Introduction
Subtribe Spiranthinae is the most species-rich clade of terrestrial orchids in the New World, with ca. 40 genera and 500 species (Salazar et al. 2018). One of the largest genera in this subtribe is Pelexia Poit. ex Lindl. (Lindley 1826), which in its broad sense includes about 77 species (Chase et al. 2015). Schlechter (1920a) recognized five sections within Pelexia: Cogniauxocharis Schltr., Centropelexia Schltr., Pachygenium Schltr., Pelexia, and Potosia Schltr. The monotypic Potosia and one of the species of Cogniauxocharis have been shown to belong to Sarcoglottis C.Presl (Presl 1827) and Pteroglossa Schltr. (Schlechter 1920a), respectively (Salazar et al. 2018). In turn, the two species assigned by Schlechter to the section Centropelexia shared many features with the type species of Pelexia, P. adnata (Sw.) Spreng. (Sprengel 1826), and therefore, there is no way to distinguish this section from the section Pelexia (Salazar et al. 2018). The remaining two sections [“Eu-”] Pelexia and Pachygenium, have received different treatments in recent classifications, either as congeneric (Balogh 1982, Garay 1982, Salazar 2003) or as two separate genera (Rutkowski et al. 2008, Szlachetko et al. 2001, Szlachetko et al. 2005).
Salazar et al. (2018) assessed the phylogenetic relationships in Spiranthinae, analyzing nuclear and plastid DNA markers of a nearly complete generic sample and about one-third of the species of the subtribe. Their results recovered two strongly supported clades corresponding to sections Pelexia and Pachygenium. Nevertheless, the low bootstrap support prevents knowing with certainty the relationships of these groups with Brachystele Schltr. (Schlechter 1920b). The authors of that article suggested a possible closer relationship between the P. sect. Pachygenium and Brachystele, based on their richness centered in eastern South America, their preference for open grassy habitats, and the pollination by bumblebees (Bombus spp.). In contrast, the diversity of Pelexia s.s. is widespread from the Andean region to Mexico and the Caribbean, its species inhabit forests, and the pollination is carried out by euglossine bees (G. Gerlach pers. comm.). All the above is consistent with the recognition of the sections Pelexia and Pachygenium (Schltr.) Szlach., R.González & Rutk. (Szlachetko et al. 2001) as distinct genera.
Morphologically, Pachygenium is distinguished from Pelexia by the presence of leaves gradually attenuating (vs. abruptly contracted into a pseudopetiole in Pelexia), the proportionately broad and wide lateral sepals, and the saccate nectary spur (vs. proportionately narrow, long lateral sepals and rounded or sharp spur). Other floral morphological differences indicated by Szlachetko et al. (2001) as distinctive, such as details of the rostellum and stigma, do not hold true as distinctive characters upon close examination (Salazar et al. 2018).
In their synopsis of the “spiranthoid” orchids of Colombia, Dueñas & Fernández-Alonso (2009) recorded 14 species of Pelexia s.l., including two assignable to Pachygenium sensu Szlachetko et al. (2001), namely P. hirta (Lindl.) Schltr. (Schlechter 1920b) and P. orobanchoides (Kraenzl.) Schltr. (Schlechter 1920b). The specimen quoted by Dueñas & Fernández-Alonso (2009) as P. orobanchoides (M. Schneider 679/1, COL!) had been designated by Szlachetko as the type of his new species, Pelexia cundinamarcae Szlach. (Szlachetko 1993), which appears to have been overlooked by Dueñas & Fernández-Alonso (2009). On the other hand, the specimen determined as P. hirta (R. Schnetter 494, COL!) corresponds to the new species here described as Pachygenium muyscarum.
Here, we propose a new species of Pachygenium which is described and illustrated. Its possible relationships to other species are discussed on morphological grounds, and information about its distribution, ecology, and conservation status is provided.
Materials and methods
We conducted fieldwork at the Parque Ecológico Cerro Seco, Ciudad Bolívar locality, Bogotá D.C., in May-June 2019, May-June 2021, December 2021, and January-February 2022. The vouchers were deposited at JBB. To determine the generic identity of the species, we consulted the pertinent literature on the taxonomy of the subtribe Spiranthinae (Szlachetko et al. 2001, Dueñas Gómez & Fernández-Alonso 2007, 2009, Salazar et al. 2018). For the circumscription of Pachygenium, we followed Salazar et al. (2018). We followed the terminology of Balogh (1982), and for the seed coat morphology that of Molvray & Kores (1995); we measured the organs with a digital caliper and observed the specimens under a stereomicroscope Motic SMZ 168. We examined the protologues of the types deposited in JSTOR PLANTS website (http://plants.jstor.org), the collections of COL, HUA, HUQ, HUAZ, JBB, JAUM, MEDEL, MEXU, and the virtual collections (digital photographs) of A, BHBC, F, HBG, K, MBM, NY, RB, and U (acronyms follow Thiers 2022). We followed the morphological species concept (McDade 1995, Wiens & Servedio 2000, De Queiroz 2007). The Geospatial Conservation Assessment Tool (GeoCAT) application (http://geocat.kew.org/) was used to establish the conservation status, according to the categories and criteria of IUCN (2019).
Taxonomic treatment
Pachygenium muyscarum Rinc.-González, FonsecaCortés & Salazar, sp. nov.Fig. 1 - - 4.
TYPE: Colombia. Cundinamarca: Bogotá D.C., Ciudad Bolívar, Arborizadora Alta, Cerro Seco, 2800 m, 15 July 2021, M. Rincón-González, A.I. Díaz & M. Pinzón 1842 (holotype: JBB!).
Diagnosis: Pachygenium muyscarum is morphologically similar to P. tamanduense (Kraenzl.) Szlach., R.González & Rutk., from which it differs by flowering without leaves (vs. flowering with leaves), labellum 1.4-1.6 cm long (vs. 1.2-1.4 cm long), apical labellum lobe trilobate (vs. entire, rounded) and labellum nectar glands 0.2-0.4 cm long (vs. up to 0.1 cm long).
Terrestrial herb, 3-5 cm in height without the inflorescence and 15-30 cm in height, including the inflorescence. Roots 10-20 × 0.5-0.8 cm, fasciculate, terete, attenuating slightly towards the apex, pale dull yellowish. Leaves 6-16 × 1-2 cm, 4-6 when present, elliptic-lanceolate, 5-6 parallel nerved, base attenuate, margin entire, apex acute, dark green adaxially, pale green abaxially, forming a rosette, with the leaves ascending; pseudopetioles sublinear, channeled, white adaxially, tawny abaxially. Inflorescence 15-25 cm long, racemose, erect, glabrous below the middle, becoming increasingly villose towards the apex, hairs multicellular and clavate; peduncle 12-20 cm long; sheaths 3.7-5.0 × 0.7-1.0 cm, 5-12, lanceolate, chartaceous, yellowish green at the base, green on the rest, glabrous; floral bracts 1.8-2.5 × 0.6-0.8 cm, elliptic-lanceolate, concave, cuneate, 3-4 veined, olive green, tomentose with multicellular, clavate, caducous hairs, margins irregularly ciliate and becoming papillose near the apex. Flowers 0.8-1.4 cm long, ascending, resupinate, dorsal sepal green, lateral sepals green with the margins white, tomentose abaxially, glabrous adaxially, petals green with the apex white, labellum green at the base, white in the middle at the apex, with greenish dorsal vein. Dorsal sepal 8-1.2 × 0.4-0.6 cm, ovate-lanceolate, concave, 3-5 veined. Lateral sepals 1.2-1.4 × 0.3-0.5 cm, lanceolate-falcate, concave, 3-5 veined, base fused into a saccate nectary. Petals 1.0-1.2 × 0.2-0.3 cm, oblong, spatulate, 3-5 veined, fused internally at the base with the dorsal sepal, apex acute, external margin irregularly papillose above the middle. Labellum 1.4-1.6 × 0.4-0.6 cm, panduriform, with 3-9 veins, base provided at each side with one subulate, uncinate nectar gland, 0.2-0.4 × to 0.1 cm, apical lobe 0.4-0.6 × 0.4-0.6 cm, rounded. Column 0.8-1.2 × 0.1-0.2 cm, terete, papillose, canaliculate; anther 0.2-0.4 cm long, ellipsoid; rostellum 0.1-0.3 cm long, triangular; ovary 1.3-1.6 × 0.3-0.5 cm, fusiform, cylindrical, green, tomentose; stigma concave, slightly bilobed. Pollinarium 4.0-4.1 × 1.9-2.0 mm, wishbone-shaped, with two creamy-white pollinia; pollinia 3.0-3.2 × 0.8-1.0 mm, ellipsoid, creamywhite, viscidium 0.3-0.5 × to 0.1 cm, rhomboid, grey. Capsule 1.6-1.7 cm long × 0.6-0.7 cm wide, elliptic, tomentose, with 3 longitudinal keels, perianth persistent, brown. Seeds 0.5-0.8 × 0.15-0.20 mm, seed coat spiranthoid type, with 12-15 cells in length and 5-7 in width, cells rectangular with one of both ends arched and prominent, embryo 0.25-0.30 mm, ovoid.
Etymology: The specific epithet “muyscarum” commemorates the indigenous ethnic group Muysca, who inhabit the Cundinamarca-Boyacá highlands where this species grows.
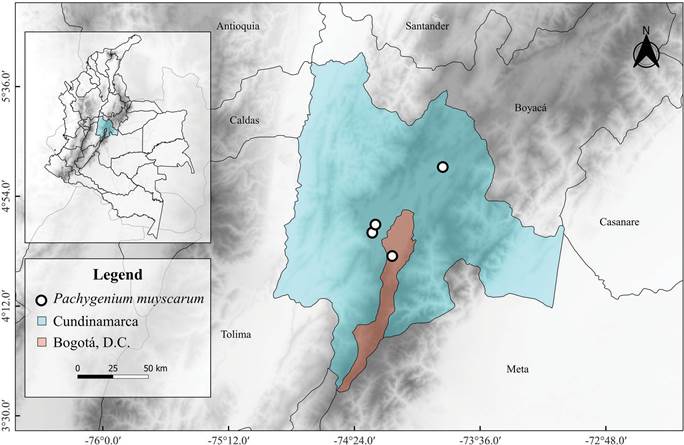
Distribution and habitat: To the date, P. muyscarum only has been found in four localities of Cundinamarca, located in subxerophytic enclaves between 2600- 2800 m a.s.l. (Fig. 6). This species grows on hills with slopes with an inclination of 15-20° and well-drained soils, with an A or A/E horizon formed from the root activity within a well-drained, silty sand, thick eluviated horizon (originally formed from altered volcanic ash), which became enriched in quartz (>70%), feldspars, hornblende, and cristobalite, with some remnant (about 10%) of halloysite clay. It exhibits a pH close to 6, about 2% organic carbon, base saturation close to 60%, and low K and P contents. Below this horizon, a sequence of intercalated clayey and silty horizons with variable thickness follows, which are richer in K and P (León Linares et al. unpubl. data). Pachygenium muyscarum grows under the shadow of Dodonaea viscosa Jacq. (Jacquin 1760) or exposed to the sun besides Anthoxanthum odoratum L. (Linnaeus 1753), Bidens andicola Kunth (Kunth 1818), Cuphea ciliata Ruiz & Pav. (Ruiz & Pavón 1794), Desmodium molliculum (Kunth) DC. (De Candolle 1825), Echeandia flavescens (Schult. & Schult.f.) Cruden (Cruden 1981), Euphorbia orbiculata Kunth (Kunth 1817), Evolvulus bogotensis Ooststr. (Ooststroom 1934), Hypericum brevistylum Choisy (Choisy 1821), Jarava ichu Ruiz & Pav. (Ruiz & Pavon 1798), Laennecia gnaphalioides (Kunth) Cass. (Cassini 1822), Myriopteris myriophylla (Desv.) J.Sm. (Smith 1854), Nassella mucronata (Kunth) R.W.Pohl (Pohl 1990), Pombalia parviflora (L.f.) Paula-Souza (Paula-Souza & Ballard 2014), Rhynchospora nervosa (Vahl) Boeckeler (Boeckeler 1869), Selaginella sellowii Hieron. (Hieronymus 1900), and Stenandrium dulce (Cav.) Nees (De Candolle 1847).

Photographs and elaboration by Milton Rincón-González.
Figure 1 Lankester Composite Dissection Plate (LCDP) of P. muyscarum.
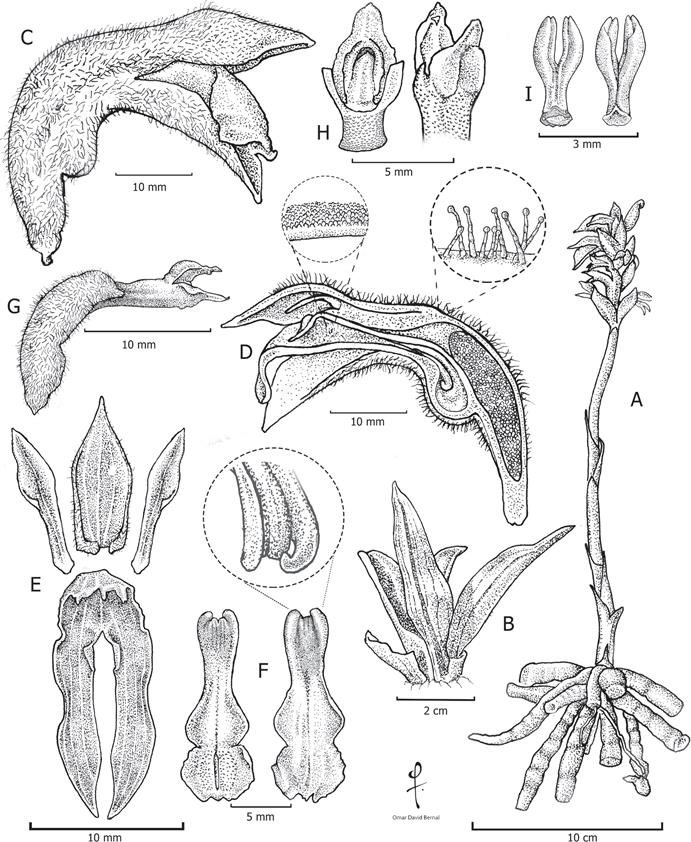
Illustration based on Milton Rincón-González et al. 1842 (JBB).
Elaborated by Omar David Bernal.
Figure 2 Illustration of P. muyscarum.

Photographs and elaboration: Andrés Fonseca-Cortés.
Figure 3 Habitat and vegetative parts of P. muyscarum.
Conservation status: The extent of occurrence (EOO) is 916.306 km2, and the area of occupancy (AOO) is 20 km2; hence, P. muyscarum meets the requirements under criterion B for threatened species (AOO < 2000 km). Additionally, it is only known from four localities, the populations are declining due to the presence of goats, cattle, and horses, quarrying, adventure sports (ATV and motorcycle tracks), and establishment of invasive neighborhoods. In this sense, the Endangered (EN) category is proposed (B2aiii) (IUCN 2019).
Additional specimens examined: Colombia. Cundinamarca: Bogotá D.C. Ciudad Bolívar, Parque Ecológico Cerro Seco, 2800 m, 26 V 2021, B. Villanueva et al. 6022, JBB! municipio Nemocón, vereda Moguá, 2650 m, 31 July 2007, S.P. Cortés 4970 (JBB!); municipio Madrid, Hacienda Casa Blanca, cúspide del cerro, frente a la casa principal, 11 July 1999, Y.A. Mora & R. Sánchez 155 (COL!); municipio de Mosquera, zona xerofítica de la laguna de la Herrera, 22 June 1972, R. Schnetter 494 (COL!).
Ecology: The places in which this species grows have a bimodal precipitation pattern, with the dry seasons usually occurring in December-March and July-September and the humid seasons in October-November and April-June. Pachygenium muyscarum produces leaves and flowers in the humid seasons, whereas during the dry seasons most individuals lose the leaves. The plants have a strong radicular system (Fig. 1D) which probably stores starch and allows this species to survive the dry season. We recorded Bombus atratusFranklin (1913) pollinating the flowers of P. muyscarum (Fig 3); when the bee visits the flower, it introduces its mouth parts deeply into the flower to reach the nectar, and the pollinarium attaches to the bee’s labrum (Fig. 5D). This bumblebee species has been reported as a pollinator of Pachygenium oestriferum (Rchb.f. & Warm.) Szlach., R.González & Rutk. in a similar manner to P. muyscarum (Singer & Sazima 1999).

Photographs and elaboration by Andrés Fonseca-Cortés.
Figure 5 Bombus atratus visiting P. muyscarum.
Discussion
Pachygenium muyscarum is similar to P. ekmanni (Kraenzl.) Szlach., R.González & Rutk. (Szlachetko et al. 2001), P. longibracteatum (Pabst) Szlach., R.González & Rutk. (Szlachetko et al. 2001)., P. tamanduense (Kraenzl.) Szlach., R.González & Rutk. (Szlachetko et al. 2001) and P. ventricosum (Cogn.) Szlach., R.González & Rutk. (Szlachetko et al. 2001), but it is clearly distinguished from them by the characters listed in Table 1.
Table 1 Differences among P. muyscarum and morphologically similar species.
| Characters | P. ekmanni | P. longibracteatum | P. muyscarum | P. tamanduense | P. ventricosum |
|---|---|---|---|---|---|
| Leaves present at flowering | Yes | Yes | No | Yes | No |
| Length of labellum (cm) | 1.4−1.6 | 1.0−1.2 | 1.4−1.6 | 1.2−1.4 | 1.2−1.4 |
| Apex of the labellum apical lobe | Entire | Entire | Trilobate | Entire | Entire |
| Length of the labellum spur (cm) | 0.2-0.3 | 0.1-0.2 | 0.2-0.4 | to 0.1 | to 0.1 |
| Distribution | Brazil | Brazil | Colombia | Brazil | Paraguay |
In their synopsis of the Colombian Spiranthoideae, Dueñas Gómez and Fernández-Alonso (2009) recorded 14 species of Pelexia, without considering the combinations in Pachygenium done by Szlachetko et al. (2001). Of those species, only two are now included in Pachygenium, P. hirtum, and P. orobanchoides. Pachygenium hirtum was recorded based on the specimen Mora & Sánchez 155 (COL!) and P. orobanchoides on the specimen Schneider 679/1 (COL!). Close study of those specimens allowed us to determine that Mora & Sánchez 155 corresponds to the species described here as P. muyscarum. At the same time, Schneider 679/1 is the holotype of Pelexia cundinamarcae, a species described in 1993 by Szlachetko but not reported by Dueñas Gómez and Fernández-Alonso (2007). Therefore, the only species of Pachygenium present in Colombia is P. muyscarum. Photographs of flowers of the three species are presented above (Fig. 7) because P. hirtum, P. orobanchoides, and P. muyscarum could be confused, and the former two were attributed to Colombia by Dueñas and Fernández-Alonso (2009).












 uBio
uBio