Introduction
Crustaceans are economically important, being the aim of many fisheries, and also ecologically important, occupying the role of transferring nutrients to the higher trophic levels (Verdi, 1995). Caridean crustaceans, including the families Atyidae and Palaemonidae, are crucial components of limnological ecosystems located in tropical and subtropical regions (De Grave & Fransen, 2011).
The family Atyidae, represented near exclusively by freshwater prawns, is characterized by the presence of unique feeding chelipeds, which count on complex structures on the first and second pair of pereiopods. These structures are used for capturing suspended matter (de Grave, Cai, & Anker, 2008). This family is represented by four species in Brazil, two of the Potimirim (Holthuis, 1954) genus and two of the Atya Leach, 1815 genera (Melo, 2003). This family is commonly associated with rocky substrates and under vegetation for covering (Hobbs & Hart, 1982).
The genus Atya is known for its specific habitat requirements, which consists of shallow oxygen-rich water (Hobbs & Hart, 1982). The species Atya scabra (Leach, 1815),along with many species of the family Palaemonidae, isfrequently associated with fisheries in many regions in Brazil, like Southern Bahia, North of Brazil (Almeida, Coelho, Luz, Santos, & Ferraz, 2008; Almeida, Mossolin, & Luz, 2010) and around the neotropical region (Martínez-Mayén, Román-Contreras, Rocha-Ramírez, & Chazaro-Olvera,2000; Lorán-Núñez, Valdéz-Guzmán, & Martínez-Isunza, 2009), having its occurrence also reported in Puerto Rico, Panama and the dutch island of Curaçao, located in the Caribbean (Covich, 1988; Torati, Page, De Grave, & Anker, 2011; Debrot, 2003). It has documented preference for high speed streams, which is linked with their capacity to resist the currents and foraging strategy (de Grave et al., 2008; Girard et al., 2014). This species is considered vulnerable in 10 Brazilian states, including the state of Bahia (Amaral, 2005).
Knowledge of allometric growth patterns is essential to determine sexual dimorphism in shrimps by comparison of growth parameters (Hartnoll, 1974). Also, it is an important tool when analyzing intraspecific variation among different populations (Costa & Soares-Gomes, 2008). Data on relative growth can also be used as a diagnostic device to identify evolutionary relationships between species (Rahman & Ohtomi, 2018).
Studies that deal with population structures and reproductive biology are important in the sphere of fishery management and conservation, being crucial to determine conservation status in the studied environment and tracing management strategies (Almeida et al., 2010). Thus, this study aimed to determine the population structure of the prawn Atya scabra in the Contas River, Bahia, Brazil, as well as its reproductive period, patterns of allometric growth, sexual dimorphism and size classes distribution among the sexes, helping to establish a minimum capture size in fisheries along the region.
Materials and methods
Study site: The Contas River is located in southern Bahia, Brazil (Fig. 1). The water depth rarely exceeds 1 m and the sediment is composed mostly of rocks, but with a few points of sand. The marginal vegetation presents a variable composition of macrophytes.
Field work: Shrimps were sampled between January and December 2009, by using drag nets for 20 minutes and passing sieves along the marginal vegetation, after removing rocks from the substrate, during 1 h in each sampling site. This type of sampling method has been utilized in other studies (Almeida et al., 2010; Galvão & Bueno, 2000). We used a mercury thermometer to measure temperature twice in each sampling point and then means were calculated. The organisms were collected in containers with 70 % alcohol and tagged.
Laboratory: In the laboratory, we determined the sex of the individuals by checking the presence (for males) or absence (for females) of an appendix in the second pair of pleopods. Individuals were also measured using a pachymeter with 0.001 mm precision. The following dimensions were measured: carapace width (CW), as the distance between the medium points at the side of the carapace (mm); carapace length (CL), as the distance between the post-orbital margin to the posterior margin of the carapace (mm); and total length (TL), as the distance from the post-orbital margin to the beginning of the telson. TL was preferred for most analyses.
Data analysis: Linear models (y = ax + b) for the CL × CW relationships were constructed. When b > 1, positive allometry is registered. When b < 1, negative allometry is recorded and when b = 1, the relationship is isometric. CW (carapace width) was considered the independent variable and CL (carapace length) was considered the dependent variable. Individuals of both sexes were grouped into size classes of 5 mm for TL. A chi-square test (χ2) was used to check if there was a significant deviation from the 1:1 normal sex ratio, which was considered as the number of males divided by the number of females. The presence of ovigerous females was computed for each month, considering as breeding period the months in which it was detected. Mean carapace length (CL) of males and females was compared by means of the Mann-Whitney U test. An analysis of covariance (ANCOVA) was utilized in order to test whether there were differences in the equation’s parameters between males and females, using sex as a covariant. A t-test was used to verify whether the equation’s slope was different from 1 (H0: b = 1). The Spearman’s correlation test was used to verify the existence of any correlation between temperature variation and abundance of ovigerous females throughout the year. The level of confidence for rejecting the null hypothesis was considered to be below 5 %. All data was tested for normality and homogeneity of variance prior to any test by means of the Shapiro-Wilk test and Levene’s test, respectively. We used Prism version 8.0.1 (GraphPad, CA, USA) and R version 3.6.3 (R Core Team, 2020) for graphing and analysis.
Results
Abiotic factors: Temperatures in the river varied throughout the year (Table 1). The highest temperature registered was 27.6 oC in December, while the lowest value (25.3 oC) was registered in June.
TABLE 1 Means of temperature readings by month in 2009
| Month | Temperature (oC) |
| Jan | 27.1 |
| Feb | 26.7 |
| Mar | 26.8 |
| Apr | 27 |
| May | 26.4 |
| Jun | 25.3 |
| Jul | 25.5 |
| Aug | 25.8 |
| Set | 26.4 |
| Oct | 26.9 |
| Nov | 27.5 |
| Dec | 27.6 |
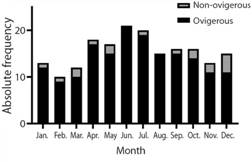
Population structure: In total, 517 individuals were collected, representing 328 males and 186 females (169 ovigerous females). The smallest female was ovigerous, allowing us to assume that all the individuals sampled were fully developed (Table 2). There was a significant deviation from the 1:1 normal sex ratio towards more males in all but three months (Table 3).
TABLE 2 Minimum, maximum, mean and standard deviation (SD) of the carapace length (CL) and total length (TL) for males and females
| Sex | TL (mm) | CL (mm) | ||||||
| Min. | Max. | Mean | SD | Min. | Max. | Mean | SD | |
| Males | 37.45 | 96.60 | 73.04 | 10.829 | 13.60 | 47.50 | 31.43 | 5.222 |
| Fem. | 38.00 | 87.40 | 57.65 | 8.648 | 13.00 | 39.20 | 22.69 | 3.804 |
TABLE 3 Number of males and females in each month and sex ratio (males: females)
| Month | Number of males | Number of females | Sexual proportion | χ2 test |
| Jan | 30 | 13 | 2.5:1 | P < 0.05 |
| Feb | 30 | 10 | 3:1 | P < 0.05 |
| Mar | 28 | 12 | 2.33:1 | P < 0.05 |
| Apr | 31 | 18 | 1.72:1 | P < 0.05 |
| May | 25 | 17 | 1.47:1 | P < 0.05 |
| Jun | 24 | 21 | 1.14:1 | NS |
| Jul | 23 | 20 | 1.15:1 | NS |
| Aug | 24 | 15 | 1.14:1 | NS |
| Sep | 30 | 16 | 1.87:1 | P < 0.05 |
| Oct | 30 | 16 | 1.87:1 | P < 0.05 |
| Nov | 25 | 13 | 1.92:1 | P < 0.05 |
| Dec | 28 | 15 | 1.86:1 | P < 0.05 |
| Total | 329 | 186 | 1.76:1 | P < 0.05 |
NS = Non-significant.
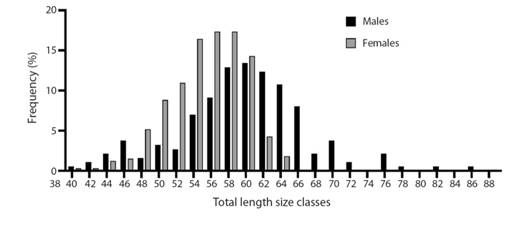
The males were dominant in the size classes 25 - 29.9, 30 - 34.9 and 35 - 39.9 mm, while females showed highest frequency in the 20 - 24.9 mm size class (Fig. 2). Patterns were unimodal for females and polymodal for males. The non-parametric Mann-Whitney U test demonstrated that there are differences between the sexes in TL (P < 0.001) (Fig. 3).
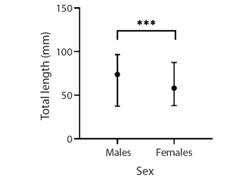
Fig. 3 Medians and ranges of total length (TL) for males and females. Asterisks (***) denote significance level (P < 0.001).
The allometry relationships (Fig. 4) demonstrated to be negative (b < 1) in males and females (t-test, P < 0.05), which means that the carapace length increases in a higher rate than the carapace width. The ANCOVA analysis demonstrated that the slope (b) and intercept (a) were significantly different among males and females (P < 0.01), denoting that the patterns of growth are different.
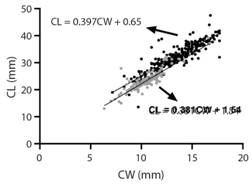
Fig. 4 Relationshipbetween carapace length (CL) and carapace width (CW) for males (black) and females (gray).
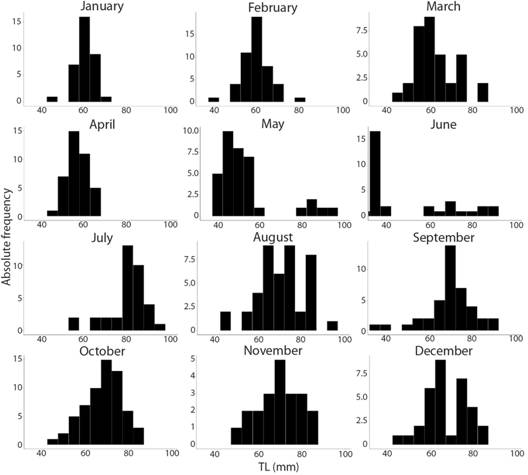
Reproduction: According to the proportion of ovigerous females per month (Fig. 5), it is possible to note that the reproductive season is continuous. The Spearman’s correlation test found a significant positive correlation between the monthly abundance of females with eggs and the temperature in each month (P < 0.05). Recruitment was also observed to be continuous. However, a major peak was observed in May (Fig. 6).
Discussion
The carapace length for males and females were similar to the observed in other studies, with males also being generally larger than females. Almeida et al.(2010) observed that males ranged 5 - 38.18 mm and females 5.4 - 23.06 mm of carapace length while Herrera-Correal, Mossolin, Wehrtmann and Mantelatto (2013) sampled females that ranged 13.5 - 25.1 mm (data only obtained for females). Galvão and Bueno (2000) also found out that males are able to achieve higher sizes when compared to females.
Almeida et al.(2010) also collected ovigerous females of Atya scabra throughout the entire year, characterizing the reproduction as continuous. The breeding period in the Atyidae family demonstrated to be continuous in many studies (Benzie & De Silva, 1988; De Silva, 1990; Idrisi & Salman, 2005; Leuven, Van Der Brink, & Van Der Velde, 2008). It is possible that the rainy season is able to influence peaks (Hoarau, 2018; Hoffman & Negreiros-Fransozo, 2010). Galvão and Bueno (2000) found out higher abundance of ovigerous females of A. scabra in the months that registrated the higher temperatures, while Lopes, Frédou, Silva, Calazans and Peixoto (2017) reported positive correlation between the percentage of mature females and temperature in the species Xiphopenaeus kroyeri Heller 1862, suggesting that this abiotic factor does influence reproductive peaks in caridean prawns. In this study, a different pattern was observed, which consisted in a higher abundance of ovigerous females in the months with lower temperatures. A correlation with the rainfall is possible, probably caused by the increase in the amount of nutrients in the rainy season. Similar patterns of reproductive period were also observed in other caridean shrimps (Mossolin & Bueno, 2002; Terossi & Mantelatto, 2010), suggesting that these patterns are common for this group of crustaceans. The fact that the smallest female (13.00 mm of carapace length) was ovigerous is an indicative that much probably the juveniles in this specific population are below this size.
The different patterns of relative growth observed among the sexes emphasizes that the males, during development, invest energy in the development of structure related to sexual behavior, while females invest their energy in developing its gonads, explaining the fact of females being predominant in lower size classes (Correa & Thiel, 2003a). It is a concern that caridean shrimps show sexual guarding behavior, which consists in the male “protecting” the female with the chelipeds (Gualberto, Almeida & Menin, 2012). It is clear that this pattern is shown by the studied species. Lorán-Núñez et al.(2009) also found higher size and abundance in males of two rivers in México, a pattern also shown by the population analyzed in this study.
The sex ratio favorable to males almost the entire year can be explained as a result of different configurations of migration and mortality between the sexes, which can be explained as relations between age at sexual maturity and its relationship with the life expectancy (Rondeau & Saint-Marie, 2001; Siegel, Damm, & Neudecker, 2008). Sex ratio can also be an important factor when it comes to determining parameters of sexual selection, like direction and intensity (Correa & Thiel, 2003b). This result was also found for Atya scabra by other authors (Galvão & Bueno, 2000; Lorán-Núñes et al., 2009; Almeida et al., 2010).
The data exposed in this article is essential to expand the knowledge about the freshwater crustaceans that live alongside rivers in Northeast Brazil, contributing to draw sustainable fishery managing strategies by the local government, in order to ensure that the population continues to be stable. The correlation between variation of temperature and the abundance of ovigerous females was clear. Although, it is possible that variation in rainfall may also affect the frequency of ovigerous females, caused by an increase in nutrient availability. We suggest that restrictions in the months of May and June must be implemented. This is based on the observations that recruitment reaches its peaks in these periods. These measures can be valuable when it comes to conservation of the fishing stock. The sexual dimorphism present in the species was reinforced by our data, being highly similar to other studies with the animal. The male-biased sex ratio can possibly indicate clues for sexual selection occurring in the species and is a common pattern in Atyid prawns, something that deserves investigation.
Ethical statement: authors declare that they all agree with this publication and made significant contributions; that there is no conflict of interest of any kind; and that we followed all pertinent ethical and legal procedures and requirements. All financial sources are fully and clearly stated in the acknowledgements section. A signed document has been filed in the journal archives.












 uBio
uBio