Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.48 n.4 San José Dec. 2000
Athayde Tonhasca Jr.1 and Marcos Antonio Lima Bragança
Received: 2-XI-99 Corrected: 2-VI-00 Accepted: 16-VI-00
Abstract
We determined the size characteristics of foragers of the leaf-cutting ant Atta sexdens in a mature eucalyptus forest in Campos dos Goytacazes, Rio de Janeiro State, Brazil, at daytime (7: 30 to 10: 00 hr) and nighttime (19: 00 to 23: 00 hr). There were no significant differences between daytime and nighttime ant mass (Ma), but leaf fragment mass (Ml) and burden (B = [Ma + Ml]/Ma), which indicates relative load capacity, were significantly greater at daytime. There was a positive linear relationship between Ma and Ml for the combined daytime and nighttime data, and increases in Ma resulted in lower B. We compared A. sexdens characteristics with published results for Atta cephalotes, a closely related species. A. sexdens is larger and therefore able to carry heavier loads, but its burden is about 72% of the average value for A. cephalotes. We suggest that the lower load capacity of A. sexdens in comparison to A. cephalotes is related to its relatively larger size.
Key words
Atta sexdens, Atta cephalotes, ecology, foraging, leaf-cutting ants.
The leaf-cutting ant Atta sexdens (L., 1758) is common in primary forest throughout most of South America, but it readily invades new areas of agriculture or forestry, where it may become a serious pest (Cherrett 1986). The range and economical importance of A. sexdens have been increasing, probably because of the decline of native forests (Fowler 1983). Foraging of A. sexdens is predominantly nocturnal, although daytime foraging may occur sparsely throughout the day or during short periods of intense activity (personal observation). During daytime foraging, A. sexdens is exposed to harsh climatic conditions such as high temperatures and low humidity (Pereira da Silva 1975, Fowler and Robinson 1979), as well as parasitism from several species of phorid flies (Feener and Moss 1990).
Because of the high degree of polymorphism of Atta spp. (Wilson 1980a), it could be expected that A. sexdens colonies undergo a reduction in the average size of foragers from nighttime to daytime to cope with higher risks of desiccation (Wetterer 1990) and phorid parasitism (Orr 1992). This study was conducted to determine the size characteristics of A. sexdens foragers and whether there were significant differences on mass and load capacity between diurnal and nocturnal foragers. We also compared A. sexdens and Atta cephalotes (L., 1758) regarding the sizes of foragers and their loads. Research on the ecology of Atta spp. has focused mainly on A. cephalotes in natural forests, with considerably less available information on A. sexdens in disturbed habitats. Because both species have the widest distributions of Atta spp. in South America (Fowler et al. 1989), we considered these comparisons worthwhile.
Material and methods
The study was conducted from August 1995 to February 1996 in a forest of eucalyptus (Eucalyptus spp.) of approximately 1,300 ha located 35 km from Campos dos Goytacazes (21o48'S, 41o20'W), Rio de Janeiro State, Brazil. The forest undergrowth was composed mostly of grasses, which are rarely harvested by A. sexdens. Foraging trails of adult A. sexdens colonies were selected according to the ants' foraging activity, which was determined by the presence of fresh pieces of eucalyptus leaves along trails and around tunnel entrances. Because foraging activity varies among colonies and among trails within colonies, the location of observed trails varied during the study period, but at least five trails from five nests were used at each sampling date.
Leaf-cutting ants were sampled on three pairs of daytime (from 7: 30 to 10: 00 hr) and nighttime (from 19: 00 to 23: 00 hr) sampling dates: August 29 (n = 51) and 31 (n = 107), September 6 (n = 88) and 5 (n = 80), and February 7 (n = 47) and 6 (n = 107). Additional, non-paired samples were obtained at daytime on August 25 (n = 51), September 27 (n = 38), and February 16 (n = 125), and at nighttime on September 19 (n = 114). Each sample consisted of loaded ants collected randomly at approximately equal numbers from each foraging trail. The same trails were used on each pair of dates. Ants and their loads were placed individually in glass vials, which were held in an ice chest to reduce loss of water of the leaf fragments. In the laboratory, ants and leaf fragments were weighed to the nearest 0.1 mg on a precision balance. The ants' relative load capacity was evaluated with burden (B): B = (Ma + Ml)/ Ma, where Ma and Ml are ant and leaf fragment masses, respectively (Rissing 1982).
To compare day and night results of Ma, Ml and B, we used mixed-model ANOVA's on the paired data only. Periods (day and night) were considered fixed effects and dates were considered blocks of random effects. Thus, F tests for period effects were calculated with the interaction between period and date. When interactions were not significant at a probability level of at least 0.25, interaction and residual mean squares were pooled (Sokal and Rohlf 1995). Analyses of Ma and Ml were done after a log(x) transformation to correct heterogeneity of variances. In the results, means are followed by ± 1 standard deviation.
Because division of labor is associated with polymorphism of Atta spp., head size is a good indicator of the workers' foraging capability (Wilson 1980a, 1980b). Head size of 161 ants selected at random from our samples was measured by the standard head width, which is the greatest width of the head viewed face on (Wilson 1980a). Measurements were taken to the nearest 0.1 mm with a dissecting microscope equipped with an ocular micrometer. The values of head size were correlated with mass for determination of an allometric relationship.
Results
The ANOVAs indicated that the effects of dates were not statistically significant for Ma, Ml or B, but there was a significant interaction period x date for Ma (F2,474 = 6.71, P = 0.001). As the interactions period x date for Ml(P = 0.44) and B (P = 0.93) were not significant, residual and interaction mean squares for those variables were pooled. Daytime Ma was not significantly different from nighttime Ma, but Ml and B were significantly higher during the day (Table 1).
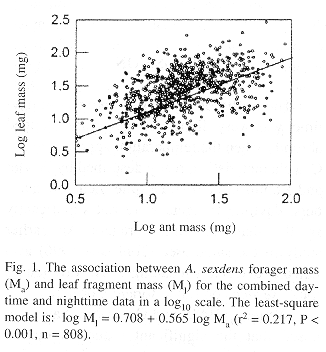
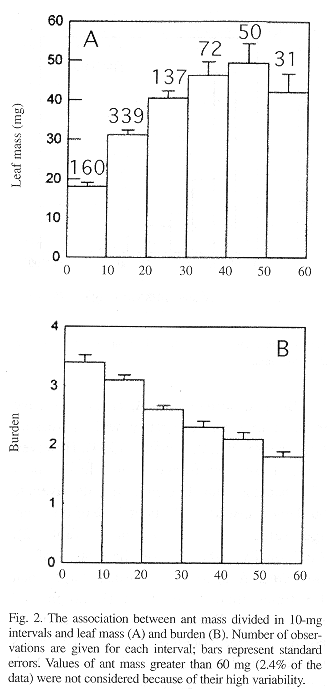
Similar to other leaf-cutting ant species (Burd 1995 and references therein), there was a positive linear relationship between mass of A. sexdens foragers and their loads (Fig. 1). The least-squares method was not adequate to describe the association between B and Ma because regression diagnostics demonstrated that the assumption of homogeneity of variances did not hold despite transformations. However, histograms of Ma data divided in 10-mg intervals clearly demonstrated that an increase in Ma resulted in higher Ml and lower B (Fig. 2). The frequency of the mass-class distribution of all ants indicates that most foragers are in the 10-20 mg interval (Fig. 3). The allometric relationship between head size (HS) and Ma for A. sexdens can be expressed as HS = 0.70 + 0.53 Ma1/2 (r2 = 0.983, P < 0.001, n = 161). By converting mass values to head sizes, we determined that less than 20% of foragers have head widths between 2.2 and 2.6 mm, which was proposed by Wilson (1980b) as the most efficient size for A. sexdens foragers.
Both A. sexdens and A. cephalotes have reduced transport efficiency with increasing forager size (Fig. 2; Wetterer 1994). However, while A. sexdens foragers carry loads about 50% heavier than A. cephalotes, the average burden of A. sexdens is about 72% of the average value for A. cephalotes (Table 2). Additionally, while more than 80% of A. cephalotes foragers carry burdens larger than 1.5 times their body sizes (Rudolph and Loudon 1986, Wetterer 1994), almost half of all A. sexdens foragers (47.2%) had loads smaller than 1.5 times their body sizes, which corresponds to B = 2.5 (Fig. 2).
Discussion
The traffic of A. sexdens foragers at daytime was very light and generally stopped abruptly when the temperature approached 30o C, a characteristic already described by Fowler and Robinson (1979). Despite their low numbers, daytime foragers were not significantly smaller than nighttime foragers. An earlier study in the same area (Tonhasca 1996) also failed to detect any meaningful diel differences for A. sexdens on trails, foragers or not.
Wetterer (1990) and Orr (1992) have proposed that the significant reduction on the average daytime mass of A. cephalotes foragers could be a strategy to escape parasitism from phorid flies, which prefer larger ants. Although we expect a greater impact of phorids on A. sexdens during daytime (Bragança et al.1998), the effect of these parasitoids in response to diel differences has to be better evaluated, as we have observed strong phorid activity at nighttime (unpublished). The similarity between day and night foragers size may be related to the two-stage foraging system deployed by A. sexdens. While arboreal workers cut the petioles and drop the leaves, other workers cut leaf fragments on the ground and transport them to the nest (Fowler and Robinson 1979). Thus, efficient foraging either at daytime or nighttime require workers of a minimum size for cutting the hard tissue of petioles, and this should be particularly important in homogeneous, mature commercial forests. Nonetheless, if foraging during the day is a riskier activity than at night because of greater chances of desiccation or parasitism, it could be expected that foragers optimize their daytime excursions by carrying greater loads. In fact, leaf fragment weight and burden of A. sexdens was heavier for daytime foragers.
Despite the significant relationship between ant and fragment masses, the linear model was a poor predictor of this relationship, even with a large number of observations and the log scale used. Equivalent results were obtained for A. cephalotes and Atta colombica (Guérin-Méneville, 1844)(e.g., Wetterer 1990, Shutler and Mullie 1991, Wetterer 1994, Burd 1995). It is clear from field observations that foraging of A. sexdens is subject to a great deal of "noise" from the environment. Ants drop their loads when they are disturbed by high wind speed, other arthropods and by hovering phorid flies (Bragança et al. 1998). Abandoned fragments may be picked up by other ants, and in these cases there is no apparent load size selection. Fragments also are abandoned when they become stuck in obstacles along trails, although occasionally foragers remove pieces of these fragments and resume their trips. As traffic along trails is routinely impaired by litter, larger fragments are more likely to be dropped because of obstacles. It has been demonstrated that leaf-cutting ants carry loads below their capacity, possibly because of a trade off between load mass and ant velocity (Rudolph and Loudon 1986, Lighton et al. 1987) or an overall gain of efficiency at the colony level (Burd 1996). Transport difficulties may be another factor that contribute to the relatively lower burden of larger workers of A. sexdens and A. cephalotes (Lighton et al. 1987) and the lower burden of A. sexdens in relationship to A. cephalotes. If that is the case, the lower foraging capacity of A. sexdens in comparison with A. cephalotes is associated with the larger size of A. sexdens foragers.
Acknowledgements
Thanks are expressed to Milton Erthal Jr. for helping with the field work. Jackie Blackmer, Gilberto Albuquerque and Og de Souza made important contributions on early drafts of this manuscript. A. Tonhasca Jr. was supported by a grant from the International Foundation for Science.
Resumo
Nós determinamos as características de tamanho de forrageiras da saúva Atta sexdens em uma floresta adulta de eucalipto em Campos dos Goytacazes, estado do Rio de Janeiro, Brasil, nos períodos diurno (7: 30 a 10: 00 hr) e noturno (19: 00 to 23: 00 hr). Não houve diferença significativa entre o dia e a noite para a massa das formigas (Ma), mas a massa dos fragmentos de folhas (Ml) e o esforço ([Ma + Ml]/Ma), o qual indica a capacidade relativa de carga, foram significativamente maiores durante o dia. Houve uma correlação linear positiva entre Ma e Ml para os dados diurnos e noturnos combinados, e o aumento em Ma resultou em esforço menor. Nós comparamos as características de A. sexdens com resultados publicados para Atta cephalotes, uma espécie correlata. A. sexdens é maior e portanto capaz de carregar cargas mais pesadas, mas seu esforço é cerca de 72% do valor médio obtido para A. cephalotes. Nós sugerimos que a menor capacidade de carga de A. sexdens em relação a A. cephalotes é devida ao seu tamanho relativamente maior.
References
Bragança, M. A. L., A. Tonhasca Jr. & T. M. C. Della Lucia. 1998. Reduction in the foraging activity of the leaf-cutting ant Atta sexdens caused by the phorid Neodohrniphora sp. Entomol. Exp. Appl. 89: 305-311. [ Links ]
Burd, M. 1995. Variable load size-ant size matching in leaf-cutting ants, Atta colombica (Hymenoptera: Formicidae). J. Insect Behav. 8: 715-722. [ Links ]
Burd, M. 1996. Server system and queuing models of leaf harvesting by leaf-cutting ants. Amer. Natur. 148: 613-629. [ Links ]
Cherrett, J. M. 1986. History of the leaf-cutting ant problem, p. 10-17. In C. S. Lofgren & R. K. Vander Meer (eds.). Fire ants and leaf-cutting ants: biology and management. Westview, Boulder, Colorado. [ Links ]
Feener, D. H., Jr. & K. A. G. Moss. 1990. Defense against parasites by hitchhikers in leaf-cutting ants: a quantitative assessment. Behav. Ecol. Sociobiol. 26: 17-29. [ Links ]
Fowler, H. G. 1983. Distribution patterns of Paraguayan leaf-cutting ants (Atta and Acromyrmex) (Formicidae: Attini). Stud. Neotrop. Fauna Env. 18: 121-138. [ Links ]
Fowler, H. G. & S. W. Robinson. 1979. Foraging by Atta sexdens (Formicidae: Attini): seasonal patterns, caste and efficiency. Ecol. Entomol. 4: 239-247. [ Links ]
Fowler, H. G., M. I. Pagani, O. A. Silva, L. C. Forti, V. Pereira da Silva & H. L. Vasconcelos. 1989. A pest is a pest is a pest? The dilemma of Neotropical leaf-cutting ants: keystone taxa of natural ecosystems. Environmental Manage. 13: 671-675. [ Links ]
Lighton, J. B. R., G. A. Bartholomew & D. H. Feener, Jr. 1987. Energetics of locomotion and load carriage and a model of the energy cost of foraging in the leaf-cutting ant Atta colombica Guer. Physiol. Zool. 60: 524-537. [ Links ]
Orr, M. R. 1992. Parasitic flies (Diptera: Phoridae) influence foraging rhythms and caste division of labor in the leaf-cutter ant, Atta cephalotes (Hymenoptera: Formicidae). Behav. Ecol. Sociobiol. 30: 395-402. [ Links ]
Pereira da Silva, V. 1975. Contribuição ao estudo das populações de Atta sexdens rubropilosa Forel, e Atta laevigata (Fr. Smith) no Estado de São Paulo (Hym.: Formicidae). Stud. Entomol. 18: 201-250. [ Links ]
Rissing, S. W. 1982. Foraging velocity of seed-harvester ants, Veromessor pergandei (Hymenoptera: Formicidae). Environ. Entomol. 11: 905-907. [ Links ]
Rudolph, S. G. & C. Loudon. 1986. Load size selection by foraging leaf-cutter ants (Atta cephalotes). Ecol. Entomol. 11: 401-410. [ Links ]
Shutler, D. & A. Mullie. 1991. Size-related foraging behaviour of the leaf-cutting ant Atta colombica. Can. J. Zool. 69: 1530-1533. [ Links ]
Sokal, R. R. & F. J. Rohlf. 1995. Biometry. Freeman, New York. 887 p. [ Links ]
Tonhasca Jr., A. 1996. Interactions between a parasitic fly, Neodohrniphora declinata (Diptera: Phoridae), and its host, the leaf-cutting ant Atta sexdens rubropilosa. Ecotropica 2: 157-164.
Wetterer, J. K. 1990. Diel changes in forager size, activity, and load selectivity in a tropical leaf-cutting ant, Atta cephalotes. Ecol. Entomol. 15: 97-104. [ Links ]
Wetterer, J. K. 1994. Forager polymorphism, size-matching, and load delivery in the leaf-cutting ant, Atta cephalotes. Ecol. Entomol. 19: 57-64. [ Links ]
Wilson, E. O. 1980a. Caste and division of labor in leaf-cutter ants (Hymenoptera: Formicidae: Atta). I. The overall pattern in A. sexdens. Behav. Ecol. Sociobiol. 7: 143-156. [ Links ]
Wilson, E. O. 1980b. Caste and division of labor in leaf-cutter ants (Hymenoptera: Formicidae: Atta). II. The ergonomic optimization of leaf cutting. Behav. Ecol. Sociobiol. 7: 157-165. [ Links ]
and nighttime A. sexdens foragers. Means are followed by ± 1 standard deviation.
| Period | | | |
| Daytime | | | |
| Nighttime | | | |
| ANOVA | | | |
of A. cephalotes and A. sexdens. Means are followed by ± 1 standard deviation.
| Species | | | | | |
| A. cephalotes | | | | | |
| A. sexdens | | | | | |
a value estimated from Table 1 in Wetterer (1994).
Centro de Ciências e Tecnologias Agropecuárias, Universidade Estadual do Norte Fluminense, Av. Alberto Lamego, 2000, Campos dos Goytacazes, RJ 28015-620, Brazil.
1corresponding author Athayde Tonhasca Jr. UENF-CCTA. Av. Alberto Lamego, 2000, Campos dos Goytacazes, RJ 28015-620, Brazil. Fax: +55-24-726-3746. E-mail: tonhasca@uenf.br