Introduction
Dental biofilm is formed by a microbial community of multiple species (1, 2, 3, 4) and can be defined as a non-calcified film, strongly adhered to tooth surfaces, which resists the presence of salivary flow (5, 6). This phenomenon is recognized as an important factor in the triggering and progression of caries lesions which compromise healthy teeth (7, 8).
The basic structure of normal, healthy enamel is composed of tightly joined hydroxyapatite crystals and has a translucent surface (9). When enamel is exposed to acids, such as those produced during the formation of dental biofilm, a dynamic process of imbalance between tooth mineral and adjacent biofilm fluid can occur, resulting in mineral loss or demineralization (10, 11).
Mineral loss enlarges the intercrystalline spaces, and can be observed as an increase in tissue porosity, as a result of the removal of mineral salts that are more soluble than hydroxyapatite (carbon and magnesium) (12). This loss leads to a change in the optical properties of the enamel (13), making it less translucent, which clinically can be observed as opaque and chalky whitish changes, with a chalky appearance commonly called "white spot" (14). When diagnosed in its initial phase, the lesion is still capable of remineralization (15).
There are several types of treatments to control the progression of dental caries, and their choice is directly related to the patient's disease activity (16). In some individuals, removal of biofilm by the patient himself, combined with the use of fluoride toothpaste, is sufficient to maintain demineralization in subclinical stages (17). In individuals with very rapid progression of caries lesions, more specific measures are necessary (18).
Dental biofilm control carried out by the patient through brushing and flossing requires motivation, oral hygiene instruction and the individual's manual skills to be effective (19).
As a way to compensate for such limitations, there are professional prophylactic methods, carried out in the office by the dentist to seek to prevent cavities in the most natural way possible, providing balanced conditions for the biodiversity of the oral cavity (20).
Professional dental biofilm removal, when carried out by a dentist, has three main objectives: to facilitate the judgment of real carious activity, to favor the control of visible active carious lesions and to form the basis for patient education (21, 22).
Among the prophylactic methods carried out by professionals, the most commonly used are the rubber cup, brush, ultrasonic scraping, manual scraping and sodium bicarbonate jet (23).
Prophylaxis carried out by brush and cup is generally done together with pumice stone, but in some current clinical procedures, the use of pumice stone has been replaced by prophylactic paste. Despite the benefit provided by professional prophylaxis in controlling microorganisms, there is a question about the amount of sound dental enamel that is removed during such a procedure, and this situation has been researched and studied by several authors (24,25,26). However, this effect on surfaces with white spot lesions is still poorly understood (27).
Therefore, the objective of this study was to evaluate, in vitro, the effect of different methods used in dental prophylaxis on the mineral content of demineralized enamel surfaces, using quantitative light-induced fluorescence (QLF).
Methodology
The research protocols were reviewed by the Human Research Ethics Committee of the São Paulo State University (UNESP), Faculty of Dentistry of Araraquara (CAAE: 02744412.5.0000.5416).
Sampling design
The sample was made up of 4 groups: G1 - Robinson brush and pumice stone; G2 - Robinson brush and prophylactic paste; G3 - Rubber and pumice bowl; G4 - Rubber cup and prophylactic paste. Forty healthy premolars were used, extracted for periodontal or orthodontic indications, donated by the Bank of Human Teeth - Permanent of the Faculty of Dentistry of Araraquara - UNESP, which were randomly divided into the four experimental groups.
The evaluations were carried out at 3 levels: with the tooth healthy, immediately after the demineralization process and after application of the proposed treatments.
Demineralization process
The methodology for demineralization was carried out according to Marquezan et al. (28).
The teeth were washed in running water and left to dry at room temperature. Subsequently, they were waterproofed with the application of two layers of acid-resistant varnish (Colorama, CEIL Com. Exp. Ind. Ltda, SP, Brazil) leaving an area corresponding to 50% of the buccal surface of each tooth exposed. The teeth, in groups of ten, were then immersed in 300 mL of distilled water deposited in a beaker, and the set was sterilized in an autoclave at 120°C, 1 kgf/cm2, for 20 minutes.
The cariogenic medium used for the artificial induction of caries was composed of a solution for bacterial growth containing 100 mL of distilled water, 3.7 g of BHI, 1 g of glucose (D-Glucose Anhydrous, Synth; LabSynth, São Paulo, SP, Brazil), 2 g of sucrose (Cristalçúcar União, Cia União, São Paulo - Brazil) and 0.5 g of yeast extract (Becton Dickinson and Company, Sparks, MD, USA). After sterilization in an autoclave and cooling, the teeth were transferred from the beaker containing distilled water to a beaker containing the cariogenic medium (25 mL/tooth) to which 2% of the BHI broth culture medium containing S. mutans (100 μL) were inoculated. Finally, the beaker containing the teeth immersed in the inoculated cariogenic medium was placed in an anaerobic jar for growth under microaerophilic conditions and taken to the oven at 37°C.
The total period of contact between the enamel and the cariogenic medium was 9 days, and every 48 hours, the cariogenic medium was exchanged for a freshly prepared one. Once the caries induction protocol was completed, the biofilm formed on the teeth was removed with sterilized gauze and waterproofing materials with the help of a scalpel, followed by abundant washing in running water.
Prophylactic treatment
The teeth were inserted into individual mannequins containing condensation silicone (Zetaplus, Zhermack, Germany), thus avoiding manual contact with the teeth during treatment. The time for each treatment was 10 uninterrupted seconds, controlled by a stopwatch. The procedure was performed with a contra-angle at a speed of 1500 rpm, according to Honorio et al. (27), with pressure controlled by a tourniquet attached to the wrist connected to a dynamometer (29).
The rubber cup or brush was replaced with each treated tooth. The amount of prophylactic paste (Herjos-f, Vigodent SA) and extra fine powdered pumice stone (Asfer) used for each specimen was standardized, being 5g of water for 6g of powder (pumice) and 6g of prophylactic paste. Then, all specimens were washed with a water/air spray for 10 seconds and then dried with an air jet for 10 seconds (29).
Surface analysis
The device previously made with silicone also served to standardize the position of the teeth during exams with the QLF, which directly contributed to the quality of the images obtained, as well as standardizing the reading of the surfaces of the teeth to be evaluated.
Quantitative light-induced fluorescence-QLF
To capture images of the dental structure (emitted fluorescence observed through a yellow high-pass filter - 520 nm), an intra-oral fluorescence camera from the Inspektor™ Pro device (Inspektor Dental Care BV, Amsterdam, Netherlands) was used to measure the fluorescence intensity of enamel lesions, indicating the amount of mineralization. A spacer was used to control the distance and inclination of the blue light emitting source, which was kept parallel to the analyzed surface, with a distance of 5 mm, avoiding distortions in the latent image. The images were obtained in a room with window glass having a film that prevented the incidence of direct sunlight, which could cause reflections on the enamel surface and, thus, compromise the reading of the image.
The teeth were exposed to blue-violet light, which has a peak intensity at 404 nm. The image was captured with QLF focusing on the white spot lesion. Mineral quantification was performed non-invasively and allowed monitoring of lesions (mineral loss and gain) over time. The images obtained were examined using the Inspektor™ Pro version 2.0.0.32 software (Inspektor Dental Care BV, Amsterdam, Netherlands) and delta F (DF) values were calculated considering a threshold of 5%. In other words, variations in fluorescence between healthy and demineralized enamel, less than 5%, were ignored. To ensure the reliability of the measurements, the analyses were conducted by a single examiner following a set of standardized rules for image analysis. Additionally, triplicate analyses and periodic calibrations of the fluorescence device were performed (30).
Results
The Friedman analysis was conducted to compare the mineral content among three different conditions: Healthy Tooth (HT), Demineralized Tooth (DT), and Tooth treated with Robinson brush and pumice stone (GI). The analysis provided the sum of ranks, medians, and mean ranks for each condition, along with the statistical significance of the differences observed (Table 1).
The results indicate a statistically significant difference (p<0.001) between the mineral content of the HT and the DT. This significant difference confirms that demineralization successfully reduced the mineral content, as expected, resulting in the formation of a white spot, evidenced by a decrease in fluorescence intensity.
However, when comparing the HT to the tooth treated with Robinson brush and pumice stone (GI), there is no statistically significant change (p=0.082). This suggests that the treatment with Robinson brush and pumice stone did not result in any detectable changes in mineral content. In summary, demineralization significantly altered the mineral content of the teeth, while the subsequent treatment did not produce further significant changes.
The following images illustrate the different stages of tooth treatment evaluated in this study using the Quantitative Light-induced Fluorescence (QLF) method (Figure 1).
Below are the GII results, there is a statistically significant difference (p<0.001) between the mineral content of the healthy tooth (HT) and the demineralized tooth (DT). This significant difference confirms that demineralization successfully reduced the mineral content, resulting in the formation of a white spot, evidenced by a decrease in fluorescence intensity.
However, when comparing the healthy tooth (HT) with the tooth treated with a Robinson brush and prophylactic paste (GII), there is no statistically significant change (p=0.092). This suggests that prophylactic treatment did not result in any detectable changes in mineral content compared to the demineralized state (Table 2).
The following images illustrate the different stages of dental treatment evaluated in the experimental group GII using the Quantitative Light- Induced Fluorescence (QLF) method (Figure 2).
In the experimental group GIII, the results indicate a statistically significant difference (p<0.001) between the mineral content of the healthy tooth (HT) and the demineralized tooth (DT). This significant difference confirms the successful reduction of mineral content due to demineralization, resulting in the formation of a white spot.
Additionally, a statistically significant difference (p<0.001) was observed between the healthy tooth (HT) and the tooth treated with prophylaxis using a rubber cup and pumice stone (GIII). This finding suggests that the prophylactic treatment with a rubber cup and pumice stone can partially restore the mineral content of demineralized enamel, although it does not completely return to the original healthy state (Table 3).
The following images, obtained using the Quantitative Light-induced Fluorescence (QLF) method, visually represent the different stages of tooth treatment in Group III (Figure 3).
These images support the findings from the Friedman analysis, visually confirming the reduction in mineral content due to demineralization and the partial improvement in mineral content following prophylactic treatment.
The Friedman analysis in Table 4 compares the mineral content among three conditions in Group IV: Healthy Tooth (HT), Demineralized Tooth (DT), and Tooth treated with prophylaxis using a rubber cup and prophylactic paste (GIV). The table provides the sum of ranks, medians, and mean ranks for each condition, along with the statistical significance of the observed differences.
The results indicate a statistically significant difference (p<0.001) between the mineral content of the healthy tooth (HT) and the demineralized tooth (DT) in Group IV. This significant difference confirms the successful reduction in mineral content due to demineralization, resulting in the formation of a white spot.
Additionally, a statistically significant difference (p<0.001) was observed between the healthy tooth (HT) and the tooth treated with prophylaxis using a rubber cup and prophylactic paste (GIV). This finding suggests that the prophylactic treatment with a rubber cup and prophylactic paste can partially restore the mineral content of demineralized enamel, although it does not completely return to the original healthy state.
The following images, obtained using the Quantitative Light-induced Fluorescence (QLF) method, visually represent the different stages of tooth treatment in Group IV (Figure 4).
These images support the findings from the Friedman analysis, visually confirming the reduction in mineral content due to demineralization and the partial improvement in mineral content following prophylactic treatment.
Table 1 Friedman analysis of the experimental group GI.
| Friedman Analysis | HT | DT | GI |
|---|---|---|---|
| Sum of Ranks | 29 | 13 | 18 |
| Median | -6,84 | -13,75 | -8,82 |
| Mean of Ranks | 2,9 | 1,3 | 1,8 |
| Degrees of Freedom | 2 | ||
| Friedman (Fr) | 13,4 | ||
| p | 0,0012 | ||
| Comparison | Difference | p | |
| DH x DD | 16 | < 0.001 | |
| DH x GI | 11 | 0.082 |
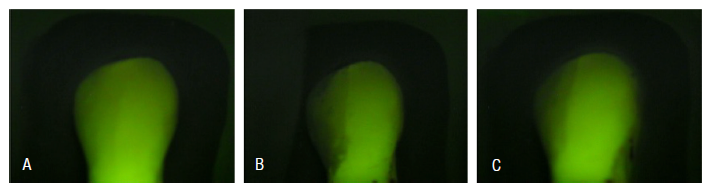
Figure 1 A. A healthy tooth, displaying the baseline mineral content and fluorescence; B. A tooth after the demineralization process, showing a reduction in mineral content and fluorescence intensity, evidenced by the appearance of a white spot; C. (GI) A tooth after prophylaxis with a Robinson brush and pumice stone, indicating the effect of the treatment on the tooth's mineral content.
Table 2 Friedman analysis of the experimental group GII.
| Friedman Analysis | HT | DT | GII |
|---|---|---|---|
| Sum of Ranks | 29 | 13 | 18 |
| Median | -6,57 | -11,95 | -9,83 |
| Mean of Ranks | 2,9 | 1,3 | 1,8 |
| Degrees of Freedom | 2 | ||
| Friedman (Fr) | 13,4 | ||
| p | 0,0014 | ||
| Comparison | Difference | p | |
| DH x DD | 17 | < 0.001 | |
| DH x GII | 10 | 0.092 |
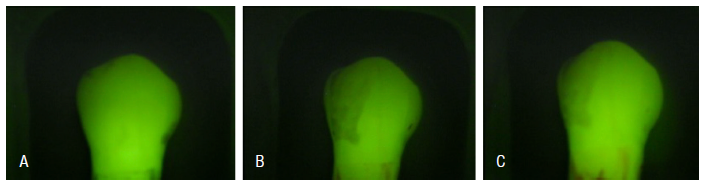
Figure 2 A. A healthy tooth, displaying the baseline mineral content and fluorescence; B. A tooth after the demineralization process, showing a reduction in mineral content and fluorescence intensity, evidenced by the appearance of a white spot; C. (GII) Tooth after prophylaxis with Robinson brush and prophylactic paste, indicating the effect of treatment on the mineral content of the tooth.
Table 3 Friedman analysis of the experimental group GIII.
| Friedman Analysis | HT | DT | GIII |
|---|---|---|---|
| Sum of Ranks | 28 | 12 | 20 |
| Median | -6,66 | -10,95 | 9,00 |
| Mean of Ranks | 2,8 | 1,2 | 2,0 |
| Degrees of Freedom | 2 | ||
| Friedman (Fr) | 12,8 | ||
| p | 0,0017 | ||
| Comparison | Difference | p | |
| DH x DD | 16 | < 0.001 | |
| DH x GIII | 8 | < 0.001 |
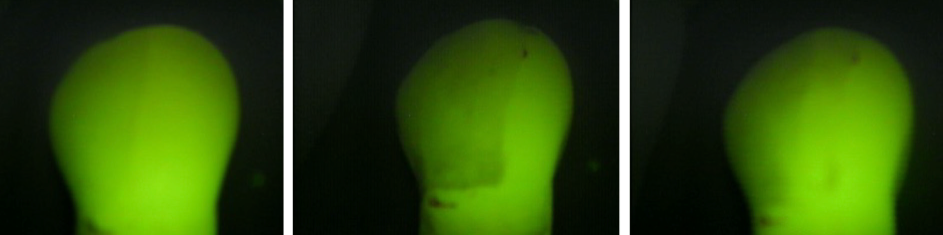
Figure 3 A. A healthy tooth, displaying the baseline mineral content and fluorescence; B. A tooth after the demineralization process, showing a significant reduction in mineral content and fluorescence intensity, evidenced by the appearance of a white spot; C. (GIII) A tooth after prophylaxis with a rubber cup and pumice stone, indicating the partial restoration of mineral content compared to the demineralized state.
Table 4 Friedman analysis of the experimental group GIV.
| Friedman Analysis | HT | DT | GIV |
|---|---|---|---|
| Sum of Ranks | 28 | 14 | 18 |
| Median | -7,59 | -12,35 | -10,6 |
| Mean of Ranks | 2,8 | 1,4 | 2,0 |
| Degrees of Freedom | 2 | ||
| Friedman (Fr) | 10,4 | ||
| p | 0,0055 | ||
| Comparison | Difference | p | |
| DH x DD | 14 | < 0.001 | |
| DH x GIII | 10 | < 0.001 |
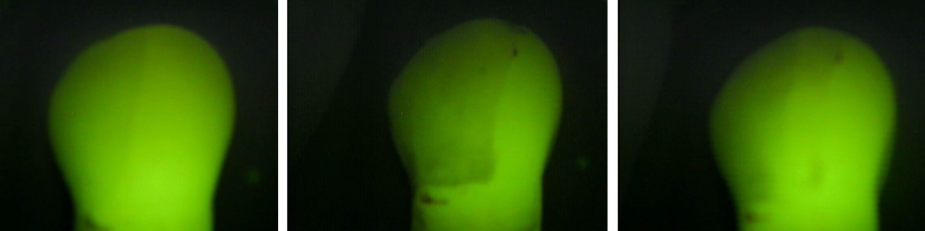
Figure 4 A. A healthy tooth, displaying the baseline mineral content and fluorescence. B. A tooth after the demineralization process, showing a significant reduction in mineral content and fluorescence intensity, evidenced by the appearance of a white spot. C. (GIV) A tooth after prophylaxis with a rubber cup and prophylactic paste, indicating the partial restoration of mineral content compared to the demineralized state.
Discussion
The necessity of dental prophylaxis prior to certain dental procedures has been substantiated by various studies (27), and these procedures become even more essential in individuals with rapid progression of carious lesions (31). However, despite the numerous benefits provided by professional prophylaxis, there are concerns about the potential removal of healthy dental enamel during this technique, a matter that has been researched and studied by various authors (24, 25, 32).
Different devices and instruments are used to study alterations on the dental surface before and after prophylaxis. QLF (quantitative light-induced fluorescence) has been employed in several studies (33,34, 35 36), demonstrating high sensitivity and excellent results in detecting changes in the mineral content of dental enamel. For this reason, we used this device to analyze the healthy dental surface, post-demineralization, and post-prophylactic treatment, respectively, achieving satisfactory results consistent with previous studies.
Regarding prophylaxis performed on healthy teeth, many authors (26, 37) have concluded in their studies that prophylaxis does not cause damage to the dental surface. Honório et al. (27) argue that even though prophylactic techniques may have disadvantages, such as wear, the benefits they provide, such as cavity prevention, seem to be worthwhile. Covey et al. (38) demonstrated that prophylactic cups without paste do not significantly affect enamel surface roughness.
The present study showed results similar to those previously described, despite being conducted on demineralized dental enamel, as no reduction in the amount of minerals in the teeth was observed after prophylaxis. Conversely, some authors (5, 39) have concluded that regardless of the prophylactic method used, there is always a deterioration of the surface when analyzing the roughness of teeth and restorative materials post-prophylaxis.
A comparative study on the remineralization of white spot lesions through professional teeth cleaning concluded that there were no significant changes in the amount of minerals, although a significant difference in the tooth's fluorescence change was observed (37). In the present study, an improvement in mineral content was observed between the demineralized tooth and the experimental group treated with a rubber cup and pumice stone, as well as a significant improvement post-treatment with a rubber cup and prophylactic paste. These results suggest that this treatment provides better outcomes when seeking recovery from the initial stage of caries, characterized by the white spot.
Another study evaluated the wear produced by professional prophylaxis with a sodium bicarbonate jet and with a brush and pumice stone on intact and artificially demineralized bovine enamel. The results revealed that the average wear of demineralized bovine enamel fragments was greater compared to intact specimens, regardless of the prophylaxis method used (27).
In the present study, after prophylactic treatment with a Robinson brush associated with pumice stone or prophylactic paste, no changes were observed on the demineralized tooth surface regarding the amount of minerals. However, when using the rubber cup associated with prophylactic paste or pumice stone, an improvement was found in the quantification of minerals present in the tooth structure. These results suggest that this treatment provides better results when seeking to recover from the initial stage of caries, characterized by the white spot.
The use of QLF provided high sensitivity and excellent results in detecting changes in the mineral content of dental enamel. The study comprehensively analyzed different prophylactic treatments and their effects on demineralized dental enamel. The findings are consistent with previous studies, enhancing the reliability of the results.
The study was conducted on artificially demineralized enamel, which may not perfectly replicate natural conditions. The sample size and diversity could be expanded to increase the generalizability of the results. Additionally, further research is needed to evaluate the long-term effects and clinical relevance in a broader population sample.
Conclusion
From the analysis and discussion of the results obtained, it is concluded that:
Dental prophylaxis through treatment with a Robinson brush associated with pumice stone or prophylactic paste presents similar results, not causing statistically significant changes in the amount of minerals in demineralized tooth enamel in the analysis carried out by QLF.
Dental prophylaxis using rubber cup and pumice or prophylactic paste has demonstrated potential partial remineralization of demineralized tooth enamel.
Author contribution statement
Conceptualization and design: F.F.E.T. and A.V.J.
Literature review: F.L.R. and L.E.G.
Methodology and validation: F.L.R., S.R.C.S. and E.P.S.T.
Formal analysis: F.F.E.T.
Investigation and data collection: F.F.E.T.
Resources: F.L.R.
Data analysis and interpretation: F.F.E.T.
Writing-preparation of original draf: F.E.P. and L.E.G.
Writing-review and editing: L.E.G.
Supervision: F.L.R.
Project administration: F.L.R.
Funding acquisition: F.L.R.















