Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.48 n.1 San José Mar. 2000
biology of Perditomorpha brunerii and Cephalurgus anomalus
(Hymenoptera: Colletidae, Andrenidae)
Maria Cristina Gaglianone 1
Abstract
Nesting and foraging behavior were studied in Cephalurgus anomalus and Perditomorpha brunerii, two oligolectic bee species on Malvaceae flowers, in the University of São Paulo campus, Ribeirão Preto, Brazil. Bees were monitored on flowers of Sida, Malvastrum, Gaya and Wissadula. Perditomorpha brunerii females collect pollen with the forelegs and abdomen to transport it in short-branched hairs on the hind tibia and metasomal sterna; males patrol flowers and spend the night in closed flowers of Sida cerradoensis and Sida sp. C. anomalus females lean the dorsal region of the body against the petals and collect pollen with the forelegs, accumulating the grains on the mesepisterna before placing them into sparse unbranched hairs on the anterior outer side of the hind tibiae; males patrol and wait for females in flowers, where the copulate throughout the foraging season. Both species nest in the soil among the host plants. P. brunerii bees build solitary nests; whereas in C. anomalus one or more foraging females are associated with an individual nest. The nests, in both species, consist of a descent straight main tunnel and cells arranged singly and horizontally at the end of the branches, which are filled with soil. The efficient foraging behavior, location of the nests among the host plants and scopal setae that allow the transport of large pollen grains in large amounts indicate an intimate association between these two bee species and Malvaceae flowers.
Key words
Behavior, Cephalurgus, foraging, Malvaceae, nests, oligolectic bees, Perditomorpha.
Most bees depend on flowers for their food. Further, flowers serve as mating and resting place to many bee species (Eickwort and Ginsberg 1980). Oligolectic bees restrict pollen collection to a few related plants (Linsley 1958) and frequently have behavioral strategies and morphological or physiological adaptations associated with the gathering and transport of pollen of certain flowers. The oligolectic behavior seems to have appeared several times through the evolution of the bees. Several species were considered to be oligoleges among the Colletinae as well as among the Panurginae in South America.
Perditomorpha brunerii Ashmead (Colletidae, Colletinae) is distributed in the Southern and Southeastern Brazil and in the Northern Argentina (Moure 1954). This species was captured on flowers with large pollen grains of Malvaceae: Abutilon pauciflorum St. Hil. (Schlindwein 1995) and Sida rhombifolia L. and Onagraceae (Wilms 1995). In the region of Ribeirão Preto, such bees were captured only on Malvaceae flowers (J.M.F. Camargo, pers. inf.).
Michener and Lange (1957) studied nests of Lonchopria cingulata Moure and other Colletinae. They observed P. brunerii digging a vertical hole at the base of a bank in Parana and a female in a 15 cm deep vertical burrow.
Different aspects of the behavior of oligolectic Panurginae bees in South America were studied by Rozen (1989), Wittmann et al. (1990), Ruz and Rozen (1993), Rozen and Ruz (1995) and Schlindwein & Wittmann (1997b).
Cephalurgus anomalus Moure and Oliveira (Andrenidae, Panurginae) was originally reported in Southeastern Brazil and Paraguay. Camargo and Mazucato (1984) found this species to be an oligolege on Sida rhombifolia L. (Malvaceae). Additional studies have also registered it on Sida flowers (Rozen 1989, Silveira et al. 1993).
Observations on the behavior of the bees on flowers and the nesting biology of P. brunerii and C. anomalus, as well as, those on the morphological structures involved in the transport of pollen are reported in this paper.
Materials and Methods
The study was conducted in the University of São Paulo campus, in Ribeirão Preto, Brazil (21°1022 S, 47°51 W). Visits of bees were monitored on flowers of the following Malvaceae herbs or shrubs: Sida rhombifolia L., Sida glaziovii Schum., Sida acuta Burm. f., Sida carpinifolia L., Sida cordifolia L., Sida cerradoensis Krapov., Sida urens L., Sida glomerata Cav., Sida linifolia Cav., Sida sp.1, Sida sp.2, Malvastrum coromandelianum (L.) Garcke, Gaya domingensis Urb. e Wissadula hernandioides (LHér.) Garcke.
Flowers and visiting bees were observed between 9:00 and 17:00 h, from October,1994 to June,1995, which included the activity period of P. brunerii and C. anomalus (Gaglianone, in press). Bees behavior was observed directly or analysed from video recordings and photographs.
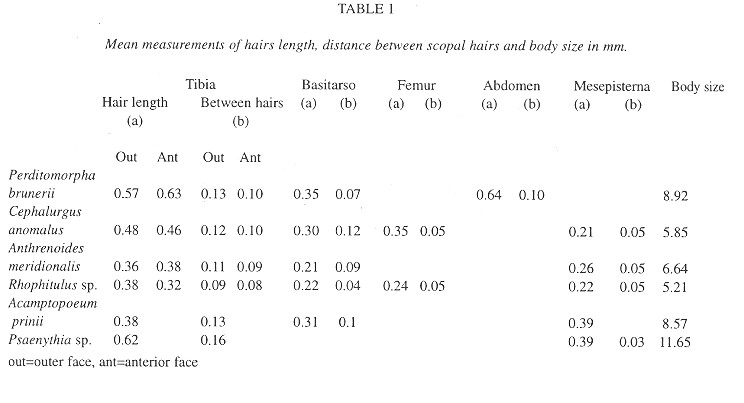
Structures associated with pollen transport and body size were measured on at least eight bees from each species. In order to compare the morphology of the scopa of C. anomalus to that of other Malvaceae-visiting Panurginae occurring in the same area, measurements were also taken on specimens of Anthrenoides meridionalis (Schrottky), Rhophitulus sp., Acamptopoeum prinii (Holmberg) and Psaenythia sp. Photomicrographs of the scopae of P. brunerii and C. anomalus were taken at magnification of 150x.
The intermandibular distance was used to represent head width for males of C. anomalus; 50 males were measured. Gut content of a few males and females was analysed in order to check the presence of pollen grains.
Some females were captured when they left their nests and received individual marks in order to monitor their activities. Nests were marked and excavated after they had been closed by the bees. Cells were brought to the laboratory in blocks of sediment to take their measurements.
Bee specimens are deposited as vouchers in RPSP (Coleção Camargo) and plant vouchers are in Herbarium FFCLRP, both in the University of São Paulo, Ribeirão Preto (SP), Brazil.
Results
Behavior on flowers and structures associated to pollen transport: The studied flowers are shallow, pentamerous, radially symmetric, diameter ranging from 8 to 22.4 mm; petals predominantly white to yellow, or in some cases pinkish, and their basal portion has a different coloration, forming a color ring, whose diameter varies from 3 to 8.4 mm; staminal tube formed by the concrescence of the filaments, involving the styles. The flower last only a few hours and fall at the following day.
Females of P. brunerii landed directly onto the anther and stigma surfaces and gathered pollen grains with the forebasitarsi, transferring them to the anterior region of the mid tibiae through ipsilateral movements. They grasped the staminal tube with the forelegs, bent their abdomen forward onto the anthers and vigorously moved mid and hind legs, transferring the pollen to the scopal setae in the hind tibia and metasomal sterna. Pollen grains were also scraped directly from the anthers with the scopal setae on the abdominal sterna.
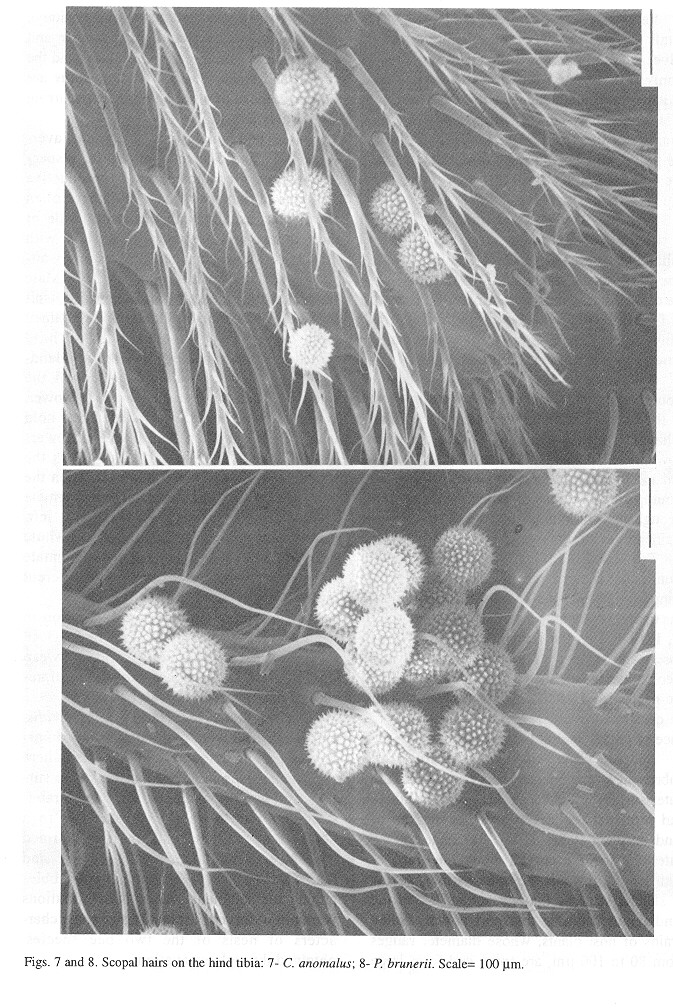
The scopal setae on the outer and anterior surface of the hind tibia are stout, 0.4 to 0.8 mm in length, and short-branched (table 1, fig. 8). The setae on the anterior surface are curved toward the outer surface; branches are 0.05 to 0.075 mm long and the basalmost ones 0.15 mm from the setal base. Scopal setae on the metasomal sterna III to VI , 0.56 to 0.76 mm long, are stout and have short branches along two-thirds of their distal length. Large pollen grains of Malvaceae, 80 to 100 µm in diameter, are carried by the scopal setae almost along their entire length, forming large pollen loads on the tibial and abdominal surfaces.
Females of P. brunerii gathered pollen from most of Malvaceae species studied (Materials and Methods), except from Wissadula hernandioides, Sida urens, Sida glomerata and Gaya domingensis, which were not very common in the study area. Individual flower visits lasted 2 to 7 s, and, in most cases, bees did not visit many flowers in the same plant. Sometimes the female landed on leaves and cleaned its antennae, wings and head with forelegs. Females also collected nectar in the same pollen plants (fig.1). Males and females collected nectar in several nectaries in each flower by folding their bodies onto the floral reproductive elements. Body size averaged 8.24 mm for males and 8.92 mm for females; both sexes always contacted stigma and anthers and could pollinate the flowers.
Males of P. brunerii patrolled flowers in circular flights without landing. Each flower was patrolled by several males, which showed no territorial behavior. Copulation was not observed. The males spent the night in flowers of S. cerradoensis and Sida sp.1; they landed between 15:00 and 15:40 h, ate pollen and nectar and, after that, remained there motionless (fig.2), while the petals rolled back and closed the flower. The male stayed within the flower until the next morning, leaving between 9:00 and 10:00 h, without damaging the petals and before the flower fell. One female was observed once in a closed flower of Sida sp.1.
Females of C. anomalus landed on the petals or on the anthers, sustaining themselves on the staminal tube, or more commonly, they leaned the dorsal side of the abdomen and wings against the basal region of the corolla. This position allowed the female to collect and simultaneously transfer the pollen (fig.4). With its forebasitarsi, the bee collected pollen from the open anthers or exposed grains on the anther surface or between the filaments, and transferred them to the mesepisterna, where there were unbranched and sparse setae (table 1), and from there to the scopae in the hind tibiae by the mid basitarsi. Individual flower visits lasted from a few seconds to approximately one minute and a half. Females have to visit many flowers in order to get a full pollen load.
Pollen grains adhered to antennae, head, mouthparts, wings and thorax were removed by the fore and mid legs by means of cleaning behavior, which was mainly observed on the flower after pollen collection. Both females and males were observed to move pollen to mouthparts. Ingestion of pollen was verified through the observation of Malvaceae pollen grains in the intestinal tube.
For nectar collection, C. anomalus females and males folded their bodies onto the staminal tube in order to reach the nectariferous tissue, where they inserted their proboscises. Periodically, the female stretched its proboscis and scraped its forebasitarsi on it and then on its scopae. Such behavior suggests that the nectar is added to pollen mass, which can be confirmed by observing the moist appearance of pollen load in the scopae.
Scopal setae of C. anomalus are unbranched and sparsely distributed on the outer surface and anterior edge on hind tibiae and proximal region of the outer surface of hind basitarsi (table 1, fig. 7). Setae on the anterior edge are curved along one-third of their length, towards the outer surface. A row of slender setae on the anterior surface of the hind femur can also transport pollen. Pollen grains of host plants, whose diameter ranges from 80 to 100 µm, are spiny and form large masses around the hind tibiae and basitarsi. The position of the scopa on the outer and anterior face of the tibiae and basitarsi and the simple and sparse setae of C. anomalus are similar to those of other Panurginae occurring in the study area (Table1).
Males and females of C. anomalus, averaging 5.45 and 5.85 mm in length, respectively, always contacted floral reproductive elements during their visits to gather pollen and nectar, usually with the ventral side of their thorax and abdomen as well as with their legs. Males patrolled flowers and waited for females inside them (fig.5). Male stayed on the flower floor, supporting itself on the staminal tube on its legs and remained there for up to 15 minutes with its head turned to flower opening. If no female landed, the male went out and performed the same behavior or patrolled another flower. When a female landed, the male tried to hold it quickly. Copulation took place on flowers of food plants (fig. 6) and many times the female continued collecting pollen when the pair was in copula. After that, the female stayed on the flower and the male left. Copulation was observed through the whole period of adult activities. One marked female was observed in copula with two different males in sequence.
C. anomalus males showed variation in head width (1.18 to 1.84 mm, averaging 1.48 mm), but there was no association between this measure and the two behavioral strategies of males, patrol or wait on flowers.
Nesting behavior: Nests of C. anomalus and P. brunerii were found on unshaded horizontal ground among the host plants, where plant cover was not very dense. At the surface, soil was dry and compact, finely granulated, but moist when excavated; up to a depth of approximately 30 cm, it was formed by fine and homogeneous sediment and below this layer, it was brighter, with pebbles among the sediment grains. Nest initiations were not observed. Table 2 shows some characters of nests of the two bee species, obtained through excavation.
The nesting site contained ten nests of P. brunerii observed from March to May 1995. Only one foraging female was active in each nest. The simple nest entrance, without tumuli, was approximately 4.5 mm in diameter and followed in a main tunnel, filled with soil up to a depth of 8 mm, descending 15 to 25 cm. Lateral canals were closed. In two excavated nests, four and eight cells were found, arranged singly and horizontally in the soil (table 2). The internal surface of the cells was smooth and the walls were indistinguishable from the substrate. One imago was found in an excavated cell on April, 19. A dead female was found at the end of the main tunnel, in a nest that had been closed for nine days before excavation.
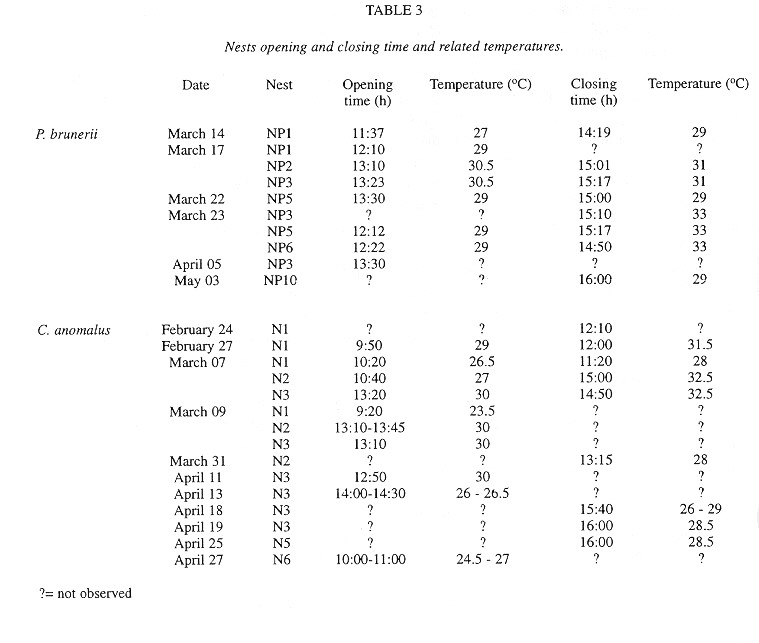
Females of P. brunerii opened their nests from 11:37 to 13:30 h, with temperatures ranging from 27 to 30.5°C (table 3). They pulled closing earth into the burrow, and nest entrance remained open until the last foraging trip, between 14:19 and 16:00h, with temperatures from 29 to 33°C (table 3). Female seemed to spend the night in the nest. Foraging trips lasted from 1 min 30 s to 9 min, averaging 2 min 48 s. Between two succeeding foraging trips, the female stayed from 1min to 28 min 30 s, averaging 4 min 48 s in the nest. It took from 14 to 19 foraging trips in a day.
Upon leaving and returning, females flew over the nest entrance (fig. 3), following circular paths and reaching a circle of approximately 50 cm in diameter. They took such orientation flights mainly before the first trips of a day and whenever the surroundings changed.
Six nests of C. anomalus in two small aggregations among the host plants were observed from February to May 1995. There was from one to several foraging females in each nest. The circular nest entrance had no tumuli. The main tunnel had a diameter ranging from 2.5 to 3.3 mm; it was filled with soil in the first 2 to 4 cm and descended open up to 27 to 63 cm. Two nests had a lateral branch at 23 and 42 cm, respectively, which reached 4 cm in length. It was not possible to distinguish two nests because they were adjacent and only one main tunnel was found. They are indicated in table 2 as nest N2/N3.
Cells were horizontally on the ground, distant 1.0 to 4.5 cm from the main burrow and lateral branches were, in most cases, filled with soil. Cells were 3.9 mm in diameter and 4.9 mm in length. In one closed nest, excavated in March, there was a live female in the main tunnel (table 2, N1), whose wings were not wasted and whose oocytes were in initial stage of development. There were no traces of egg-laying and it was probably a recently emerged female. In another closed nest (table 2, N2/N3), there was an older female at the end of the main tunnel, which had wasted wings and pollen in its intestine. In both cases (with live females in N1 and N2/N3), nests had been closed for at least four and 18 days, respectively, before excavation.
Nest entrance was opened by one female between 9:20 and 14:30 h, with temperatures from 23.5 to 30°C (table 3). In the same day, different nests were opened at different times, like N1, N2 and N3 on March 07 and 09. The female uncovered the nest entrance by pulling earth that was closing it. It showed orientation behavior similarly to that described in this paper for P. brunerii. The females foraged during 3 to 21 min (n=21), averaging 6 min 50 s, while the nest entrance remained open. In general, they brought pollen load into the nest and stayed there for 1 to 3 min (n=19), averaging 1 min 50 s, before going out on another foraging trip. One female took approximately 12 foraging trips in a day. After the last one, between 11:20 and 16:00 h (table 3), entrance was closed by pushing earth from the burrow and the female stayed in the nest until the next day. During heavy rains, entrance was also closed and it was opened again if the weather improved.
In two monitored composite nests, periods of activity were observed when the bees went out to collect and came back to nest with provisions, intercalating with periods of inactivity, during which the nest remained closed. The longest observed period of continuous activity for one nest was 19 days. One marked female in the nest N2 was observed foraging on flowers of Sida acuta when the nest N2 had been closed for, at least, 35 days.
Discussion
Behavior on flowers and structures associated to pollen transport: Scopal structures of P. brunerii differ from those of most other Colletinae, which usually have long branched hairs (Pasteels and Pasteels 1976, Michener 1989). The hairs branches of P. brunerii are short and allow large pollen grains to get attached to them. Large pollen grains are also collected by other Colletinae like Sarocolletes sp., Cephalocolletes rugata (Schlindwein and Wittmann 1997b), Tetraglossula bigamica (Gimenes 1991) and Brachyglossula sp. (Michener 1989). These species have sparse scopal hairs with slender and short branches, except T. bigamica, whose simple hairs are associated with the transportation of interconnected pollen grains of Onagraceae.
C. anomalus females have very different strategies to collect resources on the same flowers from those of P. brunerii. They stay on the flower for several minutes and collect nectar to add to the pollen load. The support by the wings and abdomen on the petals enables the female to collect and transfer pollen to the scopae simultaneously.
Perdita opuntiae Cockerell and Perdita texana (Cresson), Panurginae species oligolectic on Cactaceae, also deposit pollen on the mesepisterna before transporting it to the scopae (Bennett and Breed 1985, Neff and Danforth 1991, respectively). But they differ from C. anomalus, whose paths of deposition on the mesepisterna and accummulating on the scopae are simultaneous by using the fore and mid legs.
Copulation of C. anomalus occurs on flowers, which, according to Eickwort and Ginsberg (1980), are the most common encounter site used by bees. Males have two behavioral patterns associated with mating: patrol and waiting. These patterns were also observed for other Panurginae, such as, Calliopsis (Hypomacrotera) subalpina (Cockerell) (Rozen 1970), P. texana (Barrows et al. 1976), Panurgus sp. (Tengö et al. 1988) and Callonychium petuniae Cure and Wittmann (Wittmann et al. 1990). It seems that C. anomalus males can differentiate the more attractive flowers for the females and wait more frequently in them. I could not see any association between head width and the behavioral strategies of the males. Tengö et al. (1988) refer to the association between different amounts of pollen in flowers and different behavioral strategies of Panurgus sp. males, as well as, different body size, age or physiological aspects of such males. According to Alcock et al. (1978), sit-and-wait behavior would be more probable when the males are capable of identifying the most attractive place for females. Patrolling would be favored when females visit flowers for a relatively long time in areas with few flowers and males competing with each other.
Foraging while in copula was observed for C. anomalus females in this work and for other Panurginae (Barrows et al. 1976, Tengö et al. 1988, Wittmann et al. 1990). Rozen (1989) analysed some behaviors of Panurginae and considered foraging while in copula as an apomorphic character for this group.
The analysis of the scopa of C. anomalus showed similar structures to those of Rhophitulus sp., Psaenythia sp., Acamptopoeum prinii and Anthrenoides meridionalis, which occur in the same area and also visit Malvaceae flowers (Gaglianone, 1997). According to Ruz (1991), A. prinii (Calliopsini) and C. anomalus, A. meridionalis, Rhophitulus sp. and Psaenythia sp. (Panurgini) belong to two different phylogenetic clades. Another Panurginae bees associated with flowers with large pollen grains are Anthemurgus passiflorae Robertson, oligolectic on Passifloraceae (Neff and Rozen 1995), and Arhysosage sp., on Cactaceae (Schlindwein and Wittmann 1997b), whose foraging behavior is very different from that performed by C. anomalus.
The monitored nests of C. anomalus showed asynchronism in nest opening and closing in the same day. This must be related to intrinsic factors of the nest rather than to climatic conditions. This asynchronism was also observed by Neff and Danforth (1991) for nests of Perdita texana.
Live females of C. anomalus were found inside two closed nests. Besides, the nests could be reactivated after they had been closed. It is possible that the observed females inside the closed nests would reopen them. In the case of recently emerged females, the emergence time could correspond to a reactivation of the nest. This is possible, since this species has more than one generation in a year (Rozen 1989). But the monitoring of more nests of C. anomalus would be necessary to solve this question.
Acknowledgements
I am grateful to João M. F. Camargo for his advisement and for his comments and corrections of a previous manuscript. I thank Luisa Ruz for carefully reading the manuscript and comments; Fernando C. V. Zanella and Marcio L. Oliveira for their helpful suggestions; Gabriel A. R. Melo for suggestions and corrections in English; Menderson Mazucato for help during fieldwork; Gerleni L. Esteves for identifying the plants and Sôicho Yamane for the photomicrographs. This work was granted by CAPES.
References
Alcock, J., E.M. Barrows, G. Gordh, L.J. Hubbard, L. Kirkendall, D.W. Pyle, T.L. Ponder & F.G. Zalom. 1978. The ecology and evolution of male reproductive behaviour in the bees and wasps. Zool. J. Linn. Soc. London 64: 293-326. [ Links ]
Barrows, E.M., M.R. Chabot, C.D. Michener & T.P. Snyder. 1976. Foraging and mating behavior in Perdita texana (Hymenoptera : Andrenidae). J. Kansas Entomol. Soc. 49: 275-279. [ Links ]
Bennett, B. & M.D. Breed. 1985. The nesting biology, mating behavior, and foraging ecology of Perdita opuntiae (Hymenoptera : Andrenidae). J. Kansas Entomol. Soc. 58: 185-194. [ Links ]
Camargo, J.M.F. & M. Mazucato. 1984. Inventário da apifauna e flora apícola de Ribeirão Preto, SP, Brasil. Dusenia 14: 55-87. [ Links ]
Eickwort, G.C. & H.S. Ginsberg. 1980. Foraging and mating behavior in Apoidea. Ann. Rev. Entomol. 25: 421-446. [ Links ]
Gaglianone, M.C. 1997. Estudo sobre as interações de Cephalurgus anomalus Moure & Oliveira (Andrenidae) e Perditomorpha brunerii Ashmead (Colletidae) com flores de Malvaceae. Dissertação de Mestrado. Universidade de São Paulo, 167 p. Ribeirão Preto, SP, Brazil. [ Links ]
Gaglianone, M.C. 2000. Biologia floral de espécies simpátricas de Malvaceae e suas abelhas visitantes. Biociências 8. (in press). [ Links ]
Gimenes, M. 1991. Some morphological adaptations in bees (Hymenoptera, Apoidea) for collecting pollen from Ludwigia elegans (Onagraceae). Revta bras. Ent. 35: 413-422. [ Links ]
Laroca, S. & M.C. Almeida. 1985. Adaptação dos palpos labiais de Niltonia virgilii (Hymenoptera, Apoidea, Colletidae) para coleta de néctar em Jacaranda puberola (Bignoniaceae), com descrição do macho. Revta bras. Ent. 29: 289-297. [ Links ]
Linsley, E.G. 1958. The ecology of solitary bees. Hilgardia 27: 543-597. [ Links ]
Michener, C.D. 1989. Classification of American Colletinae. Univ. Kans. Sci. Bull. 53: 662-703. [ Links ]
Michener, C.D. & R.B. Lange. 1957. Observations on the ethology of some brazilian colletid bees. J. Kansas Entomol. Soc. 30: 71-80. [ Links ]
Moure, J.S. 1954. Novas notas sobre abelhas do antigo gênero Pasiphae (Hymenoptera - Apoidea). Dusenia V: 165-190. [ Links ]
Neff, J.L. & B.N. Danforth. 1991. The nesting and foraging behavior of Perdita texana (Cresson) (Hymenoptera : Andrenidae). J. Kansas Entomol. Soc. 64: 394-405. [ Links ]
Neff, J.L. & J.G. Rozen. 1995. Foraging and nesting biology of the bee Anthemurgus passiflorae (Hymenoptera: Apoidea), descriptions of its immature stages, and observations on its floral host (Passifloraceae). Amer. Mus. Novitates 3138: 1-19. [ Links ]
Pasteels, J.J. & J.M. Pasteels. 1976. Étude au microscope életronique á balayage des scopas collectrices de pollen chez les Colletidae et les Oxaeidae (Hymenoptera, Apoidea). Arch. Biol. 87: 79-102. [ Links ]
Rozen, J.G. 1970. Biology and immature stages of the panurgine bee genera Hypomacrotera and Psaenythia (Hymenoptera, Apoidea). Amer. Mus. Novitates 2416: 1-15. [ Links ]
Rozen, J.G. 1989. Life history studies of the primitive Panurgine bees (Hymenoptera : Andrenidae : Panurginae). Amer. Mus. Novitates 2962: 1-27. [ Links ]
Rozen, J.G. & L. Ruz. 1995. South american panurgine bees (Andrenidae : Panurginae), Part II. Adults, immature stages, and biology of Neffapis longilingua, a new genus and species with an elongate glossa. Amer. Mus. Novitates 3136: 1-15. [ Links ]
Ruz, L. 1991. Classification and phylogenetic relationships of the panurgine bees: the Calliopsini and allies (Hymenoptera : Andrenidae). Univ. Kans. Sci. Bull. 54: 209-256. [ Links ]
Ruz, L. & J.G. Rozen. 1993. South american panurgine bees (Apoidea : Andrenidae : Panurginae), Part I. Biology, mature larva, and description of a new genus and species. Amer. Mus. Novitates 3057: 1-12. [ Links ]
Schlindwein, C. 1995. Wildbienen und ihre Trachtpflanzen in einer südbrasilianischen Buschlandschaft : Fallstudie Guaritas, Bestäubung bei Kakteen und Loasaceen. Dissertation Eberhard-Karls-Universität Tübingen, Fakultät für Biology. Tübingen, Germany. [ Links ]
Schlindwein, C. & D. Wittmann. 1997a. Micro-foraging routes of Bicolletes pampeana (Colletidae) and bee-induced pollen presentation in Cajophora arechavaletae (Loasaceae). Bot. Acta. 110: 177-183. [ Links ]
Schlindwein, C. & D. Wittmann. 1997b. Stamen movements in flowers of Opuntia (Cactaceae) favour oligolectic pollinators. Pl. Syst. Evol. 204: 179-193. [ Links ]
Silveira, F.A., L.B. Rocha, J.R. Cure & M.J. Oliveira. 1993. Abelhas silvestres (Hymenoptera, Apoidea) da Zona da Mata de Minas Gerais. II. Diversidade, abundância e fontes de alimento em uma pastagem abandonada em Ponte Nova. Revta bras. Ent. 37: 595-610. [ Links ]
Tengö, J., J. Eriksson, A. Borg-Karlson, B.H. Smith & H. Dobson. 1988. Mate-locating strategies and multimodal communication in male mating behavior of Panurgus banksianus and P. calcaratus (Apoidea, Andrenidae). J. Kansas Entomol. Soc. 61: 388-395. [ Links ]
Vieira, M.F. & N.A.S. Lima. 1997. Pollination of Echinodorus grandiflorus (Alismataceae). Aquatic Botany 58: 89-98. [ Links ]
Wilms, W. 1995. Die Bienenfauna im Küstenregenwald Brasiliens und ihre Beziehungen zu Blütenpflanzen: Fallstudie Boracéia, São Paulo. Dissertation Eberhard-Karls-Universität Tübingen, Fakultät für Biologie. Tübingen, Germany. [ Links ]
Wittmann, D., R. Radtke, J.R. Cure & M.T. Schifino-Wittmann. 1990. Coevolved reproductive strategies in the oligolectic bee Callonychium petuniae (Apoidea, Andrenidae) and three purple flowered Petunia species (Solanaceae) in southern Brazil. Z. zool. Syst. Evolut.-forsch. 28: 157-165. [ Links ]