Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.57 suppl.1 San José Nov. 2009
In vitro plant regeneration system for tropical butternut squash genotypes (Cucurbita moschata)
Marta Valdez-Melara1, Alexander García1, Marlon Delgado1, Andrés M. Gatica-Arias1 & Pilar Ramírez-Fonseca1,2
1. Escuela de Biología, Universidad de Costa Rica, 11501-2060, San Pedro, Costa Rica. Tel: (506) 2075091, Fax: (506) 2074216; marta.valdez@ucr.ac.cr; andresgat@gmail.com
2. Centro de Investigación en Biología Celular y Molecular (CIBCM), Universidad de Costa Rica, 11501-2060, San Pedro, Costa Rica. Tel: (506) 2075091, Fax: (506) 2074216; pilar.ramirez@ucr.ac.cr
Abstract: An efficient and reproducible method for regeneration of commercial and pure lines of tropical butternut squash (Cucurbita moschata) plants via somatic embryogenesis was developed. The influence of genotype, explant source, N6-benzylaminopurine (BAP), 2,4-dichlorophenoxyacetic acid (2,4-D) and 2,4,5-trichlorophenoxyacetic acid (2,4,5-T) concentration on somatic embryogenesis induction was investigated. Friable embryogenic calli was produced from zigotic embryos (53-56%) and cotyledons from seedlings (70%) of C. moschata cv. Sello de Oro cultured on callus induction medium (CIM) supplemented with 0.5 mg/l or 3.5 mg/l 2,4-D. No embryogenic calli was obtained from leaf segments of C. moschata cv. Sello de Oro cultured on CIM supplemented with different concentrations of BAP and 2,4-D and cotyledons from seedlings of C. moschata cv. PVG 04 cultured on CIM with BAP and 2,4,5-T. Embryogenic calli induction was achieved in 75% C. moschata pure lines evaluated and calli percentage frequency range from 5% to 34%. Successful acclimatization of squash in vitro plants was achieved in the greenhouse and in the field. Regenerated plants appeared morphologically normal and set flowers and fruits with seeds that could germinate normally. Rev. Biol. Trop. 57 (Suppl. 1): 119-127. Epub 2009 November 30.
Key words: cucurbits, Cucurbita moschata, auxins, cytokinin, explant source, genotype, Costa Rica.
The Cucurbitaceae is a large family that includes a number of valuable edible crop species. Collectively world production of cucurbits in 2004 reached 19.7 million metric tons (FAOSTAT 2005). Squash (Cucurbita moschata) is an important crop for countries of tropical and temperate zones (Chee 1991). In Costa Rica, squash is a crop of increasing economic and social importance. In 2006, Costa Rica exported 8 348 kg of squash with a total value of US$ 2 509 472 (BCCR 2006).
Despite its importance, this species has not been subject of sufficient genetic or biotechnological investigations. Improvement of this crop to resist viruses, insects, weeds and somat to improve its growth and vigor would be of considerable commercial value. Plant biotechnology techniques could help overcoming these problems, but their use requires a reliable and efficient in vitro culture system (Debeaujon & Branchard 1993).
Plant regeneration from in vitro cultured cells can be accomplished thought somatic embryogenesis or organogenesis. In the first case, somatic embryogenesis is the developmental process by which bipolar structures that resemble zygotic embryos are developed from haploid or diploid somatic cell through an orderly embryological stage without gametes fusion. This process can be achieved via direct somatic embryogenesis (DSE) or indirect ic embryogenesis (ISE). DSE is characterized by the induction of somatic embryos directly from pro-embryogenic cells of the leaf, stem, microspores or protoplasts in the absence of conspicuous embryogenic calli proliferation, whereas in ISE somatic embryos are developed from friable embryogenic calli (Jiménez 2005).
On the other hand, organogenesis is the developmental pathway by which shoots and roots are formed in response to culture (concentration and type of plant growth regulators) and environmental (light intensity, temperature and photoperiod) conditions. Contrasting to somatic embryogenesis pathway, organogenesis is characterized by the presence of vascular connections between mother tissue and the regenerating sections (Jiménez 2001, 2005).
In the Cucurbitaceae family, in vitro plant regeneration has been reported by organogenesis in squash (Cucurbita pepo L.) (Abrie & Staden 2001, Ananthakrishnan et al. 2003, Kathiravan et al. 2006, Selvaraj et al. 2006, Ananthakrishnan et al. 2007), pumpkin (Cucurbita maxima L.), melon (Cucumis melo L.) and cucumber (Cucumis sativus L.) (Abrie & Staden 2001) or somatic embryogenesis in pumpkin (Cucurbita pepo L.) (Chee 1991, Gonsalves et al. 1995, Kintzios et al. 2002, Leljak-Levani et al. 2004) and melon (Abrie & Staden 2001, Kintzios et al. 2002). Several protocols have been described in the literature for plant regeneration of squash via ISE (Debeaujon & Branchard 1993), nevertheless, specific conditions and protocols developed for a particular genotype are not necessarily reproducible for others.
The influence of BAP, 2,4-D and 2,4,5-T concentration, explant source and genotype on ISE induction of Cucurbita moschata has not been reported. The aim of this work was to set up a protocol for the callus induction and plant regeneration thought ISE, as well as for the acclimatization of tropical butternut squash (C. moschata) plants.
Materials and methods
Plant material and explant preparation.
Mature seeds of commercial butternut squash (C. moschata) cultivars Sello de Oro, Birris, PVG 01 and PVG 04 were used as source of explants. Also, seeds of C. moschata pure lines 7036, 18933, 15049A, 5896, 5998, 14895, 12130, 5978, 18938, 7211, 11048, 11442, 9284A, 9206, 9212, 11316, 5996, 9316A, 6368 and 11428, provided by the Centro Agronómico de Investigación y Enseñanza (CATIE, Turrialba, Costa Rica) were used. After removal of the seed coat, the de-coated seeds were washed in 70% (v/v) ethanol for 10 min, disinfected in 4.5% (v/v) sodium hypochlorite (NaOCl) supplemented with eight drops of Tween 20 (Sigma, St. Louis, MO, USA) for 20 min followed by an immersion in the fungicide Benomyl (Piscis, Costa Rica) at a concentration of 100 mg/l for 5 min. Finally, the seeds were washed three times with sterile distilled water.
The disinfected seeds were cultured in 125 ml flasks, closed with polyethylene food wrap (Glad, Costa Rica), containing 20 ml of half strength Murashige & Skoog (1962) medium (MS) with 30 g/l sucrose and 3 g/l Gelrite. The pH was adjusted to 5.6 with NaOH before autoclaving for 21 min at 121 °C and 1.05 kg cm-2. Explants were cultured with 16 h light photoperiod (30 µmol/m2 s1) at 26±2 °C.
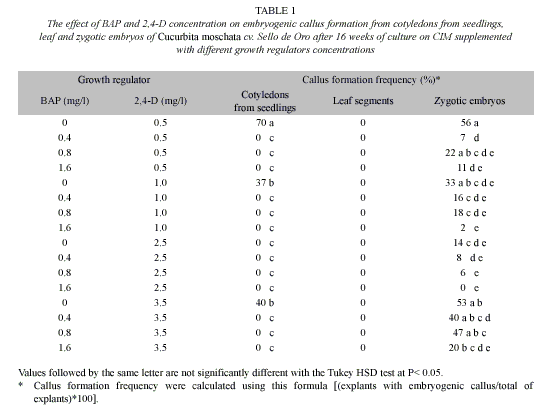
Induction of somatic embryogenic and plant regeneration. Cotyledons (1 cm2) and leaf segments (1 cm2) of C. moschata cv. Sello de Oro were excised from in vitro grown seedlings seven days after germination. Moreover, the zygotic embryos excised from disinfected seeds were used as explant. In a first experiment, these explants were cultured on Petri dishes (100 mm x 20 mm) containing 20 ml of CIM composed of MS mineral salts and vitamins, BAP (0, 0.4, 0.8 and 1.6 mg/l) and 2,4-D (0.5, 1.0, 2.5 and 3.5 mg/l) (Table 1), 30 g/l sucrose and 3 g/l Gelrite. The pH was adjusted to 5.6 with NaOH before autoclaving for 21 min at 121 °C and 1.05 kg cm-2. Cultures were maintained in the dark at 26±2 °C. Embryogenic calli were transferred to fresh medium every four weeks during 16 weeks.
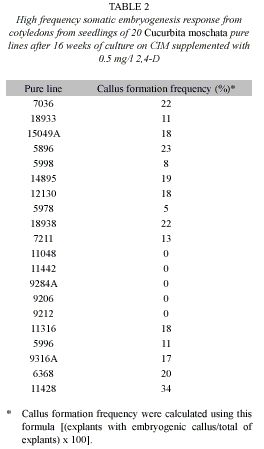
Once the best explant source and culture media composition was determined, in a second experiment, cotyledons from seedlings of C. moschata cv. Birris, PVG 01 and PVG 04 were cultured on CIM supplemented with 0.5 mg/l 2,4-D. To evaluate the influence of 2,4,5-T on embryogenic calli induction, in a third experiment, cotyledons from seedlings of C. moschata cv. PVG 04 were cultured on MS basal medium supplemented with BAP (0, 0.4, 0.8 and 1.6 mg/l) and 2, 4, 5-T (0.5, 1.0, 2.5 and 3.5 mg/l) (Table 2). Moreover, the cotyledons from seedlings of 20 pure lines of C. moschata provided by CATIE were cultured on MS basal medium supplemented with 0.5 mg/l 2,4-D. Callus formation frequency [(explants with embryogenic callus/total of explants)*100] was evaluated after 16 weeks of culture.
After 16 weeks of culture on CIM, embryogenic calli were transferred to regeneration medium (RM) composed of MS mineral salts supplemented with 0.05 mg/l napthaleneacetic acid (NAA) and 0.05 mg/l kinetin (Chee 1991).
After six weeks of culture RM, the regenerated plantlets were transferred to half strength MS medium. The explants were cultured under 16 h light photoperiod (30 µmol/m2 s1) at 26 ±2 °C. The regeneration percentage [(number of plants regenerated from shoots/total of embryogenic calli)*100] was recorded after five weeks of culture on RM.
Scanning electron microscopy (SEM). Embryogenic callus pieces were fixed in 2.5% glutaraldehyde and 2.5% paraformaldehyde in 0.1 M sodium phosphate buffer with pH 7.4 for 24 h at 4 ºC. The samples were washed twice in the same buffer and were postfixed in 2% osmium tetroxide solution for 2 h. Fixed tissues were dehydrated in a graded ethanol solutions for 15 min each. After that, the dehydrated specimens were critical point dried and then dried specimens were mounted on aluminium blocks. Finally, the mounted specimens were coated with a very thin layer of gold-palladium and subsequently the coated specimens were examined in a scanning electron microscope (S-570, Hitachi, Japan) at an accelerating voltage of 15 KV.
Acclimatization and field transfer. Regenerated shoots (approximately 3 cm long) with well developed leaves and roots were transferred to pots containing Peat Moss and organic soil, covered with plastic bags and maintained in the growth room under 12 h light photoperiod (30 µmol m-2s-1) at 26 ±2 °C. The plantlets were watered twice a week with water. Then, after two week, the plantlets were transferred in soil under field conditions. The plant regeneration percentage [(number of plants successful acclimated/total number of acclimated plants)*100] was determined. Moreover, a phenotypic analysis was conducted using flowers, fruits and seeds production.
Statistical analysis. The statistical analysis was performed using one-way ANOVA and the significance of differences among treatment means were contrasted with Tukeys Honestly Significant Difference Test (HSD) at p<0.05. The program STATISTICA (StatSoft, Tulsa, OK, USA) version 6.0 was used.
Results
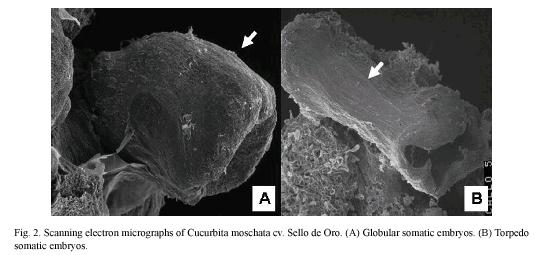
ISE began with the development of a translucid primary callus on the cut edges of explants after two weeks of culture on CIM. Subsequently, yellow friable embryogenic callus formed on the edges of primary callus after 16 weeks of culture on CIM (Fig. 1A). Shoots developed after five weeks of culture on RM (Fig. 1B). Squash somatic embryos developed into plants with two to four pairs of leaves (Fig. 1C) and 100% of the regenerated plants were successful acclimated and developed into adult plants with normal flowers and fruits (Fig. 1D and 1E). Scanning electron micrographs showed formation torpedo shape somatic embryos on the embryogenic calli surface (Fig. 2).
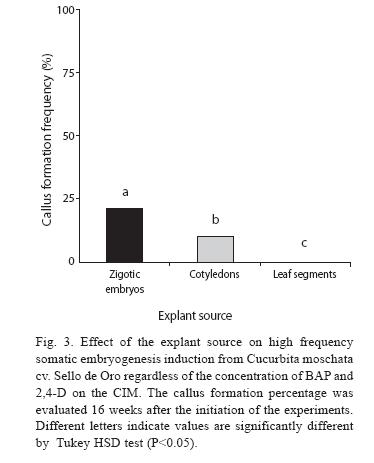
Cucurbita moschata cv. Sello de Oro was used in initial experiments. Regardless of the concentration of BAP and 2,4-D on the CIM, the calli formation frequency differed significantly among the three types of explants evaluated. The higher percentage of callus formation was obtained using zigotic embryos (21%), cotyledons from seedlings (10%) and leaf segments (0%) (P<0.05) (Fig. 3).
Culture of C. moschata cv. Sello de Oro cotyledons from seedlings on CIM supplemented with a range concentration of BAP and 2,4-D resulted in a differential response. The best results were obtained when cotyledons from seedlings were cultured on CIM with 0.5 mg/l 2,4-D (Table 1). Embryogenic calli were obtained from C. moschata cv. Sello de Oro zygotic embryos cultured on CIM supplemented with all the concentrations of BAP and 2,4-D evaluated, except with 1.6 mg/l BAP and 2.5 mg/l 2,4-D. The best results were obtained when zygotic embryos were cultured on CIM with 0.5 or 3.5 mg/l 2,4-D (Table 1).
Independently of the 2,4-D concentration on the CIM, it was observed that as the BAP concentration on the CIM increase, the callus formation frequency decreased (Table 1).
On the other hand, no embryogenic calli were observed in cotyledons from seedlings of C. moschata cv. PVG 04 after 16 weeks of culture on the CIM supplemented with different BAP and 2,4,5-T concentrations (data not show).
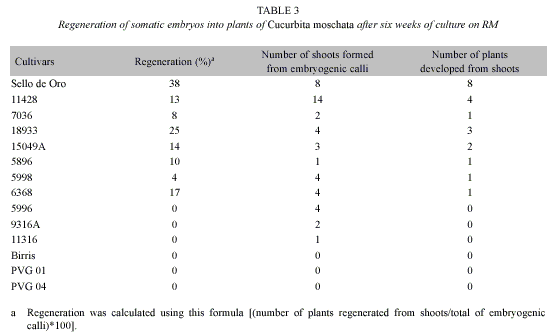
Embryogenic calli induction was achieved in 75% (15 of the 20) C. moschata pure lines evaluated. No embryogenic calli were obtained in 11048, 11442, 9284A, 9206 and 9212 pure lines after 16 weeks of culture on CIM supplemented with 0.5 mg/l 2,4-D. In the reactive C. moschata pure lines, callus formation frequency range from 5% to 34% and it was lower than the frequency obtained using C. moschata cv. Sello de Oro (Table 2).
In relation to the regeneration of shoots from embryogenic calli and their conversion into plants, only C. moschata cv. Sello de Oro and the pure lines 6368, 11428, 7036, 18933, 15049A, 5896 and 5998 developed plants from shoots. In the C. moschata pure lines 5996, 9316A and 11316, the shoots regenerated from embryogenic calli did not develop into plants (Table 3). The higher regeneration percentage and number of plants developed from shoots was obtained with C. moschata cv. Sello de Oro (Table 3).
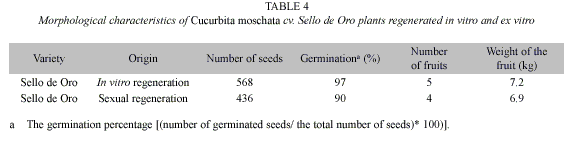
The number of seeds per fruit, the number of fruits per plant and the weight of the fruits were very similar among plants regenerated in vitro from embryogenic calli and plants regenerated ex vitro from sexual seed (Table 4).
Discussion
A protocol for the induction of C. moschata embryogenic callus and for the formation of somatic embryos was established in the present study. Germination and development of butternut squash somatic embryos into plants was also successfully obtained. Moreover, the results obtained in this work corroborated the influence of the genotype, the source of explant and the type and concentration of plant growth regulator on ISE induction using commercial and pure lines of C. moschata.
The influence of the genotype was observed since the higher calli formation frequency was obtained with Sello de Oro cultivar than with Birris, PVG 01, PVG 04 and the C. moschata pure lines (Table 1 and 2). In this sense, it has been reported that the embryogenic capacity is genetically determined in cucumber (Nadolska-Orczyk & Malepsky 1989 reviewed by Debeaujon and Branchard 1993) melon (Oridate et al. 1992 reviewed by Debeaujon & Branchard 1993), black peper (Piper nigrum L.) (Ramakrishnan et al. 2005) rice (Oryza sativa L.) (Hoque & Mansfield 2004), cotton (Gossypium hirsutum L.) (Wu et al. 2004) and coffee (Coffea sp.) (Bieysse et al. 1993, Molina et al. 2002, Santana et al. 2004).
In addition, our results corroborated that the explant source influenced ISE since higher calli formation frquency was obtained using zigotic embryos and cotyledons from seedlings. Success in regeneration thought somatic embryogenesis depends on the origin, physiological state and age of the explant. Generally, immature organs and meristematic tissues, which contain undifferentiated cells, are more suitable for plant morphogenesis (Hoque & Mansfield 2004). In cucurbits, diverse types of explants have been used to induce somatic embryogenesis, nevertheless, cotyledons and hypocotyls tissue are especially successful (Debeaujon & Branchard 1993). In C. moschata cv. Seoul Dabagi and PM 143, somatic embryos and plants have been obtained by DSE from nucellar cells from unfertilized ovules cultured on half strength MS (Kwack & Fujieda 1998 reviewed by Debeaujon & Branchard 1993). To our knowledge, this is the first report of somatic embryogenesis induction in C. moschata using zigotic embryos and cotyledons as explants. However, due to the lower availability of zigotic embryos, this tissue was considered less appropiate for ISE induction and plant regeneration in C. moschata. Regarding the influence of plant growth regulators (PGR), the best results were obtained using
0.5 and 3.5 mg/l 2,4-D (Table 1). In this sense there are several reports that relate the endogenous hormone levels in the initial explants and the application of exogenous plant growth regulators to the morphogenetic competence of a particular explant type and genotype (Jiménez 2001). On squash somatic embryogenesis induction, investigations have been focused on the type, concentration and time of application of PGR. The process of somatic embryogenesis in the Cucurbitaceae family requires an induction culture medium followed by a maturation medium. Generally, the former medium is supplemented with an auxin, such as 2,4-D, NAA, IBA (indole-3-butyric acid) and 2,4,5-T (Debeaujon & Branchard 1993).
On the other hand, no embryogenic calli were obtained from C. moschata PVG 04 cotyledons using different BAP and 2,4,5-T concentrations. In contrast, Chee (1991, 1992) obtained embryogenic calli from shoot tips and cotyledons of C. pepo L. cv. YC60 cultured on MS supplemented with 1.2 mg/l 2,4,5-T, 0.8 mg/l BAP and 0.1 mg/l kinetin. Because cotyledons from seedlings of C. moschata cv. PVG 04 were the only source of explants evaluated, further studies are required to determine the influence of BAP and 2,4,5-T on somatic embryogenesis induction from different C. moschata genotypes and explants.
The PGR used at the induction phase can play an important role on somatic embryogenesis process. Somatic embryogenesis is generally promoted by auxins, either alone or in combination with cytokinins (Pacheco et al. 2007). Althought, BAP is the most frequently used cytokinin for somatic embryogenesis induction in cucumber and melon (Debeaujon & Branchard 1993), in C. pepo L. BAP reduced embryogenesis efficiency (Noël et al. 1992 reviewed by Debeaujon & Branchard1993).Asimilarresultwas observedin the present study when C. moschata Cv. Sello de Oro cotyledons were cultured with different BAP concentrations (Table 1).
Based on the protocol developed in this study, C. moschata in vitro plants could be obtained in 20 weeks after culture initiation.
Moreover, the in vitro protocols reported in this work could be used for mass production of somatic embryos in bioreactor for use in artificial seeds and to obtain competent target tissue (embryogenic calli and plants) for genetic modification thought biolistic, Agrobacterium or mutagenesis.
Acknowledgments
The authors are grateful to Paul Hanson (School of Biology, UCR) for languague
correction of the manuscript. Financial support was provided by Consejo Nacional para Investigaciones Científicas y Tecnológicas (CONICIT, Costa Rica) and the Vicerrectoría de Investigación of the University of Costa Rica (UCR).
Resumen
En este estudio se desarrolló un método eficiente y reproducible para la regeneración de líneas puras de la planta tropical Cucurbita moschata mediante la vía de embriogénesis somática. Además se investigó acerca de la influencia del genotipo, transplante, y la concentración de N6-benzylaminopurina (BAP), 2,4-diclorofenoxyacetico ácido (2,4-D) y 2,4,5-triclorofenoxyacetico ácido (2,4,5-T) en la inducción de embriogénesis somática. Los callos embriogenéticos viables fueron producidos de embriones zigóticos (53-56%) y cotiledones de semillas (70%) de C. moschata cv. Sello de Oro cultivados en un medio de inducción de callos (CIM) suplementado con 0.5 mg/l o 3.5 mg/l 2,4-D. Los callos no embriogénicos fueron obtenidos de segmentos de hojas de C. moschata cv. Sello de Oro cultivados con CIM suplementado con diferentes concentraciones de BAP y 2,4-D y cotiledones de semillas de C. moschata cv. PVG 04 cultivado con CIM con BAP y 2,4,5-T. La inducción de callos embriogenéticos fue exitosa en un 75% de las líneas puras evaluadas de C. moschata y el porcentaje de frecuencia de los callos fue de 5% a 34%. Se logró una adecuada aclimatización de las plantas in vitro tanto en el invernadero como en el campo. Las plantas regeneradas fueron normales morfológicamente, y las flores y frutos poseen semillas que pueden germinar normalmente.
Palabras clave: cucurbitas, Cucurbita moschata, auxinas, citoquinina, transplante, genotipo, Costa Rica.
Received 03-X-2007. Corrected 01-IX-2009. Accepted 04-X-2009.
References
Abrie, A.L. & J. Van Staden. 2001. Development of regeneration protocols for selected cucurbit cultivars. Plant Growth Regul. 35: 263-267. [ Links ]
Ananthakrishnan, G.,· X. Xia, C. Elman, S. Singer, H.S. Paris, A. Gal-On & V. Gaba. 2003. Shoot production in squash (Cucurbita pepo) by in vitro organogenesis. Plant Cell Rep. 21: 739-746. [ Links ]
Ananthakrishnan, G.,· X. Xia, S. Amutha, S.Singer, M. Muruganantham, S. Yablonsky, E. Fischer & V. Gaba. 2007. Ultrasonic treatment stimulates multiple shoot regeneration and explant enlargement in recalcitrant squash cotyledon explants in vitro. Plant Cell Rep. 26: 267-276. [ Links ]
Bieysse, D., A. Gofflot & N. Michaux-Ferriere. 1993. Effect of experimental conditions and genotypic variability on somatic embryogenesis in Coffea arabica. Can. J. Bot. 71: 1496-1502. [ Links ]
Chee, P. 1991. Somatic embryogenesis and plant regeneration of squash Cucurbita pepo L cv. YC 60. Plant Cell Rep. 9: 620-622. [ Links ]
Chee, P. 1992. Initiation and maturation of somatic embryos of squash (Cucurbita pepo). HortScience. 27: 59-60. [ Links ]
Debeaujon, I. & M. Branchard. 1993. Somatic embryogenesis in Cucurbitaceae. Plant Cell Tiss. Organ Cult. 34: 91-100. [ Links ]
Leljak-Levani, D., N. Bauer, S. Mihaljevi & S. Jelaska. 2004. Somatic embryogenesis in pumpkin (Cucurbita pepo L.): Control of somatic embryo development by nitrogen compounds. J. Plant Physiol. 161: 229-236. [ Links ]
Gonsalves, C., B. Xue & D. Gonsalves. 1995. Somatic embryogenesis and regeneration from cotyledon explants and six squash cultivars. HortScience.30: 1295-1297. [ Links ]
Hoque, M.D. & J.W. Mansfield. 2004. Effect of genotype and explant age on callus induction and subsequent plant regeneration from root-derived callus of Indica rice genotypes. Plant Cell Tiss. Organ Cult.78: 217-223. [ Links ]
Jiménez, V. 2001. Regulation of in vitro somatic embryogenesis with emphasis on the role of endogenous hormones. R. Bras. Fisiol. Veg. 13: 196-223. [ Links ]
Jiménez, V. 2005. Involvement of plant hormones and plant growth regulators on in vitro somatic embryogenesis. Plant Growth Regul. 47: 91-110. [ Links ]
Kathiravan, K., G. Vengedesan, S. Singer, B. Steinitz, H.S. Paris & V. Gaba. 2006. Adventitious regeneration in vitro occurs across a wide spectrum of squash (Cucurbita pepo) genotypes. Plant Cell Tiss. Organ Cult. 85: 285-295. [ Links ]
Kintzios, S., E. Sereti, P. Bluchos, J.B. Drossopoulos, C.K. Kitsaki &· A. Liopa-Tsakalidis. 2002. Growth regulator pretreatment improves somatic embryogenesis from leaves of squash (Cucurbita pepo L.) and melon (Cucumis melo L.). Plant Cell Rep. 21: 1-8. [ Links ] [ Links ]
Murashige, T. & F. Skoog. 1962. A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plant. 15: 473-497. [ Links ]
Raghavan, R. & D. G. Snehasish. 2005. Effect of Explants and Genotypes on Primary Somatic embryogenesis in Black Pepper (Piper nigrum L.). Cytologia. 70: 195-202. [ Links ]
Santana, N., M.E. González, M. Valcárcel, A. Canto-Flick, M. Hernández, C.F.J. Fuentes- Cerda, F. Barahona, J. Mijangos- Cortés & V.M. Loyola-Vargas. 2004. Somatic embryogenesis: a valuable alternative for propagating selected robusta coffee (Coffea canephora) clones. In Vitro. Dev. Biol. Plant. 40: 95-101. [ Links ]
Selvaraj, N., A. Vasudevan, M. Manickavasagam & A. Ganapathi. 2006. In vitro organogenesis and plant formation in cucumber. Biol. Plant. 50: 123-126. [ Links ]
Wu, J., X. Zhang, Y. Nie, S. Jin & S. Llang. 2004. Factors affecting somatic embryogenesis and plant regeneration from a range of recalcitrant genotypes of Chinese cottons (Gossypium hirsutum L.). In Vitro. Dev. Biol. Plant. 40: 371-375. [ Links ]
Internet references
BCCR (Banco Central de Costa Rica). Estadísticas de exportaciones: ayote. Available http://www.bccr.fi.cr/ flat/bccr_flat.htm. 2006. [ Links ]
FAOSTSAT. 2005. World agricultural data. Available in: http://apps.foa.org (March 20 2007). [ Links ]