.
G. Ethylene evolution by fading flowers of Vanda R. Br. Miss Joaquim, a natural hybrid, which is now the National Flower of Singapore.
H. Effects of pollination and removal of the rostellum (all of which are known to produce ethylene) on anthocyanin production by the labellum (lip) of Cymbidium Jungfrau flowers.
Patterns of ethylene evolution by all orchids (A-E, G, and H) are similar.
Timing and level of ethylene evolution differ.
Source: A, Burg and Dijkman, 1967 with permission from Dr. Yunde Zhao, Editor in Chief of Plant Physiology; C, D, courtesy Dr. Helen Nair; E, Nair et al., 1991; F, Wikipedia; G, graph prepared for this article from data in Akamine, 1963; H, Arditti & Flick, 1974.
Sources of photographs: A, D, G, courtesy Dr. Tim Wing Yam; B, E, H, Joseph Arditti; C, courtesy Dr. Helen Nair.
Figure 17 Ethylene evolution and its effects.
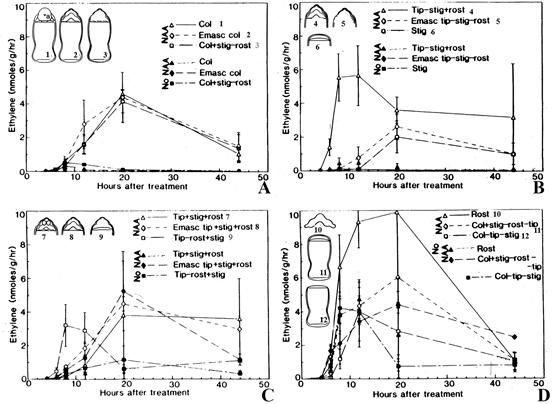
A. Auxin application following emasculation or excision of the rostellum.
B. Auxin application following excision of the stigma.
C. Auxin application following excision of the rostellum.
D. Auxin application following excision of rostellum and gynostemium tip or the gynostemium tip and stigma.
Explanation of symbols: a, anther cap; Col, column (gynostemium); Emasc, emasculation; rost, rostellum; stig, stigma; tip, tip of the gynostemium.
Numbers following captions (upper right of A-D) correspond to the numbers of surgical treatment (upper left of A-D).
Bars at data points are standard deviations, some of which are large.
Source: Chadwick et al., 1986.
Figure 18 Effects of auxin applications (naphthaleneacetic acid, NAA) and surgical treatments of gynostemia on ethylene evolution by the column and its parts of Cymbidium Jungfrau (for a photograph see Fig. 17H).
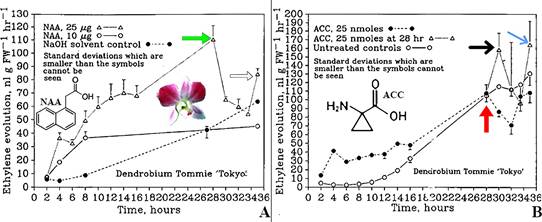
A. Evolution of ethylene after application of auxin (naphthaleneacetic acid, NAA) to the stigma.
Green arrow points to peak of evolution 28 hours after treatment.
The open arrow points to a secondary peak at 35 hours.
B. Production of ethylene after application of the ethylene precursor, 1-aminocyclopropane-1-carboxilic acid (ACC).
Red arrow points to the initial peak of ethylene production at 28 hours, and the time at which additional ACC was applied to the stigma.
The black arrow points to a peak resulting from the application of the additional ACC.
A secondary peak resulting from the application of ACC is marked with a blue arrow.
Source: Graphs, Nair et al, 1991; photograph, Joseph Arditti.
Figure 19 Ethylene production by flowers of Dendrobium Sw. Tommie ‘Tokyo’.
Adding ACC at 28 hours restored ethylene evolution, resulting in a peak at 30 hours, a drop at 32 hours, and a second peak at 34 hours (Fig. 19B). Both new peaks were higher than the initial peak at 28 hours (Fig. 19B). These findings suggest that either the ethylene production pathway becomes temporarily impaired at 28 hours and is restored by ACC, or that the decrease in ethylene production is due to ACC depletion.
Conclusions
Despite its small size and often concealed position, the rostellum plays a crucial role as a floral organ because it: A) Separates the pollinia from the stigma, and with few exceptions, prevents self-pollination. B) Produces an adhesive, facilitating pollination. C) Develops the antennae in Catasetum, enabling a unique pollination mechanism. D) Varies in size and shape, making it useful in orchid systematics. E) Plays an important role in orchid pollination phenomena by producing ethylene, which induces the senescence and death of floral organs that are no longer needed, thereby conserving energy and allowing the reutilization of substances and resources (Harrison & Arditti, 1976).
Despite its significance in orchid biology, the rostellum has not been extensively studied, and much remains to be discovered. This lack of information required several speculations and assumptions in this review, which may lead to differing opinions and may need revision as more is learned about the rostellum.












 uBio
uBio