Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO  uBio
uBio
Share
Revista de Biología Tropical
On-line version ISSN 0034-7744Print version ISSN 0034-7744
Rev. biol. trop vol.48 n.4 San José Dec. 2000
the Atlantic versant of northern Central America
A new species of green, prehensile-tailed pitviper of the genus Bothriechis is described from the Atlantic slopes of eastern Guatemala and western Honduras. This species appears to be most closely related to B. bicolor of the Pacific versant of Chiapas (Mexico) and Guatemala. Several other species of Bothriechis occur on the Atlantic versant of northern Central America, including two montane species, B. aurifer and B. marchi but, with one possible exception, these are not known to be sympatric with the new species and occur in different mountain ranges. The widespread B. schlegelii occurs up to at least 900 m on the Sierra de Caral, where the lowest elevation recorded for the new species is 885 m.
Key words
Reptilia, Squamata, Viperidae, Pitvipers, Bothriechis, New species, Izabal, Guatemala, Honduras.
Species of the arboreal pitviper genus Bothriechis range from the highlands immediately east of the Isthmus of Tehuantepec to western Panama, with one species extending into northern South America (Crother et al. 1992). Despite this wide distribution, the actual habitat in which most of these species occur is remarkably restricted. The only species having a moderately wide distribution is B. schlegelii, which occurs in the mesic tropical forests of the Atlantic lowlands and foothills from Chiapas (Mexico) through Central America to northern South America and on parts of the Pacific versant from Costa Rica to Peru (Campbell and Lamar 1989, 1992). All other species of Bothriechis occur in isolated, wet, montane forests, usually above 1 000 m, but two species descend to about the 500 m contour.
The genus Bothriechis is defined by being arboreal, prehensile-tailed, New World pitvipers that are usually mostly greenish in color, have the middle preocular and supralacunal coalesced into a single scale, and have an exceptionally short tail spine that is usually not longer than about the preceeding subcaudal. Several species may approach or even slightly exceed a meter in total length, but these snakes generally have maximum total lengths between 700 and 850 mm. The tail comprises >15% of the total length. There are 7-12 supralabials, 8-13 infralabials, 137-175 ventrals, 42-75 undivided subcaudals, and 17-25 dorsal scale rows at midbody. The canthus is angular, the snout is unelevated and broadly rounded in dorsal view, and the rostral is broader than high. The scales covering the crown of the head are extremely variable, with the number of scales between the supraoculars being as few as one (in some B. aurifer) to as many as 11 (in some B. bicolor).
Materials and methods
Specimens were preserved in formalin (diluted to 10% of stock solution) and subsequently transferred to 70% ethanol for permanent storage. Comparative material of New World pitvipers has been examined at one time or another from most of the major museums in North America. Individuals of the new species described here are curated in the collections of the University of Kansas (KU), Louisiana State University (LSU) and the University of Texas at Arlington (UTA). We have examined Bothriechis bicolor from the American Museum of Natural History (AMNH), University of Michigan Museum of Zoology (UMMZ), the United States National Museum (USNM), and UTA (see Appendix–Specimens Examined).
Measurements were taken using digital calipers or a meter stick. Dimensions of scales and head were taken to the nearest 0.1 mm; those of snout-vent length (SVL), tail length, ot total length (TL) were taken to the nearest mm.
Terminology for scales in most instances follows Klauber (1972) or as defined in Gutberlet and Campbell (2000). It is worth noting that the canthals include the large, flat scales that border the crown between the internasal(s) and the supraoculars. In some species of Bothriechis there are only two of these scales on each side, but in other species there may be one or two smaller sales intervening between the supraocular and the last large scale in this series; these are considered canthals also, even when they are excluded from the edge of the canthus proper by the dorsalmost preocular. The method of counting ventrals is that of Dowling (1951). Interrictal counts include the ultimate supralabial on each side.
Systematic account
Recent incursions into several mountain ranges along the Guatemala-Honduras border have revealed the presence of a greenish blue, arboreal species of pitviper heretofore unknown from Guatemala. Comparison of this Guatemalan material with a few individuals known from Honduras confirms that these populations are conspecific with each other but distinct from B. bicolor of the Pacific versant of southern Mexico and Guatemala.
Bothriechis bicolor–Campbell and Lamar 1989:163, Crother et al. 1992:1. [In part.]
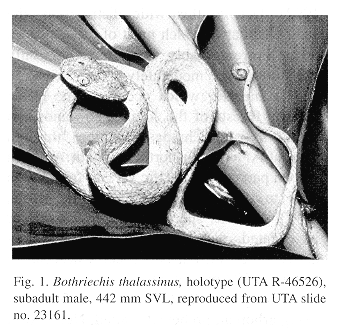
Holotype.–The University of Texas at Arlington (UTA) R-46526, a subadult male from Finca La Firmeza, Sierra de Caral, Izabal, Guatemala, 900 m (15û22'29" N, 88û41'44" W), collected on 30 Aug. 1997 by E. N. Smith (Fig. 1).
Paratypes.–All from the Sierra de Caral, Departamento de Izabal, Guatemala. UTA R-38891 and 42259, Aldea Negro Norte, NE slope of Cerro del Aguacate, ca. 1 200 m, Sierra de Caral; UTA R-39251, Finca La Firmeza, on trail to Aldea Negro Norte, 885 m, Sierra de Caral; Cerro del Mono, 1 420-1 450 m, near La Unión, Sierra del Merendón (UTA R-37223, 38220, 44438).
Referred specimens.–All from Honduras. KU 203094, Quebrada Grande, 1 300 m, Copán; LSU 23821, 21.7 km E Nueva Ocotepeque, 1 730 m, Depto. Ocotepeque; LSU 11638, SE slope of Cerro Santa Bárbara, 1 524 m, Depto. Santa Bárbara.
Definition and diagnosis.–A moderately sized (up to almost a meter in total length), green, arboreal pitviper in which the tail comprises between 16-18% of the TL in males and females. There are 5-9 (x = 7.7) intersupraoculars that are distinctly keeled, 23-26 (x = 24.1) interrictals that are also keeled, 161-168 ventrals, 60-67 undivided subcaudals, and usually 21 (rarely 23) dorsal scale rows at midbody (Table 1).
Bothriechis schlegelii occurs on the same slopes as B. thalassinus but generally at lower elevations. Small, spinelike supraciliary scales between the eye and supraoculars and usually at least 23 dorsal scale rows at midbody distinguish B. schlegelii from B. thalassinus. Two other congeners that occur on the Atlantic slopes of northern Central America, but rarely, if ever, are sympatric with B. thalassinus, include. Bothriechis aurifer has a dorsal pattern of yellow blotches edged with a black border that is often fused between adjacent blotches, only 1-5 intersupraoculars, and usually 19 dorsal scale rows at midbody. Bothriechis marchi has irregular, mostly flat scales on the crown, at least a few of which are usually conspicuously larger than surrounding scales, 3-7 intersupraoculars, and usually 19 scales at midbody. Bothriechis rowleyi of Oaxaca and Chiapas, Mexico, may be distinguished from B. thalassinus in having 3-5 intersupraoculars that are irregularly shaped, the adjacent scales on the crown lacking parallel keeling, and 19 dorsal scale rows present at midbody.
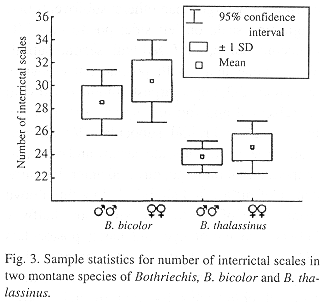
Bothriechis thalassinus most closely resembles B. bicolor of the Pacific slopes of southern Mexico and Guatemala, but B. bicolor differs in having more intersupraoculars (8-10, x = 8.7, in males; 8-11, x = 10.0 in females) and interrictals (27-31, x = 28.5, in males; 27-33, x = 30.3, in females) (Fig. 2). There is also a subtle difference in the shape of the first pair of infralabials. In B. bicolor these scales are relatively broad, whereas in B. thalassinus they are narrower and the exposure of the scales along the lingual margin is often reduced.
Two montane congeners occur south of the Nicaraguan Depression in Costa Rica and Panama. Bothriechis nigroviridis has a green body heavily speckled or mottled with black on the sides and the dorsum, fewer ventrals (139-158) and subcaudals (47-58), and usually 19 dorsal scale rows at midbody. Bothriechis lateralis has distinctive yellow paravertebral vertical bars and yellow paraventral stripes on the body. Further, there are usually 23 midbody dorsal scale rows in this species.
Description of holotype.–Subadult male; rostral broader than high (2.9 X 1.7 mm), nasal large, partially divided on top above naris, undivided below; loreals 1/1, contacting upper two preoculars; prefoveals 6/5, lower scale considerably larger than others; subfoveals 0/1, single tiny scale on left side; postfoveals 2/1; prelacunal large, not fused with second supralabial; sublacunal small; postlacunal elongate; preoculars 3/3, upper largest, middle fused with supralacunal and forming upper border of pit, lower small and round; suboculars 2/2, anterior scale large, posterior scale narrow and crescent-shaped; loreal pit large, directed anterolaterally, center of pit located slightly below line drawn from center of eye to naris and about halfway between center of eye and naris; postoculars 4/3; supralabials 9/10; mental broader than long (3.2 X 1.6 mm); infralabials 11/11; three pairs of chinshields flanking mental groove, anterior pair large, in contact with first three pairs of infralabials, followed by two pairs of smaller chinshields about half size of anterior scales; canthals 2/2, anterior above nasal, posterior contacting posterior portion of nasal, loreal, and anterior portion of upper preocular; single small, keeled head scale between posterior canthal and supraocular; three scales between anterior pair of canthals; four scales between posterior pair of canthals; supraoculars large and relatively narrow (4.3 X 1.7 mm); intersupraoculars 8; scales in parietal region small and keeled; interrictals 24; two gulars between posterior pair of chinshields and first preventral; dorsal scale rows 21-21-15; ventrals 161 (first ventral most anterior scale of series contacting paraventral row on both sides); anal undivided; subcaudals 67, undivided; tail spine short, rounded, and curved downward, about as long as preceeding subcaudal, two dorsal capping scales; 5 palatal, 10 pterygoid, and 15 dentary teeth.
Measurements.–Total length 442 mm; tail length 79 mm, comprising 17.9% of total; head length 20.7 mm; head width 13.7 at broadest point; neck 5.2 mm immediately behind jaws; fang length 4.0 mm from upper lumen to tip.
Color in preservative.–Dorsum of head and body dull blue-green with a few irregular pale bluish blotches on posterior third of body; distal tip of tail suffused with dark gray; rostral, lingual border of upper jaw, most of lower jaw, and throat pale yellow; venter of about anterior third of body pale yellow, becoming slightly pale greenish yellow on posterior part of body and subcaudals; upper and anterior border of pit with some yellow; iris pale gold heavily speckled with black.
Variation.–Contrary to the condition in many snakes, little or no sexual dimorphism is apparent in the number of ventrals or subcaudals. Males have 161-168 (x = 165.0) ventrals and females have 162-165 (x = 163.0); males have 61-67 (x = 63.6) subcaudals, whereas females have 60-64 (x = 62.0). The tail comprises 16.8-17.9 % (x = 17.5%) of the total length in males and 16.3-17.2% (x = 16.9%) in females. On the basis of ten specimens (7 males and 3 females), it is not possible to carefully assess variation, but it is possible that, compared to males, females have a greater number of intersupraoculars (9 versus 5-8, respectively) and interrictals (24-26 versus 23-25, respectively). Females may also average more supralabials (9-10, x = 9.6, in males, 10-12, x = 11.2, in females) and infralabials (10-12, x = 10.9, in males; 11-13, x = 11.9, in females). An identical trend is apparent for these particular head scales in B. bicolor (Table 1), a species for which a more adequate sample exists.
In B. thalassinus, dorsal scales are arranged in 21-21-17 rows in seven specimens and 23-23-17, 21-21-15, and 22-21-15 each in a single specimen.
Two males, including a juvenile (UTA R-37223, 357 mm TL) and an adult (UTA R-44438, 879 mm TL) had umbilical scars on Ventrals 147-149 and 149-151, respectively. Two juvenile females (KU 203094, 333 mm TL, and UTA R-38220, 430 mm TL) had prominent umbilical scars on Ventrals 144-146 and 145-147, respectively.
Statistical Analysis.–Differences between B. bicolor and B. thalassinus were further investigated statistically. The variables used were sex, SVL, head length, tail length, lacunalabial condition, number of intersupraoculars, number of interrictals (Fig. 3), total number of supralabials, total number of infralabials, ventrals, subcaudals, three dorsal scale row counts (one head length posterior to head, midbody, one head length anterior to vent). The head and tail lengths were regressed on SVL and the resulting residuals were used in the analyses, SVL was not incorporated directly in any analysis. In order to use the characters in comformity with assumptions of parametric analyses, their normality and heteroscedasticity were tested. The tail and head length residuals and subcaudal scales count needed no transformations. Of the other characters, number of interrictals and total number of infralabials were reciprocally transformed, and total number of supralabials and number of ventral scales were square-root transformed. Lacunolabial condition, number of intersupraoculars and number of dorsal scale rows could not be transformed. As a first exploration of the data, a discriminant function analysis was performed on the residual characters, the number of subcaudals, the transformed characters, and those characters that could not be transformed. The analysis showed high discrimination (Wilks' Lambda = 0.187, p < 0.0001) and a classification matrix with no errors. The single root obtained possessed a higher correlation with, in descending order, the number of interrictals, intersupraoculars and the first count for dorsal scale rows. The correlation suggests a generalized high number of scales on the dorsum of the head and neck region of B. bicolor, as compared to B. thalassinus. The number of dorsal scale rows on the anterior of the body is obviously associated with the number of interrictals. The lacunolabial condition between the two species showed no apparent distinction on a Kruskal-Wallis test (H [1, N = 61] = 2.563, p = 0.109).
An analysis of variance was performed on the variables that did not violate the parametric assumptions, using sex as a covariate. This procedure found a significant multivariate difference between the two species (Wilks' Lambda = 0.240; Rao's R = 18.504; df = 7, 41; p < 0.0001), where the only significant effect is that of the number of interrictals. The multivariate test for parallelism (effect of the covariate) was also significant (Wilks' Lambda = 0.673; Rao's R = 2.773; df = 7, 40; p = 0.019). Another analysis of variance was performed, this time making sex the independent variable and species the covariate. As expected, there was sexual dimorphism (Wilks' Lambda = 0.394; Rao's R = 9.006; df = 7, 41; p < 0.0001). This dimorphism was present in all characters analyzed, except number of ventrals (F(1, 47) = 2.25, p = 0.140). Ranking of the means of these characters (Table 2) shows a trend in which males have, on average, proportionately smaller heads with fewer scales and a longer tail with more ventral scales (taking into account that B. thalassinus usually has fewer interrictal scales, in general).
Color in life.–The dorsum of the head and body of the subadult male (UTA R-46526, 442 mm TL) was leaf green, grading to yellowish green on sides of body and the lateral portions of the ventrals and subcaudals. A series of irregular pale turquoise blotches was present on the top of the head and on the back. The sides of the body were also marked with turquoise but these were small spots (occupying 1-3 scales). A narrow turquoise postocular stripe extended from behind the eye to the angle of the jaw. The sides of the head below the canthus and below the postocular stripe were yellowish green. The distal portion of the tail was mostly turquoise, but heavily suffused with dark gray. The lower jaw, throat, and ventrals on the anterior part of the body were cream, grading to pale yellow at about midbody and then to pale green over most of the posterior half of the body and tail. The iris was green with an infusion of gold around the pupil, heavily speckled with fine black peppering.
A slightly smaller subadult male (UTA R-39251, 399 mm TL) from the Sierra de Caral was similar in color to the holotype, but the turquoise blotches on the dorsum were relatively larger and the iris was mostly yellowish.
Color notes in life for large adult Guatemalan specimens from the Sierra de Caral are not available, but judging from recently preserved individuals (UTA R-38891, trade;, 808 mm TL; UTA R-42259,
, 640 mm TL), the dorsum is uniformly dark green and the venter is likewise mostly dark green except for the lower jaw and throat which are yellowish.
We have seen a total of four specimens from the Sierra del Merendón in Zacapa, Guatemala. Two of these were juveniles and two were adults. Juveniles had a pale leaf-green to yellow-green ground color on the sides and dorsum of the head and body. The dorsum of the head and body was marked with black mottling that became progressively less evident on the posterior of the body. A pair of black stripes was present on top of the head, with a stripe extending from the frontal region near a supraocular posteriorly to the posterior temporal region. A black postocular stripe extended from behind the eye to the angle of the jaw and was bordered below by turquoise. The sides of the body were marked with turquoise spots, with each spot comprising 1-3 scales. The distal tip of the tail was black. In one adult, the black dorsal mottling was somewhat muted (UTA R-44438), and in the other (UTA slide nos. 15987-16012) the black pattern was well developed. Both adults had larger turquoise markings on the sides of the body than the juveniles.
A juvenile male (LSU 23821, 273 mm TL) from southwestern Honduras was described in life as "dorsum chartreuse green, dorsal blotches grayish-green, lateral blotches sky blue; venter very light green, finely peppered with dark flecks, lateral edges of most ventrals chartreuse green; head chartreuse green with two dark grayish-green bands extending from the snout to the angle of the jaw, a similar but lighter band extending from the posterior edge of the eye to the angle of the jaw; some additional dark grayish-green mottling between the two bands on top of the head; eye chartreuse green with heavy black reticulations" (Meyer and Wilson 1971).
Another Honduran specimen, the largest individual of the species known (LSU 11638, , 967 mm TL), was described by the collector as having a grass-green dorsum with powder blue chevrons and a yellowish green venter (Monroe inMeyer and Wilson 1971).
Etymology.–The specific epithet is derived from the Greek thalassinos, meaning blue-green, in allusion to the striking coloration of this snake.
Habitat, distributon, and habits.–In Guatemala, this species occurs in Lower Montane Wet Forest (sensu Holdridge 1959) at elevations of 885-1 450 m (Fig. 4). In Honduras, it has been recorded to occur also in Lower Montane Moist Forest (Meyer and Wilson 1985) and up to 1 730 m in the mountains east of Nueva Ocotepeque (LSU 23821).
Bothriechis thalassinus is known from several mountain ranges, including the Sierra de Caral (Izabal) and the Sierra del Merendón (Zacapa) in eastern Guatemala. In western Honduras, the species occurs in the Sierra Gallinero, which forms a continuous highlands with the Sierra de Caral. These highlands on the Guatemala-Honduras border are often referred to as the Sierra del Merendón or the Sierra del Espíritu Santo, although differences between the uses of these two names are considerable among maps. Bothriechis thalassinus is also known in Honduras from the mountains to the east of Nueva Ocotepeque and from the Cerro de Santa Bárbara, which flanks the western side of Lago de Yojoa.
Bothriechis thalassinus is sympatric with B. schlegelii, which occurs on the lower slopes of many of the same mountain ranges inhabited by B. thalassinus. On the Sierra de Caral B. thalassinus has been found as low as 885 m and B. schlegelii has been taken as high as 900 m. Some records for B. marchi from the Departamento de Santa Bárbara are imprecise (i.e., town of "Santa Bárbara") and it is possible that these specimens originated from the Cerro de Santa Bárbara, which would place them in sympatry with B. thalassinus. However, it seems equally plausible that these specimens may have been taken in the mountains to the west of Santa Bárbara where B. thalassinus is unknown.
Bothriechis thalassinus has been found while active at night. UTA R-39251 was collected at 2357 h as it crawled across a rock at the bottom of a forested gorge. At the time, it was raining and the ambient temperature was 19û C. UTA R-46526 was collected at 2300 h as it crawled among the smaller branches of a large fallen tree limb; the night was clear and the temperature was 23û C. Collecting notes associated with four individuals (UTA R-38891, 39251, 46526, LSU 23821) indicate these snakes were collected very near small, mountain steams flowing through cloud forest or secondary growth near cloud forest. UTA R-46526 and LSU 23821 were on branches directly over streams.
KEY TO SPECIES OF THE
GENUS BOTHRIECHIS
1. Series of small, usually spinelike superciliary scales present between ocular and supraocular ........... 2
No superciliary scales present.......................................................................................................... 3
2. Color pattern of small rust-colored triangular spots or vertical bars on a greenish background; or ground color
yellow to reddish brown, sometimes essentially patternless but with numerous small dots or flecks of secondary pigmentation.......................................................................................................Bothriechis schlegelii
Color pattern of middorsal markings of variable shapes (circular, rhomboidal, bands), rarely large paravertebral oval spots; ground color usually uniform ........................................................................................Bothriechis supraciliaris
3. Dorsal coloration green with many heavy black mottling; iris blackish ..................Bothriechis nigroviridis
Dorsal coloration mostly green, sometime with yellow or black dorsal or paravertebral markings; iris yellow, greenish, or reddish brown .............................................................................................................................................4
4. Scales on head (intersupraoculars forward) irregular in shape, many large and flat or with irregular and multiple keels at different angles ........................................................................................................................................... 5
Scales on top of head small, each scale with a single keel, adjacent keels parallel ................................7
5. A black postocular stripe; usually a dorsal pattern of small, yellow blotches bordered with black ... Bothriechis aurifer
Postocular stripe, if present, bluish; dorsum of adults usually uniformly green, subadults with
irregular blue or blackish blotches .................................................................................................... 6
6. Scales (excludive of supraoculars) on top of head anterior to level of posterior edge of supraoculars < 20..Bothriehcis rowleyi
Scales (excludive of supraoculars) on top of head anterior to level of posterior edge of supraoculars >.. 20 Bothriechis marchi
7. Dorsal pattern of paravertebral bars and a pale paraventral stripe ............................................Bothriechis lateralis
No pattern of paravertebral bars or paraventral stripe ........................................................................ 8
8. Interrictals 27-33 ................................................................................................Bothriechis bicolor
Interrictals 23-25 ............................................................................................. Bothriechis thalassinus
Discussion
Our understanding of New World pitvipers systematics has undergone dramatic advances recently, especially over the last decade (Campbell and Lamar 1989, 1992, Crother et al. 1992, Werman 1992, 1999, Kraus et al. 1996, Vidal et al. 1997, Gutberlet 1998, Solórzano et al. 1998, Parkinson 1999, Werman et al. 1999). A number of new species has been discovered and distinct clades of closely releated species have been identified although, in some instances, the evolutionary relationship of these lineages to other lineages remains controversial.
Although the presence of a uniquely derived heat-sensitive loreal pit provides convincing evidence for the monophyly of the crotalinae, previous efforts of deriving phylogenetic hypotheses for the group were hampered, in part, by widespread convergent evolution suggesting a close relationship between multiple Old and New World groups. More recently, DNA sequence data have suggested that New World crotalines are monophyletic (Kraus et al.1996, Vidal and Lecointre 1998, Parkinson 1999) and are the result of single invasion from the Old World into North America with subsequent dispersal southward.
Several studies have clearly indicated that the genus Bothriechis comprises a monophyletic group of species (Campbell and Lamar 1989, Crother et al. 1992, Werman 1992, 1997) whose evolution is inextricably associated with the historical geology of Middle America. Further, there is little doubt that snakes of the genus Bothriechis are relatively distantly related to the arboreal species of pitvipers inhabiting South America (Werman 1992, 1997, Kraus et al. 1996, Vidal et al. 1997, Vidal and Lecointre 1998, Parkinson 1999; however, see Schätti et al. 1990, Schätti and Kramer 1991, 1993, and Golay et al. 1993, for a contrasting view). It is also becoming increasingly apparent that most of the South American prehensile-tailed pitvipers form a clade that may be recognized as Bothriopsis (Parkinson,, 1999; but for contrasting views see Golay et al. 1993, Shätti and Kramer 1993, and Salomão et al. 1997). In contrast with Bothriechis, this clade contains members isolated in the Atlantic and Pacific lowlands of South America and on various mountain slopes, and whose evolutionary history and distributions are explained primarily by the Andean orogeny. Given the widespread distributions of pitvipers in diverse forested tropical habitats, it is not surprising that a strongly prehensile tail associated with arboreality has evolved multiple times.
Acknowledgments
This material is based in part upon work supported by a grant from the National Science Foundation (DEB-9705277) to J. A. Campbell. We thank M. E. Acevedo and C. Guirola for help in the field. We appreciate the loan of material by F. T. Burbrink (LSU) and W. E. Duellman (KU). Permits for conducting research in Guatemala were granted by officials of the Consejo Nacional de Areas Protegidas (CONAP).
Resumen
Se describe una nueva especie de víbora de foseta, verde, arborícola y de cola prensil, del género Bothriechis. Esta nueva especie se encuentra en las laderas boscosas de la vertiente Atlántica del Este de Guatemala y el Oeste de Honduras, y al parecer está cercanamente relacionada a B. bicolor, de la vertiente Pacífica de Chiapas (México) y Guatemala. Algunas otras especies de Bothriechis también habitan la vertiente Atlántica del norte de Centro América, incluyendo dos especies montanas, B. aurifer y B. marchi. Sin embargo, estas dos especies no ocurren en simpatría con la nueva especie, e incluso, habitan distintos sistemas montañosos. La especie de distribución más amplia, B. schlegelii, sí ocurre en simpatría con la nueva especie en la Sierra de Caral, a 900 m sobre el nivel del mar.
References
Bogert, C. M. 1968. A new arboreal pit viper of the genus Bothrops from the Isthmus of Tehuantepec, Mexico. Am. Mus. Novit. 2341: 1-14. [ Links ]
Campbell, J. A. & W. W. Lamar. 1989. The Venomous Reptiles of Latin America. Cornell, Ithaca. 425 pp. [ Links ]
Campbell, J. A. & W. W. Lamar. 1992. Taxonomic status of miscellaneous Neotropical viperids, with the description of a new genus. Occas. Pap. Mus. Texas Tech Univ. 153:1-31. [ Links ]
Crother, B. I., D. M. Hillis & J. A. Campbell. 1992. Biochemical and morphological studies on the phylogeny of palm-pitvipers, genus Bothriechis, pp. 1-20. In J. A. Campbell & E. D. Brodie, Jr. (eds.). Biology of the Pitvipers. Selva, Tyler, Texas. [ Links ]
Dowling, H. G. 1951. A proposed standard system of counting ventrals in snakes. Brit. J. Herpetol. 1: 97-99. [ Links ]
Golay, P., H. M. Smith, D. G. Broadley, J. R. Dixon, C. J. McCarthy, J.-C. Rage, B. Schätti & M. Toriba. 1993. Endoglyphs and other major venomous snakes of the world. A checklist. Azemiops, Geneva. 478 pp. [ Links ]
Gutberlet, R. L. Jr. 1998. The phylogenetic position of the Mexican black-tailed pitviper (Squamata: Viperidae: Crotalinae). Herpetologica 54: 184-206. [ Links ]
Gutberlet, R. L., Jr. & J. A. Campbell. 2000. Generic recognition for a neglected lineage of South American pitvipers (Squamata: Viperidae: Crotalinae) with the description of a new species from the Colombian Chocó. Am. Mus. Novit. (In press). [ Links ]
Holdridge, L. R. 1959. Mapa ecológico de Guatemala, C. A. 1:1,000,000. Instituto Interamericano de Ciencias Agrícolas de la Organización de Estados Americanos, Proyecto 39, Prog. Coop. Téc., San José, Costa Rica, in 2 sheets. [ Links ]
Klauber, L. M. 1972. Rattlesnakes: Their Habits, Life Histories, and Influence on Mankind, Second edition., 2 vols. Univ. California, Berkeley. 1533 pp. [ Links ]
Kraus, F., D. G. Mink & W. M. Brown. 1996. Crotaline intergeneric relationships based on mitochondrial DNA sequence data. Copeia 1996: 763-773. [ Links ]
Meyer, J. R. & L. D. Wilson. 1971. Taxonomic studies and notes on some Honduran amphibians and reptiles. So. California Acad. Sci. 70: 106-114. [ Links ]
Parkinson, C. L. 1999. Molecular systematics and biogeographical history of pitvipers as determined by mitochondrial ribosomal DNA sequences. Copeia 1999: 576-586. [ Links ]
Salomão, M. De G., W. Wüster, R. S. Thorpe, J.-M. Touzet, and BBBSP. 1997. DNA evolution of South American pitvipers of the genus Bothrops. Pp. 89-98 in R. S. Thorpe, W. Wüster, and A. Malhotra (eds.), Venomous Snakes: Ecology, Evolution, and Snakebite. Symp Zool. Soc. London, Clarendon, Oxford. [ Links ]
Schätti, B. & E. Kramer. 1991. A new pitviper from Ecuador, Bothriechis mahnerti n. sp. Rev. Suisse Zool. 98: 9-14. [ Links ]
Schätti, B. & E. Kramer. 1993. Ecuadorianische grubenottern der gattungen Bothriechis, Bothrops, and Porthidium (Serpentes: Viperidae). Rev. Suisse Zool. 100: 235-278. [ Links ]
Schätti, B., E. Kramer & J.-M. Touzet. 1990. Systematic remarks on a rare crotalid snake from Ecuador Bothriechis albocarinata (Shreve), with some comments on generic arrangements of Neotropical pit-vipers. Rev. Suisse Zool. 97: 877-885. [ Links ]
Solórzano, A., L. D. Gómez, J. Monge-Nájera & B. I Crother. 1998. Redescription and validation of Bothriechis supraciliaris (Serpentes: Viperidae). Rev. Biol. Trop. 46: 453-462. [ Links ]
Vidal, N. & G. Lecointre. 1998. Weighting and congruence: A case study based on three mitochondrial genes in pitvipers. Mol. Phylogen. Evol. 9: 366-374. [ Links ]
Vidal, N., G. Lecointre, J. C. Vié & J. P. Gasc. 1997. Molecular systematics of pitvipers: Paraphyly of the Bothrops complex. C. R. Acad. Sci. Paris, Science de la vie 320: 95-101. [ Links ]
Werman, S. D. 1992. Phylogenetic relationships of Central and South American pitvipers of the genus Bothrops (sensu lato): Cladistic analyses of biochemical and anatomical characters, pp. 21-40. In J. A. Campbell & E. D. Brodie, Jr. (eds.). Biology of the Pitvipers. Selva, Tyler, Texas. [ Links ]
Werman, S. D. 1997. Systematic implications of lactate dehydrogenase isozyme phenotypes in Neotropical pitvipers (Viperidae: Crotalinae), pp. 79-88. In R. S. Thorp, W. Wüster & A. Malhotra (eds.). Ecology, Evolution and Snakebite: Venomous Snakes. Clarendon, Oxford, England. [ Links ]
Werman, S. D. 1999. Molecular phylogenetics and morphological evolution in Neotropical pitvipers: An evaluation of mitochondrial DNA sequence information and the comparative morphology of the cranium and palatomaxillary arch. Kaupia 8: 113-126. [ Links ]
Werman, S. D., B. I. Crother & M. E. White. 1999. Phylogeny of some Middle American pitvipers based on a cladistic analysis of mitochondrial 12S and 16S DNA sequence information. Contemp. Herpetol. 1999 (3). [ Links ]
Wilson, L. D. 1983. Update on the list of amphibians and reptiles known from Honduras. Herpetol. Rev. 14: 125-126. [ Links ]
Wilson, L. D. & J. R. Meyer. 1985. The snakes of Honduras. Milwaukee Pub. Mus., Milwaukee, Wisconsin. 150 pp. [ Links ]
Bothriechis aurifer.–GUATEMALA: Alta Verapaz: Carchá, Caquipec, Aldea Chirrucbiquim (UTA R-37224); vicinity of Cobán (UTA R-4494); Finca El Volcán (UMMZ 91081); Baja Verapaz: 0.3 km SE by road Biotopo "Mario Dary Rivera" (UTA R-8778); 3.2 km SE by road Purulhá (UTA R-8777); 7.7 km SSE Purulhá, Plantación Santa Teresa (UTA R-6241, 6275-76, 6459, 6504-05, 6525); Biotopo "Mario Dary Rivera" (UTA R-6553); 1.6 km SE Biotopo "Mario Dary Rivera" (UTA R-7716); Cerro Quisís (UTA R-6562, 7039-45, 7763-68, 9608-09, 10434-36, 12552, 22448, 26574); Cerro Verde (UTA R-7047, 9366, 12783, 14687, 16064-65); vicinity of La Unión Barrios (UTA R-7046, 7048, 7635-36, 7762, 7788, 12949-51, 13030-31, 13614-16, 14223, 16060-61, 17906, 21842, 22439-47, 22449, 26163-64, 26388-91, 2855152, 31207, 31978, 32085-86, 32424, 34486, 35031, 36211); Niño Perdido (UTA R-39219-23, 45870-71); vicinity of Purulhá (UTA R-16062-63, 37226); Sierra de las Minas (UTA R-35600); Quiché: Finca El Soché [Soch], 40 km W Cobán (CAS 67049); Zacapa: 7.8 km NNW San Lorenzo (KU 191201).
Bothriechis bicolor.–GUATEMALA: Chimaltenango: Finca Pacayal, near Pochuta (MCZ 31941); Yepocapa (USNM 127973); Escuintla: S slope Volcán de Agua, Finca Rosario Vista Hermosa, 900-1370 m (UTA R-9353, 16066-67, 18365, 21843-46, 21848, 24758-59, 26932, 31977, 34535, 39238); Sacatepéquez: S slope Volcán de Atitlán, ca. 1200 m (UTA R-21847, 22223-24, 23060); San Marcos: Aldea Patí, cerca de San Rafael Pie de la Cuesta, ca. 1140 m (UTA R-38149); Municipio Esquipulas Palo Gordo, Aldea La Fraternidad, lado oeste, ca. 1700-1800 m (UTA R-39412); Municipio San Rafael Pied de las Cuesta, Aldea Feria, Finca America El Vergel, 1480-1600 m (UTA R-39413-22, 42273-76, 42281, 45902); Area La Trinidad, Aldea La Fraternidad, 12001940 m (UTA R-42277-80); Suchitepéquez: Olas de Mocá, near Finca La Moká (FMNH 20612); No Other Data: (UTA R-34156). MEXICO: Chiapas: "Chicharras" [probably from Cerro Chicharras, a mountain near the village of San Juan Chicharras] (USNM 46511); Cerro Ovando (UMMZ 94644).
Bothriechis lateralis.–COSTA RICA: Alajuela: Cordillera de Tilarán (UTA R-17017); La Balsa (KU 140086); 11.2 km SW Villa Quesada (KU 30961); Cartargo: Navarro (KU 35549); 3.2 km above Sant Cruz, Volcán Turrialba (KU 25163); Guanacaste: Volcán Cacao (UTA R-32455); Limón: Pico Blanco (KU 180261); Puntarenas: Monteverde (UTA R-32457); San José: Acosta, Cangrejal (UTA R-24822-24, 24831-33); Km 122 on Ruta 2, N of San Isidro de Pérez Zeledón (UTA R-12941); Patarra (UTA R-2799-800, 2811, 2845-46, 3659-62, 5132-36, 5291, 5708, 7490, 7634, 825556); near San Antonio de Escazu (UTA R-16069-71, 16350); near Tarbaca (UTA R-12784, 14536, 14537); No Other Data: (UTA R-2804, 7583, 8176, 9001, 14602, 16072-74, 16351, 25378-84, 31203-04). PANAMA: Chiriquí: Río Chiriquí Viejo (MCZ 25211); Finca Santa Clara (KU 112589); El Hato del Volcán (AMNH 75636); Quebrada Chevo, S slope Cerro La Pelota (KU 112590-95).
Bothriechis marchi.–HONDURAS: Atlántida: Tela (AMNH 46949); Cortés: Sierra de Omoa, La Cumbre (AMNH 46954-57, MCZ 32029-31); Sierra de Omoa, N San Pedro Sula (UTA R-7158-59, 8175, 8258, 8333, 8336, KU 180263); "San Pedro Sula" [probably from the Sierra de Omoa which flanks this town to the north and east] (MCZ 33334-36, 33561-64, USNM 83454); Santa Bárbara: CofradiaSanta Bárbara road (MCZ 27567-68); Quimistán (MCZ 27260); Santa Bárbara (MCZ 28014); Yoro: Montañas de los Mataderos (MCZ 38785-86); Portillo Grande (MCZ 38790-91).
Bothriechis nigroviridis.–COSTA RICA: Alajuela: Río Poasito, 1 km W Poasito (KU 63919-20); Volcán Poás (UTA R-21852); Cartago: El Empalme (UTA R-24841); Volcán Irazú (UTA R-16075, 16077-78); Heredia: Vara Blanca (UTA R-21926); Volcán Barba (AMNH 17283); Puntarenas: Monteverde (UTA R-32458); San José: Bajo La Hondura (UTA R-21853); Cascajal de Coronado (UTA R-21851); Providencia, Río Brujo (KU 128994); vicinity of San Isidro del General (UTA R-2801, 2808, 2850, 6799, 7327, 7463, 9364-65, 9635-37, 10432-33); No Other Data: (UTA R-14058, 16076, 21850, 22523). PANAMA: Bocas del Toro: N slope Cerro Pando (KU 112598); Chiriquí: Río Chiriquí Viejo (MCZ 39655).
Bothriechis rowleyi.–MEXICO: Chiapas:Hwy 195, 50.2 km N Bochil (UF 52553); Oaxaca: Cerro Baúl (UTA R-6207, 6636, 7707-09, 12565); 8 km W Cerro Baúl (AMNH 100669); W slope Cerro Baúl (AMNH 102894-95).
Bothriechis schlegelii.–COSTA RICA: Cartago: San Juan de Dios (KU 34001-02, 34004); Turrialba (KU 25162, 30953-54, 30980, 31999, 34000, 3560405; Guanacaste: Tenorio, Las Flores (KU 34003); Heredia: Puerto Viejo (KU 63921); 1.5 km N Puerto Viejo (KU 63922); Limón: 7 km W Guapiles, Río Toro Amarillo (KU 86589); near Penshurst (UTA R-6293); San Clemente (UTA R-5628); near Suretka (KU 356606); Puntarenas: Golfito (KU 31996, 31998). ECUADOR: Pichincha: Santo Domingo de los Colorados (KU 179509); Tandapi (KU 121349). GUATEMALA: Izabal: Aldea Vista Hermosa (KU 187441, 191217-24); Petén: Paso Subín (KU 58105). PANAMA: Bocas del Toro: Almirante (KU 80248-49); 3.2 km W Almirante (KU 112603-04); E half Isla Escudo de Veraguas (KU 112605-07); Darién: N slope Cerro Quia (KU 112600); NE slope Cerro Sapo (KU 112601-02); Laguna (KU 75766); Panamá: Alto de Panamá (KU 80604).
Bothriechis supraciliaris.–COSTA RICA: Puntarenas: San Vito de Coto Brus, 1200 m (UTA R-30289, 35192-93, 35240); San José: mountains near San Isidro el El General (KU 31997).
Bothriechis thalassinus.–See Holotype, Paratypes, and Referred Specimens.
Table 1.
Comparison of certain characteristics of Bothriechis bicolor and B. thalassinus.
| Characteristic | Bothriechis bicolor (N = 25 | Bothriechis thalassinus
(N = 7 | ||||||
| Intersupraoculars
|
8-11 (x = 10.0) | 9 | ||||||
| Interrictals |
27-33 (x = 30.4) | 24-26 (x = 24.7) | ||||||
| Supralabials |
9-12 (x = 10.4) | 9-10 (x = 9.6) | ||||||
| Infralabials |
10-14 (x = 11.7) | 10-12 (x = 10.9) | ||||||
| | | | ||||||
| Lacunolabial |
Present 7% | Absent | ||||||
| Ventrals Subcaudals | 157175 (x = 164.9)
156174 (x = 163.2) 58-75 (x=66.5) | 161-168 (x = 165.0) 162-165 (x = 163.0) 61-67 (x = 63.6) | ||||||
Table 2
Ranking of character means for males and female Bothriechis bicolor and B. thalassinus.
Ranking is from smallest to largest (one tie recorded for number of infralabials)
| Species/sex | (residuals) | interrictals | supralabials | infralabials | (residuals) | subcaudals |
| B. bicolor | ||||||
|
4 | 4 | 3 | 2 | 2 | 1 | |
| B. thalassinus | ||||||
|
3 | 2 | 4 | 3 | 1 | 2 |
1 Department of Biology, The University of Texas at Arlington, Arlington, TX 76010, USA. Fax 817-272-2406. e-mail (JAC) campbell@uta.edu, (ENS) e.smith@uta.edu