Introduction
Flowering in plants indues a succession of subsequent events, including anthesis, fructification, development, and fruit maturation, which determine the propagation of species and can contribute to crop improvement via genetic recombination (Bhagwan & Reddy, 2014). In fruit trees, flowering is an important reproductive phenomenon that marks the commencement of fruit production (Olubode et al., 2018). It typically progresses in four stages: floral induction, floral initiation, floral differentiation, and flowering. The length of the flowering phase is dependent on both genotype and environmental conditions (Saroj et al., 2014). In cashew, flower production is a gradual process that can be influenced by morphological mechanisms in the panicles, interfering in the final proportion of male and hermaphrodite flowers that remain until the final development of the fruits in the crop.
The plant family Anacardiaceae comprises more than 700 species (Barros, 1988; Nakasone & Paul, 1998). Prominent among these is the cashew tree Anacardium occidentale Linnaeus, a species native to Northeastern Brazil (Barros et al., 2002), although is now cultivated in more than 30 countries worldwide (FAOSTAT, 2021). Classified as an andromonoic species, the cashew tree has both male and hermaphroditic flowers (Barros & Crisóstomo, 1995). The inflorescence of A. occidentale is a panicle, with variable shapes, sizes, quantities, and proportions of male and hermaphrodite flowers (Adiga et al., 2019; Sousa et al., 2007).
Flowering in cashew is directly associated with vegetative growth fluxes, both of which occur concomitantly during certain periods and with different intensities. The period of maximum floral differentiation and flowering occurs mainly during the dry season (Frota & Parente, 1995; Wunnachit & Sedgley, 1992) and can extend to up to 7 months (Sousa et al., 2007) due to the heterogeneity of the orchards, which undergo variations in the length of the flowering period (Barros, 1988). Under suitable growing conditions with well-distributed rainfall, cashew trees can flower more than once a year (Nambiar, 1979; Ohler, 1979; Wait & Jamieson, 1986), with flowering and fruiting during the dry season conferring the distinct advantage of a lower incidence of pests and diseases, thereby reducing potential damage during these vulnerable phenological phases (Wunnachit & Sedgley, 1992). However, low levels of relative humidity during flowering and fruiting can reduce not only stigma receptivity but also pollen viability (Sturtz, 1981), and increase the likelihood of the abortion of developing fruits (Wunnachit & Sedgley, 1992).
Cashew flowering normally begins at the end of the rainy season, after the emergence of a new growth stream, although its onset and duration are strongly dependent on external environmental factors, including temperature, radiation and precipitation, which occur in the planting area during the period. of flowering (Saroj et al., 2014). Such factors can compromise the structural aspects of flowering, interfering with the sexual expression of flowers (Sousa et al., 2007), promoting the development of panicles that present a large number of male flowers and low emission of hermaphrodite flowers, negatively influencing production (Madail et al., 2012).
Although cashew trees produce an abundance of flowers, less than 10 % of these are hermaphroditic and, among these, approximately 85 % are fertilized. Of these fertilized flowers, however, only 4 % to 6 % will subsequently undergo development to maturity and bear fruit, with the remainder being eliminated at different stages of production (Mog et al., 2018). Accordingly, the characteristics of flowers and flowering affect not only those of the fruits, but also their production. Nevertheless, despite the important practical implications of floral biology, this is an aspect that has hardly been investigated in A. occidentale. In this study, we tried for the first time to answer some behaviors. How can the association between the expression of hermaphrodite flowers and other morphological parameters of panicles in dwarf cashew trees be explained? Furthermore, from an evolutionary point of view, how is the influence of the evaluation period of flowering and harvests established on the floral parameters of the culture? These parameters are important, because the time of flowering evaluation is important to explain the production of hermaphrodite and male flowers during the different flowering periods of the species, and they can also exhibit different behaviors under the influence of the crops and their respective environmental characteristics.
Materials and methods
Experimental area: Data were collected from dwarf-type cashew plants that were vegetatively propagated and cultivated under rain-fed conditions in an experimental field of Embrapa Agroindústria Tropical, in the municipality of Pacajus in Ceará, Brazil (4º10'21'' S & 38°27'50'' W). The predominant climate of the region is humid hot tropical (Aw, in the Köppen Geiger climate classification), with an average annual temperature of 26 °C and an average annual rainfall of 1 020 mm (Kottek et al., 2006). The meteorological data recorded in the experimental area in 2018 and 2019 are presented in Fig. 1.
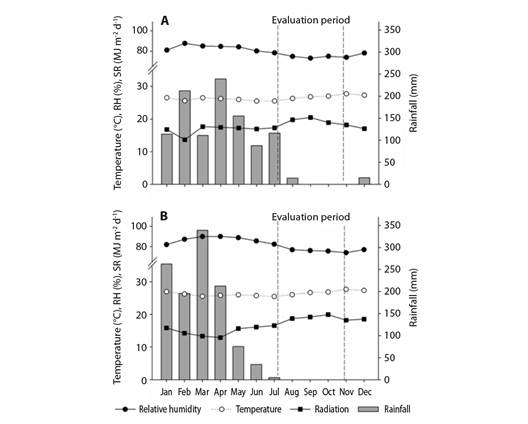
Fig. 1 Meteorological information from the experimental area in the two crops evaluated for the years A. 2018. B. 2019 in the municipality of Pacajus-CE, Brazil, 2021.
Genetic material: Ten clones of the dwarf cashew tree were selected in progeny experiments based on phenotypic evaluations of their suitability to edaphoclimatic conditions in the state of Ceará (Table 1). The selected clones differed with respect to several characteristics, including chestnut and peduncle productivity, average weight of chestnut and peduncle, peduncle color and shape, plant size, early flowering, fruiting, and the duration of these productive periods and almond yield.
Table 1 Identification of dwarf cashew clones and their referred origins.
| Clones | Origins |
| END II 6-9 | Dwarf x Dwarf hybrid |
| PRO 555/2 | Free-pollinated progeny |
| PRO 553/2 | Free-pollinated progeny |
| A + A 134/1 | Dwarf x Dwarf hybrid |
| HB 116/4 | Dwarf x Dwarf hybrid |
| HB 33 | Dwarf x Dwarf hybrid |
| PRO 740/4 | Free-pollinated progeny |
| PRO 106/2 | Free-pollinated progeny |
| CCP 76 | Commercial Clone |
| BRS 226 | Commercial Clone |
Experimental design: Experiments were performed based on a randomized block design, with 10 treatments (genotypes), three replications, and four plants per plot. One plant in each plot was selected for examination. The block structure and the genotype factor were not considered in the analysis, thereby enabling extrapolation of results for the dwarf cashew tree. In this way, each plant represented a repetition (30 repetitions). The five-year-old plants were spaced in rows of 8 m × 6 m, and all cultural management practices, including pruning, fertilization, weed control, and pest and disease control, were carried out in accordance with the recommendation for commercial cashew cultivation under rain-fed conditions (Serrano & Oliveira, 2013).
Evaluations: For the morphological characterization of panicles, we took into consideration the period of maximum floral differentiation and full flowering that occurred in the 2018 and 2019 growth seasons. As a reference point, for the purposes of evaluations, we defined the commencement of the reproductive period of each genotype as day 30 after marking panicles in the field.
For each of the focal plants, three panicles were selected and marked at each of the cardinal points (North, South, East, and West) of the tree, one for each flowering time assessment (30, 45, and 60 days after the beginning of flowering), totaling 12 panicles per plant.
During each of the two flowering seasons, four panicles (one from each quadrant of the plant) were detached from the plants, packed in labeled plastic bags to prevent dehydration, and placed in plastic boxes. After which they were transported to the evaluation room at the Laboratory of Plant Genetics and Breeding of Embrapa Agroindústria Tropical, for morphological characterization of the floral structures.
According to Violle et al. (2007), the functional traits of plants can be defined as the morphological, physiological and phenological characteristics that directly impact the performance reproduction of individuals, thus influencing their aptitude for a given environmental condition. Based on its attributes, which represent the different values that a given functional trait can assume (Appendix), the characterization of the floral structures of A. occidentale L. (Fig. 2) was carried out through the evaluation and classification of panicles, according to the following parameters: Number of male flowers: counting the number of male flowers with approximately 10 stamens (male organ of the flower), with one stamen being more developed than the others. Number of hermaphrodite flowers: counting the number of hermaphrodite flowers with small stamens and a pistil, the latter of which is generally longer than the most developed stamen and is attached to the ovary. Number of abnormal flowers: counting the number of flowers with an arrangement similar to that of male or hermaphrodite flowers, but with no pistil or larger stamen. Panicle biomass: determined by weighing the panicles. Panicle length: the distance from the first node to the apex. Maximum panicle width: the maximum distance between the ends of the panicle branches. Number of panicle branches: counting the number of branches originating from the main axis of the panicle (primary branches) and from the branches that develop from the primary branches (secondary branches).

Fig. 2 Classification, floral structures and evaluations performed on dwarf cashew plants in the 2018 and 2019 harvests in the experimental area of the municipality of Pacajus, Ceará, Brazil. A. Male flowers on panicles. B. Floral details and structures of male flowers. C. Hermaphrodite flowers in panicles. D. Floral details and structures of hermaphrodite flowers. E. Flowers classified as abnormal for presenting malformation, absence, or duplication of floral structures. F. Biomass. G. length. H. width evaluations of panicles. I. General structure of panicles. J. Branches classified as primary. K. Branches classified as secondary. An = Anther; St = Stamen; Pt = Petals; Sg = Stigma; Sl = Style; Pb = Primary branches; Sb = Secondary branches.
Variation in floral parameters as a function of time-point and harvest: Variations in floral parameters with respect to the effect of time (time-point of flowering evaluation) and harvest were determined by analysis of variances, followed by Tukey's HSD and Fisher's exact tests (F test), respectively. Analyses of variance were conducted using the following genetic-statistical model:
Eq. (1): Yijk = m + di + sj + dsij + eijk,
where Yijk is the observation referring to the i-th flowering time-point, in the j-th harvest, in the k-th replication (block); m is the general mean; di is the effect of the i-th season (i = 30, 45, or 60 days after flowering), considered as fixed; sj is the effect of the j-th harvest (j = 2018 and 2019), considered as random, where sj~NID (0, s2); dsij is the effect of the interaction between the i-th flowering season in the j-th crop, considered as random, where dsij~NID (0, s2) and eijk is the random experimental error associated with the Yijk observation.
The period effect was considered as fixed in view of the particularities of each flowering period in the crop, specifically to enable conclusions to be drawn regarding panicle behavior in each of the three evaluation periods. The harvest effect was considered as random, with the aim of reaching broader conclusions regarding the responses of the cashew tree flowering, regardless of the agricultural year (harvest) considered. Block structure and the genotype factor were not considered in the analysis, to enable extrapolation of the results for the dwarf cashew tree. Thus, influences associated with flowering times of the two evaluated crops could be extrapolated to the whole, as time × harvest interactions have a random effect (Resende, 2005).
The relationships between floral parameters and the expression of hermaphrodite flowers: The data obtained for panicle morphological parameters were subjected to correlation analysis using linear regression, implemented with SAS® statistical software (SAS, 2002). Given that we detected minimal seasonal variation in these morphological parameters (7 of the 8 floral parameters showed no significant differences), they were grouped in order to construct more robust statistical models. Data were subjected to multiple regression analyses using the proc reg procedure, selection=stepwise, with hermaphrodite flowers as the dependent variable (y), and the numbers of male and abnormal flowers; the inflorescence biomass; the length and width of the inflorescence; and the numbers of primary and secondary branches as independent variables.
Initially, all eight floral morphological parameters were included, adjusted by stepwise regressions, to explain the dependent variable (number of hermaphrodite flowers). Using the statistically significant model, the parameters were selected according to the flowering season and higher coefficient values in the regression analysis, and thereafter, the data obtained for the parameters selected by stepwise regression were plotted as a function of the hermaphrodite flower number.
Results
Variation of floral parameters depending on the time-point and harvest: With the exception of panicle biomass, significant effects of the time-point (T) (P < 0.05 and P < 0.01) were observed for all other assessed floral parameters, whereas in contrast, with the exception of the number of secondary branches, no significant effects were detected with regards to harvest. However, period of evaluation × harvest interactive effects were found to be significant for most of the assessed parameters (Table 2). These significant interactive effects can be attributed to the variation in all floral parameters in the year 2018 and an absence of variation in almost all parameters in 2019 (Fig. 3).
Table 2 P-values for the effects of season, crop and interaction (season x crop) of floral morphology characters of dwarf cashew tree, evaluated in two years of cultivation, in the municipality of Pacajus, CE, 2021.
| Effects | GL | P-values | |||||||
| FH | FM | FA | BP | CP | LP | RP | RS | ||
| Season (S) | 2 | < 0.0001 | < 0.0001 | < 0.0001 | 0.0846 | 0.0003 | < 0.0001 | 0.0084 | < 0.0001 |
| Harvest (H) | 1 | 0.5739 | 0.8867 | 0.0534 | 0.6036 | 0.1374 | 0.2878 | 0.0965 | 0.0225 |
| S x H | 2 | < 0.0001 | < 0.0001 | 0.0004 | 0.0913 | < 0.0001 | 0.0042 | 0.0076 | 0.4999 |
Values in bold are significant (P < 0.05) or (P < 0.01) by the F test. FH-hermaphrodite flowers; FM-male flowers; FA-abnormal flowers; BP-panicle biomass; CP-panicle length; LP-panicle width; RP-primary branches; RS-secondary branches.
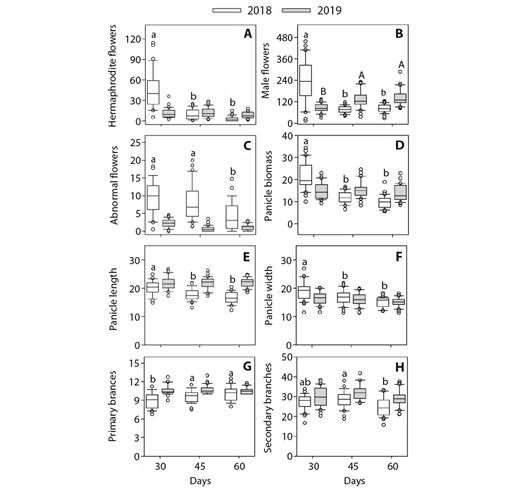
Fig. 3 Dwarf cashew plants showed higher flower production at the beginning of the flowering season. Boxplots of panicle floral morphology characters. A. Hermaphroditic flowers. B. Male flowers. C. Abnormal flowers. D. Panicle biomass. E. Panicle length; F. Panicle width; G. Primary branches. H.* Secondary branches) of dwarf cashew, analyzed in two harvests (2018 and 2019), in the municipality of Pacajus-CE, for the three periods after the beginning of flowering (30, 45 and 60 days). Only significant differences are shown. Different letters indicate significant differences between seasons for the same season by the Tukey test (P < 0.05). (*) indicates that there was a significant effect of crops (isolated effect).
During the 2018 harvest, the number of hermaphrodite and male flowers, panicle fresh biomass, and length and width of panicles showed similar trends, with higher mean values observed at 30 days after the commencement of flowering and lower mean values at 45 and 60 days after the initiation of flowering (Fig. 3A, Fig. 3B, Fig. 3D, Fig. 3E and Fig. 3F). With regards to the number of abnormal flowers, we observed mean values higher than 30 and 45 days and lower than 60 days after the onset of flowering (Fig .3 C).
The number of primary branches showed lower mean values at 30 days after the commencement of flowering and higher mean values at 45 and 60 days (Fig. 3G). Compared with 30 days after the initiation of flowering, mean values of number of secondary branches were higher at 45 days and lower at 60 days (Fig. 3H). With respect to the 2019 harvest, we detected significant variations only for the number of male flowers, with lower mean values at 30 days and higher mean values at 45 and 60 days after the beginning of flowering.
The effect of harvest was detected only with respect to the number of secondary branches (Fig. 3H), with the average number of secondary branches in the 2019 harvest being higher than that observed in the 2018 harvest.
The relationship between floral parameters and the expression of hermaphrodite flowers: Different models were obtained to explain the expression of hermaphrodite flowers at the three evaluation times (30, 45, and 60 days after the commencement of flowering) (Table 3). For the 30-day evaluation, the parameters contributing most to an explanation for hermaphrodite flower expression were panicle length and width, which together accounted for 47 % (R² = 0.47) of the numerical variations in hermaphrodite flowers. For the 60-day evaluation, panicle length and the number of primary branches were found to make the highest contribution, collectively explaining 37 % (R² = 0.37) of the numerical variation in hermaphrodite flowers. We were, however, unable to obtain a significant model for the 45-day evaluation period. Interestingly, although neither significant nor marginally significant (P = 0.102), the floral parameter that appeared to be the most important at this time-point was the number of male flowers.
Table 3 Contributions of dwarf cashew panicles morphological characters in the dynamics of explaining the emission of hermaphrodite flowers collected in two harvests, in the municipality of Pacajus, CE
| Flowering season | Characters | R² (partial) | Estimated | R² (model) | F Test | P |
| 30 | Panicle length (1) | 0.2621 | -6.3992 | - | - | - |
| Panicle width (2) | 0.2089 | 6.6411 | - | - | - | |
| (1)*(2) | - | - | 0.4710 | 25.37 | < 0.001 | |
| 45 | Male flowers (1) | 0.0454 | -0.0354 | 0.0454 | 2.76 | 0.102 |
| 60 | Panicle length (1) | 0.3449 | 1.0027 | - | - | - |
| Primary Branches (2) | 0.0333 | -1.0174 | - | - | - | |
| (1)*(2)* | - | - | 0.3782 | 17.34 | < 0.001 |
In terms of the accumulation of hermaphrodite flowers, a contrasting result was obtained with respect to panicle dimensions (length and width) during the first flowering season (30 days after the commencement of flowering), with the number of hermaphrodite flowers being positively correlated with panicle width. However, we detected a negative correlation with panicle length (Fig. 4A, Fig. 4B).
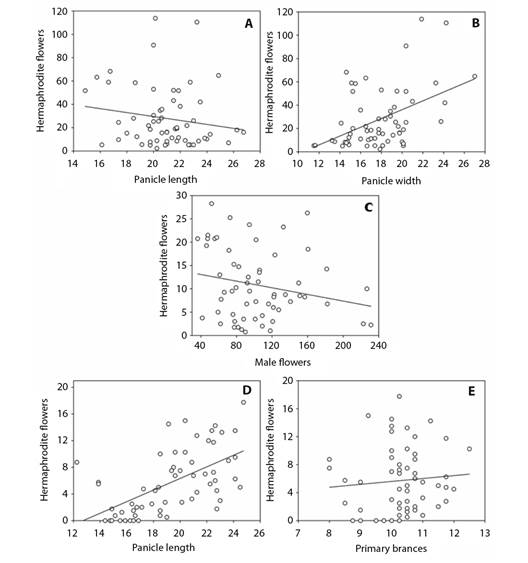
Fig. 4 Different floral morphological attributes explained the emission of hermaphrodite flowers in panicles. Stepwise regression analysis correlating morphological characters and the production of hermaphrodite flowers in dwarf cashew panicles in 2018 and 2019 crops, in the municipality of Pacajus, CE. Assessments performed at 30 days. A. Panicle length x hermaphrodite flowers, y = -1.6892x + 63.3501, R2 = 0.0267. B. Panicle width x hermaphrodite flowers, y = 3.7713x - 39.2612, R2 = 0.2095; P = <0.001), 45 days. C. Male flowers x hermaphrodite flowers, y = -0.0356x + 14.4965, R2 = 0.0460; P = 0.102), and 60 days. D. Panicle length x hermaphrodite flowers, y = 0.8702x -11.0664, R2= 0.3445. E. Primary branches x hermaphrodite flowers, y = 0.4163x + 1.4416, R2 = 0.0068; P = <0.001) after flowering starts.
Furthermore, there was a negative correlation between male and hermaphrodite flowers at 45 days after the onset of flowering (Fig. 4C). This negative effect of an increase in the production of male flowers suppresses the development of hermaphrodite flowers, the production of which was observed to be inversely proportional to the increase in the number of male flowers in panicles.
When evaluated after 60 days of flowering, it was found that the greater the panicle length, the higher the number of hermaphroditic flowers per panicle (Fig. 4D). At this time-point, we also detected an inverse correlation between number of primary branches and number of hermaphrodite flowers produced in inflorescences (Fig. 4E).
Discussion
The findings of this study revealed that with the exception of panicle biomass, all assessed morphological parameters of cashew panicles showed significant effects with respect to evaluation time-point. Contrastingly, for most parameters, we were unable to detect any significant differences between the two harvest seasons, which can be explained by the fact that the annual variations in abiotic factors were not pronounced enough to promote quantitative changes among the panicle parameters. The number of hermaphrodite flowers produced by cashew trees is dependent on parameters associated with the vegetative structures of panicles. The contribution of this factor to the expression of these flowers was found to differ when evaluated at three different time-points during the flowering period.
The cashew tree growth pattern alternates between vegetative and reproductive phases, whose onset and duration differ according to the genotype, as these phases are regulated by genetic and environmental factors (Saroj et al., 2014). Among the environmental factors, those that have the most pronounced effects on cashew growth are the amount of solar radiation and the availability of water, as they play a particularly important role in the opening of flower buds, triggered by the hormonal activity expressed in the plants (Saroj et al., 2014).
The higher rate of flower production observed at the beginning of the flowering season in the 2018 crop, with similar trends in the first evaluation period at 30 days, is consistent with corroborates the results of research carried out by Sharma (2009), who claimed to have reported a greater production of male and female flowers (hermaphrodites) in early flowering. Similarly, Wunnachit and Sedgley (1992) found that the majority of hermaphrodite flowers were produced during the first 21 days of the flowering period.
According to Parente et al. (1991), in the semi-arid regions of the tropics, dwarf cashew trees are known to display peak flowering in August, with greater flowering intensities at the beginning of the reproductive period. This behavior among panicles can be explained by the occurrence of higher temperatures during this period of the season. Our failure to detect any significant differences among the three periods evaluated in 2019 can be ascribed to the fact that transitions in the phases of the plant life cycle are regulated by genetic development programs, modulated by environmental and endogenous stimuli (Ravindra, 2014). Thus, we suspect that the environmental stimuli experienced by cashews during the 2019 season were not strong enough to promote any pronounced differences among parameters.
According to the meteorological data obtained for the experimental area during the 2018 and 2019 harvests, the period from August to November corresponds to the period with the lowest relative humidity, high average temperatures (close to 30 °C), and high solar radiation levels. These factors predictably contribute to greater variation in the floral biology of plants, affecting the emission of hermaphrodite flowers during the typical period of flowering of the crop (Fig. 1). In 2018, low amounts of rainfall were recorded during the months of August and December, which, together with the low relative humidity and high average temperatures, probably altered the microclimate of the area, thereby influencing the pattern of flowering. The occurrence of high temperatures during this period, along with the differences in humidity and solar radiation, enabled the cashew trees to channel larger amounts of photoassimilates to the formation of new branches, thus providing a physical structure of healthy branches for the support and favorable distribution of inflorescences.
At the onset of the flowering cycle, panicles assign a large proportion of the energy contained in photoassimilates to the growth of the main stem and the production of primary branches, which serve as a structural basis for the development of secondary branches. This development of secondary branches becomes more pronounced after 45 and 60 days of flowering, when there is a greater incidence of solar radiation and higher temperatures together with greater panicle growth. In this regard, several species exhibit gradual phenotypic changes during the flowering period, corresponding to variations determined by meteorological variables (Wadgymar et al., 2015). Changes in temperature during the dry season, together with a reduction in the length of the photoperiod, are considered factors most conducive to the promotion of flowering (Silvério & Lenza, 2010). The production of flowers during periods of lower rainfall is assumed to be a survival strategy, which, in addition to avoiding damage to the flowers from heavy rainfall (Janzen, 1967; Sousa & Cunha, 2018), makes the flowers more conspicuous to potential pollinators, thereby enhancing pollination.
In the present study, we found that the emission of hermaphroditic flowers was associated with the characteristics of other structural parameters of panicle morphology, which influenced the proportions of these flowers throughout the three evaluation periods. Structural parameters of panicle length and width, primary/secondary branches, and production of male flowers in panicles were identified as the most important determinants. In this sense, several studies have evaluated the influence of panicle structure on fruit production and quality, which are directly dependent on the density of fertile flowers suitable for pollination. Vidal-Neto et al. (2013) reported that panicle structure is an important factor, as it directly or indirectly influences the production of hermaphroditic flowers. Thus, the oscillation of climatic variables, mainly temperature, solar radiation, and humidity, between the evaluation periods, contributed to changes in the development pattern of hermaphrodite flowers in the panicles, reflecting changes in the available amounts of nutrients and photosyntates in the plants directed to the formation of flowers (Olsen & Martin, 1980).
We identified associations between different floral components and the emission of hermaphrodite flowers throughout the evaluation period. At 30 days after the onset of flowering, the length and width of the panicle were found to affect the number of hermaphroditic flowers, which to a large extent was expected, given that an increase in panicle dimensions provides mechanical support for better distribution of branches in the inner structure of the inflorescence. The flowering process is affected by the structural characteristics of the aerial parts (Ravishankar, 2014), with the composition of floral buds being altered by the degree of branch extension. Although this direct growth relationship is clear in cashew, the panicle does not grow in width and length simultaneously, which is taken to be a strategy of the plant to evenly distribute the photoassimilates among panicle, as the larger the panicle, the greater the potential number of hermaphroditic flowers.
An increase in the width of the inflorescences promotes a greater allocation of photosynthetic resources to the panicle structure, thereby initiating a greater expansion in the primary and secondary branches, the structures of which are essential for the effective distribution of new flowers. This broad inflorescence structure contributes to enhancing the pollination and fertilization of hermaphrodite flowers, which develop throughout the period of blooming. Conversely, the development of narrower panicles would limit the access of pollinators to flower buds, thereby leading to pollination deficits, and thus potentially compromising the desired productivity (Freitas, 2014). A further characteristic that tends to preclude the production of hermaphrodite flowers is the development of male flowers. Male flowers, when present in larger quantities in these inflorescences, can interfere with the proportion of hermaphrodite flowers exposed to pollinators.
With the progression of flowering time, we found that after 45 days, there was a direct relationship between the emission of hermaphrodite flowers and the trend in male flower production. During this period, there was peak in male flower production, which accordingly represents greater competition for photoassimilates. Flowers are normally supplied via the formation of new buds, which serve as a depot for carbohydrates in the subterranean and aerial organs of the plants (Baptist et al., 2009). Therefore, if the production of flowers is sustained by the provision of photoassimilates, large amounts of which are consumed in the production of male flowers, there will be a reduction in the availability of these resources for the production of hermaphrodite flowers. Another important feature to be highlighted in this context is that flowers of the cashew tree abort during flowering. Consequently, floral parameters that might initially contribute to flower development may no longer make a prominent contribution when evaluated later during the flowering period, as the number of hermaphrodites tends to decline as a consequence of abortion.
The production of large numbers of male flowers consumes large amounts of energy, the expenditure of which results in a reduction in production efficiency (Araújo, 2013; Pinheiro et al., 1993). In this regard, anacardiaceous species are typically characterized by prolific flower abortion and, accordingly, it can be conjectured that over time, the length and width of panicles might no longer explain the number of hermaphroditic flowers present. At 60 days into the flowering period, it is likely that the composition of the continually developing inflorescences comprise not only hermaphroditic, but also male and abnormal flowers. During this period, increases in the length and width of panicles occur as a consequence of the sustained input of photoassimilates throughout the flowering phase, which enables the different cashew genotypes to maintain the growth of vegetative structures. This is consistent with the findings of Saroj et al. (2014), who observed that the proportion and emission of hermaphrodite flowers differed significantly from the development of new branches, due to the vegetative growth of the species.
In the present study, we detected positive and negative linear correlations between the morphological attributes of panicles and the emission of hermaphrodite flowers, reinforcing the fact that cashew flowers show associations with the morphological structure of panicles, irrespective of the evaluation period. Positive linear correlations with structural components at the beginning of the reproductive period indicate that during this period, plants require a sufficient quantity of photoassimilates to strengthen development of the panicle structure, contributing to an increase in panicle width. Furthermore, the negative linear correlation between panicle length and the production of hermaphroditic flowers indicates that the greater the extension in panicle length, the lower the production of hermaphrodite flowers.
In our evaluation of flowering at day 45, we detected a negative linear correlation between the number of male and hermaphrodite flowers, indicating that a higher density of male flowers tends to suppress the production of hermaphrodite flowers, and vice versa, due to competition for plant photoassimilates. Evaluations performed at day 60 revealed positive linear correlations between the length and number of primary branches, and the production of hermaphrodite flowers in the panicles. On the basis of these findings, we can deduce that as flowering progresses, there is a conversion of energy to promote an increase in the growth of the panicle main axis, thereby positively influencing the production of hermaphrodite flowers in the branches. This, in turn, can probably be attributable to the fact that a larger panicle area is conducive to increased photosynthesis, thereby channeling larger amounts of photosynthates for flower production.
With the advance of flowering, floral structures show significant development, concomitant with a gain in fresh plant biomass and an increase in branch expansion. Given their influence on the structure of panicles, the primary branches of panicle have a notable influence on the proportion of hermaphrodite flowers, with the number of these branches showing a positive correlation with the number of hermaphrodite flowers. The greater the number of primary branches, the greater the support base for the development of new secondary branches, which in turn facilitates the development of new floral apices.
The flowering distribution in cashew was found to be strongly dependent on the development of the morphological parameters of panicles, which changed over time. These alterations promoted changes in the vegetative structures of the species associated with an elongation of the internodes, which subsequently developed into productive branches, with the production of an inflorescence in the terminal part of the newly formed bud. Thus, the emission of hermaphrodite flowers was altered due to characteristic variations in the floral and reproductive morphology of the crop, consequently determining the potential production.
Collectively, our findings thus indicate that changes in the vegetative structure at different time-points in the flowering period and the variation between different harvest seasons lead to differences in the parameters of panicle floral morphology. Another important finding is that the numbers of hermaphrodite flowers were mainly influenced by panicle length and width, the presence of primary branches, and the production of male flowers in panicles. A greater number of hermaphrodite flowers in the panicles is fundamental in the selection of early dwarf cashew tree clones.
Ethical statement: the authors declare that they all agree with this publication and made significant contributions; that there is no conflict of interest of any kind; and that we followed all pertinent ethical and legal procedures and requirements. All financial sources are fully and clearly stated in the acknowledgements section. A signed document has been filed in the journal archives.
APPENDIX Morphological characters of panicles and their relevance to the fitness of Anacardium occidentale
| Morphological Characters | Relevance to plant fitness |
| Number of male flowers | Pollen grain production. The formation of the pollen grain plays a role in fertilization and fruit production. A greater energy expenditure of the plant for the production of pollen, results in a low productive efficiency. |
| Number of hermaphrodite flowers | Fruit production. In addition to the male gamete (pollen grain), which serves as food for visiting insects, this type of flower also has the female gamete (egg), located inside the ovary. Therefore, these flowers result in the production of fruits. |
| Number of abnormal flowers | They have a stamen arrangement similar to that of staminate and hermaphrodite flowers; however, they do not present the most developed stamen or the pistil, being, therefore, denominated as anomalous. They affect the concentration of fertile flowers that are suitable for pollination. Differences related to floral structures contribute to variability in flower quality, pollination efficiency, and uneven production. |
| Panicle biomass | Allocation of photosynthetic resources that are directed to production of new floral gems. The panicle area favors the increase of photosynthesis, energy conversion and the targeting of photosyntates for the production of flowers. |
| Panicle length | Panicle length extension. The expansion of the forming panicle affects the number of flowers on the lateral and sublateral branches. |
| Maximum panicle width | Greater dimensioning of photosynthetic resources in the partitioning of the panicle structure, promoting greater expansion of primary and secondary branches, whose structure is essential for the distribution of new flowers. |
| Number of panicle branches | Physical structure of healthy branches for the support and distribution of inflorescences. They serve as a structural basis for the development of primary and secondary branches, which will support the development of flower buds. |












 uBio
uBio 


