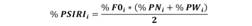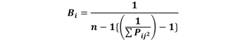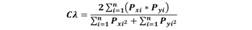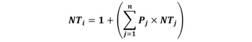Introduction
Black skipjack, Euthynnus lineatus, and Pacific sierra, Scomberomorus sierra (Family Scombridae), are fish that inhabit the pelagic-oceanic zone. They are distributed in the tropical and subtropical waters of the Tropical Eastern Pacificfrom California, USA, to Peru (Collette & Nauen, 1983). They frequently form multispecific schools and are considered highly active fish that carry out seasonal migrations associated with feeding and reproduction (Collette& Nauen, 1983).
Scombrids are of great economic importance and constitute the foundation of small-scale fisheries in several regions worldwide (Fischer et al., 1995). Although the scombrid fishery is generally more focused on tunas, skipjacks are very abundant, and due to their low price in local markets, they represent an accessible resource for the population. Due to its nutritional value, sierra is one of the most attractive species for consumers on the Mexican coast (Montemayor-López & Cisneros-Mata,2000).
According to previous trophic biology studies, scombrids are widely known as generalist opportunistic predators. That is, their niche width is composed of a wide variety of organisms at different levels on the food chain (Olson et al., 2016). Scombrids, along with dolphins, billfishes, sharks and other species, are found at the highest trophic levels, moving freely between habitats and transferring energy between food webs (Ménard, Labrune, Shin, Asine, & Bard, 2006). When populations of top predators decrease can cause a cascade effect on the ecosystem, defined as a reciprocal effect between predator-prey that alters the abundance, biomass or productivity of a community or trophic level in more than one link in the trophic chain (Pace, Cole, Carpenter, & Kitchell, 1999).
In general, feeding habits of fish represent an integration of many ecological components that include behaviour, condition, habitat use, niche overlap, energy intake, intraspecific and interspecific interactions. Thus, diet composition information of high-level predators is particularly important to understand the functional role of this species and to elaborate ecosystem models that consequently provides information that contributes to the management of fishing resources (Hyslop, 1980; Christensen & Pauly, 1992; Caragitsou & Papaconstantinou, 1993).
In the present study we characterize the diet of Black skipjack, Euthynnus lineatus, and Pacific sierra, Scomberomorus sierra, from a poorly studied area, Bahía de Acapulco. The specific objectives were i) to determine the niche width, ii) the trophic level, and iii) the trophic overlap between the two species, by size, that is juveniles and adults, and season, dry and wet.
Materials and methods
Study area: Bahía deAcapulco is located at (16º52’ - 16º41’ N & 99º29’ - 100º11’ W) in Guerrero, Mexico, in the Eastern Tropical Pacific.The climate in this area is characterized by a wet season extending from May to October and a dry season extending from November to April (Gutiérrez-Zabala & Cabrera-Mancilla 2012). Some of the important characteristics of Bahía de Acapulco is its proximity to two coastal lagoons (Laguna de Coyuca and Laguna de Tres Palos), the flow of several streams to the bay, the different types of substrate found within the bay (sandy, with patches of coral reefs, rocky reefs, islets and seamounts, resulting in a highly heterogeneous system (Palacios-Salgado et al. 2014).
Sampling: Biological samples were obtained weekly, according to the species availability in the catches made by the small-scale fishery in Bahía de Acapulco. The E. lineatus sampling period occurred from October 2016 to October 2017, whereas S. sierra was sampled from October 2016 to June 2018. The total length (TL) and total weight (TW) of all organisms were obtained. Stomachs were extracted and frozen for later analysis.
Laboratory work: Stomach contents were separated by prey type; each type was counted, weighed, and identified to the lowest possible taxonomic level using specialized keys according to the prey digestion state. Fish at an initial state of digestion were identified using the keys by Allen and Robertson (1994) and Fischer et al. (1995). Fish at an advanced state of digestion were identified based on vertebral characteristics, using keys by Clothier (1950).
Data analysis: Samples were classified as juveniles or adults, based on the work by Schaefer (1987), who reported that the size at first maturity of E. lineatus was 45.7 cm TL (total length), and on the results by Aguirre-Villaseñor, Morales-Bojórquez, Morán-Angulo, Madrid-Vera, & Valdez-Pineda (2006), who indicated that the size at first maturity of S. sierra was 44.3 cm FL (fork length). Samples were divided into two characteristic seasons of the area: dry (November to April) and wet (May to October). Sex was not included due to the absence of mature gonads in the collected specimens.
Sample size: Prey accumulation curves were constructed to determine whether sample sizes were representative of E. lineatus and S. sierra diets. Curves were created using the Vegan package (Oksanen, 2016) in R (R Core Team, 2015), which consists in randomly sampling the species richness found in the stomachs of the predators and graphing the average accumulated prey according to the number of analyzed stomachs. A linear regression of the last four points of the curve was performed to verify whether the best adjusted slope was significantly different from a zero slope line; this indicated that the curve had reached the asymptote (Bizzarro, Robison, Rinewalt, & Ebert, 2007).
Diet composition: The Frequency of Occurrence (% FO = number of stomachs containing prey i / total number of stomachs containing food * 100), the Numeric Method (% N = number of prey i / total number of prey * 100), and the Gravimetric Method (% W = weight of prey i / total weight of prey * 100), were used to analyzed the diet composition (Hyslop, 1980). The Prey-specific Index of Relative Importance (% PSIRI) (Brown, Bizzarro, Cailliet, & Ebert, 2011) was used to evaluate in an integrated manner the importance of each prey item in the diet of the predator. This index was calculated in the following manner:
Where % FOi is the Frequency of Occurrence and % PNi and % PWi are the indices of Prey-specific Abundance (% PA = ∑ % Aij / ni), where % Aij is the abundance (expressed in number or weight) of the prey i category in the jth stomach, ni is the number of stomachs containing prey i, and n is the total number of stomachs.
Niche breadth: Levin´s index (Bi) (Krebs, 1999) was used as a measure of niche width. This index evaluates quantitatively whether organisms are generalists (varied food sources without selection) or specialists (preference for certain prey). It was calculated as follows:
Where Pij is the proportion of the diet predator i that includes prey j, and n is the number of prey categories. The predator is considered a specialist if Bi is close to zero and a generalist if Bi is close to one.
Diet overlap by size and season: The EcosimR package for niche superposition (Gotelli, Hart, & Ellison, 2014) was used to evaluate diet overlap by size (juvenile or adult) and season (dry or wet), as well as between the two predators. The package was modified by using the Morisita-Horn index (Horn, 1966) instead of the Pinkas index due to Morisita’s measure of overlap gives the most accurate results (Smith & Zaret, 1982). The Morisita-Horn index values range from zero, when diets are completely different, to one, when diets are identical. It was calculated using the following equation:
Where Pxi is the proportion of prey i from the total number of prey consumed by predator x; Pyi is the proportion of prey i from the total number of prey consumed by predator y; and n is the total number of prey. To assess whether differences in diet were the result of biological processes and not random, we compared a null distribution with the overlap index values. A total of 1 000 simulations of the index were performed using the RA3 randomization algorithm in EcosimR (Gotelli, Hart, & Ellison, 2014).
Trophic level: The trophic level (TL) of predators was calculated using the method proposed by Cortés (1999), calculated as follows:
Where Pj is the proportion of each prey category in the diet of the predator and NTj is the trophic level of each prey category j. The trophic levels of prey were obtained from the database of the Sea Around Us project (http://www.seaaroundus.org/data/#/marine-trophic-index).
Results
The size of E. lineatus specimens ranged between 33.8 and 59 cm TL, the average length was 45.6 cm TL and standard deviation was 6.3 cm TL. While, S. sierra specimens measured between 34 and 65.5 cm TL, the average length was 51.2 cm TL and standard deviation was 10.1 cm TL.
Diet composition: A total of 262 E. lineatus stomachs were analyzed (42 % were empty and 58 % contained food) and a total of 209 S. sierra stomachs were analyzed (74.6 % were empty and 25.3 % contained food) (Table 1).
TABLE 1 Stomach contents analysis of the black skipjack Euthynnus lineatus and the Pacific sierra Scomberomorus sierra in the Tropical Eastern Pacific
| Euthynnus lineatus | Scomberomorus sierra | ||||||
| Stomachs with food | Empty stomachs | N | Stomachs with food | Empty stomachs | N | ||
| Oct-2016 | 1 | 2 | 3 | 1 | 2 | 3 | |
| Nov-2016 | 5 | 1 | 6 | 3 | 1 | 4 | |
| Jan-2017 | 26 | 13 | 39 | 3 | 4 | 7 | |
| Feb-2017 | 20 | 24 | 44 | 3 | 34 | 37 | |
| Mar-2017 | 13 | 6 | 19 | 20 | 27 | 47 | |
| Apr-2017 | 33 | 2 | 35 | 4 | 2 | 6 | |
| May-2017 | 3 | 1 | 4 | 7 | 36 | 43 | |
| Jun-2017 | 5 | 6 | 11 | ||||
| Jul-2017 | 8 | 21 | 29 | 2 | 1 | 3 | |
| Aug-2017 | 3 | 8 | 11 | ||||
| Sep-2017 | 1 | 3 | 4 | ||||
| Oct-2017 | 34 | 23 | 57 | 2 | 2 | ||
| Jan-2018 | 3 | 17 | 20 | ||||
| Feb-2018 | 3 | 3 | |||||
| Apr-2018 | 2 | 31 | 33 | ||||
| Jun-2018 | 1 | 1 | |||||
| TOTAL | 152 | 110 | 262 | 53 | 156 | 209 | |
The prey accumulation curve of E. lineatus became asymptotic at stomach number 89, with 94.3 % of the items that theoretically are expected to find, this indicates that the number of stomachs analyzed was sufficient to represent the diet (P > 0.05). The prey accumulation curve of S. sierra became asymptotic at stomach number 28, with 83.1 % of the items that theoretically are expected to find, however, the analyzed sample size was close to being sufficient for an adequate diet description (P < 0.05). The overlap value obtained between the two species was low, which indicated that the diets were different (Cλ = 0.2).
Diet variation by size: Of the total number of E. lineatus specimens sampled, 107 were juveniles and 155 were adults. Black skipjack juveniles fed on five food items. The most important prey in the diet were unidentified fish (% PSIRI = 57.4) and fish from the families Engraulidae (% PSIRI= 30.1) and Clupeidae (% PSIRI = 12.3) (Table 2).
TABLE 2 Diet composition of the black skipjack Euthynnus lineatus in the Tropical Eastern Pacific
| Juveniles | Adults | |||||||
| Prey | % FO | % N | % W | % PSIRI | % FO | % N | % W | % PSIRI |
| MOLLUSCA | ||||||||
| Bivalvia | 1.98 | 0.19 | 0.01 | 1.98 | ||||
| PISCES | ||||||||
| Family Engraulidae | 30.95 | 45.31 | 38.35 | 29.06 | 33.66 | 18.21 | 16.51 | 14.24 |
| Anchoa spp. | 2.38 | 6.25 | 0.54 | 1.09 | 39.60 | 55.09 | 39.04 | 24.65 |
| Centegralis mysticetus | 5.94 | 0.75 | 1.08 | 1.07 | ||||
| Family Clupeidae | ||||||||
| Lile stolifera | 4.95 | 1.68 | 2.98 | 1.99 | ||||
| Lile sp. | 15.84 | 6.91 | 8.13 | 5.21 | ||||
| Opisthonema libertate | 2.38 | 1.56 | 11.91 | 2.38 | 7.92 | 0.84 | 0.94 | 4.30 |
| Opisthonema sp. | 11.90 | 7.81 | 2.35 | 9.98 | 9.90 | 3.36 | 3.24 | 3.21 |
| Family Exocoetidae | 0.99 | 0.56 | 0.28 | 0.37 | ||||
| Fodiator acutus rostratus | 0.99 | 0.09 | 0.33 | 0.99 | ||||
| Family Serranidae | 1.98 | 0.28 | 0.37 | 0.12 | ||||
| Fish larvae | 7.92 | 7.38 | 3.92 | 7.45 | ||||
| Unidentified fish | 59.52 | 39.06 | 46.86 | 57.50 | 49.50 | 4.67 | 23.17 | 34.42 |
| TOTAL | 107.1 | 100 | 100 | 100 | 181.1 | 100 | 100 | 100 |
Where % FO = Frequency of Occurrence, % N = Numerical method, % W = Gravimetric method and % PSIRI = Prey-Specific Index of Relative Importance.
Black skipjack adults fed on 13 food items. The most important items in the diet were the family Engraulidae (% PSIRI = 39.9), unidentified fish (% PSIRI = 34.4), and the family Clupeidae (% PSIRI = 14.7) (Table 2). According to the Morisita-Horn index, there was no significant overlap between juveniles and adults of E. lineatus (Cλ = 0.3).
Of the total number of S. sierra specimens sampled, 75 were juveniles and 134 were adults. Pacific sierra juveniles fed on five food items. The most important items in the diet were unidentified fish (% PSIRI = 66.8), unidentified crustaceans (% PSIRI = 17.6), and the families Engraulidae (% PSIRI = 9.6) and Clupeidae (% PSIRI = 5.8) (Table 3).
TABLE 3 Diet composition of the Pacific sierra Scomberomorus sierra in the Tropical Eastern Pacific
| Juveniles | Adults | |||||||
| Prey | % FO | % N | % W | % PSIRI | % FO | % N | % W | % PSIRI |
| CRUSTACEA | ||||||||
| Unidentified crustaceans | 17.65 | 15.79 | 0.90 | 17.65 | 15.15 | 10.87 | 5.27 | 15.15 |
| PISCES | ||||||||
| Family Engraulidae | 5.88 | 5.26 | 17.96 | 5.88 | 9.09 | 15.22 | 13.78 | 7.39 |
| Anchoa spp. | 5.88 | 5.26 | 4.06 | 3.75 | 3.03 | 2.17 | 0.16 | 1.19 |
| Family Clupeidae | ||||||||
| Opisthonema libertate | 5.88 | 10.53 | 35.20 | 5.88 | ||||
| Opisthonema sp. | 6.06 | 17.39 | 11.13 | 4.52 | ||||
| Unidentified fish | 70.59 | 63.16 | 41.88 | 66.84 | 75.76 | 54.35 | 69.66 | 71.75 |
| TOTAL | 105.8 | 100 | 100 | 100 | 109 | 100 | 100 | 100 |
% FO = Frequency of Occurrence, % N = Numerical method, % W = Gravimetric method and % PSIRI = Prey-Specific Index of Relative Importance.
Pacific sierra adults fed on five food components. The most important prey in the diet were unidentified fish (% PSIRI = 71.7), unidentified crustaceans (% PSIRI = 15.1), and the families Engraulidae (% PSIRI = 8.5) and Clupeidae (% PSIRI = 4.5) (Table 3). According to the Morisita-Horn index, there was high diet overlap between S. sierra juveniles and adults (Cλ = 0.9).
Seasonal variations in diet: A total of 143 stomachsof E. lineatus were collected during the dry season and 119 stomachs were collected during the wet season. During the dry season, black skipjack fed mainly on the family Engraulidae (% PSIRI= 39.5), unidentified fish (% PSIRI= 32.6), and the family Clupeidae (% PSIRI= 16.4). During the wet season, black skipjack fed mainly on unidentified fish (% PSIRI= 57) and the families Engraulidae (% PSIRI= 31.2) and Clupeidae (% PSIRI= 9.8) (Fig. 1).
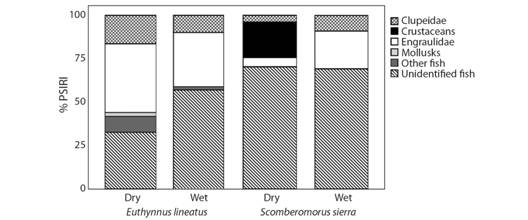
Fig. 1 Prey-Specific Index of Relative Importance (PSIRI) of the black skipjack Euthynnus lineatus and the Pacific sierra Scomberomorus sierra in the Tropical Eastern Pacific, during dry and wet season.
A total of 157 stomachs of S. sierra were collected during the dry season, whereas 52 stomachs were collected during the wet season. The most important food items during the dry season were unidentified fish (% PSIRI = 70.3), unidentified crustaceans (% PSIRI = 20.5), and the families Engraulidae (% PSIRI = 5.3) and Clupeidae (% PSIRI = 3.8). The main diet items during the wet season were unidentified fish (% PSIRI = 69.1) and the families Engraulidae (% PSIRI = 21.7) and Clupeidae (% PSIRI = 9) (Fig. 1). There was high trophic overlap between the seasons for the two species (Cλ = 0.6 and Cλ = 0.9, respectively).
Niche width: Values of Levin´s index (Bi) obtained for the two species were below 0.6 for juveniles as well as for adults. Values found for E. lineatus were 0.4 for juveniles and 0.1 for adults, whereas for S. sierra values were 0.3 for juveniles and 0.4 for adults, which indicated that the two species are specialist predators.
Trophic level: The trophic levels calculated were 4.2 for E. lineatus juveniles and 4.1 for E. lineatus adults. The trophic level of S. sierra was 4 for juveniles and adults. Results found for the two species placed them as tertiary predators.
Discussion
Diet composition: Black skipjack and Pacific sierra fed mainly on fish. The most important items in the diet were unidentified fish (with an advanced state of digestion) and the families Engraulidae and Clupeidae. These results coincide with what has been previously reported for the two species. For example, according to Chiou and Lee (2004), fish comprised 90 % of the general diet of E. affinis. Griffiths, Kuhnert, Fry, & Manson (2009) reported that pelagic clupeids and demersal fish were the main food components in the diet of that same species. Bahou et al. (2007) found that E. alletteratus fed on 23 prey taxa, including mainly flying fish, squid, and crustaceans. Ruíz-Pérez, Cerdenares-Ladrón de Guevara, López-Herrera, & Altamirano-Ramírez (2016) indicated that E. lineatus feed mainly on crustaceans in the coast of Oaxaca. Miranda-Delgado et al. (2019) found that the E. lineatus trophic spectrum included 14 prey items in the study area, among which were fish, crab, penneid larvae, mollusk larvae, isopods, ostracods, juvenile squid, amphipods, and polychaetes.
Artunduaga (1972) reported that S. sierra is a carnivorous fish found at the upper levels of the food chain, whose existence depended mainly on planktivorous fish. Previous results for this species coincide with the present study in that it consumed mainly small pelagic fish that form dense schools, especially anchovies and sardines (Fischer et al., 1995; Moreno-Sánchez, Quiñonez-Velázquez, Abitia-Cárdenas, & Rodríguez-Romero, 2012).
One of the most notable differences between the present study and what has been reported in previous studies is that the E. lineatus diet did not include cephalopods and crustaceans, and the diet of S. sierra did not include cephalopods, which could be related to geographical variations in the abundance and availability of prey on which these species feed (Rojas-Herrera, Mascaró, & Chiappa-Carrara, 2004).
It should be mentioned that a considerable proportion of stomachs of the two studied species were found empty or with a large proportion of contents at an advanced state of digestion. Several factors can influence the vacuity including the type of fishing, timing of capture and gastric emptying rates, influenced at the same time by the size of specimens, environmental temperature, and chemical properties of ingested prey (Cortés, Papastamatiou, Carlson, Ferry, & Wetherbee, 2008; Wetherbee, Cortés, & Bizarro, 2012; Flores-Martínez, Torres-Rojas, Galván-Magaña, & Ramos-Miranda, 2016; Leigh, Papastamatiou, & German, 2017; Ehemann, Abitia-Cardenas, Navia, Mejía-Falla, & Cruz-Escalona, 2019). Arrington et al. (2002) indicated that organisms with nocturnal feeding habits and piscivorous predators show a larger percentage of empty stomachs compared with fish with diurnal habits that prefer other food categories. Schaefer (1984) showed that gastric evacuation times are faster for scombrids than for most other fishes. This would explain the high frequency of fish and crustaceans at an advanced state of digestion found in both species, which made identification difficult.
Diet variations by size: In the present study, although fish constituted the largest proportion of the diet of juveniles and adults, we observed that adult fish presented a wider diet that included other fish groups such as flying fish and grunts, as well as fish larvae. In this regard, Bahou et al. (2007), explained three factors involved in size-based feeding strategies of E. alletteratus: (i) juveniles are constrained by their small size to take relatively small fish as prey; (ii) only following a probable period of initial growth it can switch to feeding on both crustaceans and fish, and (iii) adults tend to prefer consuming fish to crustaceans or cephalopods. Likewise, Griffiths et al. (2009) observed that the diet of small and medium sized of E. affinis (400-580 mm long) consisted mainly in small pelagic crustaceans (stomatopod larvae and brachyura megalopa) and fish larvae (engraulids, clupeoids, dactylopterids, monacanthids, and tetraodontids); whereas large fish ( > 580 mm) consumed mainly larger specimens belonging to a different set of teleost prey (carangids, Trachurus declivis, belonids and Sardinops sagax). Changes in diet according to size may be simply attributable to evolution in feeding habits with increasing size of predator and provide evidence that size-based feeding strategies of scombrids may be related to limiting factors other than mouth-gape morphology, such as prey availability and behavioural differences of both predators and prey (Ménard et al., 2006; Bahou et al., 2007). In addition, the presence of fish during development may be due to the fact that they provide more energy for faster growth (Saucedo-Lozano et al., 2012).
Bakhoum (2007) indicated that teleosts were the most important food item for S. commerson of all sizes. Naughton and Saloman (1981) reported that fish occurred in 74 % of the stomachs of S. cavalla, and invertebrates were of minor importance in the juvenile mackerel diet. Also, they reported that fishes occurred in 95 % of the stomachs of S. maculatus, and invertebrates (squid) had a frequency of occurrence of 6 %. Naughton and Saloman (1981) explained that the smaller size and diameter of engraulids (Anchoa sp.) would be more readily ingested by juvenile predators with smaller mouth parts. No differences in diet were found between S. sierra juveniles and adults. However, the highly proportion of unidentified fish in its diet, represents uncertainty regarding dietary changes that this species might present with growth.
Seasonal variations in diet:Griffiths et al. (2009) pointed out that the season is the most important factor explaining variability in the diet of E. affinis. Bahou et al. (2007) indicated that E. alletteratus respond to seasonal changes in food availability, which reflects the opportunistic behaviour and trophic adaptability, allowing them to take advantage of the most readily available prey in the environment at any time. The diet of E. lineatus and S. sierra comprised the same prey groups during the wet and dry seasons. In the study area, Rojas-Herrera et al. (2004) also reported that two species of lutjanids (Lutjanus peru and L. guttatus), did not show seasonal qualitative variations in the use of the different components of their diets. They explained that the availability of prey in each region is what determines the feeding of these species, rather than the selection of prey. Likewise, Hernández-Aguilar, Abitia-Cárdenas, Moreno-Sánchez, Arellano-Martínez, & González-Rodríguez (2012) reported that the sailfish Istiophorus platypterus, there were no differences in prey preference between the warm and cold seasons. They infer that in the southern Mexican Pacific region there is stability in prey abundance caused by the oceanographic characteristics of the area. In this sense, the feeding behavior of E. lineatus and S. sierra seems to depend on the most readily available preys, which are sufficiently diverse and abundant to allow their coexistence (Rojas-Herrera et al., 2004).
Niche width: Among scombrids, tunas are widely known as generalist opportunistic predators that consume a wide prey spectrum at different trophic levels, which allows them to move freely among habitats (Olson et al., 2016). However, according to the FAO (1994), the black skipjack is not considered a “highly migratory” species, although it carries out movements associated with continental platforms. This probably leads to its diet containing epipelagic as well as demersal prey (Griffiths et al., 2009).
According to Levin´s index, E. lineatus and S. sierra are specialist predators, because their diets are narrow, with marked preference for a few prey species (anchovies and sardines). This behavior has been observed by other authors, who have also reported opportunistic behavior in these two species. This allows them to take advantage of the most easily available prey in the environment at a given time (Bahou et al., 2007; Moreno-Sánchez et al., 2012).
Trophic level: The consumption of species at different trophic levels in the food chain resulted in E. lineatus and S. sierra occupying relatively high trophic positions; this indicated that the two species act as tertiary consumers, or mesopredators, in the food web (Cortés, 1999). The energy transfer from lower trophic levels to upper levels and vice versa that these species present mean that their predatory habits could be used to detect important changes in the structure of the ecosystem (Olson et al., 2014).
According to our results, we can conclude that E. lineatus and S. sierra are tertiary predators and present specialist behavior, feeding mainly on fish from the families Engraulidae and Clupeidae. Moreover, although a degree of similarity in the dietary food components of these species was observed, there was not a high degree of interspecific competition for food.
Ethical statement: authors declare that they all agree with this publication and made significant contributions; that there is no conflict of interest of any kind; and that we followed all pertinent ethical and legal procedures and requirements. All financial sources are fully and clearly stated in the acknowledgements section. A signed document has been filed in the journal archives.












 uBio
uBio