Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.54 n.4 San José Dec. 2006
A.A. Ortega-Salas1 & H. Reyes-Bustamante2
1 Instituto de Ciencias del Mar y Limnología, UNAM. Calz. Joel M. Camarena s/n, Mazatlán 82040, Sinaloa Apdo.
Post. 811; ortsal@mar.icmyl.unam.mx
2 Facultad de Ciencias del Mar, Universidad Autónoma de Sinaloa, Sinaloa, México
Key words: seahorse, Hippocampus ingens, aquaculture, growth, mortality.
Several species of Hippocampus have been commercially exploited for several years without proper knowledge of culture techniques. Seahorses have been mainly caught in China, Thailand, India, and the Philippines for many years (Mi 1992). Commercial demands for seahorses, mainly for medical purposes, are increasing by 20 % per year and the price per kilogram of dried seahorse varies from US$ 400.00 to 1 300.00 (Vincent 1994).
Liang (1992) studied H. ramalassus reproduction and assured that artificial production would allow the preservation of this Hippocampus species. The aim of this paper was to count and calculate fecundity, survival, and growth of the seahorse Hippocampus ingens under semi-controlled conditions.
Materials and methods
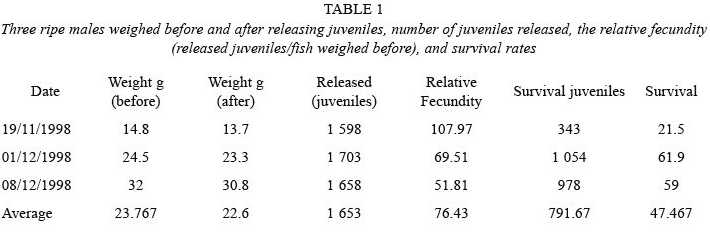
Three wild brood stock mature male were used in this study. They were weighed just before they released the juveniles, and after they released them to estimate the total weight. The average individual weight was estimated dividing the total larval weight by the number of juveniles released. All the released juveniles per each mature male were counted; the relative fecundity was calculated dividing the number of juveniles per gram of seahorse body weight before release of the juveniles. After 35 days the juveniles were counted to calculate mortality.
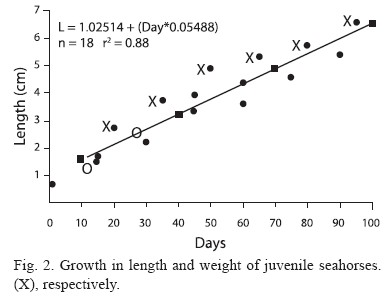
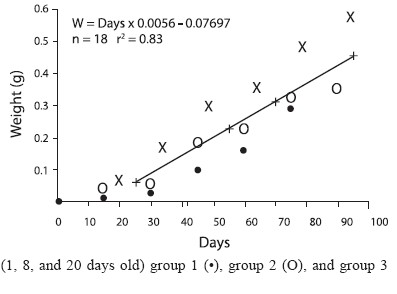
Three groups of juveniles aged 1, 12, and 20 days old were used (60 organisms each) to calculate growth every 15 days for the 95 daysduration of this study (Fig. 2). The length (mm) and weight (g) parameters from the three groups of released juveniles were used to calculate growth by the least square method to estimate changes in length and weight with time.
The seahorse juveniles at a stocking density of 12 were settled in 60 l aquariums in groups of 1, 8, and 20 days old organisms with a mixture of microalgae culture of Chaetoceros and Tetracelmis between 500 000and 100 000 cells/ml; they were fed at a density rate of 75 rotifers/ml, and Artemia nauplii at a density of 1 to 2/ml for 35 days. Then they were transfer to a cement tank of 100 000 l at a density of 50/1 000 l and fed with live Artemia adult at a rate of 2/ml for 60 days more. The seawater temperature registered in this study varied from 17 to 23 oC.
Results
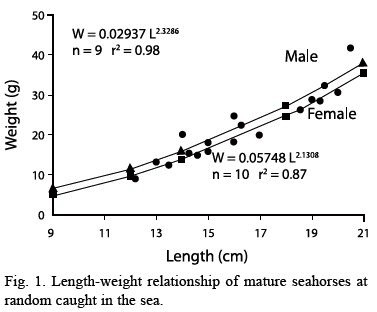
Figure 1 shows the length-weight relationship of 9 males and 10 females caught at random in the sea.
The equations show that for a given length males are heavier. Average length of males was 16.6 cm (± 2.83 SD). Average length of females was 15.3 cm (± 3.21 SD). A t-test and mean differences revealed a significant difference between sexes according to the mean lengthwith 6.01; 95 % confidence interval; d.f.= 17; t= -1.51; significance level= 0.34, even with mean weight there were significant differences between sexes with –1.3; 95 %; d.f.= 17; t= - 096; significance level= 0.14, at p< .05.
Three mature males released 1 598, 1 703, and 1 658 juveniles giving a total of 4 959 juveniles in 20 days, which gave an averageweight male of 23.7 g, a larval release of 1 653, and a relative fecundity of 76.43 juveniles per gram of seahorse (Table 1). Survival registered for 1 598, 1 703 and 1 658 released juveniles was 343, 1 054 and 978 juveniles, respectively, which means 78.5, 38.1 and 41.0% survival rates, respectively.
Figure 2 shows the increase of the average length and weight from the three groups of juveniles, every 15 days. The equations indicate that the average length and growth rate from the three groups were 0.05 cm and 0.005 g per unit of time (days), respectively.
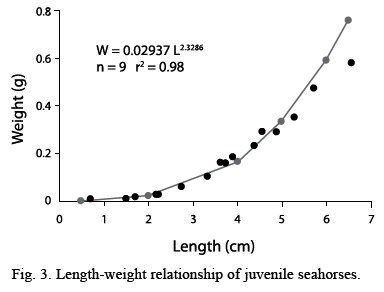
Figure 3 shows the average growth in length and weight of juveniles from the three groups, from which a length-weight relationship was calculated; starting with the 3 cm length, weights increased geometrically.
Sexual differences in juveniles were observed at 95 days of age at a length range between 6 and 7 cm.
Discussion
The temperature of the seawater reported in this paper ranged between 17 to 23 oC,although Tawil (1994) reported temperatures for this species from 3 to 27 oC. In other species Vincent and Clifton (1989) found an optimal temperature of 27 oC for H. erectus, and Fam (1992) reported an optimal temperature of 21 oC for H. kuda.
Vincent (1994) calculated an average length at hatching of 0.9 cm for H. ingens; Graff (1968) estimated 2.5 cm in H. enano; Karel and Karel (1991) found 2.52 cm in H. gluttatus; Minelli (1985) reported a length of 0.3 cm in H. hippocampus, and Fam (1992) calculated 1 cm for H. kuda. This study showed that H. ingens juveniles were hatched with a length of 0.7 and reach 1.5 cm the first week, and 2.18 cm in one month; they were fed with rotifers. Under similar conditions, Reyes-Bustamante and Ortega- Salas (1999) calculated a better growth from an initial length of 0.69 cm to reach 2.84 cm in one month; they were fed with a variety of live food such as rotifers, copepods, and Artemia nauplii. In H. erectus, Correa et al. (1989) registered 1.3 cm of initial length that grew to 3.47 cm in 35 days; they were fed with Artemia nauplii. Liang (1992) mentioned that H. japonicus is larger than 4.5 cm after one month. Tawil (1994) estimated that H. ingens reached 3.5 cm in one month.
As observed H. ingens and H. kuda are the largest species reaching a 30 cm length but hatching smaller juveniles as compared with the other species that are smaller in adult length but hatch bigger juveniles, such as H. enano. Reyes-Bustamante and Ortega-Salas (1999) calculated from the length-weight relationship of juveniles, a slope of 2.98, which compared with the 3.09 slope obtained in this study, indicates a slightly heavier body weight for a given length.
In this study, mortality was 38.1, 41.0, and 78.5 % in 95 days (Table 1). Reyes-Bustamante and Ortega-Salas (1999) calculated a mortality rate between 20 to 40 % in 63 days; juveniles were fed with live food in both cases. In other species Fam (1992) and Liang (1992) mentioned for Hippocampus kuda that food supply such as fresh and live copepods at the fry stage is good for growth promotion, disease resistance, and survival. Correa et al. (1989) calculated a mortality of 50.67 % for H. erectus in 35 days; however if water and quality food improved, survival rate increased to 97.08 % for juveniles and 70 % for adults.
The success of rearing at least 50% of the seahorse juveniles to young commercial stages at temperatures between 17 and 23 ºC was due to the good quality and quantity of live food under appropriate seawater conditions in the laboratory and using open-air tanks.
Acknowledgments
We thank A. Nuñez P, J.L. Beltrán M., J.A. Ortega R. and C. Ramírez for their devoted participation in this study.
Resumen
Estudiamos la fecundidad, supervivencia, y crecimiento del caballito de mar, Hippocampus ingens en condiciones semi-controladas. Tres machos reproductores silvestres de 14.8, 24.5 y 32.0 g produjeron 1 598, 1 703 y 1 658 jóvenes. La densidad utilizada fue de 12 jóvenes por acuario de 60 l. Se agruparon en 1, 12 y 20 días de nacidos por acuario. La supervivencia fue de 78.5, 38.1 y 41.0 % en 35 días. Se les alimentó con una mezcla de rotíferos B. plicatilis y nauplios de Artemia para después transferirlos a estanques de 100 000 l a una densidad de 50/1 000 l, donde se les alimentó con Artemia adulta durante 60 días más. Crecieron de un promedio de 0.7, 1.5, y 2.7 a 4.5, 5.4 6.7 cm, respectivamente, en 95 días. La temperatura del agua marina utilizada varió de 17 a 23 oC.
Palabras clave: caballito de mar, Hippocampus ingens, acuicultura, crecimiento, mortalidad.
References
Axelrod, H., W. Burges & C. Emmens. 1969. Exotic marine fishes. TFH, New York, USA. p. 400-409. [ Links ]
Correa, M., K.S. Chung & R. Manrique. 1989. Experimental culture of the seahorse, Hippocampus erectus. Bol. Inst. Oceanog. de Venezuela 28: 91-196. [ Links ]
Fam, T.M. 1992. Rearing of the seahorse Hippocampus kuda. Biol. Morya Mar. Biol. 56: 93-96. [ Links ]
Graff, F. 1968. Marine Aquarium Guide. Pet Library, London, England. 184-187 p. [ Links ]
Karel, P. & C. Karel 1991. Caballito de Mar Hippocampus guttatus. El Gran Libro de los Peces. Susaeta, Madrid, Spain. 176 p. [ Links ]
Liang, B. 1992. Research on the culture of Hippocampus. J. Oceanol. Univ. Qingdao Peopl. Rep. China 22: 39-44. [ Links ]
Mi, P.T. 1992. Raising the sea horse Hippocampus kuda. Russ. Jour. Mar. Biol. 18: 93-96. [ Links ]
Mi, P.T. 1993. Raising the sea horse Hippocampus kuda. Biol. Mor. 18: 203-205. [ Links ]
Minelli, A. 1985. Nueva enciclopedia del reino animal. Inst. Biól. Anim., Universidad de Padua, Padua, Italy. 180 p. [ Links ]
Pivnicka, K & K. Cemy. 1991. Caballito de mar, Hippocampus guttulatus. El gran libro de los peces. SUSAETA, Madrid, Spain. 304 p. [ Links ]
Reyes-Bustamante, H. & A.A.Ortega-Salas. 1999. Cultivo del caballito de mar, Hippocampus ingens (Pisces: Syngnathidae) (Girard, 1859) en condiciones artificiales. Rev. Biol. Trop. 47: 1045-1049. [ Links ]
Tawil, G.A. 1994. Aqua Guía Méx. 2: 28-33. [ Links ]
Vincent, A. 1994. The improbable Sea horse. Natl. Geogr. 186: 126-140. [ Links ]
Vincent, A.C.J. & H.R.S. Clifton. 1989. Parasitic infection of the sea horse Hippocampus ingens a case report. J. Wild Dis. 25: 404-406. [ Links ]
 All the contents of this journal, except where otherwise noted, is licensed under a Creative Commons Attribution License
All the contents of this journal, except where otherwise noted, is licensed under a Creative Commons Attribution License