The Myrtaceae is a pantropical family that comprises 17 tribes, about 144 genera, and over 5500 species (Wilson, 2011; Vasconcellos et al., 2017) distributed through southern regions of the world (with a few representatives in Africa). This family is composed mainly of shrubs and trees with most genera occurring in Australia and tropical and subtropical America. One of the characteristics of this family is the presence of oil glands that produce essential oils, mainly constituted by terpenoids.
Myrcianthes O. Berg is a genus composed of 39 recognized species ranging from southern Florida and Mexico to Bolivia and northern Argentina, Uruguay and north-central Chile and the Caribbean (McVaugh, 1963; Tucker et al., 1992; Tucker et al., 2002, World Flora Online (WFO), 2023). Myrcianthes storkii (Standl.) McVaugh (Synonyms: Eugenia rigidissima Cufod.; E. storkii Standl.; Myrcianthes rigidissima (Cufod.) W.D. Stevens) is a native shrub or tree of about 4 to 30m tall, with a distributional range from Costa Rica and western Panama. In Costa Rica, it is distributed in wet to very rainy, cloud, and oak forests, from 1300 to 3150m of elevation and it is known vernacularly as ‘guayabillo’ (Barrie, 2007). These forests can be found on mountain slopes, varying in the intensity of rainfall. The leaves are elliptic or obovate to broadly elliptic or broadly obovate, coriaceous, and glabrous on both sides. When the leaves are crushed, they give off a scent with aromatic flavor. Young twigs are coarsely sericeous.
Many studies on the chemical composition of essential oils of diverse species of Myrcianthes
have been reported. Some of these studies are summarized in Table 1 in Appendix. The species and the morphological part from which the studied essential oil was isolated, the location, and the major compounds that constitute the oils are indicated. In general terms, the studied oils are constituted mainly of terpenes and terpenoids.
There is no information about possible traditional uses of M. storkii.
To the best of our knowledge, no previous reports on the chemical composition of essential oils of this species have been published.
Materials and methods
Plant materials: We collected leaves, floral buds, and twigs of Myrcianthes storkii from a single tree in the locality of Pacayas de Alvarado, Province of Cartago, Costa Rica (09°55'03"N 83°48'29"W, at an elevation of 1 700m). A voucher specimen is Luis J. Poveda Álvarez 4915 (F).
Extraction of essential oils: We isolated the oils from fresh plant material by hydrodistillation at atmospheric pressure, for 3 h using a Clevenger-type apparatus. The distilled oils were collected and dried over anhydrous sodium sulfate, filtered, and stored between 0°C and 10°C in the dark, until further analysis. The essential oil yields (v/w) were 0,05% (leaves), 0,09% (floral buds), and (0,01% twigs).
Gas chromatographic analyses (GC-FID): We analyzed the essential oils of M. storkii by capillary gas chromatography with a flame ionization detector (GC/FID) using a Shimadzu GC-2014 gas chromatograph. Data have been collected on a poly (5% diphenyl/95% dimethylsiloxane) fused silica capillary column (30m x 0,25mm; film thickness 0,25μm), (MDN-5S, Supelco). The GC integrations were performed with LabSolutions, Shimadzu GCsolution™ Chromatography Data System software, version 2.3. Operating conditions used were carrier gas N2, flow 1,0mL/min; oven temperature program: 60 to 280°C at 3°C/min, 280°C (2 min); sample injection port temperature 250°C; detector temperature 280°C; split 1:60.
Gas chromatography-mass spectrometry (GC-MS): GC-MS analyses were performed with a Shimadzu GC-2010 Plus gas chromatograph coupled with a GCMS-QP2010 SE apparatus and with GCMSsolution™ software (version 4.20), with NIST and Wiley 139 computerized databases. The analyzes were performed with two fused-silica-capillary columns with stationary phases of different polarities: 1,4-bis(dimethylsiloxy) phenylene dimethylpolysiloxane and polyethylene glycol. The data were obtained with a non-polar SLB™-5ms (Supelco) fused silica column (30m x 0,25mm; film thickness 0,25μm). Operating conditions were: carrier gas He, flow 1,4 mL min-1 with constant pressure; oven temperature was programmed linearly from 60°C to 280°C at 3°C min-1; sample injection port temperature 250°C; interface temperature 260°C; ionization voltage: 70 eV; ionization current 60μA; scanning speed 0,30s over 35 to 400 amu range; split 1:70. Also, the data were obtained with a second polar Supelcowax™10 (Supelco) fused silica column (30m x 0,25mm; film thickness 0,2 μm). Operating conditions were carrier gas He, flow 1,4mL min-1; oven temperature program: 60-220°C at 3°C min-1; sample injection port temperature 240°C; transfer line temperature 230°C; ionization voltage: 70 eV; ionization current 60 μA; scanning speed 0,30s over acquisition mass range, 35 to 400 amu; split 1:70.
Compound identification: We identified the essential oil constituents by comparison of their linear retention indices which were calculated in relation to a homologous series of n-alkanes, on a poly (5% diphenyl/95% dimethylsiloxane) type column (van den Dool & Kratz, 1963) and on polyethylene glycol capillary column and, by comparison of their mass spectra with those published in the literature (Adams, 2007), or those of our own homemade MS library, or comparing their mass spectra with those available in the computerized databases (NIST 107 and Wiley 139) or in a web source (Wallace, 2021). To obtain the retention indices for each peak, 0,1 μL of an n-alkane mixture (Sigma, C8-C32 standard mixture) was co-injected under the same experimental conditions reported above. Integration of the total chromatogram (GC/FID), expressed as area percent, without correction factors, has been used to obtain quantitative compositional data.
Results
The essential oils from different parts of Myrcianthes storkii presented a complex mixture of compounds. The constituents identified, their experimental retention indices on two columns of diverse polarity, their relative percentage concentrations, and the methods used for their identification are presented in Table 2 in Appendix. The constituents are listed in order of elution on a non-polar poly-(5% phenyl 95% dimethylsiloxane) type column and for comparison purposes, previously published values of the retention indices are included (Adams, 2007; Wallace, 2021).
Myrcianthes storkii gave essential oils that were predominantly terpenoid in nature. The leaf and floral bud oils were dominated by monoterpenoids (42,66% and 38,63%, respectively) and sesquiterpenoids (44,67% and 44,69%, respectively), whereas twig oil was dominated by sesquiterpenoids (46,45%) and aliphatic compounds (18,77%). From the hydrodistilled oils, a total of 281 compounds were identified using GC/FID and GC/MS, accounting for 91,27% (leaves), 86,65% (floral buds), and 74,56% (twigs) of the total composition of the essential oils.
The leaf essential oil consisted largely of monoterpene hydrocarbons (36,98%) and sesquiterpene hydrocarbons (34,06%) with minor amounts of oxygenated derivatives. The main constituents were myrcene (17,44%), cis-calamenene (12,60%), α-pinene (5,48%), (E)-caryophyllene
(5,16%), limonene (3,91%), p-cymene (3,71%), 1,8-cineole (2,80%), α-humulene (2,80%), cubenol (2,45%), α-copaene (2,22%), α-cubebene (2,10%), linalool (2,05%), (E)-caryophyllene oxide (2,04%), and β-phellandrene (2,00%). See the total ion chromatogram (TIC) in Figura 1. The chemical structures of some of these compounds are shown in Figura. 3.
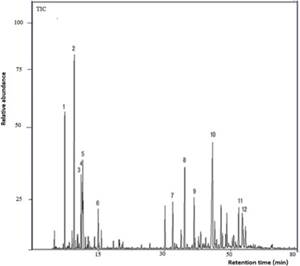
Figura. 1 GC-MS chromatogram (TIC) of Myrcianthes storkii leaf essential oil (1. α-pinene; 2. myrcene; 3. p- cymene; 4. limonene; 5. 1,8-cineole; 6. linalool; 7. α-copaene; 8. (E)-caryophyllene; 9. α-humulene; 10. cis-calamenene; 11. 1-epi-cubenol; 12. cubenol).
The composition of the floral bud essential oil also was dominated by sesquiterpene hydrocarbons (32,36%), and monoterpene hydrocarbons (30,75%) with α-pinene (15,23%), cis- calamenene (12,70%), myrcene (8,59%), 1,8-cineole (4,26%), germacrene B (3,65%), α-humulene
(3,55%), (E)-caryophyllene oxide (2,93%), α-copaene (2,24%), hinesol (2,16%), and α-cubebene (2,14%) as main constituents.
The twig essential oil was constituted mainly of sesquiterpenoids (46,45%) and aliphatic compounds (18,77%), with a minor quantity of monoterpenoids (6,84%). The major compounds found were cis-calamenene (11,31%), hexadecanoic acid (7,99%), (E)-caryophyllene (4,68%), 𝛿- cadinene (3,28%), cubenol (3,24%), (Z)-caryophyllene oxide (2,94%), 1-epi-cubenol (2,45%), α- humulene (2,38%), and α-copaene (2,19%). The aliphatic mixture of compounds was constituted of acids (palmitic acid as the main compound), aldehydes, alcohols, esters, and hydrocarbons. See the total ion chromatogram (TIC) in Figura 2.
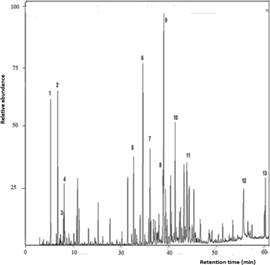
Figura. 2 GC-MS chromatogram (TIC) of Myrcianthes storkii twig essential oil (1. α-pinene; 2. myrcene; 3. p- cymene; 4. limonene; 5. α-copaene; 6. (E)-caryophyllene; 7. α-humulene; 8. δ-cadinene; 9. cis-calamenene; 10. (E)-caryophyllene oxide; 11. cubenol; 12. palmitic acid; 13. (E)-phytol).
Discussion
Analyzing the data in Table 1, the chemical composition of essential oils obtained from leaves of Myrcianthes is very varied. However, there seem to be some common, widespread compounds, such as the monoterpenes α-pinene, β-pinene, p-cymene, and limonene; the sesquiterpenes (E)-caryophyllene and α-humulene, and the terpenoids 1,8-cineole, linalool, terpinen-4-ol, α-terpineol, and (E)-caryophyllene oxide. Some of them are ubiquitous natural products that display ecological roles such as assisting in pollinator attraction, deterrent action against certain herbivores, and antimicrobial or allelopathic activities (Anaya et al., 2001; Gershenzon & Dudareva, 2007; Yazaki et al., 2017; Boncan et al., 2020; Escobar-Bravo et al., 2023). Observing the data provided in Table 1, differences are found in the composition of the essential oils of samples of the same species that grow in different places. The oils of M. fragrans from Jamaica (Tucker et al., 1992) and Cuba (Pino et al., 2000) were rich in limonene, α-pinene, α-terpineol, and 1,8-cineole, whereas the essential oil from Venezuela (Mora et al., 2009) was rich in myrcene, β-caryophyllene, and other sesquiterpenoids. The oil from Ecuador (Armijos et al., 2018) differed from all the other samples and species in that it contained large amounts of geranial and neral. The two Costa Rican samples of this species gave oils with the unique fact of presenting as main compounds the benzenoid 1,3,5-trimethoxybenzene and (E)-methyl isoeugenol (Cole et al., 2008) and the phenylpropanoid ester, methyl (E)-cinnamate (Chaverri & Cicció, 2017).
The essential oil of M. storkii leaves is mainly constituted of terpenoids (87,75%) and small amounts of aliphatic compounds (2,55%) and benzenoids (0,91%). This oil is characterized by the dominant compounds myrcene (17,44%) and cis-calamenene (12,60%). In the studies conducted to date, only the oils of M. rhopaloides and M. leucoxyla from Colombia (Silva et al., 2016; Quijano- Célis et al., 2016), and M. fragrans from Venezuela (Mora et al., 2009) presented myrcene in significant quantities (17,7%, 17,4% and 8,9% respectively). Myrcene possesses sedative and anxiolytic properties (Rao et al., 1990), anti-inflammatory (Rufino et al., 2015), as well as antioxidant and cytoprotective properties (Xanthis et al., 2021); it also has anti-aging properties (Surendran et al., 2021) and anti-invasive activity on a human breast cancer epithelial cell line, MDA-MB-231 (Lee et al., 2015). This compound is a valuable renewable material for the industrially sustainable synthesis of many fine chemical products, which have high added value and are used in multiple applications (Behr & Johnen, 2009).
cis-Calamenene appears to be a distinctive compound in the essential oils of M. storkii from Costa Rica, accompanied by a large amount of myrcene. Of the studied species, only M. rhopaloides from Costa Rica (Cole et al., 2008) and M. myrsinoides from Ecuador (Montalván et al., 2018) presented significant amounts of the diastereoisomer, trans-calamenene (2,5% and 15,9% respectively). The cis-calamenene, an aromatic cadinene, is a major constituent (2,1-9,1%) of the essential oil of Cupressus bakeri Jeps. (Cupressaceae) foliage (Rafii et al., 1992; Kim et al., 1994) and is present in the commercial Baccharis dracunculifolia DC. essential oil (1,0%) (Weyerstahl et al., 1996). Also, this compound was identified in cuticular waxes of the stingless bees Nannotrigona testaceicornis and Plebeia droryana (Pianaro et al., 2009).
In summary, we have shown, for the first time, the chemical composition of Myrcianthes storkii essential oil from different morphological parts (leaves, flower buds, and twigs). The presence of a high amount of myrcene and cis-calamenene in the essential oils seems to be characteristic of this species.
Ethical, conflict of interest and financial statements
The authors declare that they have fully complied with all pertinent ethical and legal requirements, both during the study and in the production of the manuscript; that there are no conflicts of interest of any kind; that all financial sources are fully and clearly stated in the Acknowledgements section; and that they fully agree with the final edited version of the article. A signed document has been filed in the journal archives.












 uBio
uBio 



